Research Article, Res J Zool Vol: 1 Issue: 1
Appearance of B Chromosomes like Structures in Apodemus Peninsulae Primary Cell Culture
Ostromyshenskii DI1,2, Kuznetsova IS3, Podgornaya OI14,5 and Kartavtseva IV5*
1Institute of Cytology RAS, St Petersburg 194064, Russia
2Far Eastern Federal University, Vladivostok 690922, Russia
3School of Biomedical Sciences, The Chinese University of Hong Kong, Shatin, Hong Kong
4St. Petersburg State University, St Petersburg 199034, Russia
5Federal Scientific Center of the East Asia Terrestrial Biodiversity, Far East Branch of RAS, Vladivostok 690022, Russia
*Corresponding Author : Irina V. Kartavtseva
Federal Scientific Center of the East Asia Terrestrial Biodiversity, Far East Branch of RAS, Vladivostok 690022, Russia
Tel: +7-4232-318198
E-mail: kartavtseva@biosoil.ru
Received: January 16, 2018 Accepted: February 20, 2018 Published: March 05, 2018
Citation: Ostromyshenskii DI, Kuznetsova IS, Podgornaya OI, Kartavtseva IV (2018) Appearance of B Chromosomes like Structures in Apodemus Peninsulae Primary Cell Culture. Res J Zool 1:1.
Abstract
B chromosomes (Bs) have been found in all major eukaryotic clades. Specimens of Korean field mouse (Apodemus peninsulae) from populations inhabiting the mainland of the Russian Far East, Sakhalin and Stenina Islands were used. Animals from wild populations and Hokkaido Island in the Japan contain Bs while ones of Russian island populations do not have Bs. Cell cultures have been established from mouse tails fibroblasts from both island populations; in 3-5 passages, Bs-like elements appear in cultures cells. CENP-A chromatin immunoprecipitation (AChIP probe) on cell culture A. peninsulae from the Stenina Island has been undertaken. CENP-A precipitated DNA and probe of mouse centromere were used to mark all the heterochromatic regions of A. peninsulae and A. agrarius species. Bs-like elements from cultured cells of A. peninsulae show the same tendency as in wild populations: there are weakly and strongly labelled by AChIP probe amongst them Bslike elements, which appear in cultured cells of island populations (Stenina, Sakhalin) and wild populations of Hokkaido Island, are more similar in morphology compared to the Bs from the animals from Russian Far East. The data obtained suggest that the mice of the three islands 7-10 thousand years ago belonged to a single population, have a similar gene pool, and differ from the gene pool of continental populations of mice from the Far East.
Keywords: Heterochromatin; CENP-A chromatin immunoprecipitation (ChIP); Islands populations
Introduction
B chromosomes (Bs or supernumeraries, extra chromosomes) have been found in all major eukaryotic clades. In mammals, where the majority of about 5000 known species are karyotyped, Bs was reported for 75 species [1,2]. Traditionally, Bs were considered to be totally heterochromatic (i.e. transcriptionally inactive) or, if they had evolved recently, undergoing the heterochromatisation process. The heterochromatic nature of Bs agrees with their high variability and these elements often demonstrate typical C-positive staining [3].
The studies of Bs, involving Bs isolation by either flow sorting or micro-dissection followed by reverse painting, provide some information about homologous regions between autosomes and Bs [2,4-6]. Based on the presence of the same orthologous protooncogenes in canids and brocket deer Bs, it is supposed that genomic regions involved in Bs formation are not random [7]. The highly repetitive content of Bs remains enigmatic as well as the content of the heterochromatic regions [8]. Both centromeric (CEN) and pericentromeric (periCEN) regions are enriched with tandem repeats (TR, satellite repeats, satDNA), so these regions remain unassembled in the majority of the genomes sequenced due to the lack of adequate bioinformatics methods and algorithms. Besides TR, the repetitive sequences of transposable elements (TE) class are members of heterochromatic regions, but the investigation of their state is still at an early stage [9].
Cytogenetic studies of Bs molecular composition used repetitive probes to find specific sequences localised on Bs. Amongst the sequences found were tandemly arranged repetitive elements [10], LINEs (long interspersed nuclear elements) and SINEs (short interspersed nuclear elements) [11], interstitial telomeric sequences [12,13], ribosomal DNA clusters [14,15] or histone genes [16]. There is a growing body of evidence that Bs, once regarded as totally heterochromatic and genetically inert, harbour clusters of ribosomal RNA genes, processed pseudogenes and protein-coding genes [2].
Bs of both deer investigated bear a higher proportion of repeats compared to bovine autosomes. Repeat family composition of grey brocket deer Bs was similar to cattle autosomes, while the largest repeat clusters from Siberian roe deer Bs were often unannotated or comprised of low complexity and satellite repeats. There is a cluster that contained virtually intact copies of LTR retrotranspons from the ERVK family, indicating a recent repeat expansion in brocket deer Bs [7].
In natural populations of the genus Apodemus, three species have Bs: A. argenteus, A. peninsulae and A. agrarius, but only the Korean field mouse A. peninsulae has normal karyotype (48 acrocentric) with Bs in almost all populations; Bs representation can reach 100 percent in the population [17]. The Korean field mouse is a convenient species to study the supernumerary chromosomes. Bi-armed Bs is different from the basic set of acrocentric chromosomes and they are very frequently present in the karyotype of individuals from different continent populations. Sizes of Bs vary from large bi-armed to small and micro chromosomes [18-20]. Extensive cytological studies of Bs by fluorescent in situ hybridisation (FISH) in the Korean field mouse have been on-going for many years [6,21-24] but their functions and content remain obscure. A. peninsulae exhibits Bs polymorphism with individual variants of six B classes throughout its vast range (from Altai to the Pacific Ocean coast) except for Sakhalin and Stenina Islands [20,25-27]. The number of Bs of A. peninsulae varied up to 7 in continental and up to 14 in insular populations (Hokkaido) of the Far East [26].
The colonisation patterns on islands have long been of major interest for studying evolutionary processes [28,29]. Islands are considered natural laboratories for new adaptations due to their restricted scale, isolation and sharp boundaries. Colonisation is usually accompanied by adaptive changes, but it is a long standing question whether fast adaptations are constrained by the initial paucity of variation caused by a genetic bottleneck of only a few arriving individuals [28]. A. peninsulae has been the constant inhabitant of the three Islands (Sakhalin, Stenina and Hokkaido) for about 7,000, 11,500 and 12,000 years respectively [30] and therefore have been long established in their environment.
The main purpose of this work was to study the appearance of Bs-like elements in the cell culture of A. peninsulae from Sakhalin and Stenina Islands, their preliminary characteristics and comparison with Bs in bone marrow cells from the wild population of A. peninsulae from the Russian Far East as well as from Hokkaido Island. In addition, it was attempted to determine the types of pericentromeric regions in Bs-like elements in the cell culture with autosomal pericentromeric heterochromatic regions of A. peninsulae and Mus musculus by FISH.
Material and Methods
Specimens
A. peninsulae mice were captured in 2006: (1) in the vicinity of lake Evoron (mainland, Khabarovskii krai, (51º 22.08’N; 136º 31.09’E), 3 females: 2043, 2053, 2055); (2) on the Stenina Island (Primorskii krai, (42º 43.54’ N; 131º 30.09’E), 4 females: 2-06, 3-06, 4-06, 5-06); (3) captured in 2007 on the Sakhalin Island (near Okha city (53º 51.43’N; 142º 53.33’E), 3 females: 6-06, 7-06, 8-06). A. agrarius was trapped in 2006 in the vicinity of lake Evoron (mainland, 2 males: 1398, 1406). The Mus musculus line has been kept in the Institute of Cytology RAS (INC RAS, St. Petersburg, Russia) animal house. Male and female CBA mice (3-6 month-old) were housed and maintained according to the approved standards in the Laboratory Animal Resources facility at INC RAS. Bone marrow metaphase plates from wild populations were made by the standard method. Bone marrow metaphase plates were undertaken by using an injection of colcemid (200 μg/mL) into the peritoneum of mice. Bone marrow cells were flushed with 0.06M potassium chloride and fixed with methanol: acetic acid (3:1) solution.
Primary culture and chromosomes preparation
The primary fibroblast cultures were set up from pieces of the tail of A. peninsulae from Stenina and Sakhalin Islands. The sample of skin was placed in a sterile Petri dish and cut into small pieces using scissors. Then pieces of dissected skin were washed a number of times by DMEM/F12 medium with antibiotic/antimycotic solution (Sigma) and seeded into cell culture dishes in DMEM/F12 medium, supplemented with 10% foetal calf serum with antibiotic and antimitotic solution (Sigma). Cells cultures were grown at 37ºC with 5% CO2. The cell went through 3-5 passages until Bs-like elements appeared, then the culture was frozen and stored, but it could not be recovered in a viable state after thawing. After formation of monolayer cell went through passages and was examined at 3-5 and up to 10 passages. Metaphase chromosomes were isolated from cells blocked in mitosis by 0.5 mg/ml colcemid (Gibco). Chromosome suspensions were fixed in methanol/acetic acid (3:1).
Mus musculus panCEN probe
A total CEN (panCEN) probe from M. musculus was obtained by micro-dissection of centromeric and pericentromeric regions of several chromosomes (3-5, a kind gift from Dr. V. Trifonov, Institute of Molecular and Cell Biology, Novosibirsk, Russia) followed by DOP-PCR (intervals) amplification (primer 5’-CCGACTCGAGNNNNNNATGTGG-3’) [31]. The obtained product underwent an additional amplification cycle with biotin-16-dUTP to insert the label. The resulting probe was used for FISH.
CENP-A ChIP and Probe
Chromatin immunoprecipitation has been undertaken with the fibroblast cell culture of A. peninsulae (5th passage, Stenina Island). 7×106 cells per ml were used; formalin was added to the cells’ suspension up to 1%; in 10 min. Glycine, up to 0.025 M was added for 5 min. to stop crosslinking; cells were precipitated at 1500 rpm for 5 min and sediment was re-suspended in cold 1×PBS with 10 üÃÂœ PMFS. After further centrifugation under the same conditions, the cells’ pellet was re-suspended in lysis buffer 1 (5 mM PIPES, 85 mM KCl, 10 üÃÂœ PMSF) and kept in ice for 10 min. The nuclei, precipitated at 5000 rpm for 3-5 min, were re-suspended in lysis buffer 2 (50 mM TrisHCl рà8.0, 10 üÃÂœ EDTA, 1% SDS, 10 üÃÂœ PMSF) and kept in ice for 10 min. Chromatin was sonicated to the average length 1000 bp. 10 μl of pretreated (blocked) sepharose A was added to the fragments’ mixture and kept in ice for 15 min. Chromatin fragments were cleared by centrifugation at 13000 rpm for 15 min. The supernatant fluid was transferred to a new tube and diluted in dilution buffer (0.01% SDS; 1.1% Triton; 1.2 mM EDTA; 16.7 mM Tris; 16.7 mM NaCl). The supernatant with dilution buffer was incubated with 2 μg of anti-CENP-A antibody (Abcam) overnight at 4º C with rotation. Immune complexes were separated on 10 μl of protein A Sepharose. Sepharose A was added and mixture incubated for 15 min. After centrifugation at 13000 rpm for 15 min, the sepharose A was washed 3 times for 5 min. with elution buffer (50 mM TrisHCL pH 8.0, 1% SDS, 1 üÃÂœ EDTA). DNA was eluted by adding 150 μl elution buffer and shaken on a vortexer for 15 min. Sepharose was sediment by centrifugation at 13000 rpm for 5 min. DNA after DOP-PCR amplification was used for FISH.
FISH (fluorescent in situ hybridization)
Slides with metaphase spreads were treated in RNAse (SigmaAldrich): stock solution (10 μg/ml) diluted 1:200 by 2х SSC for 45-60 min. at 37 º C, washed 3 times for 5 min. with 2х SSC. DNA denaturation was conducted by incubating the slides in 70% formamide, 2x SSC for 3-5 min. at 65º C. After the denaturation, the slides were dehydrated in an ethanol series at -20º C, quickly dried and incubated in the hybridisation mixture (5 ng/μl probe, 25% formamide, 4x SSC) for 17-24 h at 37º C. After the hybridisation, the slides were washed three times for 5 min. with 2х SSC at RT. If the biotin labelled probes were used, the slides were incubated with streptavidin- Alexa 568 (Invitrogen) (1:200 in 2x SSC, 5% BSA) for 30 min. at 37ºC and then washed 3 times for 5 min. with 2х SSC at RT. Signal amplification was then performed by treating the slides with a biotinylated antistreptavidin (Vector Laboratories) 1:200 in 2 x SSC, 5% BSA, 30 min. at 37ºC) , washing with 2x SSC (3 times for 5 min. at RT), incubating again in streptavidin-Alexa 568 (30 min. at 37ºC) and finally washing with 2x SSC (3 times for 5 min. at RT). In case of immune-FISH the hybridization procedure preceded by incubation with CENP-A antibodies and fixation. The slides were finally mounted in Prolong Gold Antifade with DAPI (Invitrogen) and stored in a refrigerator in the dark. Images were captured with a Nikon (CCD) camera on a Zeiss/MetaMorph epifluorescence microscope and prepared for publication using Adobe Photoshop CS2.
Results
Cell cultures have been established from mouse tails fibroblasts of A. peninsulae; in 3-5 passages, Bs-like elements appear in cultures cells. Primary cell cultures have been established from Stenina and Sakhalin Island populations and both lines show a similar type of karyotypes’ rearrangement (Figure 1a). Bs-like elements, once appearing, did not change in quantity and morphology in advanced passages of each cell line. There are 2-5 Bs-like elements in cell cultures from both Sakhalin and Stenina Islands (Figures 1a and 2a). The cultivation results from both cultures were polyploidisation (Figure 1a). The karyotype has been determined as 2n=48+2-5 Bs-like elements during first 3-5 passages (Figure 2a). Karyotypes of cell culture on 10 passages have 56 or 58 acrocentric chromosomes and one metacentric chromosome with multiple heterochromatic regions and 2-5 Bs-like elements (2n=57-59+2-5 Bs-like elements) (Figures 1a and 2c). Most probably, the metacentric chromosome with multiple heterochromatic regions was rearranged from X chromosomes. None of the cultures underwent spontaneous malignization to produce permanent cell lines and all of them ceased (terminated) in 12 passages.
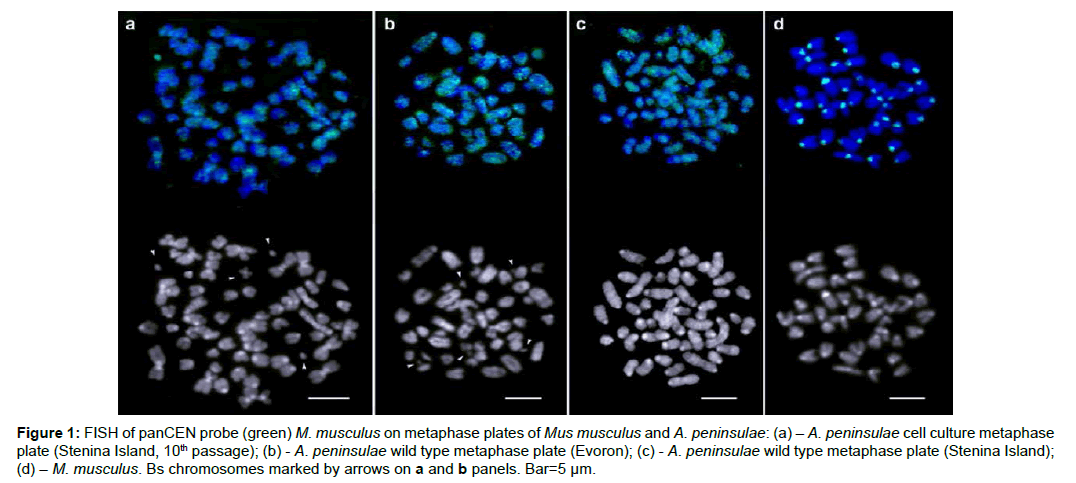
Figure 1: FISH of panCEN probe (green) M. musculus on metaphase plates of Mus musculus and A. peninsulae: (a) – A. peninsulae cell culture metaphase plate (Stenina Island, 10th passage); (b) - A. peninsulae wild type metaphase plate (Evoron); (c) - A. peninsulae wild type metaphase plate (Stenina Island); (d) – M. musculus. Bs chromosomes marked by arrows on a and b panels. Bar=5 μm.
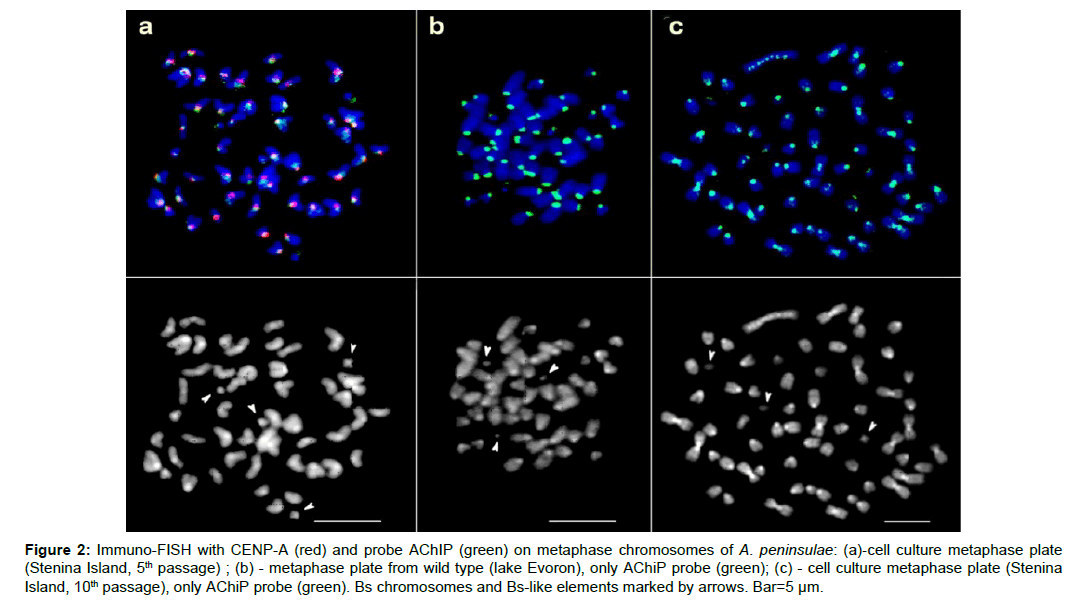
Figure 2: Immuno-FISH with CENP-A (red) and probe AChIP (green) on metaphase chromosomes of A. peninsulae: (a)-cell culture metaphase plate (Stenina Island, 5th passage) ; (b) - metaphase plate from wild type (lake Evoron), only AChiP probe (green); (c) - cell culture metaphase plate (Stenina Island, 10th passage), only AChiP probe (green). Bs chromosomes and Bs-like elements marked by arrows. Bar=5 μm.
The microdissected probe (panCEN) contains centromeric and pericentromeric regions and is assumed to include all the sequences from both regions. On the M. musculus, the metaphase plate probe labels all the primary constriction regions though the intensity of signal differs on different chromosomes (Figure 1d). The same probe, applied to the A. peninsulae metaphase plate, went to the sites along chromosome arms with no label at the primary constriction regions.
A weak hybridisation signal is visible only at some periCEN (close interval between paragraphs) regions from the wild type karyotype (Figures 1b and c) and a label of the same kind is visible on chromosomes from the cell culture (Figure 1a). The panCEN probe of genus Mus stains the region expected though it is not applicable for the genus Apodemus. The genus specific content of the CEN-periCEN region has been confirmed in the current work and the necessity for a specific probe for A. peninsulae is evident.
A. peninsulae heterochromatic probe (AChIP)
The mammalian centromere formation is dependent on the CEN protein CENP-A (Centromere protein A) which is the CEN-specific histone H3 variant [31,32]. CENP-A chromatin immunoprecipitation (ChIP) has been successfully used for heterochromatic sequences isolation [33-36]. Thus, CENP-A ChIP on cell culture A. peninsulae from Stenina Island was undertaken. CENP-A precipitated DNA (AChIP) was purified and labelled by the DOP-PCR procedure. The probe AChIP was obtained. Immuno-FISH with CENP-A and probe AChIP show that DNA, precipitated on the protein during biochemical isolation, labelled the area wider than CENP-A itself (Figure 2a). However the position of the AChIP probe, label corresponds to the CEN-periCEN region, so it is sufficiently specific for A. peninsulae. The probe AChIP, while hybridised to the very culture from which it originated, stained all the heterochromatic regions (Figure 2a). There was some difference in staining between cell lines of different passages (Figures 2a and 2c). On the advanced cell culture (10th passage), the label was visible even within chromosomes rearranged and within Bs-like elements formed (Figure 2). The heterochromatic sub-telomeric regions of some chromosomes were also stained. Bs-like elements did not contain CENP-A, indicating the absence of functional CEN (Figure 2a). Bs-like elements labelled with the AChiP probe, were weak and diffuse at the beginning of their formation (Figure 2a), became more intensive during cell passaging (Figure 2c), but never reached the intensity of the AChiP label on wild type Bs (Figure 2b)
AChiP probe is genus specific
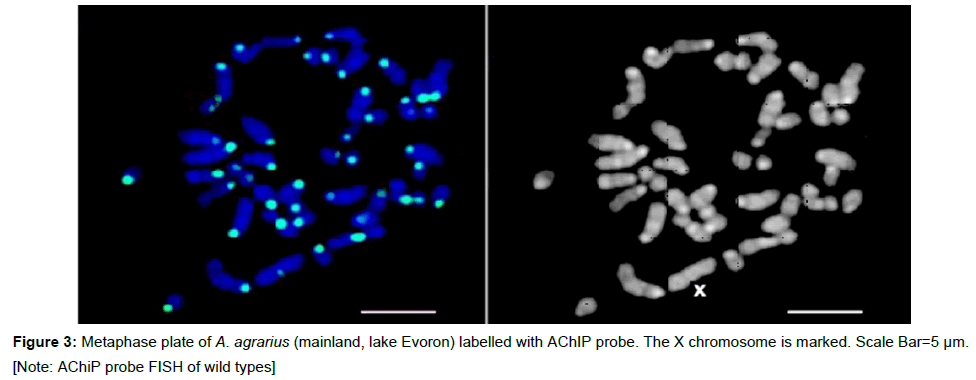
The genus specificity of the A. peninsulae heterochromatic probe was checked on metaphase chromosomes of A. agrarius which is the related species usually without Bs. A perfect CEN-periCEN label was visible on A. agrarius metaphase chromosomes (Figure 3). It indicated the genus specificity of the AChiP probe (Figure 3).
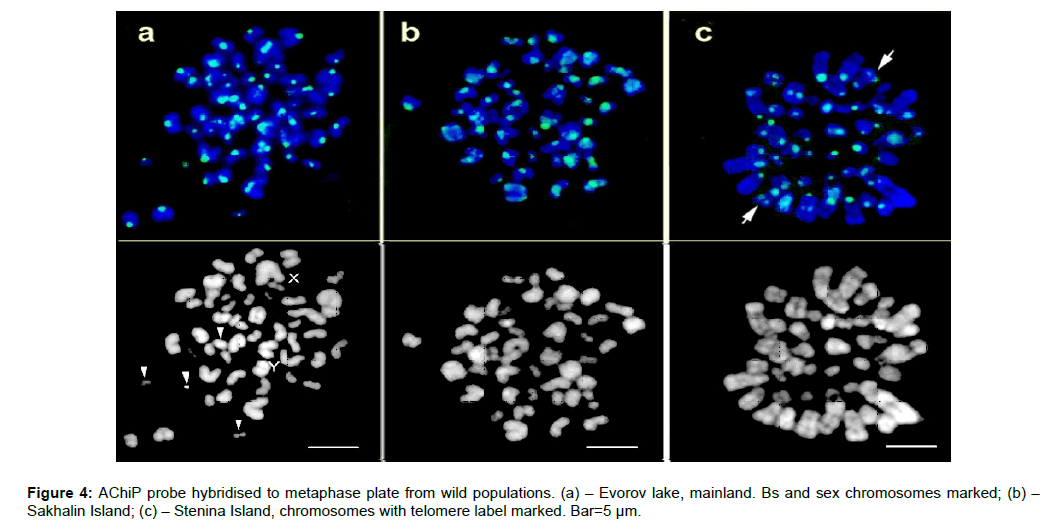
The probe AChIP was hybridised to the chromosomes of wild type populations of A. peninsulae (2n=48) with and without Bs (Figure 4). All the chromosomes’ sets from mainland, Stenina and Sakhalin Islands were labelled at the CEN regions as expected. The intensity of labelling was variable for different chromosomes. Some of the small chromosomes were weakly stained. The Stenina Island metaphase plate contains a label at the telomeric region (Figure 4c, arrows). However, the overall label pattern of Apodemus populations looks similar. Most of the Bs from the mainland was labelled (Figure 4a, arrows). Bs of the wild population show the same tendency as in cultured cells: there are weakly and strongly labelled Bs amongst them (Figures 2 and 4).
Bs–like elements in culture versus nature Bs
The cultured cells with Bs-like elements were initially raised from the island populations, so it appears reasonable to compare Bs appearing with the other island populations. The modal karyotype of A. peninsulae specimens from Hokkaido Island comprised 48 acrocentrics plus 0-13 Bs [18]. The Bs was C-positive and had different morphology: metacentric, acrocentric and dot-like morphology. Some Bs had a heterogeneous structure with black and grey C-blocks [37]. The periCEN regions of autosomes specimens from all geographic regions always had black C-blocks [20], sometimes a third pair of autosomes had grey C-blocks on telomeric regions [25].
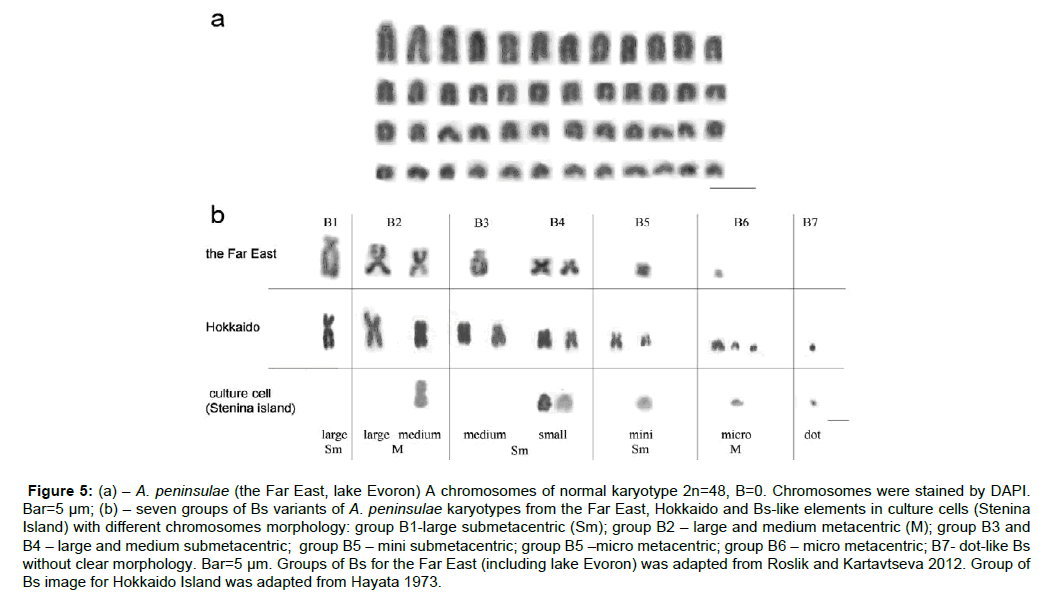
The Stenina and Sakhalin Island populations did not have Bs in the wild type karyotype (Figures 4b and 4c, Figure 5a). Cultivation of cells from Stenina and Sakhalin induced Bs-like elements formation (Figures 2a and 2c). A. peninsulae (2n=48, 0-30 Bs for the Siberia and 0-7 Bs for the Far East) from the Far East possessed varying sizes of B-chromosomes, from large bi-armed (larger than the largest autosome) [25], to small dot-like (Volobujev 1981) (Figure 5b). The Bs from the Far East populations of A. peninsulae differ in appearance from those obtained in cultured cells, which are similar to Bs of the Hokkaido Island wild population: the chromosomal group B7-dotlike Bs is the most similar beside the rest (Figure 5b).
Figure 5: (a) – A. peninsulae (the Far East, lake Evoron) A chromosomes of normal karyotype 2n=48, B=0. Chromosomes were stained by DAPI. Bar=5 μm; (b) – seven groups of Bs variants of A. peninsulae karyotypes from the Far East, Hokkaido and Bs-like elements in culture cells (Stenina Island) with different chromosomes morphology: group B1-large submetacentric (Sm); group B2 – large and medium metacentric (M); group B3 and B4 – large and medium submetacentric; group B5 – mini submetacentric; group B5 –micro metacentric; group B6 – micro metacentric; B7- dot-like Bs without clear morphology. Bar=5 μm. Groups of Bs for the Far East (including lake Evoron) was adapted from Roslik and Kartavtseva 2012. Group of Bs image for Hokkaido Island was adapted from Hayata 1973.
Discussion
Constitutive heterochromatin based on TR, a main constituent of various genomes, is the most enigmatic chromatin component. Satellite DNA (satDNA, TR) were localised in key chromosome regions. TR is basic components of structurally important chromosome areas, such as sub telomeres, centromeres and heterochromatin regions. The functional significance of TR in the genome remains obscure due to the limited tools for their study. The large scale assay aimed at determining the main TR for each species with a genome read was undertaken [38]. This study is a comprehensive assay of the major TR content in 282 species carried out by bioinformatics methods. It was shown that main TR is similar only in phylogenetically close species. In evolutionarily remote species, the main TR has practically nothing in common. SatDNA (TR) is a very variable genome component, possibly species specific.
The genus specificity of the heterochromatic probes was suspected when the M. musculus panCEN probe was used on 8 species of Murine subfamily [39]. The bioinformatics search for TR was also undertaken in poorly assembled genomes of two hamsters and it was found that TR sets differ in two close species Mesocricetus auratus and Cricetulus griseus [40]. It appears that TR sets are rather genus specific. The time of deviation for the Mus and Apodemus ancestors was about 9.7 million years [41]. The results of the current work are in accordance with the following assumptions
1. The Mus pan-CEN probe does not recognise Apodemus periCEN regions (Figure 1)
2. AChip probe does recognise periCEN regions of both species from genus Apodemus (Figures 3 and 4).
AChip probe and DOP primer
The main TR in genomes of various species differ in practically all parameters but the proteins, which bind CEN and periCEN TRs, are conservative. The CENP-A ChIP probe from A. peninsulae (Stenina Island) was produced in the hope of having the probe enriched with heterochromatic sequences specific for this species.
For both probes used (M. musculus pan-CEN and A. peninsulae AChiP), the DOP-PCR primer, as it was initially developed, was used. This method for amplifying DNA has been widely used [9,21-24,42,43].
This method can be applied to many species including those with the genomes not yet read, where interspersed repeats are not easily available for general amplification [44] but DOP-PCR has its limitations. This limitation was noticed in the reverse chromosome labelling technique: samples undergoing DOP-PCR produced a relatively even signal for euchromatin, but it often failed to label highly repetitive sequences in the centromere and other heterochromatic regions. It is rather difficult to predict whether these repetitive regions will hybridise to DOP-PCR processed probe [42]. It seems likely that the DOP primer used prefers to amplify transposable elements (TE) rather than TRs, suggesting that AChiP was depleted of TRs and enriched in TE. The cloning and library analysis of M. musculus chromocenters’ fragments proved that feature of DOP-PCR. The results lead to the conclusion that the content of the AChiP probe is definitely heterochromatic, but the representation of TR and TE fragments could be shifted to the TE enrichment in comparison with constitutive chromatin [9]. The AChiP probe staining, which gave a weak signal on some chromosomes (Figures 2-4) could be due to the TE enrichment proved [9].
Bs in A. peninsulae wild populations
Bs is considered to be highly variable genomic elements, which include C-positive pericentric chromosomal regions predominantly consisting of DNA repeats. Extensive cytological studies of Bs in the Korean field mouse (A. peninsulae), performed on the basis of FISH with micro-dissected Bs from the Siberian population [21], resulted in assumption that Bs diversity is low in Far East populations and high in Siberia and Japanese populations [22,24].
The molecular cytogenetic analysis of Bs chromosomes of A. peninsulae based on the DNA content of their periCEN regions shows, that there are two main types [24]. Bs of the first type (type I) consisted of Bs with sequences homologous to the sequences of autosomal periCEN regions due to FISH staining. The second type (type II) comprised of Bs characterised by the absence of DNA homologous to DNA of autosomal periCEN regions. The type I was typical for animals from Siberia, Transbaikalia and Hokkaido Island, while type II was typical for animals from the Russian Far East.
In the process of cloning and characterisation of the repetitive DNA sequences that comprise the constitutive heterochromatin of the A and B chromosomes of the Korean field mouse (A. peninsulae) apart from TR centromeric sequences, 1.8 kb band from the EcoRI digest has been cloned and produced sequences of 1720 bp and 48.3% GC for EcoRI-1 (AB427143; [45]). Different lines of evidence confirmed that the EcoRI-1 fragment is an interspersed type of repetitive sequences: EcoRI-1 showed homology with a long terminal repeat of rat’s endogenous beta retrovirus ERVB4_5, internal sequence (AC106444). Centromeric TR stains CEN of some Bs in different patterns, suggesting that the Bs of A. peninsulae were derived from a chromosome and that the different families of repetitive sequences amplified independently on each Bs [45]. EcoRI-1 produced weak signals both in the CEN regions and interspersed weak signals on all Bs [45,46] in the same way as in the current work.
All kinds of probes used to reveal the Bs content identified that the degree of TR and TE representation was different for Bs from different populations and that the TE enriched probe was specific for some Bs (Figure 2).
Bs-like elements formation in cell culture
Bs-like elements obtained in cell cultures from the Sakhalin Island have been reported [47]. The modal number of chromosomes in cultured cells from heart, lung and skin tissues was, as a rule, constant, but specimens from insular population showed growing fibroblasts with polyploid cells and with Bs-like elements even in the primary cell cultures. The observed bi-armed (Mm-sm) and micro Bs-like elements presumably appeared de novo and became visible after polyploidisation in the fibroblast cell culture. Bi-armed Bs contained GC-rich heterochromatin in the CEN regions and AT-rich heterochromatin in the arm regions, but micro Bs-like elements were completely GC-rich [47].
Most of the micro and acrocentric Bs-like elements, obtained in the culture in the current work, display the signal (Figure 2) which resembles that from the ERV derived sequence (EcoRI-1; [45]). It suggests that Bs-like elements in the cell culture (Figure 2) possess TE, probably of ERV nature. It is assumed that newly formed Bs-like elements contain mainly TE elements and later are populated with TR. Several types of Bs’ specific DNA [6,22,24,45] probably reflect TE or TR enriched heterochromatic Bs’ components. The ability of cell cultures from island populations to produce Bs-like elements is confirmed in the current work for two Islands, Stenina and Sakhalin. Surprisingly, the morphology of Bs-like elements in cultured cells differs from Bs usually persisting in the Far East populations of A. peninsulae but resemble Bs from the Hokkaido Island wild population (Figure 5b, [24]). The dot Bs (group 7, Figure 5) has been found in the current work and natural population of the Hakkaido Island. The main feature of group seven Bs-like elements from the culture cell and Hokkaido population was hybridisation with autosome centromere DNA (Figure 5, [24]).
A. peninsulae species geography (distribution in nature)
Geological data and data of molecular-genetic analysis draw the following picture of the A. peninsulae species distribution over an extended period. In Pleistocene, the uniform (united) areal of the species existed through Siberia, Transbaikalia, Russian Far East, China, Korea and Japan (Hokkaido) [48,49]. Glaciation and separation of the islands divided the populations. Sakhalin was separated from the continent ~7,000 years ago and Stenina Island ~11,500 years ago as well as Hokkaido (~12,000 years ago). Remarkably, karyotyping studies reveal features more common for the Russian Far East and Hokkaido versus Siberia and Korea [48-50]. It was assumed that, at the end of late Pleistocene to Holocene, the land link periodically appeared between the mainland, Sakhalin and Hokkaido due to the ocean lowering [51,52]. In this very period (8,000-15,000 years ago), the formation of Sakhalin’s theriofauna occurred [53] including rodents [53]. A. peninsulae species could be considered as an emigrant from the mainland to Sakhalin and then to Hokkaido. Hokkaido happens to be the last point of A. peninsulae emigration. There was no paleontological evidence of this species on the other Japanese islands [52].
Bs, which appear in cultured cells from island populations (Stenina, Sakhalin) and from the island wild population (Hokkaido) are more similar in morphology compared to the Bs from the mainland (lake Evoron and other populations from Russian Far East and Siberia (Figures 4a and 5) [25]. It is difficult to explain such a similarity by random coincidence. Thus it is assumed that Bs’ morphology is an argument for the scheme of species distribution described above. Early comparative mitochondrial DNA analysis of A. peninsulae has demonstrated the common origin for Sakhalin and Hokkaido Islands [48,49]. However A. penisulae from Stenina Island was not characterised earlier by FISH.
Chromatin precipitation on CENP-A, DOP amplified, produced the probe AChiP which labelled heterochromatic regions in a genus specific manner. It appears that TE sequences (probably of ERV family) are the first to produce Bs rather than TR. The island populations of A. peninsulae maintain the ability to produce Bs-like elements under stress conditions despite Bs loss in the island wild populations. Bs-like elements, which appear in cultured cells from island populations (Stenina, Sakhalin) and island wild populations (Hokkaido), are more similar in morphology compared to the Bs from mainland (Evoron lake) which could reflect the common origin. The data obtained suggest that the mice of the three islands 7-10 thousand years ago belonged to a single population, have a similar gene pool, and differ from the gene pool of continental populations of mice from the Far East.
Acknowledgements
This work was supported by the Russian Foundation for Basic Research (grants nos. 05-04-49156-ð, 06-04-48969-a, 11-04-01700, 15-04-03871-a), the Russian Science Foundation (grant no.15-15-20026) and the granting programme for “Molecular and Cell Biology” of the Presidium of Russian Academy of Sciences (no. 01.2.01457147) and program FEB RAS (no 18-4-031).
Editing and publishing costs have been paid for by a grant from the Russian Science Foundation (grant no.15-15-20026). Thanks to Dr. Y. Obara (Japan) and N.B. Rubtsov (Russia) for critical comments.
References
- Vujošević M, Blagojević J (2004) B chromosomes in populations of mammals. Cytogenet Genome Res 106: 247-256.
- Makunin AI, Dementyeva PV, Graphodatsky AS, Volobouev VT, Kukekova AV, et al. (2014) Genes on B chromosomes of vertebrates. Mol Cytogenet 7: 99.
- Camacho JPM, Sharbel TF, Beukeboom LW (2000) B-chromosome evolution. Philosophical transactions of the Royal Society of London Series B. Bio Sci 355: 163-178.
- Yang F, O’Brien PCM, Wienberg J, Neitzel H, Lin CC, et al. (1997) Chromosomal evolution of the Chinese muntjac (Muntiacus reevesi). Chromosoma 106: 37-43.
- Yang F, O’Brien PCM, Milne BS, Graphodatsky AS, Solanky N, et al. (1999) A complete comparative chromosome map for the dog, red fox, and human and its integration with canine genetic maps. Genomics 62: 189-202.
- Trifonov VA, Perelman PL, Kawada SI, Iwasa MA, Oda SI, et al. (2002) Complex structure of B-chromosomes in two mammalian species: Apodemus peninsulae (Rodentia) and Nyctereutes procyonoides (Carnivora). Chromosome Res 10: 109-116.
- Makunin AI, Kichigin IG, Larkin DM, O’Brien PC, Ferguson-Smith MA, et al. (2016) Contrasting origin of B chromosomes in two cervids (Siberian roe deer and grey brocket deer) unravelled by chromosome-specific DNA sequencing. BMC genomics 17: 618.
- Saksouk N, Simboeck E, Déjardin J (2015) Constitutive heterochromatin formation and transcription in mammals. Epigenetics Chromatin 8: 3.
- Kuznetsova IS, Ostromyshenskii DI, Komissarov AS, Prusov AN, Waisertreiger IS, et al. (2016) LINE-related component of mouse heterochromatin and complex chromocenters’ composition. Chromosome Res 24: 309-323.
- Potapov VA, Solov’ev VV, Romashchenko AG, Sosnovtsev SV, Ivanov SV (1990) Features of the structure and evolution of complex, tandemly organized Bsp-repeats in the fox genome. I. Structure and internal organization of the BamHI-dimer. Molecular biology (Moscow) 24: 1649-1665.
- Peppers JA, Wiggins LE, Baker RJ (1997) Nature of B chromosomes in the harvest mouse Reithrodontomys megalotis by fluorescence in situ hybridization (FISH). Chromosome Res 5: 475-479.
- Wurster-Hill DH, Ward OG, Davis BH, Park JP, Moyzis RK, et al. (1988) Fragile sites, telomeric DNA sequences, B chromosomes, and DNA content in raccoon dogs, Nyctereutes procyonoides, with comparative notes on foxes, coyote, wolf, and raccoon. Cytogenet Cell Genet 49: 278-281.
- Szczerbal I, Switonski M (2003) B chromosomes of the Chinese raccoon dog (Nyctereutes procyonoides procyonoides Gray) contain inactive NOR-like sequences. Caryologia 56: 213-216.
- Stitou S, Díaz de La GR, Jiménez R, Burgos M (2000) Inactive ribosomal cistrons are spread throughout the B chromosomes of Rattus rattus (Rodentia, Muridae): Implications for their origin and evolution. Chromosome Res 8: 305-311.
- Rubtsov NB, Karamysheva TV, Andreenkova OV, Bochkaerev MN, Kartavtseva IV (2004) Comparative analysis of micro and macro B chromosomes in the Korean field mouse Apodemus peninsulae (Rodentia, Murinae) performed by chromosome microdissection and FISH. Cytogenet Genome Res 106: 289-294.
- Teruel M, Cabrero J, Perfectti F, Camacho JPM (2010) B chromosome ancestry revealed by histone genes in the migratory locust. Chromosoma 119: 217-225.
- Volobujev VT (1981) B chromosomes system of the mammals. Caryologia 34: 1-23.
- Hayata I (1973) Chromosomal polymorphism caused by supernumerary chromosomes in the field mouse, Apodemus giliacus. Chromosoma 42: 403-414.
- Radzhabli, S. I. and Borisov, Y. M. 1979. Variants of the system of supernumerary chromosomes in continental forms of Apodemus peninsulae (Rodentia, Muridae). Doklady Akademii nauk SSSR 248: 979-981.
- Kartavtseva IV (2002) Karyosystematics of Wood and Field Mice (Rodentia, Muridae), Vladivostok: Dal’nauka: 142.
- Karamysheva TV, Andreenkova OV, Bochkarev MN, Borissov YM, Bogdanchikova N, et al. (2002) B chromosomes of Korean field mouse Apodemus peninsulae (Rodentia, Murinae) analyzed by microdissection and FISH. Cytoge Genome Res 96: 154-160.
- Rubtsov NB, Karamysheva TV, Kartavtseva OV, Andreenkova OV, Bochkarev MN, et al. (2005) B chromosomes: DNA, origin and evolution. Biol Membrany 22: 196-211.
- Rubtsov NB, Kartavtseva IV, Roslik GV, Karamysheva TV, Pavlenko MV, et al. (2015) Features of the B chromosome in Korean wood mice Apodemus peninsulae (Thomas, 1906) from Transbaikalia and the Far East identified by the FISH method. Genetika 51: 341-350.
- Kartavtseva IV, Roslik GV, Pavlenko MV, Amachaeva EYu, Sawaguchi S, et al. (2000) The B chromosome system of the Korean field mouse Apodemus peninsulae in the Russian Far East. Chromosome Sci 4: 21-29.
- Kartavtseva IV and Roslik GV (2004) A complex B chromosome system in the Korean field mouse, Apodemus peninsulae. Cyto Genome Res 106: 271-278.
- Roslik GV, Kartavtseva IV (2012) B chromosome morphotypes of Apodemus peninsulae (Rodentia) from the Russian Far East. Tsitilogiya 54: 66-77.
- Berry RJ (1996) Small mammal differentiation on islands. Philosophical transactions of the Royal Society of London Series B Biol Sci 351: 753-764.
- Losos JB, Ricklefs RE (2009) Adaptation and diversification on islands. Nature 457: 830-836.
- Velizhanin AG (1976) Time of Isolation of the Mainland Islands in the Northern Part of the Pacific Ocean. Doklady Akademii nauk SSSR 231: 205-207.
- Arneson N, Hughes S, Houlston R, Done S (2007) Whole-genome amplification by degenerate oligonucleotide primed PCR (DOP-PCR). Cold Spring Harbor Protocols 4919.
- Schalch T, Steiner FA (2016) Structure of centromere chromatin: from nucleosome to chromosomal architecture. Chromosoma 126: 443-455.
- Purgato S, Belloni E, Piras FM, Zoli M, Badiale C, et al. (2015) Centromere sliding on a mammalian chromosome. Chromosoma 124: 277-287.
- Henikoff JG, Thakur J, Kasinathan S, Henikoff S (2015) A unique chromatin complex occupies young α-satellite arrays of human centromeres. Sci Adv 1: e1400234.
- Cech JN, Peichel CL (2016) Centromere inactivation on a neo-Y fusion chromosome in threespine stickleback fish. Chromosome Res 24: 437-450.
- Cerutti F, Gamba R, Mazzagatti A, Piras FM, Cappelletti E, et al. (2016) The major horse satellite DNA family is associated with centromere competence. Mol Cytogenet 9: 35.
- Abe S, Han S, Kojima H, Ishibashi Y, Yoshida MC (1997) Differential staining profiles of B-chromosomes in the East-Asiatic wood mouse Apodemus peninsulae. Chromosome Sci 1: 7-12.
- Melters, D. P., Bradnam, K. R., Young, H. A., Telis, N., May M. R., Ruby, J. G, Sebra, R., Peluso P., et al. 2013. Comparative analysis of tandem repeats from hundreds of species reveals unique insights into centromere evolution. Genome Biol 14: R10
- Ostromyshenskii DI, Kuznetsova IS, Golenischev FN, Malikov VG, Podgornaya OI (2011) Satellite DNA as a phylogenetic marker: Case study of three genera of the murine subfamily. Cell Tissue Biol 5: 543-550.
- Miheev DY, Podgornaya OI, Ostromyshenski DI (2015) Large tandem repeats of Mesocricetus auratus in silico and in situ. Cell and Tissue Biology 9: 226-232.
- Michaux JR, Chevret P, Filippucci MG, Macholan M (2002) Phylogeny of the genus Apodemus with a special emphasis on the subgenus Sylvaemus using the nuclear IRBP gene and two mitochondrial markers: cytochrome b and 12S rRNA. Mol Phylogenet Evol 23: 123-136.
- Carter NP, Bebb CE, Nordenskjo M, Ponder BA, Tunnacliffe A (1992) Degenerate oligonucleotide-primed PCR: general amplification of target DNA by a single degenerate primer. Genomics 13: 718-725.
- Wells D, Delhanty JD (2000) Comprehensive chromosomal analysis of human preimplantation embryos using whole genome amplification and single cell comparative genomic hybridization. Mol Hum Reprod 6: 1055-1062.
- Telenius H, Pelmear AH, Tunnacliffe A, Carter NP, Behmel A (1992) Cytogenetic analysis by chromosome painting using dop pcr amplified flow sorted chromosomes. Genes Chromosomes Cancer 4: 257-263.
- Matsubara K, Yamada K, Umemoto S, Tsuchiya K, Ikeda N, et al. (2008) Molecular cloning and characterization of the repetitive DNA sequences that comprise the constitutive heterochromatin of the A and B chromosomes of the Korean field mouse (Apodemus peninsulae, Muridae, Rodentia). Chromosome Res 16: 1013-1026.
- Matsubara K, Nishida UC, Tsuchiya K, Nukaya D, Matsuda Y (2004) Karyotypic evolution of Apodemus (Muridae, Rodentia) inferred from comparative FISH analyses. Chromosome Res 12: 383-395.
- Sawaguchi S, Obara Y, Kartavtseva IV, Roslik G, Shin HE et al. (1998) Maintenance mode of the B chromosomes in Apodemus peninsulae from four areas bordering on the Sea of Japan. Chromosome Sci 2: 161.
- Serizawa K, Suzuki H, Iwasa M, Tsuchiya K, Pavlenko MV, et al. (2002) A spatial aspect on Mitochondrial DNA genealogy in Apodemus peninsulae from East Asia. Biochem Genet 40: 149-161.
- Héla S, Jean PQ, Irina K, Marina P, Galina C, et al. (2010) Comparative phylogeography of four Apodemus species (Mammalia: Rodentia) in the Asian Far East: evidence of Quaternary climatic changes in their genetic structure. Biol J Linnean Soc 100: 797-821.
- Hayasaka D, Ivanov LI, Leonova GN, Goto A, Yoshii K, et al. (2001) Distribution and characterization of tick borne encephalitis viruses from Siberia and Far Eastern Asia. J GenVirol 82: 1319-1328.
- Mikishin YA, Gvozdeva IG, Petrenko TI (2010) Early Holocene of Sakhalin. Aktual’nye problemy gumanitarnykh i estestvennykh nauk. Current Problems in Humanities and Natural Sciences 12: 432-437.
- Kawamura Y (1989) Quaternary Rodent Faunas in the Japanese Islands (Part 2). Memoirs Faculty Science Kyoto University Ser Geol Mineral 54: 235.
- Kirillova IV (2003) Remains of vertebrates from the Tronnyi Grotto (Central Sakhalin). Kraevedcheskii byulleten': problemy istorii Sakhalina, Kuril i sopredel’nykh territorii Local History Bulletin: History of Sakhalin, the Kuril Islands and Adjacent Territories 2: 128-137.
- Burkovskii OA (2004) The history of the rodent fauna formation (Mammalia, Rodentia) in Sakhalin. Rastitel’nyi i zhivotnyi mir Sakhalina 1: 238-248.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi