Research Article, J Mar Biol Oceanogr Vol: 7 Issue: 2
Classification of the Intertidal and Estuarine Upogebiid Shrimps (Crustacea: Thalassinidea), and Their Settlement in the Ryukyu Islands, Japan
Masayuki Saigusa1*, Yuriko Hirano1, Bong Jung Kang2, Kazuki Sekiné3, Masatsugu Hatakeyama4, Takahiro Nanri1, Masami Hamaguchi5 and Nobufumi Masunari1,6
1Section of Research and Education, The Biosphere Project, Tsushima-Fukui 1-8-71, Okayama-Kitaku, Okayama 700-0080, Japan
2Fisheries Division, Japan International Research Center for Agricultural Sciences, Owashi 1-2, Tsukuba, Ibaraki 305-8686, Japan
3Faculty of Geo-Environmental Science, Rissho University, 1700 Magechi, Kumagaya, Saitama 360-0194, Japan
4Division of Insect Sciences, National Institute of Agrobiological Sciences, Owashi 1-2, Tsukuba, Ibaraki 305-8634, Japan
5Coastal Fisheries and Environment Division, National Research Institute of Fisheries and Environment of Inland Sea (FEIS) Fisheries Research Agency (FRA), Maruishi 2-17-5, Hatsukaichi-shi, Hiroshima 739-0452, Japan
6Research Institute for Fisheries Science, Okayama Prefectural Technology Center for Agriculture, Forestry, and Fisheries, Ushimado 6641-6, Okayama- Setouchi, 701-4303, Japan
*Corresponding Author : Masayuki Saigusa
Section of Research and Education, The Biosphere Project (Non-Profit Organization), Tsushima Fukui 1-8-71, Okayama-Kitaku 700-0080, Japan
E-mail: saigusa@biodiv-p.or.jp
Received: January 23, 2018 Accepted: April 12, 2018 Published: April 20, 2018
Citation: Saigusa M, Hirano Y, Kang BJ, Sekiné K, Hatakeyama M, et al. (2018) Classification of the Intertidal and Estuarine Upogebiid Shrimps (Crustacea: Thalassinidea), and Their Settlement in the Ryukyu Islands, Japan. J Mar Biol Oceanogr 7:2. doi: 10.4172/2324-8661.1000192
Abstract
Classification of the Intertidal and Estuarine Upogebiid Shrimps (Crustacea: Thalassinidea), and Their Settlement in the Ryukyu Islands, Japan
Upogebiid shrimps (Crustacea: Thalassinidea) inhabit sandstones exposed on the intertidal zone of two of the islands of the Ryukyu Arc, Japan (islands located between 24°N, 123°E and 28°N, 129°E). The upogebiids here are morphologically similar; so, their classification is open to debate. In addition, when the upogebiids settled on the Ryukyu Arc has not been studied till date. Based on morphological features, we suspect that six stone upogebiid species are distributed in the Ryukyu Arc. This hypothesis was tested by molecular phylogenetic analysis of mitochondrial DNA markers. Furthermore, Bayesian evolutionary analysis, which was used to estimate TMRCA (time to most recent common ancestor) of each upogebiid, showed that stone upogebiids diverged 5-15 Ma (second half of the Miocene). Stone upogebiids may have occurred in the islands near the equator 5-15 Ma. On the contrary, Bayesian analysis indicated that the time of ‘intraspecific’ divergence varied from 1.15 Ma to 0 Ma, suggesting that stone upogebiids may have settled in the Ryukyu Arc from the Pleistocene to the present. In Iriomote Island, sedimentary layers suitable for burrowing occurred on the shores from the west to the northern half of the island, but not on the east. The Ryukyu Arc was a passive continental margin until the beginning of the Pleistocene, but the warm Kuroshio current completely separated the Ryukyu Arc from the Eurasian continent about 1.55 Ma. With subduction of the Ryukyu Arc northwestward, sedimentary layers for burrowing may have appeared on the ‘front’ of the island (i.e. on the shores from the west to the north), thereby providing suitable habitats for the stone upogebiids to settle, since 1.15 Ma.
Keywords: Stone upogebiids; Iriomote Island; Classification; Habitat; Bayesian evolutionary analysis; 1.55 Ma; Subduction of Ryukyu Arc
Introduction
Muddy shores appear in intertidal areas protected from open ocean wave activity, i.e. in partially enclosed bays, lagoons, harbors, and estuaries, especially [1]. While sand beaches have larger grain size, muddy areas consist of finer-grained sediment particles [2,3]. The slope of muddy shores is much flatter than that of sandy beaches; so, these areas are often referred to as mudflats.
Mudflats show high standing stocks of phytoplankton, as determined by measuring the chlorophyll concentration [3,4]. Detritus is also abundant. The inhabitants of tidal mudflats (infauna) burrow in the sediment, and they feed on detritus in the sediment and water. Thus, a number of mudflat inhabitants may be deposit and suspension feeders [5]. At low tide, their presence may be shown by small holes on the flats, small sediment mounds topped by a hole, or piles of feces [3]. Among the large number of mudflat inhabitants, thalassinidean decapods (Crustacea) make elaborate burrows in the sediment [6-10]. They may feed on detritus that is filtered from water [4].
Mud shrimps (Upogebiidae) have been described from many of the world’s marine and estuarine shores [8,11-13]. The main bases for upogebiid identification are the morphological characteristics of their exoskeleton. Nine species of upogebiid were recorded from the mudflats on the coasts of the main islands of Japan (Kyushu, Shikoku, Honshu, and Hokkaido), where the substrates were mud, muddy sand, gravels, or boulders [14].
Upogebiids are also found in partially enclosed bays and estuarine shores in the subtropical islands of Japan. A feature of some of these upogebiids is that they burrow into siltstones or sandstones lying on the shore. These upogebiids were first examined by Hirano and Sakai [15,16]. However, some important morphological features were not considered in their classification. A significant issue, therefore, is the uncertainty over the number of stone upogebiids inhabiting the subtropical islands of Japan, like the Ryukyu Islands [13,15-18]. To resolve some of these debates on classification, we collected stone upogebiids in the Ryukyu Island Arc and reclassified them. Based on morphological features, herein we propose a hypothesis on their classification (hereafter the ‘Hirano-Saigusa hypothesis’).
Genetic relatedness based on molecular phylogenetic studies may be available for the identification of similar species [19-24]. The Hirano-Saigusa hypothesis could be tested by using molecular phylogenetic studies, such as those employing mitochondria DNA (COI) markers; this was, therefore, our second task.
The third task we aimed to undertake was to determine the relation between the distribution and characteristics of stone upogebiid habitats. By collecting a number of stone upogebiids in Iriomote Island, we noticed that they inhabit the shores from the west to the northern parts (i.e. west side) of the island, and that no upogebiids burrow in the sedimentary layers exposed on the eastern shores. This observation led us to question why stone upogebiid distribution was limited to the western side of the island. To answer this question, we first collected samples of the substrate and analyzed their grain size. Then, we examined whether the sedimentary layers that are burrowed were exposed in the lower part of the intertidal zone. Distribution of the habitat of stone upogebiids might be strongly related to the subduction of Iriomote Island towards the northwest.
The Ryukyu Arc formed the eastern margin of the Eurasian continent between the Cretaceous and the Oligocene, and was subaerial as a passive continental margin, possibly up to around 13.5 Ma [25]. However, with the opening of the Okinawa Trough (back arc basin-rift valley), the Ryukyu Arc was isolated from the Eurasian continent about 1.55 Ma, and the warm Kuroshio current then passed between the Ryukyu Arc and the continent [26]. The movement of the Philippine Plate is possibly still causing subduction of Iriomote Island northwestward [26,27].
Bayesian evolutionary analysis is available for the estimation of the time to the most recent common ancestor (TMRCA) [28]. Divergence of the stone upogebiids was estimated to have occurred in the second half of the Miocene. However, this estimate does not agree with the geologic time at which the Ryukyu Arc is supposed to have separated from the Eurasian continent. Another question was, therefore, when the stone upogebiids settled in the Ryukyu Arc after it had separated from the Eurasian continent about 1.55 Ma.
Materials and Methods
Geography of the ryukyu islands and iriomote island
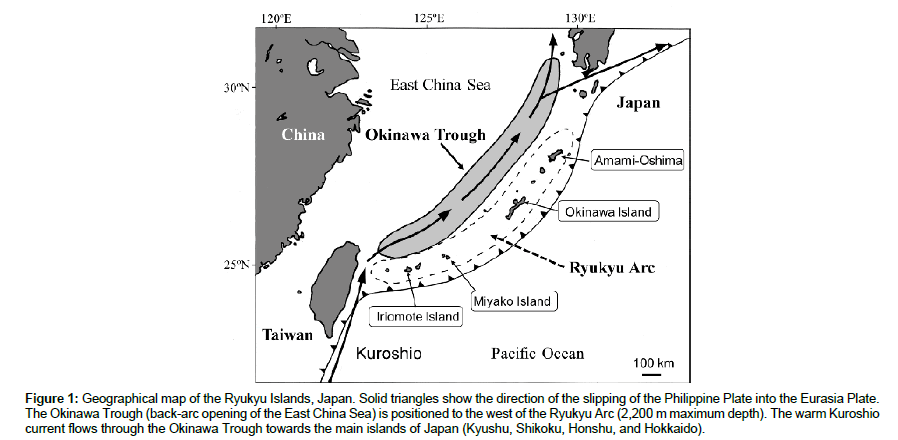
The Ryukyu Islands consist of many subtropical islands, from Yonaguni Island (24°N 123°E) in the west to Amami-Oshima (28°N 129°E) in the north (Figure 1). Major islands are Iriomote, Ishigaki, Miyako, Okinawa, and Amami. The Okinawa Trough (back arc basin-rift valley) is located between the Ryukyu Arc and the Eurasian continent. The Kuroshio current passes above the Okinawa Trough towards the main islands of Japan. The major strata of the Iriomote and Okinawa islands are sedimentary layers [27,29,30].
Figure 1: Geographical map of the Ryukyu Islands, Japan. Solid triangles show the direction of the slipping of the Philippine Plate into the Eurasia Plate. The Okinawa Trough (back-arc opening of the East China Sea) is positioned to the west of the Ryukyu Arc (2,200 m maximum depth). The warm Kuroshio current flows through the Okinawa Trough towards the main islands of Japan (Kyushu, Shikoku, Honshu, and Hokkaido).
In Iriomote Island, there are many hill chains of altitudes between 300 m and 400 m, and a number of streams flow into the sea through the subtropical rain forest on the island. Mudflats are well developed in the island’s partially enclosed bays and estuaries. In Okinawa Island, hill chains at the altitude of 200-400 m occur in the northern half, but the land is flat in the southern half. There is also much rainfall on Okinawa Island. However, because of transportation of sand by strong winds, small estuaries are closed there.
Collection of stone upogebiids and identification of morphological species
Stone upogebiids inhabit the shores of the Okinawa and Iriomote islands. These upogebiids make U-shaped burrows in relatively soft stones or sedimentary layers. Soft stones and sedimentary layers are found at the banks of the estuaries (Figure 2a), or on shores along the partially enclosed bays when the tide recedes. Stone upogebiids were collected using a chisel and hammer at these locations when the water level was low (Figure 2b). Upogebiids were fixed in 70% ethanol, and identification was made under a stereomicroscope (Stemi 2000-C, Zeiss) in the laboratory. Collection was mostly carried out every day during two weeks in May 2004, November 2005, and May 2007, and every day for one month from April to May in 2017 (Table 1).
| Species | Collection | Number* | Latitude (N) | Longitude (E) | Habitat | Distribution |
|---|---|---|---|---|---|---|
| Upogebia saigusai | Iriomote Is. | 12 | 24°35'-41' | 123°73'-77' | Shores of partially enclosed bay | Iriomote Island |
| Upogebia semicircula | Iriomote Is. | 12 | 24°35'-41' | 123°73'-77' | Shores of partially enclosed bay | Iriomote Island |
| Upogebia spinidactylus | Iriomote Is. | 12 | 24°35'-41' | 123°73'-77' | Estuarine shores | Iriomote Island |
| Upogebia iriomotensis | Iriomote Is. | 10 | 24°34'-41' | 123°73'-77' | Estuarine shores | Iriomote Island |
| Upogebia mioceni neogenii |
Iriomote Is. | 6 | 24°34'-41' | 123°73'-77' | Estuarine shores | Iriomote Island |
| Upogebia rupicola | Okinawa Is. | 12 | 26°38' | 128°02' | Shores of partially enclosed bay | Okinawa Island |
| Laomedia astacina** | Honshu | 3 | 34°22' | 133°31' | Mudflat in the estuary | Honshu and the Ryukyu Arc |
Table 1: Stone upogebiids employed for molecular philogenetic analysis.
Figure 2: Habitat of stone upogebiids and their morphological features: a) Habitat (dotted line) of U. iriomotensis, U. neogenii, and U. spinidactylus on an estuarine shore of Iriomote Island (site e in Figure 7A). HTL: high tide line. b) Collection of upogebiids in the mangrove flat (site g in Figure 7A). c) The whole body of U. spinidactylus (dorsal view). d) Lateral view of the head (the square shown in Figure 2c). es: eyestalk, st: assemblage of the seta, dp (a double arrow): depression between the hard spines (ii and iii), sp: spine, ve: ventilation hole, an: antenna, ME: merus. Hard spines arranged along both sides of the head are shown by the numbers i-viii. e) Dorsal view of the head (the square indicated in Figure 2c). st: assemblage of the seta, es: eyestalk, gv: groove, iii: the third spine. f) The outer side of male right chela. PR: propodus, DA: dactylus, st: long setae borne on the segment. A small projection characterizing this species is shown by the white arrow. g) The inner side of male right chela. ME: merus, CA: carpus, PR: propodus, DA: dactylus. Scale is shown in each panel.
Reclassification of stone upogebiids
Stone upogebiids collected from the Ryukyu Islands have previously been identified by Hirano and Sakai [15,16]. However, some characteristics were not considered in their studies. Upogebiids were re-examined by employing some new standards, and a reconstructed classification (Hirano-Saigusa hypothesis) has been proposed in this study (Table 2). Other studies [11,17,31-33] were also referred to.
| Hirano-Saigusa hypothesis | Accession number | Upogebiids that have already been registered in Gene Bank |
|---|---|---|
| Upogebia saigusai | AB771737 | Upogebia snelliisi |
| Upogebia spinidactylus | AB771740 | Upogebia spinidactylus |
| Upogebia semicircula | AB771736 | Upogebia sp. |
| Upogebia iriomotensis | AB771738 | Upogebia iriomotensis (Type 1) |
| Upogebia neogenii | AB771739 | Upogebia iriomotensis (Type 2) |
| Upogebia rupicola | AB771735 | Upogebia carinicauda |
| Laomedia astacina | AB771741 | Laomedia astacina |
Table 2: Gene Bank accession number of stone and mudflat upogebiids.
1) U. spinidactylus inhabits the estuarine shores and mudflats of the mangrove flats of Iriomote Island, as has been described by Sakai and Hirano [16].
2) U. saigusai inhabits the shores of partially enclosed bays on Iriomote Island, as has been described by Sakai et al. [15], but there the specimens were described as U. snelliusi.
3) U. iriomotensis inhabits the estuarine shores of Iriomote Island, as has been described by Sakai and Hirano [16].
4) U. neogenii (new species), inhabits the estuarine shores of Iriomote Island, as is first described in this study.
5) U. semicircula (new species), lives in the sedimentary layers on the shores of the partially enclosed bays of Iriomote Island, as is first described in this study.
6) U. rupicola inhabits the siltstone or sandstone layers on Okinawa Island, as described by Komai [18].
Distribution of stone upogebiids in Iriomote and Okinawa Islands, and grain-size analysis of stone habitats
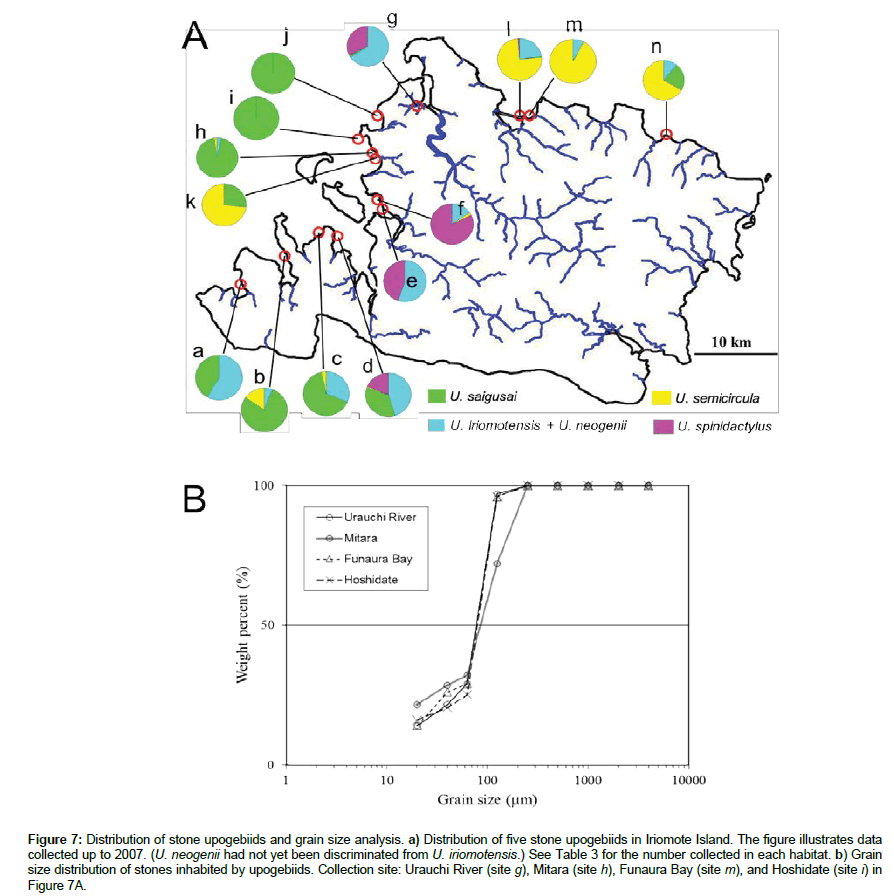
We examined the soft stones burrowed into by the upogebiids along a number of shores of several islands. Stone upogebiids were only found in Iriomote Island and Okinawa Island (Figure 1). The distribution of stone upogebiids was investigated along the shores of these islands, especially Iriomote Island (Table 3).
| Location | Shores | Type of habitat | Species (%) | Total number of individuals collected | |||
|---|---|---|---|---|---|---|---|
| U. saigusai | U. semicircula | U. iriomotenis* | U. spinidactylus | ||||
| a | Sakiyama | I | 41.7 | 0 | 58.3 | 0 | 12 |
| b | Amitori | I | 78.5 | 15.6 | 5.9 | 0 | 135 |
| c | Ida | I | 65.6 | 3.1 | 31.3 | 0 | 32 |
| d | Funauki | I, II | 35.1 | 0.7 | 45.3 | 18.9 | 148 |
| e | Nakara River | II | 0 | 0 | 55.1 | 44.9 | 107 |
| f | Shirahama | I, II | 0 | 2.3 | 15.9 | 81.8 | 44 |
| g | Mitara-1 | I | 27 | 73 | 0 | 0 | 63 |
| h | Mitara-2 | I | 94.6 | 2.2 | 3.3 | 0 | 92 |
| i | Sonai | I | 0 | 100 | 0 | 0 | 4 |
| j | Hoshidate | I | 0 | 100 | 0 | 0 | 58 |
| k | Urauchi River | II | 1.6 | 0 | 65.6 | 32.8 | 64 |
| l | Hinai River | I, II | 0.5 | 75.4 | 23.1 | 1 | 195 |
| m | Funaura Bay | I | 0 | 92.3 | 7.7 | 0 | 13 |
| n | Yutsun | I | 22.2 | 66.7 | 11.1 | 0 | 9 |
Table 3: Shores inhabited by stone upogebiids in Iriomote Island.
Sedimentary stones were classified into three categories based on their grain size: puddingstone (more than 2 mm), sandstone (from 62 μm to 2 mm), and mudstone (less than 62 μm) [2]. Stones burrowed into by the upogebiids were collected from four locations on Iriomote-island, and were pulverized using a mortar and pestle. Grain size was estimated by sifting through screens of 10 mesh sizes, from 20-μm to 4-mm.
DNA extraction, PCR, sequencing, sequence alignment, and phylogenetic analysis
Genomic DNA was extracted from the chela muscle of the specimens fixed in 70% ethanol using a DNeasy Blood & Tissue kit (Qiagen, Hilden, Germany). Partial fragments of the mitochondrial DNA Cytochrome c Oxidase Subunit I (COI) were amplified by polymerase chain reaction (PCR) using genomic DNA as template and a primer set described by Folmer et al. [34]. PCR products were purified for direct sequencing using an ExoSAP-IT (Thermo Fisher Scientific, Waltham, MA, USA) according to the manufacturer’s protocol. Sequencing reactions were performed using HCO2198 primer and a BigDye Terminator v3.1 Cycle Sequencing kit (Thermo Fisher Scientific, Waltham, MA, USA). Sequenced DNA was analyzed with a ABI 3100 automated sequencer (Applied Biosystems, Foster City, CA, USA).
Estimation of divergence time
Phylogenetic analyses were performed by Bayesian inference in MrBayes, version 3.2.6 [28] for a GTR+G+I substitution model. We estimated the time to the most recent common ancestor (TMRCA) of the different phylogenetic groupings, using the arthropod COI substitution rate of 0.0115 per site per million years in BEAST, version 1.8.4 [35,36]. The species tree prior was set to the default option of the Yule process. Markov chain Monte Carlo (MCMC) analyses were run for 1 billion steps, with trees sampled every 1,000 steps and the first 10% of the runs being discarded as burn-ins.
Results
Morphological characteristics of upogebiids
The morphological features of stone upogebiid, Upogebia spinidactylus (dorsal view) revealed that its head (rostrum) is not sharp, but round (Figure 2c). Many hard spines are arranged along both sides of the head. In U. spinidactylus, eight projections (i-viii in Figure 2d) were distinguished along the head edge, and a groove (gv in Figure 2e) was formed inside the continuous hard spines.
Some species possessed a clear depression or space (dp in Figure 2d) between the hard spines. In U. spinidactylus, a depression was found between spines ii and iii, where the tip of the eyestalk is seen from the top. Others, e.g., U. semicircula, lacked this depression, and the eyestalk was not visible from the top.
In all species of Upogebiidae, including mudflat species, the eyestalk (es) was positioned beneath the hard parts of the head (Figure 2d). In U. spinidactylus, a sharp spine was borne along the edge of the exoskeleton around the base of the eyestalk (sp in Figure 2d). Some species (U. saigusai) bore several spines along the carapace edge, but others (U. iriomotensis and U. neogenii) lacked spines there. In stone upogebiids, the tip of the propodus (PR in Figures 2f and 2g) was not degenerated. The tips of the chelae (propodus and dactylus) crossed, and this feature is common to many species of infaunal decapods, including the Axiidae, Laomediidae, and Callianassidae. Upogebiids may also be characterized by a number of long fine setae arranged on each segment of the pereiopods (st in Figures 2f and 2g).
Comparison of morphology among stone upogebiids: the Hirano-Saigusa hypothesis
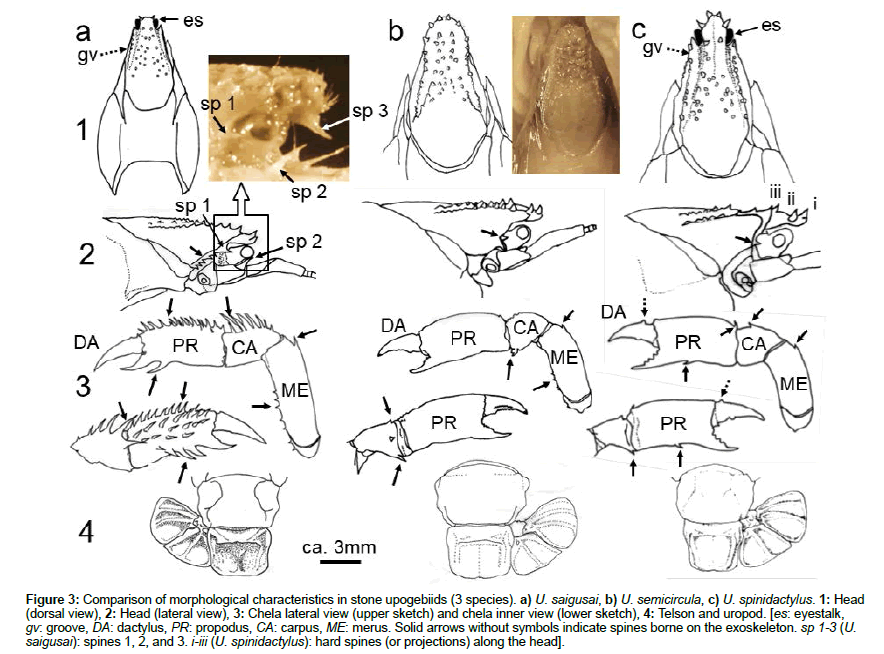
A sketch of three stone upogebiids collected from the shores of Iriomote Island is shown in Figures 3a-3c. Upogebia saigusai (Figure 3a) was most easily discriminated among the stone upogebiids because of the presence of a number of hard spines on its chelae. The head was round, and two hard spines are borne at the tip (Figure 3a-1). The tip of the eyestalk (es) was somewhat visible from the top. In lateral view (Figure 3a-2), a sharp spine (sp1) was borne from the base of the eyestalk. A large, sharp spine (sp 2) was also borne on the base of the antennae. A unique feature was a large spine (sp 3) positioned beneath the tip of the head (photo above Figure 3a-2). Five to six small spines were borne by the carapace edge (solid arrow without symbol in Figure 3a-2). Furthermore, U. saigusai was characterized by a number of small spines borne on the propodus (PR), carpus (CA), and merus (ME), as indicated by the solid arrows in Figure 3a- 3. We could not find characteristics useful for identification on the abdomen and tail of this species (Figure 3a-4).
Figure 3: Comparison of morphological characteristics in stone upogebiids (3 species). a) U. saigusai, b) U. semicircula, c) U. spinidactylus. 1: Head (dorsal view), 2: Head (lateral view), 3: Chela lateral view (upper sketch) and chela inner view (lower sketch), 4: Telson and uropod. [es: eyestalk, gv: groove, DA: dactylus, PR: propodus, CA: carpus, ME: merus. Solid arrows without symbols indicate spines borne on the exoskeleton. sp 1-3 (U. saigusai): spines 1, 2, and 3. i-iii (U. spinidactylus): hard spines (or projections) along the head].
In combination with its habitat, U. semicircula was easily identified morphologically. The head of U. semicircula was round, and several small projections were borne on the head (Figure 3b-1). The eyestalk was not visible from the top. In lateral view, a spine was seen at the base of the eyestalk (solid arrow in Figure 3b-2). Small spines were also borne at edges of the carpus and merus (arrows without symbol in Figure 3b-3). No spine was borne under the propodus (Figure 3b-3). No clear characteristics were found on either the abdomen or tail (Figure 3b-4).
A feature of U. spinidactylus was a deep depression between two spines (ii and iii) at the head, from which the groove (gv) extended (Figures 2e and 3c-1). This depression was also visible in lateral view between spines ii and iii (Figure 3c-2). A spine was borne along the carapace edge under the head (solid arrow in Figure 3c-2). U. spinidactylus was characterized by a small projection borne on the dactylus (dotted arrows in Figure 3c). Several spines were borne on the edge of the pereiopods (solid arrows in Figure 3c-3). No clear characteristics were found on either the abdomen or tail (Figure 3c-4).
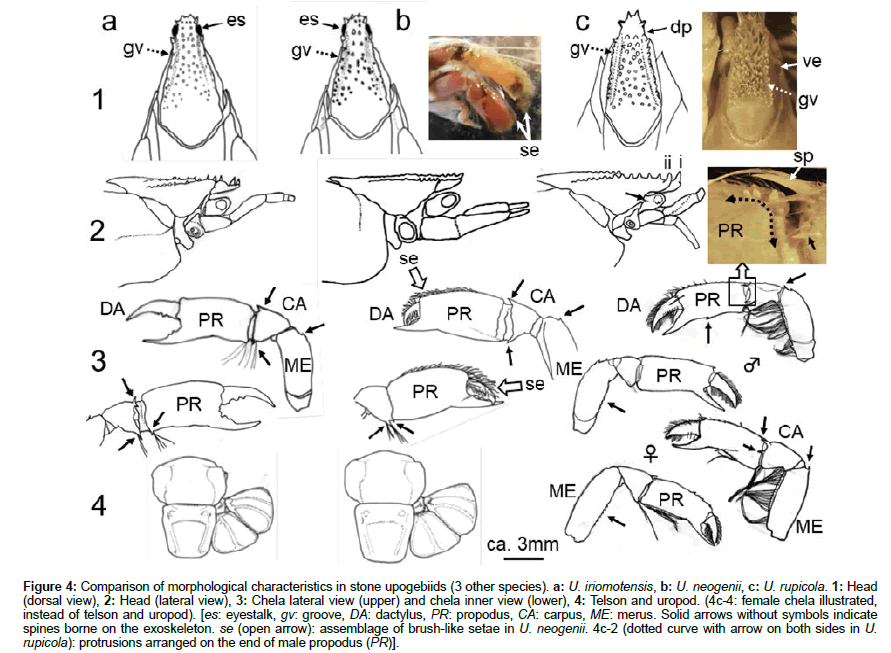
A small depression between the hard spines was also seen on the head of U. iriomotensis (Figure 4a-1). The tip of the eyestalk (e) was visible through the depression from the top. The groove (gv) on both sides of the head was weakly formed. In lateral view, no spine was borne near the base of the eyestalk (Figure 4a-2). Spines were borne at the frontal edge of the carupus, and at the lower end of the propodus (CA and PR in Figure 4a-3). No clear characteristics were found on either the abdomen or tail (Figure 4a-4).
Figure 4: Comparison of morphological characteristics in stone upogebiids (3 other species). a: U. iriomotensis, b: U. neogenii, c: U. rupicola. 1: Head (dorsal view), 2: Head (lateral view), 3: Chela lateral view (upper) and chela inner view (lower), 4: Telson and uropod. (4c-4: female chela illustrated, instead of telson and uropod). [es: eyestalk, gv: groove, DA: dactylus, PR: propodus, CA: carpus, ME: merus. Solid arrows without symbols indicate spines borne on the exoskeleton. se (open arrow): assemblage of brush-like setae in U. neogenii. 4c-2 (dotted curve with arrow on both sides in U. rupicola): protrusions arranged on the end of male propodus (PR)].
Morphology of the carapace (cephalothorax) was much the same between U. iriomotensis (Figure 4a) and U. neogenii (Figure 4b). In U. neogenii, the eyestalk was slightly visible from the top (Figure 4b- 1). No spines were borne at the base of the eyestalk (Figure 4b-2). The position and arrangement of spines were much the same as in U. iriomotensis (compare Figure 4b-3 with Figure 4a-3). The crossing of the dactylus (DA) and propodus (PR) at the tips was also the same as in other stone upogebiids. However, a unique feature of U. neogenii is the presence of long, brush-like setae borne on the dactylus and upper propodus (Figure 4b-1, photo on the right; and Figure 4b-3). Brushlike setae are possibly due to setules borne on the seta.
U. rupicola (Figure 4c) was only distributed in Okinawa Island (Figure 1). Several spines were borne along the top edge of the head. A groove was developed along both sides of the head (Figure 4c-1, left). The head (Figure 4c-1, photo on the right) resembled that of U. semicircula (Figure 3b-1, photo on the right). In lateral view (Figure 4c-2), a spine was borne along the carapace beneath the head (near the base of the eyestalk). Sexual dimorphism appeared a little stronger for this species than in the other five stone upogebiids examined (Figures 4c-3 and 4c-4). Several protrusions were arranged on the outer edge of the male propodus (PR) (photo above Figure 4c-3). A continuous arrangement of these protrusions was not obvious in the female. Many spines were borne by the edges of the propodus (PR), carpus (CA), and merus (ME) in both males and females (Figures 4c-3 and 4c-4).
Sexual dimorphism was not remarkable in these upogebiids. The only outstanding differences were those in the relative length of the merus (ME). ME length of females was comparatively greater than that of males (Figures 4c-3 and 4c-4).
Molecular phylogeny of stone upogebiids
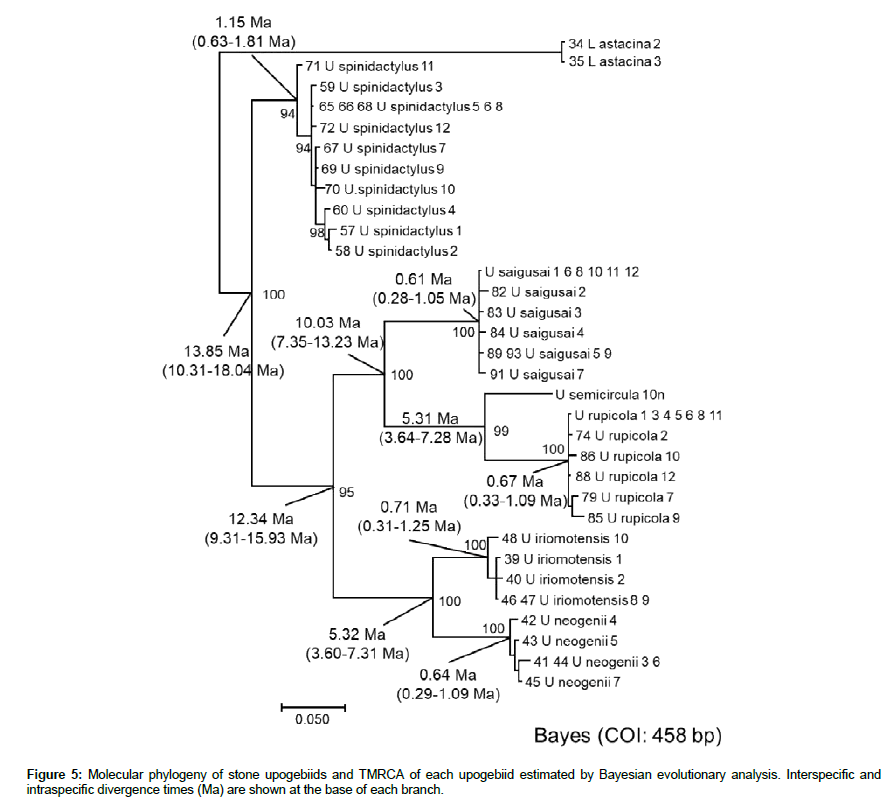
A molecular phylogenetic tree was constructed based on 458 bp of mtDNA (COI) sequences (Figure 5). U. spinidactylus was estimated to have diverged about 13.85 Ma (middle Miocene, Tertiary), suggesting that reproductive isolation may have occurred very early among the stone upogebiids. The other group may have diverged about 12.34 Ma (middle Miocene). U. saigusai was separated from U. semicircula and U. rupicola about 10.03 Ma (late Miocene). U. iriomotensis was separated from U. neogenii about 5.32 Ma (end of Miocene),
As indicated in Figure 5, the TMRCA in U. spinidactylus (1.15 Ma) was the greatest among the six stone upogebiids tested (middle Pleistocene, Quaternary), and it was 0.61 Ma for U. saigusai (late Pleistocene). In U. semcircula, mtDNA (COI) sequence data of 10 individuals were identical, suggesting that U. semicircula settled in the Ryukyu Islands most recently among all the stone upogebiids. The TMRCA of each of the other three upogebiid species (U. rupicola, U. iriomotensis, and U. neogenii) was estimated to be about 0.6-0.7 Ma (late Pleistocene).
Habitat of stone upogebiids in Iriomote and Okinawa Islands
Stone upogebiids and their habitats are summarized in Figure 6. U. saigusai was found on the shores of the partially enclosed bays on Iriomote Island (Figures 6a and 6c). The habitat was exposed by the retreat of sea water during low tide. The burrow possessed two openings and was U-shaped, as observed for all the other upogebiids (Figure 6b). The burrow was dug not only in stones lying on the shore, but also in the sedimentary layer exposed on the shores. The shores of the partially enclosed bays were also inhabited by U. semicircula (Figure 6d), and both U. saigusai and U. semicircula were also collected from the same habitat (Figure 6a).
Figure 6: Stone upogebiids and their habitat. a) Habitat of U. saigusai and U. semicircula (enclosed by a white line). Sedimentary layers and stones on the shore of a bay that is partially enclosed (site b in Figure 7A). Photographs (a,e,g,k) were taken at the time of low water. b) U-shaped burrow of U. saigusai with two openings (open arrow). Scale, 1cm. c) U. saigusai, d) U. semicircula. Scale (c, d, f), 1 cm. e) Major habitat of U. spinidactylus (enclosed by a white line) in an estuarine shore (site f in Figure 7A). g) Habitat of U. iriomotensis and U. neogenii in an estuarine shore (site e in Figure 7A). The stone wall was about 1 m high on the mudflat. h) U. iriomotensis burrowed in a decayed tree half buried in the mud. Scale, 1cm. i) U. neogenii. j) Habitat of U. rupicola on the shore (Okinawa Island). k) U. rupicola burrowing into a siltstone layer. Scale, 1 cm.
U. iriomotensis (Figure 6h) and U. neogenii (Figure 6i) inhabited the burrows dug in stones lying on estuarine shores. Figure 6g indicates a stone wall made on a stream bank around 1940 (80 years ago). Burrows of U. iriomotensis and U. neogenii were found at the bottom of the stone wall. U. iriomotensis burrowed in not only soft stones but also in a decayed tree half-buried in the mudflat (Figure 6h). U. neogenii was collected from soft sandstones lying on the river bed. Soft sandstone strata exposed along the shores of estuaries were also inhabited by U. spinidactylus (Figures 6e and 6f). U. spinidactylus was distributed as far as inside the estuary.
U. rupicola (Figure 6j) was only collected from the shores of Okinawa Island (Figure 1). The habitat (26º 6′N 127 º39′E) was the soft siltstone-strata exposed at the time of low tide (Figure 6k). These strata were 300 m distant from the shore (low tide). The siltstone strata extended 10 km along the shore (figure not shown). A second habitat (26 º40’N 128 º0’E) was found on the shore of a small island (Yagachi Island) 100 km north from the first habitat (figure not shown). Yagachi Island was also positioned on the west side of Okinawa Island.
Topography of Iriomote island and distribution of stone upogebiids
Five species of stone upogebiids were identified on Iriomote Island (Tables 1 and 2). U. saigusai and U. semicircula were found from shores in partially enclosed bays. U. iriomotensis and U. neogenii inhabited the sedimentary layers in estuaries. U. spinidactylus was found as far as inside the estuary. A noteworthy feature of their distributions was that they were limited to the western and northern shores of the island (Figure 7a and Table 3). The seacoast in the south was made up of steep cliffs, and was directly exposed to strong waves because of less developed coral reefs (figure not shown). Southern shores consisted of sand and hard rocks (limestone); so, there was no habitat for upogebiids on the shore. In the east of the island, there are extensive coral rubble flats (reef flats), and there is a distance of more than 1 km between the land and reef crest. Sandstones and sedimentary layers were found in the estuary there, but they were hard to crack by a chisel and hammer.
Figure 7: Distribution of stone upogebiids and grain size analysis. a) Distribution of five stone upogebiids in Iriomote Island. The figure illustrates data collected up to 2007. (U. neogenii had not yet been discriminated from U. iriomotensis.) See Table 3 for the number collected in each habitat. b) Grain size distribution of stones inhabited by upogebiids. Collection site: Urauchi River (site g), Mitara (site h), Funaura Bay (site m), and Hoshidate (site i) in Figure 7A.
Grain size analysis
Stones inhabited by upogebiids were collected from the shores at four sites of Iriomote Island: Mitara, Hoshidate, Urauchi River, and Funaura Bay (sites k, j, g, and m in Figure 7a, respectively). Grain size was estimated by sifting through screens of different mesh sizes (Figure 7b). Stones inhabited by upogebiids were composed of fine grain sizes of 60 μm on average. Stones with this grain size were classified as sandstone; more precisely, they were intermediate between fine-grained (125-62 μm) and ultrafine-grained (< 62 μm).
Discussion
Speciation and morphological relatedness
The time required for speciation (in terms of reproductive isolation) may be highly variable [37]. Three hundred species of cichlids in Lake Victoria may have evolved from one ancestor within the last 12,400 years [38]. At least 24 species of Drosophila may have originated on the ‘big island’ of Hawaii that formed less than 0.8 Ma. Among the snapping shrimps (Alpheus), geological evidence (i.e. the closure of the Panama seaway), combined with estimates of divergence time based on molecular data, suggests that sister species in the Caribbean and eastern Pacific achieved varying degrees of reproductive incompatibility around 3.0 Ma to 3.5 Ma [19].
The Ryukyu Arc (Figure 1) has been isolated from the Eurasian continent since 1.55 Ma [26]. Reproductive isolation between the Ryukyu Arc and the continent produced a number of endemic species in the Ryukyu Islands, including amphibians, reptiles [39], and mammals [40].
Based on morphology of the cephalothorax, Upogebia neogenii (Figure 4b-1) was very similar to U. iriomotensis (Figure 4a-1). U. neogenii was, however, characterized by an assemblage of long, brushlike setae borne on the chelae (Figure 4b-3). In addition, molecular clock data (Figure 5) suggested that U. neogenii had branched off from U. iriomotensis at 5.3 Ma, i.e. at the end of the Miocene period in the Neogene. U. semicircula (Figure 3b-1) may resemble U. rupicola in overall shape (Figure 4c-1), but molecular data (Figure 5) suggested that reproductive isolation had occurred between these two species as long ago as 5.3 Ma (late Miocene).
U. rupicola was described by Komai [18]. U. rupicola may also resemble U. carinicauda, which is widely distributed in the Indo- West Pacific [13]. In U. rupicola, intraspecific genetic divergence may have started 0.67 Ma, possibly in the Ryukyu Arc. If this is true, U. rupicola can certainly be regarded as a species different from U. carinicauda. U. saigusai was first described by Sakai et al. [15]. Morphologically, U. saigusai may be similar to U. snelliusi [17], but intraspecific genetic variation (Figure 5) did not support the possibility of both species dwelling on the same shores. The intraspecific divergence time (possibly in the Ryukyu Arc) was 0.61 Ma (Figure 5). In addition, U. snelliusi was collected from 5-6 m deep layers [13,17], showing a different habitat from that of U. saigusai (Figure 6a). Thus, U. saigusai should be regarded as a species different from U. snelliusi.
Origin of the stones burrowed into by the upogebiids
Stones burrowed into by the upogebiids in Iriomote Island were composed of fine grain sizes (mean=60 μm) (Figure 7B). This grain size is just intermediate between fine-grained sandstone (125-62 μm: sandstone) and ultra-fine grained sandstone (<62 μm: siltstone) [2]. Mean grain size of the stones in Okinawa Island may be a little smaller than that of Iriomote Island (data not shown).
The uppermost layer of the Ryukyu Arc region (Figure 1) consists of paralic sediments with sandstone-rich facies, and contains some coal-measures [29,30]. Depositional facies analysis indicated four depositional cycles in this island, when the region was on the eastern margin of the Eurasian continent [41-43]. Calcareous nannofossil biostratigraphic data suggested that the Iriomote Formation was deposited at the lower shoreface to the inner tidal flat between 14.91 Ma and 13.53 Ma in the middle Miocene [25,44,45].
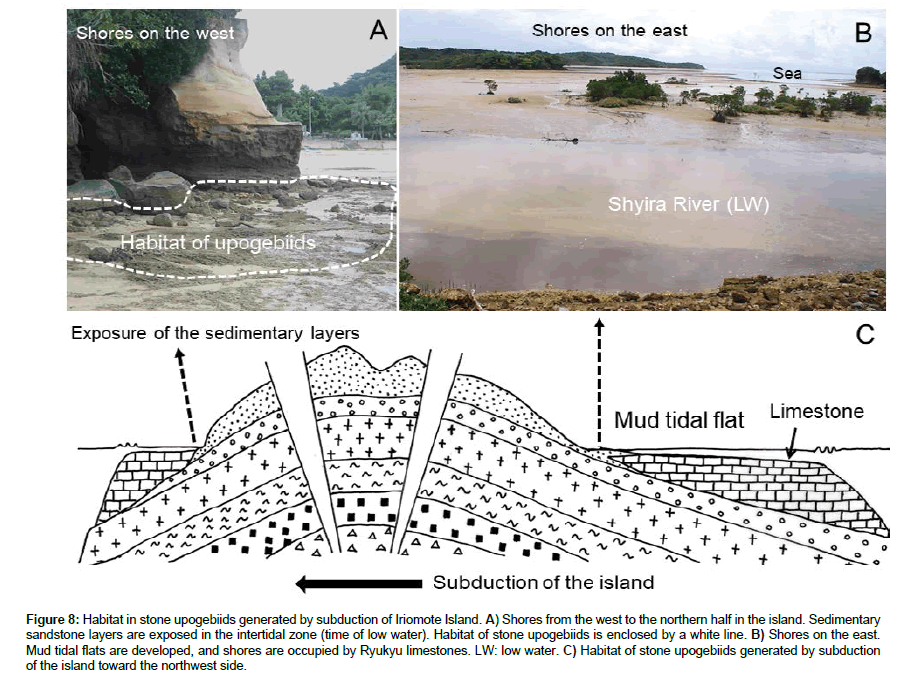
As shown in Figure 8, terrain characteristics are different between the western and eastern sides of Iriomote Island. While stratified sandstone facies are exposed on shores in the west, shores in the east are characterized by sedimentation of mud, sand, and coral rubbles. A number of stones are lying along the shores from the western to the northern parts, and these stones and the depositional layer are burrowed into by the stone upogebiids (Figures 6a,6e,6g and Figure 7A). Tidal flats composed of sedimentation of mud and coral rubbles were not used by the stone upogebiids. Thus, the distribution of stone upogebiids may be strongly influenced by difference in terrain characteristics between the western and eastern shores of the island.
Figure 8: Habitat in stone upogebiids generated by subduction of Iriomote Island. A) Shores from the west to the northern half in the island. Sedimentary sandstone layers are exposed in the intertidal zone (time of low water). Habitat of stone upogebiids is enclosed by a white line. B) Shores on the east. Mud tidal flats are developed, and shores are occupied by Ryukyu limestones. LW: low water. C) Habitat of stone upogebiids generated by subduction of the island toward the northwest side.
Divergence time and settlement of stone upogebiids in the Ryukyu Arc
Global wind patterns and the Coriolis effect produce large, circular systems of surface. In the west Pacific Ocean, the Kuroshiocurrent carries warm water from the equator to the north.
The Ryukyu Arc region (Figure 1) was originally on the eastern margin of the Eurasian continent during the Miocene (24-5 Ma). The Iriomote Formation was formed through depositional cycles between the tidal flat (shallow) and the inner shelf (deep) in the middle Miocene, and the uppermost interval was deposited from 14.91 Ma to 13.53 Ma [25]. Thus, a vast shallow sea (paleo-East China Sea) may have spread on the paleo-Ryukyu Arc region in the middle Miocene, and this region may have appeared above the sea later than 13.53 Ma. The warm, Kuroshio current may have originally flowed along the ‘east’ edge of this region. In the Quaternary, however, with the backarc spreading of the Okinawa Trough [46], the paleo-Ryukyu Arc region was separated from the continental margin, and then started to move northeastwards. The arc transition from the continental to island changed the course of the Kuroshio current from the east to the west of the Ryukyu Arc, and it started flowing the Okinawa Trough about 1.55 Ma (figure 2 in Osozawa et al. [26]). The Ryukyu Arc reached 500 km east about 1.0 Ma, and the coral reefs developed around the islands because of the influence of the Kuroshio current [26].
The back-arc spreading of the Okinawa Trough converted the Ryukyu Arch from a passive to active margin (Figure 8A). The Ryukyu Arch region may now be subducting toward the northwest (Figure 8C). Subduction suggests that depositional strata suitable for burrowing by the stone upogebiids emerge only on the shores on the western side of the islands, and habitats would not be expected on the eastern shores (Figure 8B). The new shores formed along the Eurasian continent would have become the passive margin after 1.55 Ma. Deposition of sand and mud would not produce the habitat of stone upogebiids there either.
Bayesian inference (Figure 5) showed that stone upogebiids diverged 5-15 Ma (second half of the Miocene). Since the Ryukyu Arc had not yet been isolated from the continental margin in the Miocene (24-5 Ma) and Pliocene (5-1.8 Ma), we speculate that each species occurred in many islands near the equator 5-15 Ma. In contrast, intraspecific divergence time differed from 1.15 Ma (Pleistocene) to 0 Ma (present). With enlargement of the Okinawa Trough (back arc basin-rift valley), the Ryukyu Arc was separated from the continental margin, and the warm current (Kuroshio) isolated the Ryukyu Arc at 1.55 Ma (Pleistocene). The Ryukyu Arc has been moving towards the northwestward after that, and sedimentary strata for burrowing may have been exposed on the shorefront, i.e., western shores of the island.
Stone upogebiids would have settled in the Ryukyu Arc at different times (1.15-0 Ma) since 1.55 Ma. Intraspecific genetic variation of U. spinidactylus (Figures 6e and 6f) indicated 1.15 Ma (Figure 5), suggesting that U. spinidactylus settled in the Ryukyu Arc shortly after isolation of the Ryukyu Arc from the continent. The beginning of intraspecific genetic variation in the other upogebiids (U. saigusai, U. rupicola, U. iriomotensis, and U. neogenii) was between 0.61 Ma (U. saigusai) and 0.71Ma (U. iriomotensis). These upogebiids may have settled in the Ryukyu Arc during the second half of the Pleistocene, after which genetic variation in them appeared. No intraspecific genetic variation was shown in U. semicircula (Figure 5). U. semicircula may have settled in the Ryukyu Arc most recently among the six stone upogebiids examined.
Acknowledgments
Collection of upogebiids and other decapod crustaceans was carried out in Iriomote Laboratory (Iriomote Island) and Sesoko Laboratory (Okinawa Island) of Ryukyu University. We thank Editage (www.editage.jp) for English language editing.
References
- Nybakken JW (2001) Marine Ecology. (5th edtn), Benjamin Cummings, San Francisco, USA.
- Wentworth CK (1922) A scale of grade and class terms for clastic sediments. J Geol 30: 377-392.
- Castro P, Huber ME (2005) Marine Biology. (5th edtn), McGraw-Hill, Boston, USA.
- Yamasaki M, Nanri T, Taguchi S, Takada Y, Saigusa M (2010) Latitudinal and local variations of the life history characteristics of the thalassinidean decapod, Upogebia yokoyai: a hypothesis based on trophic conditions. Est Coast Shelf Sci 87: 346-356.
- Kneer D, Asmus H, Vonk JA (2008) Seagrass as the main food source of Neaxius acanthus (Thalassinidea: Strahlaxiidae), its burrow associates, and of Corallianassa coutierei (Thalassinidea: Callianassidae). Est Coast Shelf Sci 79: 620-630.
- Dworschak PC, Ott JA (1993) Decapod burrows in mangrove-channel and back-reef environments at the Atlantic Barrier Reef, Belize. Ichnos 2: 277-290.
- Nickell LA, Atkinson RJA (1995) Functional morphology of burrows and trophic modes of three thalassinedean shrimp species, and a new approach to the classification of thalassinidean burrow morphology. Mar Ecol Progr Ser 128: 181-197.
- Dworschak PC (2000) Global diversity in the Thalassinidea (Decapoda). J Crust Biol 20: 238-245.
- Curran HA, Martin AJ (2003) Complex decapod burrows and ecological relationships in modern and Pleistocene intertidal carbonate environments, San Salvador Island, Bahamas. Palaeogeogr Palaeoclimatl Palaeoecol 192: 229-245.
- Yanin BT, Baraboshkin EY (2013) Thalassinoides burrows (Decapoda dwelling structures) in Lower Cretaceous sections of southwestern and central Crimea. Strat Geogl Corr (Stratigraphy and Geological Correlation) 21: 280-290.
- Ngoc-Ho N (1990) Nine Indo-Pacific species of Upogebia Leach (Crustacea: Thalassinidea: Upogebiidae). J Nat Hist 24: 965-985.
- Williams AB (1993) Mud shrimps, Upogebiidae, from the western Atlantic (Crustacea: Decapoda: Thalassinidea). Smith Contr Zool 544: 1-77.
- Sakai K (2006) Upogebiidae of the world (Decapoda, Thalassinidea): Crustaceana Monographs 6. Brill, Leiden, Netherlands.
- Itani G (2004) Distribution of intertidal upogebiid shrimp (Crustacea: Decapoda: Thalassinidea) in Japan. Contr Biol Lab Kyoto Univ 29: 383-399.
- Sakai K, Hirano Y, Saigusa M (2004) A new record of Upogebia snelliusi Ngoc-Ho, 1989 (Thalassinoidea, Upogebiidae) from Iriomote Island, Ryukyus, Japan. Crustaceana 77: 741-749.
- Sakai K, Hirano Y (2006) Three new species of Upogebiidae (Thalassinidea) from Iriomote Island, Ryukyus, Japan. Crustaceana 79: 977-991.
- Ngoc-Ho N (1989) Description of three new species of the family Upogebiidae (Crustacea, Thalassinidea). Bull Mus natn Hist Nat Paris 11: 865-878.
- Komai T (2005) A new species of Upogebia (Crustacea: Decapoda: Thalassinidea: Upogebiidae) from Okinawa Island, Ryukyu islands. Species Divers 10: 259-268.
- Knowlton N, Weigt LA, Solórzano LA, Mills DK, Bermingham E (1993) Divergence in proteins, mitochondrial DNA, and reproductive compatibility across the Isthmus of Panama. Science 260: 1629-1631.
- Brower AVZ (1994) Rapid morphological radiation and convergence among races of the butterfly Heliconius erato inferred from patterns of mitochondrial DNA evolution. Proc Natl Acad Sci USA 91: 6491-6495.
- Lin SM, Chen CA, Lue KY (2002) Molecular phylogeny and biogeography of the grass lizards genus Takydromus (Reptilia: Lacertidae) of East Asia. Mol Phylogenet Evol 22: 276-288.
- Tsang LM, Ma KY, Ahyong ST, Chan TY, Chu KH (2008) Phylogeny of Decapoda using two nuclear protein-coding genes: origin and evolution of the Reptantia. Mol Phylogenet Evol 48: 359-368.
- Robles R, Tudge CC, Dworschak PC, Poore GCB, Felder DL (2009) Molecular phylogeny of the Thalassinidea based on nuclear and mitochondrial genes: Decapod Crustacean Phylogenetics. CRC Press, Boca Raton, Florida, USA.
- Lin FJ, Liu Y, Sha Z, Tsang LM, Chu KH, et al. (2012) Evolution and phylogeny of the mud shrimps (Crustacea: Decapoda) revealed from complete mitochondrial genomes. BMC Genomics 13: 631-645.
- Kotake N, Kameo K, Nara M (2013) Geologic age depositional environments of the uppermost part of the Middle Miocene Iriomote Formation in the Iriomote-jima Island, Okinawa, southwest Japan. J Geol Soc Jpn 119: 701-713.
- Osozawa S, Shinjo R, Armid A, Watanabe Y, Horiguchi T, et al. (2012) Palaeogeographic reconstruction of the 1.55 Ma synchronous isolation of the Ryukyu Islands, Japan, and Taiwan and inflow of the Kuroshio warm current. Int Geol Rev 54: 1369-1388.
- Kimura M (2000) Paleogeography of the Ryukyu Islands. Tropics 10: 5-24.
- Huelsenbeck JP, Ronquist F (2001) MRBAYES: Bayesian inference of phylogenetic trees. Bioinformatics 17: 754-755.
- Saito Y, Chiba T, Miyagi H (1973) Geology of Iriomote-jima, Ryukyu Islands. Mem Natl Sci Mus 6: 9-22.
- Park JO, Tokuyama H, Shinohara M, Suyehiro K, Taira A (1998) Seismic record of tectonic evolution and backarc rifting in the southern Ryukyu island arc system. Tectonophysics 294: 21-42.
- Ngoc-Ho N (1994) Notes on some Indo-Pacific species of Upogebiidae with description of four new species (Crustacea: Thalassinidea). Mem Queensland Mus 85: 193-216.
- Sakai H, Takeda M (1995) New records of two species of decapod crustaceans from Amami-Oshima island, the northern Ryukyu Islands, Japan. Bull Natl Sci Mus (A: Zool) 21: 203-210.
- Hirano Y, Saigusa M (2008) Description of the male of Upogebia miyakei from Ryukyu Islands, Japan (Decapoda: Thalassinidea: Upogebiidae). J Mar Biol Assoc UK 88: 125-131.
- Folmer O, Black M, Hoeh W, Lutz R, Vrijenhoek R (1994) DNA primers for amplification of mitochondrial cytochrome c oxidase subunit I from diverse metazoan invertebrates. Mol Mar Biol Biotech 3: 294-299.
- Drummond AJ, Ho SYW, Phillips MJ, Rambaut A (2006) Relaxed phylogenetics and dating with confidence. PLOS Biol 4: 699-710.
- Drummond AJ, Rambaut A (2007) BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol Biol 7: 214.
- Futuyma DJ (1998) Evolutionary Biology. (3rd edtn), Sinauer Associates, Massachusetts, USA.
- Johnson TC, Scholz CA, Talbot MR, Kelts K, Ricketts RD, et al. (1996) Late Pleistocene desiccation of Lake Victoria and rapid evolution of cichlid fishes. Science 273: 1091-1093.
- Ota H (1998) Geographic patterns of endemism and speciation in amphibians and reptiles of the Ryukyu Archipelago, Japan, with special reference to their paleogeographical implications. Res Popul Ecol 40: 189-204.
- Motokawa M (2000) Biogeography of Living Mammals in the Ryukyu Islands. Tropics 10: 63-71.
- Dalrymple RW (1992) Tidal depositional systems: Facies models: response to sea level change. Geol Assoc Canada 195: 195-218.
- Einsele G (2000) Sedimentary Basins: Evolution, Facies, and Sediment Budget. (2nd edtn), Springer-Verlag, Berlin, Germany.
- Yang BC, Dalrymple RW, Chun SS (2005) Sedimentation on a wave-dominated, open-coast tidal flat, south-western Korea: summer tidal flat-winter shoreface. Sedimentol 52: 235-252.
- Lourens L, Hilgen F, Shackleton NJ, Lasker J, Wilson D (2004) The Neogene Period: A Geologic Time Scale 2004. Cambridge Univ Press, Cambridge, UK.
- Saitoh Y, Matsuda F (2004) Miocene sandstone of ‘continental’ origin on Iriomote Island, southwest Ryukyu Arc., Eastern Asia. J Asian Earth Sci 24: 137-144.
- Maruyama S, Isozaki Y, Kimura G, Terabayashi M (1997) Paleogeographic maps of the Japanese Islands: plate tectonic synthesis from 750 Ma to the present. Island Arc 6: 121-142.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi