Research Article, J Biochem Eng Bioprocess Technol Vol: 1 Issue: 2
Caffeine Induces Significant Higher Protein Activity for Cell Activation and Lower Carboxyl Esterase Activity in Male Wistar Rat’s Brain and Liver
Prabha M1*, Aditi Karanth1, Neethu P1, Vaishnavi Ramesh1 and Suneetha P2
1Department of Biotechnology, Ramaiah Institute of Technology, Bengaluru- 560054, India
2Maharani Lakshmi Ammanni College for Women Autonomous, Malleshwaram West, Bengaluru, Karnataka 560003, India
*Corresponding Author : Prabha M
Associate Professor, Department of Biotechnology, Ramaiah Institute of Technology, Bengaluru-560054, India
Tel: 91808277123125, 918023588236
E-mail: prabhamg@gmail.com
Received: February 02, 2018 Accepted: February 05, 2018 Published: March 03, 2018
Citation: Prabha M, Karanth A, Neethu P, Ramesh V, Suneetha P (2018) Caffeine Induces Significant Higher Protein Activity for Cell Activation and Lower Carboxyl Esterase Activity in Male Wistar Rat’s Brain and Liver. J Biochem Eng Bioprocess Technol 1:1.
Abstract
Specific activity of carboxyl esterase was estimated with and without caffeine on young and old aged wistar rat’s brain and liver. Carboxyl esterase (CE) is categorized as phase-I drug metabolizing enzyme that hydrolyzes carboxylic acid esters, amides and thioesters. Total Protein and CE was estimated with 3 and 6 days caffeine treated (daily dose of stock 13mg/kg) young and old male wistar rat’s brain and liver (each n=5) as compared to control. Histogram showed significantly higher protein 123.31 mg and 100.57 mg respectively for 3 and 6 days caffeine treated old rats’ brain than their control 47.72 mg of protein. While young brain caffeine treated showed higher 70.80 mg and 72.89 mg respectively than control 52.07 mg. Electrophoresis showed more protein band patterns for 6 days caffeine treated old rat brain and for 3 day treated young rat brain as compared to control which is complimentary results for spectophotometric analysis. 2D electrophoresis showed more expressed high intensity bands in caffeine treated young rat brain when compared to the control. CE specific activity was lower for 5 days caffeine treated rats 0.172(IU/mg) than their control. CE Specific activity in old control brain was higher 0.82(IU/mg) than caffeine treated rats 0.574 (IU/mg). Similar results were obtained for liver samples. CE characterization studies with time, pH, and temperature gave constantly complimentary results for higher CE activity in control than caffeine treated groups. Therefore caffeine/ coffee may interfere with drug metabolism and is not recommended for patients who are under the drug medication especially for cancer patients.
Keywords: Total protein; Caffeine; Carboxyl esterase; Specific activity; SDS PAGE
Introduction
Caffeine (1,3,7-trimethylxanthine) is a well-known plant alkaloid that is present in several food and beverage products, such as coffee and tea are commonly consumed worldwide. After oral ingestion, caffeine concentration in the plasma peaks within 30-120 min. Furthermore, the hydrophobic property of caffeine allows it to pass through all biological membranes [1]. Caffeine is highlighted as a potent dietarycomponent associated with reduced risk of several chronic diseases, including type 2 diabetes [2,3]. Caffeine and other methylxanthines are used in clinical medicine as diuretics, analgesics, and muscle relaxants, and they can aid in the treatment of brain disorders such as headaches and Parkinson’s disease [4-6]. It has been reported that caffeine acts on various tissues; in particular it has a stimulating effect on the central nervous system [7,8] and cardiac muscle [9,10]. The mechanism by which caffeine improves performance is not clear, but several possibilities have been proposed, such as antagonizing adenosine receptors and increasing catecholamine release. It is known that a chronic low dose of caffeine (10mg/kg) in rats reduces ischemic brain damage and global ischemia. Caffeine and its metabolites could have different effects on the treatment of ischemia depending on the dose, the application, and the model of ischemia [11]. The treatment of rats with a sustained lower dose of caffeine (10 mg/kg) reduced ischemic brain damage and global ischemia. Caffeine’s actions have been shown to be mediated through blocking cell surface adenosine receptors, through inhibition of cAMP phosphodiesterase (PDE) activity, and by affecting the release of calcium from intracellular stores. Each of these three mechanisms has been implicated in modulating Blood Brain Barrier (BBB) functions, and the effects of caffeine on these mechanisms are clearly concentration and dosedependent. Caffeine’s protective effects against Alzheimer’s disease (AD) and Parkinson’s disease (PD) and potential mechanisms against BBB leakage respectively [12]. In liver Karas et al. [13] found that the oral administration of caffeine (150 mg/kg) increased MDA levels in the rat liver that had been treated with allyl alcohol to reduce the hepatotoxicity from the high dose of caffeine [14]. Interestingly, coffee has been suggested to have a potential favorable impact on liver diseases. In North America and Europe, studies have shown that coffee drinking reduces the risk of liver cirrhosis and Hepato Cellular Carcinoma (HCC). In addition, the protective effect of coffee drinking on the development of HCC has been reported in a case control study in japan [15]. Caffeine is metabolized by the liver to form dimethyl and monomethylxanthines, dimethyl and monomethyl uric acids, trimethyl and dimethylallantoin, and uracil derivatives [16,17]. The demethylation, C-8 oxidation, and uracil formation occur mostly in liver microsomes. The fundamental biochemical mechanism that underlies the actions of caffeine at doses achieved in normal human consumption must be activated at concentrations between the extremes (between barely effective doses and doses that produce toxic effects). This tends to rule out the direct release of intracellular calcium probably via an action on ryanodine receptors, the only known mechanism that is significantly affected by the relevant doses of caffeine binding to adenosine receptors and antagonism actions of agonists at these receptors [18,19]. Even though the primary action of caffeine may be to block adenosine receptors this leads to very important secondary effects on many classes of neurotransmitters, including noradrenaline, dopamine, serotonin, acetylcholine, glutamate, and GABA [20]. This in turn will influence a large number of different physiological functions. Caffeine is also known to affect a number of other cell signaling molecules and physiological functions. Caffeine inhibits acetylcholinesterase which inturn responsible for release of acetylcholine, but not butyryl cholinesterase [21]. Caffeine action is also not clear with carboxyl esterase metabolism. A human liver carboxylesterase (hCE-2) that catalyzes the hydrolysis of the benzoyl group of cocaine and the acetyl groups of 4-methylumbelliferyl acetate, heroin, and 6-monoacetylmorphine was purified from human liver [22]. Hydrolysis by carboxylesterases is increasingly used as a basis for drug design, particularly on pro-drugs containing functional groups such as carboxylic acid ester [23-25]. Carboxyl esterases are a group of enzymes widely distributed in nature (mammalian tissues) and isoforms have been described in human liver, brain and placenta. CEs comprises a group of serine–dependent esterases having the highest activity in hepatic microsomes [26,27]. They catalyze the hydrolysis of carboxylic acid esters to produce free acid anions and alcohols. Since a significant number of drugs are metabolized by carboxyl esterase, altering the activity of this enzyme class has important clinical implications. Caffeine can have both positive and negative health effects. However the effects of caffeine on total protein activity and specific activity of carboxyl esterase in young and old aged rat groups is not well understood There are several known action mechanisms to explain the effects of caffeine but not with CE for its activation on drug metabolism. Hence the main objective of this study was to determine effects of caffeine on total protein and carboxyl esterase activity for drug metabolism with Caffeine treated young and old aged male wistar rat’s brain and liver were used in this study.
Materials and Methods
Procuring of rats
Male young wistar rats of 2-4 months old and weight in the range of 150-200 g and adult wistar rats of 17-20 months age and weight in the range of 450-500 g were procured. One set of young and old rats were used for treatment with caffeine while the others are used as control for reference.
Caffeine dosage
Dose of the drug was calculated by converting adult human therapeutic dose (150 mg/day) to animal dose. The average dose of caffeine was amounted to 20 mg/kg of rat’s body weight per day. To study the effects of the drug, five young and old rats were treated with modulators (caffeine) orally for 5 days.
Collection of rat tissue and processing of tissue specimens
The samples of brain and liver were collected by sacrificing the rats. The model was first anesthetized using Xylazine and Ketamine in the ratio 1:4. The models were then prepared for dissection by securing them on wax boards and a cut was made near the dorsal line of skin to take out the liver and brain, which was removed by making a cut near the skull region. Both the liver and the brain were then subjected to wash with 0.9% Phosphate Buffer Saline (PBS) to remove the residues and blood, then weight of tissues were measured.
Preparation of tissue extraction from specimens for biochemical studies
Specimens (brain and liver) were homogenized using pestle and mortar followed by glass homogenization with tris buffer saline (TBS pH=7.4 containing 1% (v/v) triton-X 100) and protease inhibitors (10 μl 0.5 mM phenyl methyl sulphonyl fluoride in ethanol). Tissue to buffer ratio was 1:10 w/v (wet weight). Denaturation was prevented by maintaining specimens at low temperatures (5oC-10oC). Then homogenized sample was centrifuged at 10,000 rpm for 15 minutes at 4°C. The supernatants were collected for biochemical analysis and stored at 4°C until the enzyme activity was assessed.
Protein estimation by lowry’s method
Protein content was estimated using Lowry’s method [28,29] using Bovine Serum Albumin solution (BSA) as a standard.
Carboxyl esterase assay
Activity was measured based on the method by Gomori [30], which was later modified by Van Asperen [31]. Enzyme reaction was initiated by adding 900 μl of 5 mM α-naphthyl acetate in phosphate assay buffer (pH 7.0) to 100 μl tissue extract then incubated for 15 min at 27°C. Subsequently, the reaction was stopped by adding 500 μl DBLS reagent and enzyme activity was measured at 600nm.
SDS PAGE gel electrophoresis
The brain tissue supernatant sample of 40 μl was electrophoresed in a native polyacrylamide gel (10 %) at a constant voltage of 50 Volts. The run was stopped when the marker dye coomassive brilliant blue reached 1-2 mm above the lower edge of the plate. The gel was carefully transferred to the PBS solution. Then gel was incubated in substrate 5 mM α-naphthyl acetate solution for 10 minutes and stopping reagent DBLS was added and incubated till intense bands were seen [32]. 2D Electrophoresis was also performed for young brain and caffeine treated samples. Native PAGE for carboxylesterase was also performed for young and old brain with their caffeine treated samples.
Statistical analysis
The mean and standard deviation was calculated for carboxyl esterase by comparing with their controls and for respective age groups of wistar rats by using ‘t’ test calculator (Quick Calcs, GraphPad Software).
Results and Discussions
Specific activity of CE was calculated by determining total activity and total protein (from rat’s brain and liver tissue samples) and expressed as nm or μm/min/mg of protein. Initially both groups of rats were treated with caffeine intraperitoneally for 150 mg/kg body weight. It was observed that the caffeine dosage given was high enough that they died after 3 days treatment due to toxicity. This confirmed that higher caffeine content is toxic to brain and liver cells. Therefore the dosage was reduced to 20mg/kg body weight for 5 days chronic period.
Total protein content in brain (with caffeine)
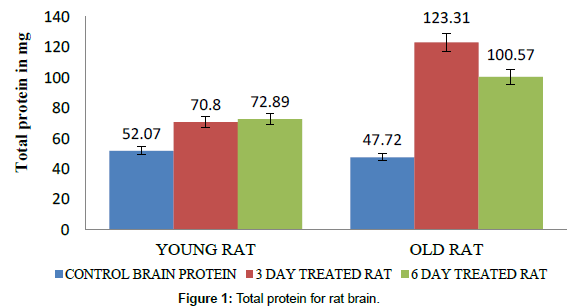
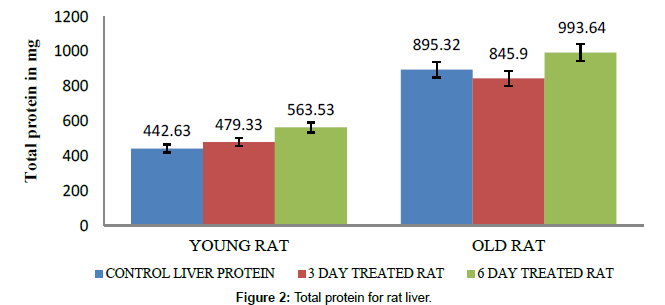
The protein content in brain and liver of young rats, old aged rats and their groups of rats treated with caffeine for 10 days at a time interval of 24 hours varies. The protein content did not shown any significant difference between young brain and old rat’s brain (brain size is almost similar (Tables 1 and 2), whereas lithium has more effect on old brain cells for elevated protein level. The protein content in young rat’s brain showed higher protein content than control, while old rat brain showed significant increase in protein levels for caffeine treatment (Figure 1). In young rat there is a gradual increased protein content 70.8 mg for the 3 day and 72.89 mg for 6 days caffeine treated young rats respectively as compared to normal young brain 52.07 mg. Whereas old rat brain showed significant increased protein content of 123.31 mg for 3 days and 100.57 mg for 6 days caffeine treated old rat brain respectively as compared to normal old rat brain of 47.72 mg. This proves that caffeine is a neurostimulant especially for old rat brain cells. However, the 6 day caffeine treated old rat brain showed a slight decrease in protein content as the assay was performed on the 7th day whereas the others were performed on the last day of their 3 day or 6 day treatment respectively. The protein content in the young and old rat liver control varied significantly as the size and weight of the old rat liver is almost twice to that of what is obtained in young rats (Figure 2 and Table 3). A gradual increased protein 479.33 mg in young rat’s liver for 3 days and 563.53 mg for 6 days caffeine treated young rat liver respectively was observed; whereas, old rat’s liver of 845.9 mg which was decreased protein content as compared to normal rat liver protein 895.32 mg. However, 6days caffeine treated old liver rats showed slight increase in the protein 993.64 mg.
| Group | Age (months) | Body weight (g) | Weight of Brain (g) | Weight of Liver (g) |
|---|---|---|---|---|
| Normal young rats | 2-4 | 100-150 | 1.3242 | 6.1363 |
| Young Treated rats | 2-4 | 100-150 | 1.2615 | 3.6034 |
| Normal old Aged rats | 16-18 | 200-300 | 1.4428 | 7.7549 |
| Old Aged Treated rats | 16-18 | 200-300 | 1.3724 | 5.9426 |
Table 1: Weight of Brain and Liver samples obtained after dissection.
| Control | 3 day treated | 6 day treated | |
|---|---|---|---|
| Old rat Brain | 47.72 mg | 123.31 mg | 100.57 mg |
| Young rat Brain | 52.07 mg | 70.80 mg | 72.89 mg |
Table 2: Total Protein content in brain tissue of male Wistar rat.
| Control | 3 day treated | 6 day treated | |
|---|---|---|---|
| Old rat Liver | 895.32 mg | 845.9 mg | 993.64 mg |
| Young rat Liver | 442.63 mg | 479.33 mg | 563.53 mg |
Table 3: Total Protein content in liver tissue of male Wistar rat.
SDS PAGE electrophoresis
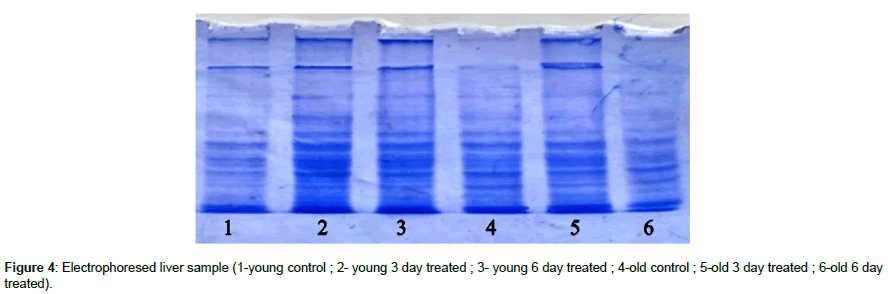
Electrophoresis protein band pattern varies for lithium treated rats as compared to control. More intense band was observed in for 6 days caffeine treated old aged rats than normal old brain and 3 days caffeine treated old rats. More intense band was observed for 3 days caffeine treated young brain than their control (Figure 3). These results are slightly different from spectrophotometric results; Since 6 days lithium treated young rat brain showed less protein band pattern as compared to 3 days treated young rat and control young brain which was not shown by spectrophotometric analysis, even 3 days lithium treated old rat’s brain showed less band protein as compared to control but the protein content was significantly high by lowry’s method. It may be due to variation in protein concentration which was loaded in vertical electrophoresis set up. However protein content for 3 days treated old brain & 6 days lithium treated young brain showed more protein band patterns which are similar to spectrophotometric results. SDS PAGE showed extra single bands for 6 days caffeine treated old rat brain and for 3 day treated young rat brain (Figure 3). The liver samples were subjected to SDS PAGE and the many protein bands were visualized obtained. SDS PAGE showed almost similar protein band patterns for 3 days and 6 days caffeine treated young liver samples than control. Whereas more band intensity was also observed in 3 days caffeine treated old rat liver as compared to respective control and 6 days caffeine treated old liver respectively (Figure 4). Young liver protein band patterns showed similar results to spectrophotometer but not for old liver. By comparing the results of 2D, SDS PAGE of control young rat brain and young 3 day lithium treated brain sample, an evident change in the intensity of bands and band pattern can be observed. 2D electrophoresis showed more expressed high intensity bands in caffeine treated young rat when compared to the control (Figure 5A,5B). Overall caffeine has positive effect on protein elevation in brain which was shown from Lowry’s method spectrophotometric method and electrophoresis which was also observed to some extent in liver samples. Hence caffeine modulates the protein content in liver and brain by binding and/or folding of protein and enhances protein activation especially in old rat brain. 2D electrophoresis showed more protein expressed high intensity bands pattern in caffeine treated young rat when compared to the control. Hence caffeine is responsible for induction of more protein content in brain cells. Higher total protein connected with cell stability and function that leads to regulation of cellular activity and cell survival.
Estimation of specific activity for carboxyl esterase
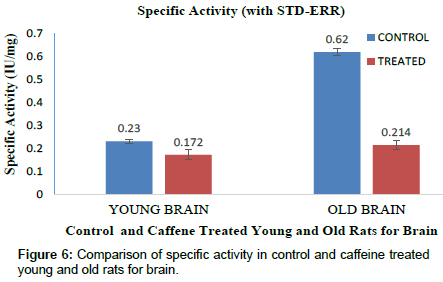
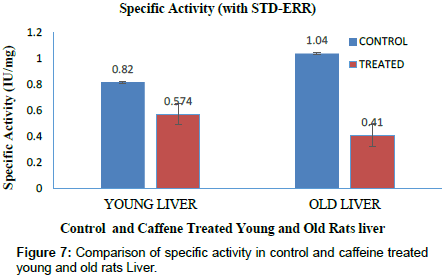
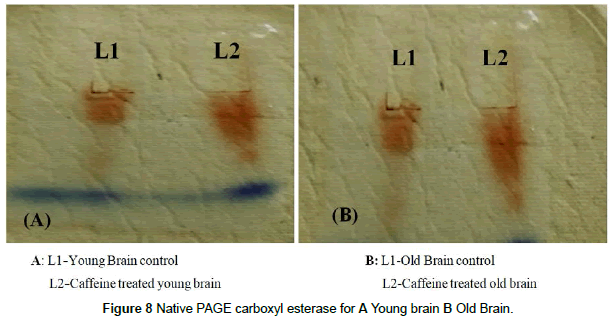
The specific activity for carboxylesterase in brain and liver tissue was determined by total activity and total protein. Specific activity of CE in young control rat brain was found to be 0.23(IU/mg) and caffeine treated rats showed 0.172(IU/mg) which was lower in brain as compared to young control rats (Fig. 6). Specific activity of old control rat’s brain was found to be 0.62(IU/mg) and caffeine treated rats showed 0.214 (IU/mg) for CE (Figure 6). In case of control young rats liver, CE showed 0.82 (IU/mg) and caffeine treated rats showed 0.574(IU/mg) a significant lower enzyme activity as compared to normal young rat (Figure 7). CE specific activity in control old liver rats was found to be 1.04(IU/mg) and 0.41(IU/mg) in caffeine treated old rats. Consequently caffeine treated rats showed significant decreased enzyme activity in both liver and brain samples when compared with control old rats. In contrast to spectrophotometric assay of CE, Native PAGE showed high CE expression in caffeine treated young and old brain rats than control. Since in vertical electrophoresis samples could not be loaded according to activity. Hence these results were opposite to spectrophotometer results since specific activity gets cancelled with the protein content whereas in native PAGE both protein and enzymatic bands were observed as intense band (Figure 8).
Characterization studies
Enzyme kinetics deals with several factors affecting the rate of enzyme catalyzed reactions. The most important factors are: time, pH, temperature, and substrate.
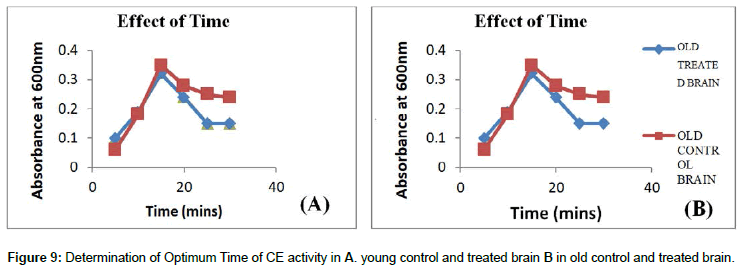
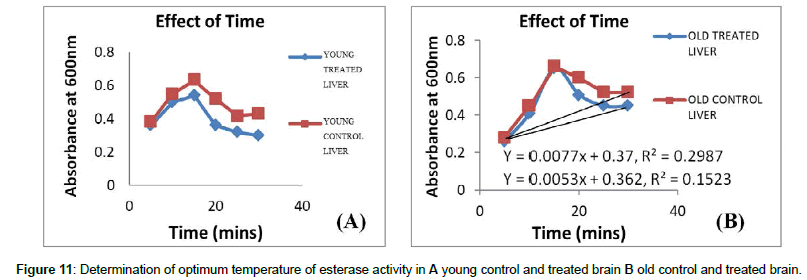
Time: Time is a characteristic feature which can indicate the activity of an enzyme. Maximum enzyme activity was observed between 15-20 minutes, where all the enzyme binding sites are occupied by the substrate; once the substrate binding sites are occupied the activity of the enzyme remains unaltered even for long period of incubation (Figures 9 and 10). Minimum activity is observed when the enzyme is incubated for a short period of time, since substrate molecules fail to bind the active sites of an enzyme.
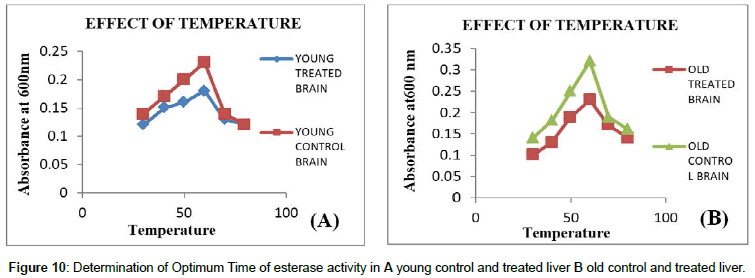
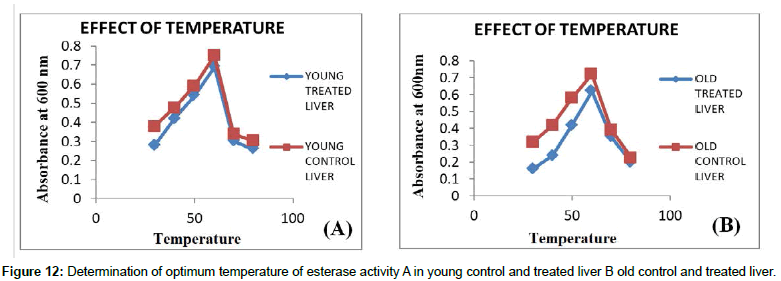
Temperature: Temperature plays a major role in activation of enzyme since higher the temperature higher the activity of an enzyme can be obtained, but at 60°C enzyme gets denatured. It was observed that every enzyme has maximum activity at a particular temperature which is characteristic of that enzyme, called the optimum temperature and hence it is the best way to do assay for an enzyme at this temperature. It was seen that there is a slight rise in temperature from 31°C- 40°C and elevated rise in temperature from 45°C- 60°C and showed decline activity in temperature at 70°C and 80°C (Figures 11 and 12). Effects of different temperatures on carboxyl esterase activity was carried out from 31°C -80°C, in which optimum temperature showed maximum activity of carboxyl esterase at 60°C which indicates the thermo stability of esterase’s at such high temperatures.
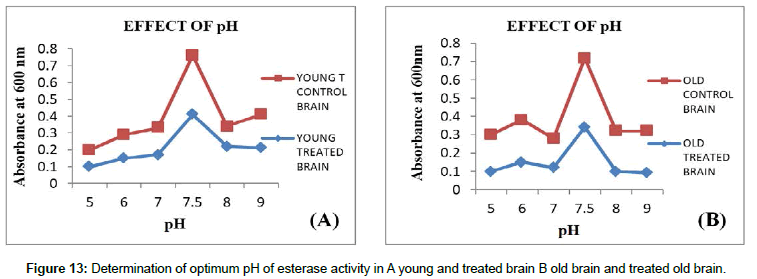
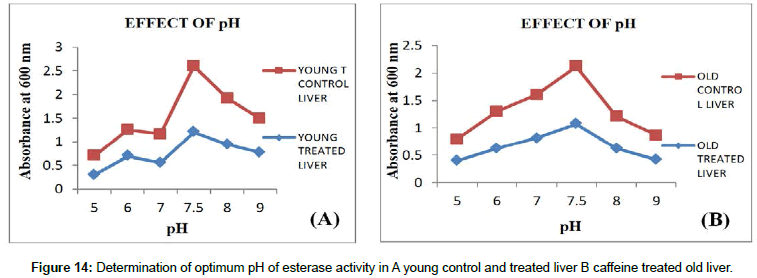
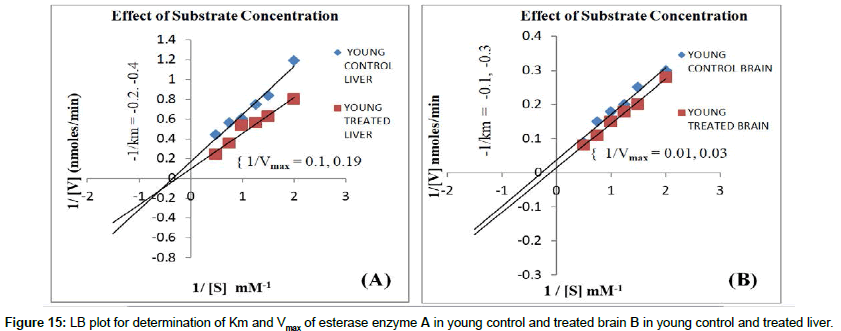
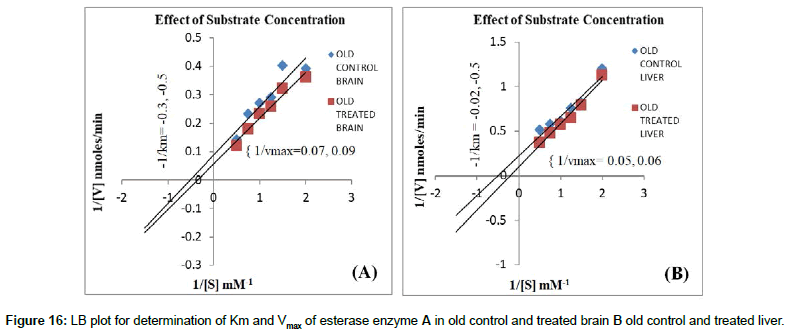
pH: Effect of different buffers with different pH on carboxyl esterase’s was carried out in the range of pH-5-9 in which optimum pH 7.5 showed maximum CE activity. During the assay, the pH is brought back to optimum to detect the maximum activity (Figures 13 and 14).
Substrate concentration: The enzyme showed maximum activity when the substrate concentration markedly increased than the Km of the native enzyme. Hence, determination of Km for any enzyme is absolutely essential during kinetic assays to determine the amount of substrate required for the maximal activity of an enzyme. Vmax value is in significant due to its information of maximum rate of enzyme activity. This is possible only when all the sites are filled with the substrate molecules. At very low substrate concentration the overall rate of the reaction will be limited by rate at which an enzyme and substrate interact to form the ES complex. Effect of Km and Vmax on carboxyl esterase’s at different substrate concentration for crude extracts of control young rats brain were determined which of Km 0.25 nM and Vmax 82.631 nmoles/min and Caffeine treated young rats showed Km 0.35 mM of and Vmax of 52.21 nmoles/min. (Figure 15A). Control old rat’s brain which showed Km of 0.33 nM and Vmax of 110.1 nmoles/min and Caffeine treated rats brain showed Km 0.2 mM of and Vmax of 98.25 nmoles/min. Therefore Lower the Km value, higher is the affinity of the substrate (Figure 15 B). Similarly LB plot for determination of Km and Vmax of esterase enzyme (Figure 16A) in young rat’s liver control and caffeine treated rat’s liver and other group old rat’s liver control and treated liver (Figure 16B) were also showed similar plot types of brain. The CE specific activity was lower in caffeine treated young and old aged rat’s brain and liver as compared to their control. These results were similar in different parameters study for CE activity. Characterization studies was carried out for CE by varying pH, time, temperature and substrate and these experiments were also showed constantly lower enzyme activity for caffeine treated brain and liver of young and old aged rats when compared to their respective control treatments (of the same parameters). The brain and liver samples of rats in young and old group control showed higher CE activity and Lower the Km value; higher is the affinity of the substrate. Hence normal young and old aged brain and liver cells were more stable with higher CE activity. In contrast native PAGE showed high CE expression in caffeine treated young and old brain rats than their control for which the exact reason is unknown. However it may be due to variation in equal activity while loading samples in vertical electrophoresis. The specific activity of CE was high for normal old brain cells rather than normal young brain and similar results were also obtained with liver cells. These results suggested that old brain and liver cells have drug metabolizing action due to higher CE specific activity; whereas, old brain and liver treated with caffeine showed significant lower CE enzyme activity as compared to their control samples. Hence this leads to failure of drug activation in models of diseases (e.g. Cancer) under medication, since CE is responsible for metabolism of drugs for an e.g. Irinotecan anticancer drug, for which CE metabolizes and gives rise to SN 38 that, is toxic to cancer cells. Therefore caffeine/coffee may interfere with drug metabolism and is not recommended for patients who are under the drug medication especially for cancer patients. Caffeine is not recommended to be consumed in high quantities for old aged people under medication with other types of drugs since it lowers the carboxyl esterase activity which in turn cannot effectively metabolize the drug. Hence CE has to be activated with other modulators such as lithium, Indian spices (cinnamon and cardamom) etc. Our three previous reports suggested that lithium treated old aged rats brain showed significantly higher CE specific activity [33] and elevated CE activity [34] as compared to young rats. In another report, CE was high with lithium treated brain tissue samples which are partially purified as compared with their control. Dietary supplementation of cardamom and cinnamon in moderate amounts was reported to aid in prevention and delay in onset of Alzheimer’s disease AD in rat model since they have shown highest anti-cholinesterase effect on acetylcholine esterase and to some extent to butryl choline esterase and modulation of CE activity may help in the treatment of AD [35]. Hence cinnamon and cardamom are inhibitors for acetylcholine esterase activity but not for CE. This information also supported the findings from other reports that caffeine inhibits Acetyl Cholinesterase (AChE), but not butyrylcholinesterase [36] and caffeine can act as a non-competitive inhibitor of AChE in the body. These findings can be expected in some of the clinically proven effects with unknown molecular mechanism. It can be emphasized that caffeine acts as a potential lead structure in drug design for protein activity that leads to cell function. Hence brain caffeine protects Alzheimer’s mice against cognitive impairment and reduces brain β-amyloid production where data demonstrates that moderate daily intake of caffeine may delay or reduce the risk of AD [37] and may provide surprising protection against, or delay, the onset of AD. These results are indicative that moderate caffeine intake (the human equivalent of 500 mg caffeine or 5 cups of coffee per day) can protect against or treat AD in a mouse model for the disease and it has a therapeutic potential against AD in humans [38]. Therefore even though caffeine does not elevates CE, it is psychoactive drug and good neurostimulant to release neurotransmitters and activation for other molecules like protein that showed higher content in our current study and hence further leads to activation of cell and regulation of brain cell function. Since the carboxyl esterase is drug metabolizing enzyme, caffeine is also referred as a drug but still it cannot elevate CE and has not shown much effect on metabolism of brain and liver cells for all groups of male wistar rats. However, caffeine is a neurostimulant as it enhances protein level in old brain cells and some extent to other groups also. Young brain and liver can withstand CE activity in caffeine treatment as compared to old brain and liver respectively. Hence during medication with other drugs for treatment of brain and liver diseases, coffee cannot be consumed in high doses especially for old people since caffeine lowers the activation of carboxyl esterase which is involved in drug metabolism for the treatments of respective diseases.
Conclusions
Total Protein and CE was estimated with caffeine treated (daily dose of stock 13 mg/kg) for 3 and 6 days young and old male wistar rat’s brain and liver (each n=5) by comparing to control. Histogram showed significantly higher protein 123.31 mg and 100.57 mg respectively for 3 and 6 days caffeine treated old rat’s brain than their control 47.72 mg of protein but not significant in young brain. SDS PAGE showed extra single bands for 6 days caffeine treated old rat brain and for 3 day treated young rat brain. This confirms that caffeine was involved in increasing protein content especially in brain cells. 2D electrophoresis also showed more expressed high intensity bands in caffeine treated young rat when compared to the control. CE specific activity was higher for young control brain 0.23 IU/mg than for 5 days caffeine treated rats 0.172 IU/mg, while old control brain showed higher 0.82IU/mg than caffeine treated rats 0.574 IU/ mg and similar results were obtained for liver samples. This suggests that caffeine interrupted CE activity. CE characterization studies with time, temperature, pH and substrate gave constantly complimentary results for assay of CE which showed highest CE activity in all groups of control than caffeine treated groups. Therefore caffeine/coffee may interfere with drug metabolism and is not recommended for patients who are under the drug medication especially for cancer patients, glaucoma and other brain diseases.
Acknowledgement
We express our sincere thanks to Prof. Nagaveni, Head of the Department, MLACW for giving us an opportunity to carryout project work in their institute. We would like to thank Dr. Channarayappa, Professor and Head, Department of Biotechnology, MSRIT for his guidance and encouragement. Authors would also like to acknowledge, Sameer, Pooja, Pooja C, who were also helped for this project.
References
- Fredholm B, Bättig K, Holmen J, Nehlig A, Zvartau E (1999) Actions of caffeine in the brain with special reference to factors that contribute to its widespread use, Pharmaco Rev, 51, 83-133.
- Keijzers B, Galan E, Tack J, Smits P (2002) Caffeine can decrease insulin sensitivity in humans, Diabetes Care 9:36.
- Duffey J, Popkin M (2007) Shifts in patterns and consumption of beverages between 1965 and 2002, Obesity 15:2739-2747.
- Paynter P, Yeh H, Voutilainen S, Schmidt I, Heiss G (2006) Coffee and sweetened beverage consumption and the risk of type 2 diabetes mellitus: the atherosclerosis risk in communities study, Am J Epidemiol 164:1075-1084.
- Tuomilehto J, Hu G, Bidel S, Lindstrom J, Jousilahti P (2004) Coffee consumption and risk of type 2 diabetes mellitus among middle-aged Finnish men and women, JAMA 291:1213-1219.
- Kolayli S, Ocak M, Küçük M, Abbasoğlu R (2004) Does caffeine bind to metal ions? Food Chem 84: 383.
- Al Deeb A, Al Moutaery A, Arshaduddin M, Biary N, Tariq M (2002) Effect of acute caffeine on severity of harmaline induced tremor in rats, Neuroscie Let 8: 216-325.
- Barbier A (2001) Caffeine protects against Parkinson’s disease? Tre Pharmaco Sci 22: 500.
- Nikolic J, Bjelakovic G, Stojanovic I (2003) Effect of caffeine on metabolism of L-arginine in the brain, Mol Cellu Biochem 8:125-244.
- Davis J, Zhao Z, Stock S, Mehl K, Buggy J, Hand G(2003) Central nervous system effects of caffeine and adenosine on fatigue. Am J Physio Regu Integ Comp Physio 284: R399-404.
- Greer F, Friars D, Graham E (2000) Comparison of caffeine and theophylline ingestion: exercise metabolism and endurance, J App Physio 89.
- Savoca R, Evans D, Wilson E, Harshfield A, Ludwig A (2004) The association of caffeinated beverages with blood pressure in adolescents, Arch Pedia Adol Medi 7:158-473
- Strong R, Grotta C, Aronowski J (2000) Combination of low dose ethanol and caffeine protects brain from damage produced by focal ischemia in rats, Neuropharmaco 39: 515-22.
- Xuesong C, Othman G, Jonathan D (2010) Caffeine Protects Against Disruptions of the Blood-Brain Barrier in Animal Models of Alzheimer’s and Parkinson’s Diseases, J Alzheimer’s Disease 127:127-141.
- Karas M, Chakrabarti K (2001) Influence of caffeine on allyl alcohol-induced hepatotoxicity in rats. In vivo study, J Envi Patho Toxi Onco 20: 141-54.
- Mukhopadhyay S, Mondal A, Poddar K(2003) Chronic administration of caff eine: eff ect on the activities of hepatic antioxidant enzymes of Ehrlich ascites tumor-bearing mice, Indian J Exp Bio 41: 283-9.
- Tanaka K, Hara M, Sakamoto T, Higaki Y, Mizuta T et al (2007) Inverse association between coffee drinking and the risk of hepatocellular carcinoma: a case–control study in Japan, Cancer Sci 98:214-218.
- Arnaud MJ (1987) The pharmacology of caffeine, Prog Drug Res 31:273-313.
- Arnaud J (1993) Metabolism of caffeine and other components of coffee in Caffeine, Coffee, and Health pp 43-96.
- Fredholm B (1980) Are methylxanthine effects due to antagonism of endogenous adenosine?, Trends Pharmacol Sci 1:129-132.
- Fredholm B, Battig K, Holmen J, Nehling A, Zvartau E (1999) Actions of caffeine in the brain with special reference to factors that contribute to its widespread use, Pharmaco Revi 51: 84-125.
- Daly W (2007) Caffeine analogs: biomedical impact, Cell Mol Life Sci 64:2153-2169.
- Miroslav Pohanka, Petr Dobes (2013) Caffeine Inhibits Acetylcholinesterase, But Not Butyrylcholinesterase, Int J Mol Sci 14:9873-9882.
- Pindel V, Kedishvili Y, Abraham L, Brzezinski MR, Zhang J (1997) Purification and cloning of a broad substrate specificity human liver carboxylesterase that catalyzes the hydrolysis of cocaine and heroin, J Biol Chem 272:14769-7.
- Bodor N, Buchwald P (2002) Designing safer (soft) drugs by avoiding the formation of toxic and oxidative metabolites, Methods Mol Biol 186:301-312.
- Peter Senter , Kevin S, Bruce Mixan , Alan F (2001) Identification and Activities of Human Carboxylesterases for the Activation of CPT-11, a Clinically Approved Anticancer Drug, Bioconjugate Chem 12:1074-1080
- Zhu W, Song L, Zhang H, Matoney L, LeCluyse E et al., (2000) Dexamethasone differentially regulates expression of carboxylesterase genes in humans and rats, Drug Metab Dispos 28:186-91.
- Hosokawa M, Maki T, Satoh T (1987) Multiplicity and regulation of hepatic microsomal carboxylesterases in rats. Mol Pharmacol 31:579-84.
- Morgan, Earl W, et al (1994) Regulation of Two Rat-Liver Microsomal Carboxylesterase Isozymes-Species-Differences, Tissue Distribution, and the Effects of Age, Sex, and Xenobiotic Treatment of Rats, Arch Biochem Biophysics 315: 513-526.
- Hartree F (1972) Determination of proteins: A modification of the Lowry method that gives a linear photometric Resp, Anal Biochem 48: 422-427.
- Gomori G (1953) Histochemical studies of the inhibition of esterases. J Cell Physiol 41: 51-63.
- Asperen K (1962) A study of housefly esterases by means of a sensitive colorimetric method, J Insect Physiol 8: 401-416.
- Laemmli K (1970) Cleavage of structural proteins during the assembly of the head of bacteriophage T4, Nature 227: 680-685
- Prabha M, Bhavana G, Sunitha P, Channarayappa, Lokesh KN (2015) The Role of Carboxyl esterase and Acid phosphatase in Aged and Lithium Treated Rats in Regulation of Neuronal Function, J Biochem 6: 889-893.
- Sushma Rao, Suneetha P, Nagaveni B, Prabha M (2015) Partial purification and characterization of carboxyl esterase in aged and lithium treated rat brain, J Biochem Tech 6: 883-888.
- Prabha M, T Anusha (2015) Esterase’s properties in commonly used Indian spices for Alzheimer’s disease model, J Biochem Tech, 6: 875-882.
- Pohanka M, Dobes P (2013) Caffeine Inhibits Acetylcholinesterase, But Not Butyrylcholinesterase, Int J Mole Sci 14:9873-9882.
- Arendash W, Schleif W, Rezai-Zadeh K, Jackson K, Zacharia L (2006) Caffeine protects Alzheimer's mice against cognitive impairment and reduces brain beta-amyloid production, Neurosci 142:941-52.
- W Arendash et al (2010) Caffeine and Coffee as Therapeutics Against Alzheimer's Disease, J Alzheimers Dis 1:117-126.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi