Case Report, Int J Cardiovasc Res Vol: 9 Issue: 3
Acute Rheumatic Fever Presenting With Severe Epistaxis in A Child in Sokoto, Nigeria
Khadijat O Isezuo1*, Usman M Sani1, Usman M Waziri1, Bilkisu I Garba1, Yahaya Mohammed2, Lukman K Coker1 and Monsurat A Falaye1
1Department of Paediatrics, Usmanu Danfodiyo University Teaching Hospital, Sokoto, Nigeria
2Department of Medical Microbiology, Usmanu Danfodiyo University Teaching Hospital, Sokoto, Nigeria
*Corresponding Author: Dr. Khadijat O. Isezuo
Department of Paediatrics,
Usmanu Danfodiyo University Teaching Hospital, Sokoto, Nigeria
E-mail: khadisez@yahoo.com
Received: March 15, 2020 Accepted: April 30, 2020 Published: May 07, 2020
Citation: Isezuo KO, Sani UM, Waziri UM, Garba BI, Mohammed Y, et al. (2020) Acute Rheumatic Fever Presenting With Severe Epistaxis in A Child in Sokoto, Nigeria. Int J Cardiovasc Res 9:3.
Abstract
Background: Rheumatic Heart Disease (RHD) is a preventable cause of cardiovascular death and disability. It is preceded by Acute Rheumatic Fever (ARF), which has some diagnostic criteria. Some cases present with atypical features including epistaxis, which occurs in about 4% of ARF. Objective: To report a case of a child who had febrile illness with epistaxis, then 2 months after developed rheumatic valvular heart disease. Case Report: A 10-year old girl with one-week history of high-grade fever, A day history of bleeding through the nostrils. She did not have sore throat, joint pains nor past history of epistaxis. She was febrile (38.70 C), moderately pale and anicteric. She was transfused within 24 hours, treated for sepsis and malaria. She had features of cardiac decompensation on 4th day of admission despite adequate blood transfusion. Results of complete blood count, peripheral blood film and clotting profile were not suggestive of bleeding abnormalities. Fever resolved after several antibiotics and antimalarials. She defaulted follow up for 2 months then presented with progressive easy fatiguability, cough and fast breathing. No prior fever, joint pains or sore throat. She was in respiratory distress and tachycardic. She had displaced apex beat and apical pansystolic murmur. Echocardiography revealed RHD with mitral and aortic regurgitation. She is presently on follow up in Paediatric cardiac clinic. Conclusion: The patient had febrile illness with epistaxis and heart failure some weeks before diagnosis of RHD supporting the possibility of the initial presentation to be probably ARF with epistaxis. At risk children presenting with fever, epistaxis and cardiac decompensation should be evaluated for ARF.
Keywords: Epistaxis; Rheumatic heart disease; Child; Sokoto
Keywords
Epistaxis; Rheumatic heart disease; Child; Sokoto
Introduction
RHD is sequelae of ARF, which is in turn preceded by streptococcal sore throat infection with Group A Streptococcus (GAS). It is disease associated with poverty, overcrowding, poor environmental sanitation and other social determinants of poor health [1]. RHD is a chronic cardiac condition that causes high disease burden in lowincome settings. It disproportionately affects children and younger adults amounting to about 30 million people worldwide with 300,000 deaths yearly [2,3].
Early recognition of features of the ARF is important to prevent RHD, however, these are similar to many other conditions that present in areas where infectious diseases predominate. The diagnostic criteria (Jones’ criteria) has undergone revisions since it was first propounded in 1944, the most recent being in 2015 with modifications for the echocardiographic era [4,5]. As laboratory medicine and diagnostics evolved, some minor clinical criteria of ARF (epistaxis, abdominal pain, pulmonary findings) were removed and replaced by more specific evidence of inflammation like elevated C-reactive protein, erythrocyte sedimentation rate and positive throat swab for GAS [6].
However, some cases of ARF may not be diagnosed early enough because they present with these non-specific features [7]. Recent reports show some patients with initial unlikely diagnosis of ARF had presented with epistaxis and febrile illness as their initial symptom. [8,9].
This is a case of a child who was managed for probable sepsis with epistaxis but later presented with established valvular heart disease few months after.
Case Report
A 10-year old girl presented to the emergency paediatric unit with a week history of high-grade fever and a day history of bleeding through the nostrils and mouth. She did not have preceding sore throat, joint pains, trauma nor bleeding from any other orifices. She did not have any risk factors for bleeding.
Relevant examination findings were that of an acutely ill-looking patient in respiratory distress, febrile with axillary temperature of 39.1oC- 39.8oC, clinically pale, with active bleeding per nostrils with clot formation. She had low volume peripheral pulses with pulse rate of 124 /min and blood pressure of 90/40 mmHg. Her precordium was not bulging, normoactive with apex beat at the left fifth intercostal space and heart sounds were normal. Respiratory rate was 36 breath/ min with normal breath sounds. There were no palpable abdominal organs.
She had two episodes of bleeding warranting blood transfusion within few hours of admission and nasal packing by the otorhinolaryngologist. The working diagnosis was probable sepsis with differentials of severe malaria with DIC and viral haemorrhagic fever (probably Dengue fever).
Results of investigations included haematocrit of 29%, complete blood count that showed normal leucocyte count with predominant granulocytes (Total leucocyte count-4 x 109/L, Granulocytes–41.1%, Lymphocytes-36.4%, Platelet count-151 x 109/L.). Haematocrit eventually declined to 26% and then 23% within 24 hours necessitating blood transfusion. Post transfusion haematocrit was 40%. Peripheral blood film showed features of mild to moderate hypochromic microcytic anaemia. Erythrocyte sedimentation rate was not done during the first admission.
There was positive blood smear for malaria parasites. Serum electrolytes, urea and creatinine were normal. Clotting profile was normal with INR of 1.21 (0.8–1.5). Liver function tests were also normal. Retroviral and viral hepatitis screening were all non-reactive. In addition, tests for Dengue and Lassa virus were negative.
She had antimalarial medications (intravenous artesunate and oral artemisinin combination therapy), antibiotics (intravenous ceftriaxone, and metronidazole and oral cefixime), but with persistence of fever was commenced on intravenous Ciprofloxacin and Gentamicin. She also had intravenous Diacynone.
She developed abdominal pain on the 4th day of admission and was still febrile. She was tachypnoeic (respiratory rate of 44 breaths/ min), tachycardic (pulse rate of 140bpm) with tenderness of the right hypochondrium (liver span of 10cm). Heart sounds were normal and there was no cardiac murmur. Haematocrit at that point was 29%.
The above treatment was continued and she responded gradually to the medications. After 9 days on admission, her caregivers requested for discharge and to complete investigations on outpatient basis. Third heart sound was noted to present even after fever resolution at point of discharge.
She defaulted follow up then presented with 2 months history of progressive easy fatiguability, cough and fast breathing. There was associated history of dyspnoea on exertion and orthopnoea. There was no fever or joint pains nor any other symptom of ARF. On examination, she was in heart failure. She had cardiomegaly (bulging, hyperactive precordium, apex impulse at 6th left interspace anterior axillary line) and cardiac murmurs of mitral and aortic regurgitation.
Erythrocyte Sedimentation Rate (ESR) was elevated at 80mmfall/hour. Anti-Streptolysin O (ASO) Titre was reactive. Electrocardiogram (ECG) revealed a prolonged PR interval with features of left ventricular hypertrophy. Echocardiography (ECHO) was consistent with RHD with mitral and aortic incompetence. It is shown in (Figure 1) that there is thickening of the mitral valve leaflets on parasternal long axis view while (Figure 2) shows the mitral regurgitant jet on colour doppler echo.
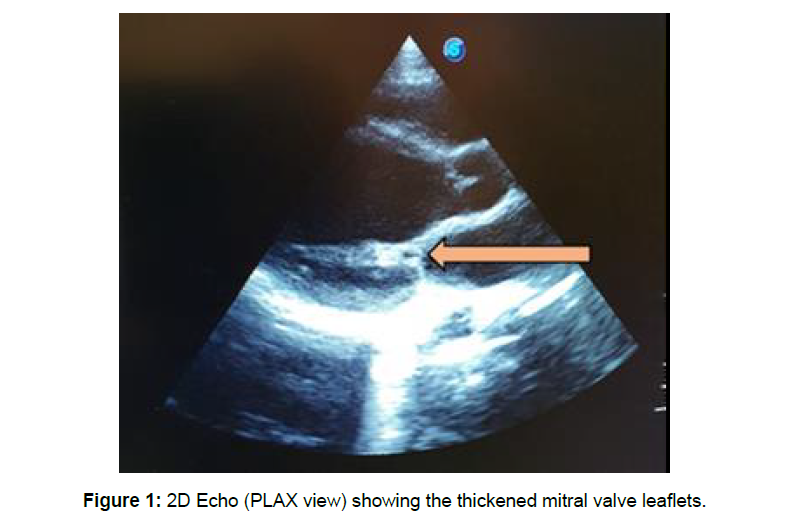
Figure 1: 2D Echo (PLAX view) showing the thickened mitral valve leaflets.
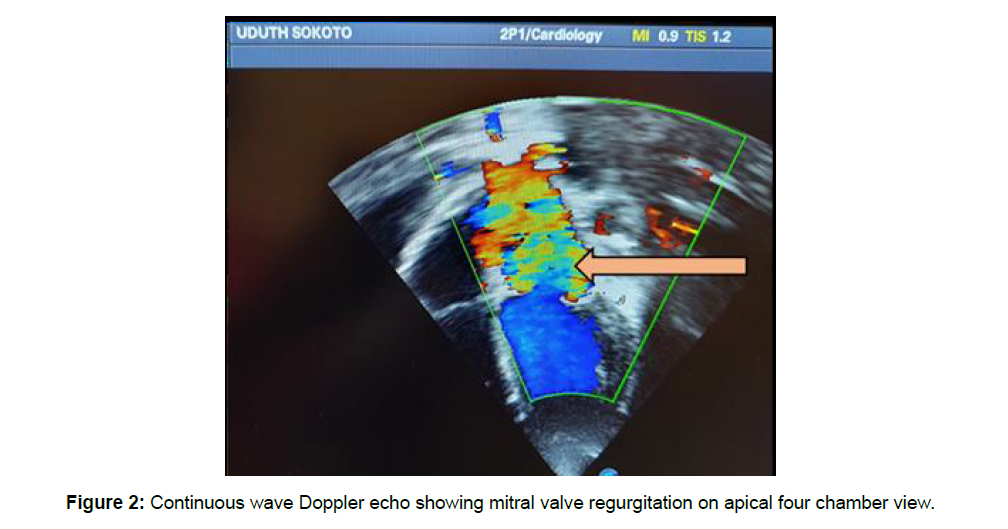
Figure 2: Continuous wave Doppler echo showing mitral valve regurgitation on apical four chamber view.
She was admitted, managed for rheumatic carditis with bed rest, prednisolone and diuretics. Secondary prophylaxis with benzathine penicillin was instituted. She improved gradually and stabilized. Counselling of the patient and caregivers on the disease condition was done.
She had two repeat episodes of epistaxis after the initial episode at onset. These were associated with low-grade fever. She presented to the otorhinolaryngologist and was managed with nasal packing. Complete blood count done was normal. There were no abnormalities on clotting profile tests and liver function tests.
Discussion
There is a 4% probability of developing epistaxis in a patient with acute rheumatic fever, which increases to 9% in those who had recurrence of RHD [3]. The pathogenetic mechanism explaining epistaxis in ARF is linked to vasculitis that occurs during the inflammatory process which makes the small fragile capillaries in the nasal mucosa to be more susceptible to minor trauma [10,11]. Following this, is inhibition of platelet aggregation by the fibrinoid degeneration of the fibrous tissue that prevents timely sealing of the ruptured capillaries to achieve homeostasis leading to prolonged and severe bleeds.
Other reasons postulated include low phospholipids in the blood and autoimmune reaction affecting platelets [10]. Mitral stenosis which is a long-term sequelae especially in adults has been associated with epistaxis and this is attributed to probable high systemic vascular resistance transmitted to the fragile nasal mucosa leading to rupture and bleeding [12]. However, our patient does not have mitral stenosis which is more of a complication in adults from long standing mitral valve damage. This patient had fever with severe episode of epistaxis warranting blood transfusion. However, there was no history of preceding sore throat nor any other criteria of ARF. She developed multivalvular disease about 2 months later as she became slowly symptomatic during her period of default. The absence of history of sore throat at initial presentation of this patient is not surprising as about 50% of streptococcal pharyngitis complicated with ARF may be unnoticed or asymptomatic [13]. Her case is similar to that reported by Pathania in a male adolescent who presented with fever and repeated epistaxis but did not have any cardiac symptomatology at presentation similar to our patient, however, he had sore throat with added findings of wide pulse pressure, collapsing pulse which led to the echocardiographic confirmation of multivalvular rheumatic heart disease [9]. It was deduced that such patients who have absence of overt cardiac symptoms may have subclinical or indolent carditis and will present later with overt cardiac failure as our patient did [9]. Our case did not have a cardiac murmur during the initial presentation with epistaxis although, there were features to suggest cardiac decompensation.
A similar case reported by Arslan (8) had fever, epistaxis and cardiac murmur. It has also been suggested that presumptive diagnosis of ARF can be made with one minor criterion with any of these features which include anaemia, abdominal pain, rheumatic pneumonia, unilateral pulmonary oedema, atypical articular involvement, silent carditis, low-grade fever, rapid sleeping pulse rate, tachycardia out of proportion to fever. Epistaxis now being increasingly seen in atypical cases of ARF, may therefore also be considered [8,9]. Our patient was not diagnosed with ARF, however, during the first admission with epistaxis because it was not suspected. The only probable clue to her diagnosis retrospectively was the presence of a third heart sound present at the point of discharge. Our patient has had 2 severe episodes after the first.
This is similar to what Kugelmass reported that epistaxis is an early sign which may herald the onset of ARF, an exacerbation or even occur during convalescence [11]. It occurs repeatedly and becomes more severe with time. There are also reports of delayed diagnosis of ARF even in patients who had the major and/ or minor criteria highlighting the need for a higher index of suspicion [6,7,14,15]. This phenomenon may also be present as an “iceberg phenomenon” with many more undiagnosed cases in the community at the lower levels of health care. These cases now present with the sequelae of RHD and are from poor families that cannot even afford the cost of surgery which is not readily available for now in the country.
Conclusion
Children with ARF may present occasionally with fever and epistaxis. High index of suspicion is necessary so as not to overlook the possibility of ARF in children presenting with prolonged or recurrent epistaxis.
References
- Meira ZM, Goulart EM, Colosimo EA, Mota CC (2005) Long term follow up of rheumatic fever and predictors of severe rheumatic valvar disease in Brazilian children and adolescents. Heart 91: 1019-1022.
- Abrams J, Watkins D, Abdullahi LH, Engel ME, Zuhlke LJ (2019) Integrating the prevention and control of rheumatic heart disease into country health systems: a systematic review protocol. BMJ Open 9: e028908.
- Watkins DA, Johnson CO, Colquhoun SM, Karthikeyan G, Beaton A, et al. (2017) Global, Regional, and National Burden of Rheumatic Heart Disease, 1990-2015. N Engl J Med 371: 713-722.
- Jones T (1944) Diagnosis of rheumatic fever. JAMA 126: 481-484.
- Gewitz MH, Baltimore RS, Tani LY, Sable CA, Shulman ST, et al. (2015) on behalf of the american heart association committee on rheumatic fever, endocarditis, and kawasaki disease of the council on cardiovascular disease in the young. revision of the jones criteria for the diagnosis of acute rheumatic fever in the era of Doppler echocardiography: a scientific statement from the American Heart Association. Circulation 131: 1806-1818.
- Kadir IS, Barker TA, Clarke B, Denley H, Grötte GJ (2004) Recurrent acute rheumatic fever: a forgotten diagnosis? Ann Thorac Surg 78: 699-701.
- Kaminecki I, Verma R, Brunetto J, Rivera LI (2018) Delayed diagnosis of acute rheumatic fever in a patient with multiple emergency department visits. Case Rep Pediatr 2018: 9467131.
- Arslan D, Guvenc O, Cimen D, Oran B (2015) An Unusual Presentation of Acute Rheumatic Fever. Eur J Gen Med 12: 358-360.
- Pathania M, Rathaur VK, Pattupara AJ, Arya S, Bansal S, et al. (2018) Kaeley N. Epistaxis as the initial presentation in a case of rheumatic heart disease. J Fam Med Prim Care 7: 1136-1138.
- Abdj Z, Abul Fadl M, El Shinnawy R, El Masri B, El Shinnawy M (1975) Mechanics of bleeding in Rheumatic fever. Sudanese J Paeditr 115–118.
- Kugelmass I (1947) Vitamin P in rheumatic epistaxis. Arch Otolaryngol 46: 684-685.
- Kumar S, Jain A, Jain S (2009) Rheumatic mitral stenosis with epistaxis as an initial symptom. Singapore Med J 50: e74-75.
- Karthikeyan G, Guilherme L (2018) Acute rheumatic fever. Lancet 392:161-174.
- Ilgenfritz S, Dowlatshahi C, Salkind A (2013) Acute rheumatic fever: case report and review for emergency physicians. J Emerg Med 45: e103-106.
- Rheumatism as a Cause of Epistaxis in Children (1904). The Hospital 36: 293.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 



