Research Article, Int J Cardiovasc Res Vol: 6 Issue: 6
Association between Albuminuria and Severity of Coronary Artery Disease detected by Angiography in Patients with Type 2 Diabetes Mellitus
Ahmed Hosny El-Adawy1*, Hoda Mahmoud Abdelaziz2, Eslam Eid2 and Tarek Gouda2
1Department of Cardiovascular Medicine, Faculty of Medicine, Mansoura University, Mansoura 35516, Egypt
2Department of Internal Medicine, Faculty of Medicine, Mansoura University, Mansoura 35516, Egypt
*Corresponding Author : Ahmed Hosny El-Adawy
Department of Cardiovascular Medicine, Faculty of Medicine, Mansoura University, Mansoura 35516, Egypt
Tel: 002-050-2202772
E-mail: dr_ahmed_hosny@msn.com.
Received: September 25, 2017 Accepted: September 25, 2017 Published: November 07, 2017
Citation: El-Adawy AH, Abdelaziz HM, Eid E, Gouda T (2017) Association between Albuminuria and Severity of Coronary Artery Disease detected by Angiography in Patients with Type 2 Diabetes Mellitus. Int J Cardiovasc Res 6:6. doi: 10.4172/2324-8602.1000335
Abstract
Background: Diabetes mellitus (DM) is the most prevalent metabolic disease worldwide and is the most potent risk factor for coronary artery disease (CAD). Consequently, finding a clue to diagnose the presence of CAD as early as possible in diabetic patients is very important. Micro albuminuria is a strong and independent indicator of increased cardiovascular risk among individuals with and without diabetes.
The aim of this study was to find any correlation between the degree of albuminuria and the severity of CAD in patients with type 2 DM.
Methods: This was a cross sectional study that included 86 patients with type 2 DM whom underwent elective coronary angiography for suspected coronary artery disease in Mansoura Specialized Medical Hospital from March 2014 till March 2015. Albumin creatinine ratio was determined for all patients and severity of CAD was assessed using the SYNTAX score.
Results: The patients were classified into 3 groups based on levels of urinary albumin creatinine ratio (ACR); group 1 consisted of patients with ACR<30 mg/gm (26 patients), group 2 consisted of patients with ACR 30-300 mg/gm (38 patients) and group 3 consisted of patients with ACR> 300 mg/gm (22 patients). Age ranged between 41 and 70 years with mean age 58.37 ± 7.20 years, sex distribution was 48 males (55.8%) and 38 females (44.2%). The SYNTAX score and ACR were significantly positively correlated (p<0.001). Regression analysis revealed ACR as an independent predictor for the severity of CAD (P<0.001).
Conclusion: There is a strong relationship between the degree of albuminuria and extent and complexity of CAD in patients with type 2 DM and this association in independent of traditional risk factors.
Keywords: Coronary artery diseases; Albuminuria; Diabetes; Risk factors
Introduction
Type 2 diabetes is a mounting health challenge associated with high rates of cardiovascular morbidity and mortality [1]. In spite of the fact that the seriousness of CVD danger changes between diabetics of distinctive ages and span of illness, numerous patients have sufficient hazard that T2DM has been believed to be a risk equivalent of coronary artery disease (CAD) [2].
Impaired endothelial function is a recognized participator in the pathophysiology of CVD, including hypertension, CAD and DM [3]. Endothelial dysfunction precedes morphological atherosclerotic alterations and has a major action in lesion progress and later clinical complications [4]. Conditions which are accompanied with impaired endothelial function (for example, DM, hypertension, vascular disease, inflammation, and insulin resistance) also may present as impaired glomerular endothelial function (changed permeability), such that micro albuminuria represents a state of vascular endothelial dysfunction, with the kidney being a window to the vasculature in other tissue parts [5]. Chronic renal dysfunction and albuminuria may be important factors determining the occurrence and the severity of CAD [6].
Microalbuminuria is a widely recognized, strong and independent risk marker of cardiovascular disease among individuals with diabetes [7,8]. The presence of microalbuminuria is not associated with endothelial dysfunction and severity of angiographic coronary atherosclerosis in non-diabetic and non-hypertensive patients with acute coronary syndromes [9].
Microalbuminuria is prescient unrelated to conventional risk elements of cardiovascular diseases and entire reasons of death in diabetes or hypertension patient groups and in the general population [10]. It is gaining importance as a marker of atherogenic milieu and indicates the target organ damage and can be a valuable tool in screening and identification of patients with cardiovascular disease [11]. It denotes underlying widespread vascular dysfunction [12]. In asymptomatic patients with type 2 diabetes, microalbuminuria was associated with increased risk of CAD and poorer clinical outcomes [13]. The patients with DM type2, microalbuminuria is an independent predictor of severity of coronary artery stenosis and reveals a positive correlation between microalbuminuria and the Gensini score [14]. Micro albuminuria is associated with the severity of CAD independent of other cardiovascular risk factors [15]. Duration of diabetes and micro albuminuria or macroalbuminuria are important predictors of severity of peripheral arterial disease and left ventricular dysfunction in a cohort of patients selected for the presence of coronary artery disease [16].
This study was performed to assess the correlation between the degree of albuminuria and the angiographic severity of CAD in patients with type 2 DM.
Methods
Patients and methodology
This study was designed as a sectional observational study that included 86 patients with type 2 diabetes mellitus (DM). The patients were underwent elective coronary angiography for suspected coronary artery disease in Mansoura Specialized Medical Hospital from March 2014 to March 2015. The patients with: type 1 diabetes mellitus, patients with eGFR > 60 ml/min/1.73 m2, decompensated liver disease, rheumatic valvular heart diseases, decompensated heart failure, previous myocardial infarction or recent cerebrovascular events (such as brain infarction or hemorrhage) within prior 6 months were excluded from this study.
Patients enrolled in this study were subjected to full clinical history and examination. Hypertension was defined as systolic blood pressure ≥140 mmHg, diastolic blood pressure ≥90 mmHg, and/or the use of antihypertensive drugs. Patients who had smoked within the previous 6 months of entry into the study were deemed current smokers. Patients who used cholesterol-lowering medications or had a total serum cholesterol level ≥200 mg/dl were classified as having hypercholesterolemia.
All participants were investigated using a 12-lead standard ECG, echocardiography and laboratory tests (CBC, serum creatinine, lipid profile and HgbA1c). The Body Mass Index (BMI) was calculated as the body weight (kg)/ the height (m). The Estimated Glomerular filtration rate (eGFR) was assessed using the Mayo Clinic Quadratic equation (MCQ equation) which doesn’t underestimate normal GFR as does the Modification of Diet in Renal Disease (MDRD) equation in diabetic subjects [17].
A morning random urine sample was collected from all patients before coronary angiography to measure Albumin/Creatinine ratio (ACR), which was tested using turbidimetric microalbuminuria kit (BioSystemsS.A.Costa Brava, 30. 08030 Barcelona, Spain).
Coronary arteriography was performed to all patients by the Judkins technique for assessment of the lesions distribution and description and assessment of the severity of atherosclerotic coronary artery disease (CAD) by using SYNTAX score. CAD was considered significant, if there was ≥ 50% diameter stenosis in ≥ one coronary artery.
Statistical analysis
Data was analyzed using SPSS version 15. Qualitative data were presented as number and percent. Comparison between groups was done using Chi-Square test. Quantitative data was presented as mean ± SD. One way ANOVA was used to compare between three groups. Correlation between ACR and SYNTAX score was tested using Spearman’s correlation analysis. P value< 0.05 was considered to be statistically significant and p value < 0.001 was considered highly significant.
Ethical statement
Study protocol was approved by Medical Ethics research Committee of the faculty of medicine, Mansoura University, Egypt and from the mangers of the hospital in which the study conducted. Informed written consent obtained from each participant in the study. Confidentiality and personal privacy respected in all levels of the study. Collected data will not be used for any other purpose.
Results
The patients were classified into three groups based on levels of urinary albumin creatinine ratio (ACR) as shown in Table 1.
| Group | ACR | Numbers of patients | prevalence |
|---|---|---|---|
| 1 | ˂ 30 mg/gm | 26 | 30.32% |
| 2 | 30 – 300 mg/gm | 28 | 44.2% |
| 3 | ˃ 300 mg/gm | 22 | 25.5% |
Table 1: Classification of patients participated in this study according to the levels of urinary albumin creatinine ratio (ACR).
The severity of CAD was evaluated by SYNTAX score as low score (0-22), intermediate score (23-32) and high score (≥ 33) as shown in Table 2. Thirty-two patients (37.2%) showing low score (0-22), 28 (32.65%) intermediate score (23-32) and 26 (30.2%) with high score (≥ 33) Table 2.
| SYNTAX score | Numbers of patients | prevalence |
|---|---|---|
| 0 - 22 | 32 | 37.2% |
| 23 - 32 | 28 | 32.6% |
| ≥ 33 | 26 | 30.2% |
Table 2: SYNTAX score of the study population.
Clinical and demographic data of all diabetic patients enrolled in the study based on ACR are demonstrated in Table 3. The following criteria were illustrated in the study population; age ranged between 41 and 70 years with mean age 58.37 ± 7.20 years, sex distribution was 48 males (55.8%) and 38 females (44.2%), smokers were 22 patients (25.6%), mean duration of DM was 10.19 ± 7.78 years, hypertension was present in 68 patients (79.1%), hypercholesterolemia was present in 62 patients (72.1%), family history of CAD was present in 18 patients (20.9%), mean BMI was 32.59 ± 4.86 kg/m2, mean HbA1c was 7.65 ± 1.49 %, mean eGFR was 94.70 ± 17.20 ml/min/1.73m2 and mean ejection fraction was 63.49 ± 7.67 %.
| Criteria | Group 1 ACR <30 mg/gm (n = 26) |
Group 2 ACR30-300 mg/gm (n = 38) |
Group 3 ACR >300 mg/gm (n = 22) |
F | P value | |
|---|---|---|---|---|---|---|
| Age | 58.00 ± 7.52 | 56.79 ± 8.07 | 61.55 ± 4.08 | 1.587 | 0.217 | |
| Sex | Male | 14 (53.8%) | 24 (63.2%) | 10 (45.5%) | 0.915 | 0.633 |
| Female | 12 (46.2%) | 14 (36.8%) | 12 (54.5%) | |||
| DM duration (years) | 8.08 ± 6.55 | 10.58 ± 9.70 | 12.00 ± 4.90 | 0.792 | 0.460 | |
| BMI (kg/m2) | 34.11 ± 5.57 | 31.96 ± 4.58 | 31.88 ± 4.48 | 0.903 | 0.413 | |
| HbA1c (%) | 7.01 ± 1.94 | 7.88 ± 1.32 | 8.00 ± 0.92 | 1.820 | 0.175 | |
| eGFR (ml/min/1.73m2) | 106.00 ± 11.53 | 95.58 ± 14.24 | 79.82 ± 17.52 | 9.891 | <0.001* | |
| Ejection fraction (%) | 65.77 ± 4.78 | 62.58 ± 7.73 | 62.36 ± 10.12 | 0.820 | 0.448 | |
| Smoking | 8 (30.8%) | 12 (31.6%) | 2 (9.1%) | 2.114 | 0.347 | |
| Hypertension | 20 (76.9%) | 28 (73.7%) | 20 (90.9%) | 1.301 | 0.522 | |
| Family history of CAD | 0 (.0%) | 12 (31.6%) | 6 (27.3%) | 5.010 | 0.082 | |
| Hypercholesterolemia# | 10 (38.5%) | 32 (84.2%) | 20 (90.9%) | 10.631 | 0.005* | |
*P value<0.05 was considered significant. #Hypercholesterolemia was diagnosed if the patient was receiving cholesterol lowering drugs or if serum total cholesterol was ≥ 200 mg/dl.
Table 3: Demographic data of the studied patients based on ACR.
Relation between number of vessels affected and ACR is shown in Table 4. The percentage of patients with no vessels affected was highest in group 1 (53.8%), while the percentage of patients with multivessel disease was highest in group 3 (63.6%) compared with (7.7%) and (57.9%) in group 1 and 2, respectively (p value<0.046).
| Number of vessels affected per patient | Group 1 ACR <30 mg/gm (n = 26) |
Group 2 ACR 30-300 mg/gm (n = 38) |
Group 3 ACR >300 mg/gm (n = 22) |
F | P value |
|---|---|---|---|---|---|
| Zero | 14 (53.8%) | 4 (10.5%) | 4 (18.2%) | 12.809 | 0.046* |
| Single vessel | 2 (7.7%) | 2 (5.3%) | 0 (.0%) | ||
| Two vessels | 8 (30.8%) | 10 (26.3%) | 4 (18.2%) | ||
| Multi-vessel | 2 (7.7%) | 22 (57.9%) | 14 (63.6%) |
*P value < 0.05 was considered significant.
Table 4: Relation between number of vessels affected and ACR.
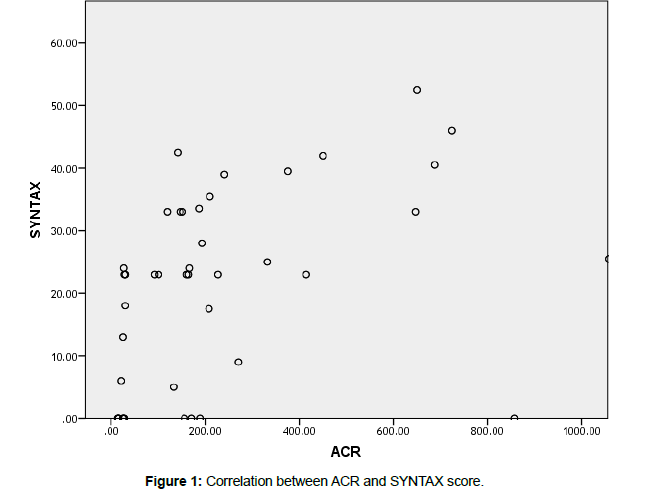
The SYNTAX score and ACR were significantly positively correlated (p value<0.001), indicating strong relationship between the degree of albuminuria and extent and complexity of CAD as shown in Table 5 and Figure 1.
| SYNTAX score | F | P value | |||||
|---|---|---|---|---|---|---|---|
| 0-22 (n = 32) |
23-32 (n = 28) |
³33 (n = 26) |
|||||
| Age (years) | 55.00 ± 7.97 | 60.57 ± 6.37 | 60.15 ± 5.83 | 3.083 | 0.057 | ||
| Sex | Male | 18 (56.3%) | 20 (71.4%) | 10 (38.5%) | 2.973 | 0.226 | |
| Female | 14 (43.8%) | 8 (28.6%) | 16 (61.5%) | ||||
| DM duration (years) | 9.19 ± 7.25 | 8.93 ± 5.85 | 12.77 ± 9.94 | 1.032 | 0.366 | ||
| BMI (kg/m2) | 32.36 ± 5.22 | 32.61 ± 5.18 | 32.85 ± 4.43 | 0.035 | 0.966 | ||
| HbA1c (%) | 7.31 ± 1.87 | 7.72 ± 1.34 | 7.99 ± 1.06 | 0.779 | 0.466 | ||
| eGFR (ml/min/1.73m2) | 103.81 ± 13.42 | 95.29 ± 17.63 | 82.85 ± 14.49 | 6.823 | 0.003* | ||
| Ejection fraction (%) | 64.63 ± 8.21 | 61.36 ± 7.14 | 64.38 ± 7.64 | 0.797 | 0.458 | ||
| Smoking | 12 (37.5%) | 8 (28.6%) | 2 (7.7%) | 3.445 | 0.179 | ||
| Hypertension | 22 (68.8%) | 24 (85.7%) | 22 (84.6%) | 1.645 | 0.439 | ||
| DM treatment | Oral drugs | 16 (50%) | 16 (57.1%) | 12 (46.2%) | 0.340 | 0.844 | |
| Insulin | 16 (50%) | 12 (42.9%) | 14 (53.8%) | ||||
| Family history of CAD | 6 (18.8%) | 4 (14.3%) | 8 (30.8%) | 1.180 | 0.554 | ||
| Hypercholesterolemia# | 18 (56.3%) | 20 (71.4%) | 24 (92.3%) | 4.640 | 0.098 | ||
#Hypercholesterolemia was diagnosed if the patient was receiving cholesterol lowering drugs or if serum total cholesterol was ≥ 200 mg/dl.
Table 5: Demographic data of the studied patients based on SYNTAX score.
Multiple linear regression analysis was performed using the SYNTAX score as an outcome (dependent variable) and the ACR as a predictor after adjusting for age, sex, smoking, hypertension, hypercholesterolemia, BMI and HbA1c. Analysis showed that ACR was independently correlated with the SYNTAX score as shown in Table 6.
| Model | B | 95% CI | P value |
| Model 1* | .021 | .009 to .033 | 0.001 |
| Model 2# | .020 | .008 to .033 | 0.001 |
*Model 1: after adjusting for age, sex, smoking and hypertension.
#Model 2: after adjusting for BMI, HbA1c and hypercholesterolemia.
Table 6: Multiple linear regression analysis using the SYNTAX score as an outcome and the ACR as a predictor.
Discussion
Type 2 diabetes mellitus (T2DM) and cardiovascular disease (CVD) are firmly connected, at least to some extent through simultaneous risk factors which often establish before the beginning of T2DM [18]. Type 2 diabetes is a growing health problem associated with high rates of cardiovascular morbidity and mortality [1,19]. Numerous patients with T2DM have sufficient hazard that it has been believed to be a risk equivalent of coronary artery disease (CAD) [2]. Moreover, it was found that the angiographically detected CAD is more severe in diabetic patients with microalbuminuria than in normoalbuminuric patients [20]. Microalbuminuria (MA) is a predictor, unrelated to conventional risk elements of cardiovascular diseases and entire reasons of death in DM or hypertension patient groups and in the general population [10]. It is gaining importance as a marker of atherogenic milieu and indicates the target organ damage and can be a valuable tool in screening and identification of patients with cardiovascular disease [11].
This study was performed to assess the correlation between the degree of albuminuria and the angiographic severity of CAD in patients with T2DM and the association between high levels of albuminuria and the severity of CAD.
The results of the current study showed that albuminuria is firmly positively correlated to angiographically detected coronary artery disease in patients with type 2 diabetes mellitus and this association was statistically significant (P<0.001).
The results of the present study showed that microalbuminuria was associated with CAD patients. In addition, the patients with microalbuminuria had great atherosclerotic burden in the form of multi-vessel disease. The results are in accordance with the previously reported findings [21-25]. It is proposed that microalbuminuria indicates early and possibly reversible glomerular damage [2].
In general, microalbuminuria is associated with hypertension, diabetes mellitus and renal dysfunction. Microalbuminuria predicts cardiovascular events and renal insufficiency in hypertensive patients [21,23,26].
Previous study was proved that the ACR can predict CAD and correlate with its severity and there was significant correlation between microalbuminuria and CAD (p value<0.001) [27].
According to study of Zand Parsa et al. that conducted in patients with T2DM, MA and Gensini score were correlated positively (P<0.001) and therefore, MA can predicts the severity of CAD independently [14].
Guo et al. investigated the relation of the urinary albumin excretion rate (UAER) with the incidence, severity and pathological properties of CAD in subjects with T2DM, and explored the effectiveness of UAER as a predictor of the risk of CHD in subjects with T2DM. The dissimilarities in the incidence of CHD, the number of coronary arteries pathologically affected, the Gensini’s score and LVEF percentages between MA group and normo-albuminuria group showed statistical significance (P<0.05). Thus MA in subjects with T2DM is an independent risk factor for CAD and there is a significant positive correlation between MA and the severity of coronary atherosclerosis [28].
El Sherif et al. performed a study to investigate the correlation between HbA1c (severity of DM) and microalbuminuria to severity of CAD assessed by coronary angiography. Gensini score showed significant higher statistically values (73.1 ± 40) (P value<0.001) in patients having microalbuminuria than those without microalbuminuria (43.6 ± 30.6) [29].
Rein et al. 2011 performed a study, which was conducted on consecutive patients whom underwent coronary angiography for evaluating established or suspected stable CAD. The prevalence of coronary artery stenosis of ≥ 50% was significantly greater in patients with albuminuria (either micro- or macro-albuminuria) contrasted with patients with normo-albuminuria (p<0.001). This association was significant in both patients with and without T2DM [30].
Conclusion
Albuminuria is firmly positively correlated to angiographically detected coronary artery disease in patients with type 2 diabetes mellitus and this association is independent of conventional risk factors. Therefore, albuminuria could be considered as a risk factor for predicting the severity of coronary artery stenosis in patients with type2 DM. Prospective studies are needed to determine with certainty the degree of risk of CAD associated with microalbuminuria.
Limitations
The major limitations of our study are that it is an observational non randomized study; also, it is a single center study, with a small number of patient subgroups.
Funding
The research received no grant from any funding agency in the public, commercial or not-for-profit sectors.
Conflict of Interest
The authors declare that there is no conflict of interest.
Acknowledgment
We express our sincere gratitude to the patients who participated in the study. We thank the technical staff at Mansoura Specialized Medical Hospital, Mansoura, Egypt for their excellent technical assistance.
References
- Bonow R, Bohannon N, Hazzard W (1996) Risk stratification in coronary artery disease and special populations. Am J Med 101: 22S-24S.
- Haffner S, Lehto S, Ronnemaa T, Pyorala K, Laakso M (1998) Mortality from coronary heart disease in subjects with type 2 diabetes and in nondiabetic subjects with and without prior myocardial infarction. N Engl J Med 339: 229-234.
- Endemann D, Schiffrin E (2004) Endothelial dysfunction. J Am Soc Nephrol 15:1983-1992.
- Deanfield J, Halcox J, Rabelink T (2007) Endothelial function and dysfunction: testing and clinical relevance. Circulation 115:1285-1295.
- Deckert T, Feldt-Rasmussen B, Borch-Johnsen K, Jensen T, Kofoed-Enevoldsen A (1989) Albuminuria reflects widespread vascular damage. The Steno hypothesis. Diabetol 32:219-226.
- Jia D, Li W, Liu J (2014) Correlation of chronic renal dysfunction and albuminuria with severity of coronary artery lesions in patients with coronary artery disease. Cell biochemistry and biophysics 69: 55-59.
- Stehouwer CDA, Smulders YM (2006) Microalbuminuria and risk for cardiovascular disease: Analysis of potential mechanisms. J Am Soc Nephrol 17: 2106-2111.
- Weir MR (2007) Microalbuminuria and Cardiovascular Disease. Clin J Am Soc Nephrol 2: 581-590.
- Duran M, Kalay N, Ardic I, Yarlioglues M, Kayaalti F, et al. (2010) Microalbuminuria is not associated with endothelial dysfunction and coronary atherosclerosis in patients with acute coronary syndromes. Renal failure 32: 659-665.
- Khan K, Khan M, Haque M (2013) Correlation between microalbuminuria with complexity of coronary artery disease in diabetic patients. Mymensingh Med J 22: 353-357.
- Sharma S, Ghalaut V, Dixit R, Kumar S, George P (2013) Microalbuminuria and C-reactive protein as a predictor of coronary artery disease in patients of acute chest pain. J Cardiovasc Dis Res 4: 37-39.
- Stehouwer C, Nauta J, Zeldenrust G, Hackeng W, Donker A, et al. (1992) Urinary albumin excretion, cardiovascular disease, and endothelial dysfunction in non-insulin-dependent diabetes mellitus. Lancet 340: 319-323.
- Kim J, Hwang B, Choi I, Choo E, Lim S, et al. (2015) A prospective two-center study on the associations between microalbuminuria, coronary atherosclerosis and long-term clinical outcome in asymptomatic patients with type 2 diabetes mellitus: evaluation by coronary CT angiography. Int J Cardiovasc Imag 31:193-203.
- Zand Parsa A, Ghadirian L, Rajabzadeh Kanafi S, Moradi Farsani E (2013) Positive correlation between microalbuminuria and severity of coronary artery stenosis in patients with type 2 diabetes mellitus. Acta Med Iran 51: 231-235.
- Ozyol A, Yucel O, Ege M, Zorlu A, Yilmaz M (2012) Microalbuminuria is associated with the severity of coronary artery disease independently of other cardiovascular risk factors. Angiol 63: 457-460.
- Escobedo J, Rana J, Lombardero M, Albert S, Davis A, et al. (2010) Association between albuminuria and duration of diabetes and myocardial dysfunction and peripheral arterial disease among patients with stable coronary artery disease in the BARI 2D study. Mayo Clinic proceedings 85: 41-46.
- Rigalleau V, Lasseur C, Raffaitin C, Perlemoine C, Barthe N, et al. (2007) The Mayo Clinic quadratic equation improves the prediction of glomerular filtration rate in diabetic subjects. Nephrol Dial Transplant 22: 813-818.
- Hu F, Stampfer M, Haffner S, Solomon C, Willett W, et al. (2002) Elevated risk of cardiovascular disease prior to clinical diagnosis of type 2 diabetes. Diabetes Care 25: 1129-1134.
- Bax JJ, Young LH, Frye RL, Bonow RO, Steinberg HO, et al. (2007) Screening for coronary artery disease in patients with diabetes. Diabetes Care 30: 2729-2736.
- Sukhija R, Aronow W, Kakar P, Garza L, Sachdeva R, et al. (2006) Relation of microalbuminuria and coronary artery disease in patients with and without diabetes mellitus. Am J Cardiol 98: 279-281.
- Bigazzi R, Bianchi S, Baldari D, Campese V (1998) Microalbuminuria predicts cardiovascular events and renal insufficiency in patients with essential hypertension. J Hyperten 16: 1325-1333.
- Gerstein H, Mann J, Yi Q, Dinneen S, Hoogwerf B, et al. (2001) Albuminuria and risk of cardiovascular events, death, and heart failure in diabetic and nondiabetic individuals. JAMA 286: 421-426.
- Ibsen H, Wachtell K, Olsen M, Borch-JohnseN K, Lindholm L, et al. (2004) Albuminuria and cardiovascular risk in hypertensive patients with left ventricular hypertrophy: The LIFE Study. Kidney Int 66: S56-S58.
- Klausen K, Borch-Johnsen K, Feldt-Rasmussen B, Jensen G, Clausen P, et al. (2004) Very Low Levels of Microalbuminuria Are Associated With Increased Risk of Coronary Heart Disease and Death Independently of Renal Function, Hypertension, and Diabetes. Circulation 110: 32-35.
- Romundstad S, Holmen J, Kvenild K, Hallan H, Ellekjær H (2003) Microalbuminuria and all-cause mortality in 2,089 apparently healthy individuals: a 4.4-year follow-up study. The Nord-Trøndelag Health Study (HUNT), Norway. Am J Kidney Dis 42: 466-473.
- Sadaka M, Elhadedy A, Abdelhalim S, Elashmawy H (2013) Albumin to creatinine ratio as a predictor to the severity of coronary artery disease. Alex J Med 49: 323-328.
- Wang Y, Yuan A, Yu C (2013) Correlation between microalbuminuria and cardiovascular events. Int J Clin Exp Med 6: 973-978.
- Guo L, Cheng Y, Wang X, Pan Q, Li H, et al. (2012) Association between microalbuminuria and cardiovascular disease in type 2 diabetes mellitus of the Beijing Han nationality. Acta Diabetol 49: 010-0205.
- El Sherif A, Khaled M, Ibrahim A, Elhattab M (2011) Association of glycosylated hemoglobin level and microalbuminuria with the severity of coronary artery disease. J Am Sci 7: 1097-2006.
- Rein P, Vonbank A, Saely C, Beer S, Jankovic V, et al. (2011) Relation of albuminuria to angiographically determined coronary arterial narrowing in patients with and without type 2 diabetes mellitus and stable or suspected coronary artery disease. Am J Cardiol 107: 1144-1148.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi