Research Article, Rrpy Vol: 1 Issue: 4
Effects of a Single Dose of Methylphenidate on Cognition in Adults with ADHD and Predictive Therapeutic Response
Ertle S*, Vanoli L, Erb A and Duval FCentre Hospitalier, Rouffach, France
*Corresponding Author : S. Ertle
Centre Hospitalier, Rouffach, France
Tel: +33 3 89 78 70 18
Fax: +33 3 89 78 74 30
E-mail: s.ertle@ch-rouffach.fr
Received Date: October 09, 2020; Accepted Date: October 26, 2020; Published Date: November 02, 2020
Citation: Ertle S, Vanoli L, Erb A, Duval F (2020) Effects of a Single Dose of Methylphenidate on Cognition in Adults with ADHD and Predictive Therapeutic Response. rrpy 1:4.
Abstract
Attention Deficit Hyperactivity Disorder (ADHD) affects about 60 % of adults who suffered from ADHD in childhood. These patients complain about organization and planning difficulties, procrastination, and thoughtlessness or memory troubles. Methylphenidate (MPH) is a common off-label treatment in European countries. The aim of this study was to assess the neuropsychological effects of a single dose of MPH on different attention components and on executive functions by using a computerized Attention Assessment Battery. Clinical and self-rating visual analogical scales were used. We also analysed if acute effects of a test dose of MPH on cognitive parameters may predict long-term outcomes with MPH medication. One hundred and one diagnosed adult patients were enrolled into this study. Neuropsychological evaluations were performed at baseline, after a single dose (“MPH Test”) and 6 months after chronic MPH treatment with an adequate dose. Compared with baseline, a single dose of MPH resulted in a statistically significant improvement in working memory performance, visual scanning, phasic and selective attention, sustained attention as well as executive functions in terms of number of mistakes and omissions. Reaction times also significantly decreased in most of these tasks. Statistically significant effects were also observed regarding the subjective assessments. These improvements on cognitive functions and subjective feelings were still observed after 6 months of MPH treatment. Positive impacts on everyday life were noticed regarding the clinical scales compared to the baseline. Our results suggest that the “MPH test” would be useful in predicting subsequent responses to methylphenidate in ADHD adult patients
Keywords: Psychology, Psychiatry, Neuroscience
Introduction
Approximately 4% to 5% of the adult population is impaired by Attention Deficit Hyperactivity Disorder (ADHD). It is largely recognized that ADHD symptoms persist into adulthood (Barkley 1998, Faraone et al. 2005) and are detected in about 60% of the adults who suffered from ADHD in childhood. Symptoms in adult patients are quite different from those observed in children and the most remarkable symptoms are organization and planning difficulties, procrastination, thoughtlessness, memory troubles, emotional disorders and other functional impairments[1-3]. Adult hyperactivity becomes less visible [4] than in childhood. Impaired neurocognitive performances have been described in adult ADHD patients [5,6]. Different studies have shown disturbances of a variety of cognitive functions such as executive functions (for instance working memory, inhibition and interference control, planning), memory (verbal learning) and attentional components including alertness, sustained attention, selective attention, divided attention and distractibility [7 -13]. These neurocognitive impairments have been related mainly to frontostriatal neuronal network dysfunction [14].
In child and adolescent psychiatry, methylphenidate (MPH) is the most common worldwide licensed treatment of ADHD. In different countries it is also licensed for the treatment in adult patients but in France it is still considered as an off-label treatment. Few studies regarding the cognitive effects of MPH on adult ADHD patients have been published during the past years compared to the literature about children. Aron et al have shown a significant improvement of response inhibition in adults after MPH intake [11]. Boonstra et al. have also observed a positive effect of MPH compared to placebo on inhibition capacities at a continuous performance test [15]. Turner et al. and later have assessed the effect of a single dose of MPH on a battery of neurocognitive tests in adult ADHD patients. Significant improvements on spatial working memory, sustained attention, selective and divided attention, flexibility and alertness were observed. Kurscheidt et al have administered a test dose of 10 mg of MPH prior to a continuous medication in ADHD adult patients and measured MPH effects using a neuropsychological test battery [16].
The first aim of our study was to assess the neuropsychological effects of a single dose of MPH (10 mg per os) on different attention components and executive functions by using a computerized test battery. We also analysed if acute effects of a test dose of MPH on cognitive parameters may predict long-term outcomes with MPH medication.
Material and Methods
Procedure
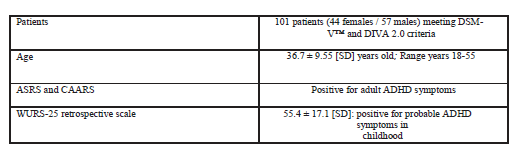
One hundred and one drug naive patients were clinically diagnosed with adult ADHD in our day hospital. Psychiatrists made the clinical diagnosis of ADHD by means of the Diagnostic and Statistical Manual of Mental Disorders (DSM V™) and the Diagnostic Interview For Adult ADHD criteria [17].Clinicalscales suchastheAdult Self- Report Scale (ASRS 1.1, Composite International Diagnostic Interview, and OMS), the Conners’ Adult ADHD Rating Scales, and the Wender-Utah Rating Scale [18-20] were also used. Major comorbidities, psychiatric disorders (Axis 1, DSM V™), medical conditionslikely to be a contraindication for the study was considered as exclusion criteria.
The procedure consisted in a 3-steps study course. A neuropsychological test battery was performed at baseline (T1) and 60 minutes after intake of 10 mg MPH per os 2 weeks after the baseline in order to avoid possible training effects (T2 called “MPH Test”). Our patients were subsequently medicated with an adequate dose of MPH (46.7 ± 23.8 mg/d) and followed-up over a period of 6 months. A neuropsychological retest was administrated after 6 months (T3).
• The Adult Self-Report Scale ASRS v1.1 consists in a symptom checklist that takes up the 18 criteria of the DSM IV. Six of the eighteen questions were found to be the most predictive of symptoms consistent with ADHD [21-23].
• The 66 items version of the self-report CAARS contains 8 subscales. It consists in different scales that assess a crosssection of ADHD-related symptoms and behavioural troubles, DSM-V symptoms (Inattentive symptoms, Hyperactivity-Impulsivity symptoms and Total ADHD symptoms) and an ADHD Index [24].
• The Wender-Utah Rating Scale, WURS-25 items version is an aid in the retrospective diagnosis of childhood attention deficit hyperactivity disorder [19].
• Concerning the neuropsychological investigations, we chose to focus the tests on attention and executive functions. Eight subtests of the Tests of Attentional Performance (TAP) 2.3 battery were administered. The most relevant parameters were selected for each task and reported in a profile [25].
Phasic alertness: The purpose of this test is to measure the reaction time of a simple or cued task, triggered by a visual stimulus and an acoustic index. The phasic arousal represents the capacity to raise the attention threshold while waiting for a stimulus.
This technique allows to measure:
• Simple reaction time, which is the best indicator for a general slowdown of reactions.
• Response time in phasic attention by calculating the average reaction time difference with and without acoustic signal.
Bimodal divided attention: this test combines 2 simultaneous tasks (visual and acoustic). The visual task consists in spotting the crosses composing a little square among all the crosses appearing randomly on the screen. The acoustic test consists in detecting an irregular sequence in a regular sequence of “beeps”.
Flexibility: This test enables to measure the capacity of attention shifting because of the necessity to alternate between two classes of critical stimuli (verbal condition: letters and numbers). For each condition, the stimuli are presented simultaneously and in a random order on the right or the left of a fixation point. Letter and then number alternately represent the critical stimuli. The subject has to press on corresponding key on side where the stimulus appears.
Incompatibility: The purpose of this test is to evidence the interference sensitivity by means of a task based on the incompatibility between the stimulus and the response. Arrows pointing sometimes to the left, sometimes to the right, appear on the right or left of a fixation point. The subject has to react to the direction indicated by the arrow: he must press with the right hand when the arrow indicates the right or with his left hand when the arrow indicates the left, and this no matter on which side of the screen where the arrow appears. There is compatibility when the side of the arrow presentation (on the screen) correspond to the hand that has to press (therefore the arrow, whichever the direction it points to, and the hand are on the same side). There is incompatibility when the arrow and the hand occupy opposite positions.
Visual scanning: This test assesses the visual scanning capacities. It consists in detecting a given critical stimulus (a square which is open at the top) among other squares (open on other sides). The squares are arranged in a matrix of five columns and five lines. The subject has to indicate if the critical stimulus is present in the matrix or not by pressing the corresponding key.
Go/no go: This test has been conceived in order to examine the capacity of the subject to inhibit inappropriate reactions (stimulus which is not pertinent) and also to measure the quickness to select the critical stimulus.
Working memory: The working memory requires a continuous monitoring of the information flow within the short-term memory. A sequence of numbers is presented to the subject on the monitor. The subject is required to determine whether each number corresponds with the last but one number.
Sustained attention: this test assesses the capacity to maintain concentration during a task lasting 15 minutes. Subjects have to press on the key as quickly as possible when two consecutive stimuli are the same colour or the same shape.
• Visual Analogic Scales (Bond and Lader, 1974): These scales assess subjective feelings. Subject has to mark a bar on 16 bipolar (two opposite adjectives) lines corresponding to how he is feeling right now. Three factors are calculated: alertness, contentedness (well-being) and calmness.
Statistical analysis
Statistical computations were performed using the Statistica software (TIBCO 13.5.017). Within-subject differences (ie. T1 vs. T2; T1 vs T3 and T2 vs. T3) were evaluated with Repeated Measures Analysis of Variance ANOVA. For statistical analysis, an alpha level of 0.01 was applied according to Bonferroni-corrected p value. Furthermore, effect sizes, which refer to the importance of the effects, were computed according to Cohen’s (1988) benchmarks for interpreting the effect size index d: negligible effects (d<0.20), small effects (0.20<d<0.50), medium effects (0.50<d<0.80) and large effects (d>0.80). Finally, linear regression equations were calculated in order to predict the differences observed on cognitive and clinical evaluations between baseline and after 6 months of MPH treatment (T1- T3) based on the differences evidenced between baseline and the MHP test (T1-T2). The adjusted correlation coefficient, denoted by R2, is a measure of the strength of the linear relationship between two variables. The most common interpretation of r-squared is how well the regression model fits the observed data. For example, an rsquared of 0.60 reveals that 60% of the data fit the regression model. Generally, a higher r-squared indicates a better fit for the model. R2 value between 0.7 and 1.0 indicate a strong positive linear relationship. R2 value between 0.3 and 0.7 indicate a moderate positive linear relationship and R2 value lower than 0.30 indicate a weak positive linear relationship.
Results
Acute effects of MPH on cognition: comparisons between neurocognitive tasks results at baseline and after a single 10 mg dose of MPH
MPH induced significant improvements using repeated measures analysis of variance in working memory, visual scanning, phasic and selective attention, sustained attention as well as executive functions in terms of number of errors and omissions. Reaction times decreased in most of these tasks. We also observed an improvement concerning the homogeneity of the performances (Table 1).
Statistically significant improvements on cognitive performances were observed comparing T1 “Baseline” vs. “MPH Test”. For the Phasic Arousal test, a significant speeding enhancement in psychomotor reactions was observed whatever the condition, with (F (1,99)=30.1; p<0.01) or without (F(1,99)=28.1; p<0.01) an acoustic warning. A better reaction time homogeneity according to the standard deviations with (F (1, 99)=39; p<0.01) or without (F(1,99)=39.4; p<0.01) an acoustic warning as well as less anticipatory responses (F(1,99)=20.3; p<0.01) were noticed. Regarding the Divided Attention test, we observed significantly less omissions errors [F (1, 99)=37.8; p<0.01) and decreased variability of reaction time both for auditory (F(1,99)=31.3; p<0.01) and visual (F(1,99)=19.2; p<0.01) stimuli. Reaction times (F(1,99)=123.5; p<0.01) were significantly improved and stabilized (F(1,99)=87.6; p<0.01) on Flexibility test and patients committed less errors (F(1,99)=31.4; p<0.01) at this task. Same significant differences were observed on Incompatibility test. Patients made fewer mistakes, both for compatibility condition (F(1,99)=18.1; p<0.01) and incompatibility condition (F(1,99)=37.4; p<0.01). Speed of performance was also ameliorated in both conditions (respectively, F(1,99)=34.9; p<0.01 and F(1,99)=55.2; p<0.01) and standard deviations were more homogeneous (respectively, F(1,99)=46.3; p<0.01 and F(1,99)=55.3; p<0.01). Concerning Visual Scanning Test, results showed no significant improvements comparing the speed (F(1,99)=4.16; p=0.043) whereas stability (F(1,99)=32.6; p<0.01) of scanning, the number of errors (F(1,99)=12.6; p<0.01) and more particularly the number of omissions (F(1,99)=105.4; p<0.01) were significantly improved between baseline and the “MPH test”. Comparison between baseline and “MPH Test” on Go no Go Test revealed a significant difference in the number of errors [F (1, 99)=21.1; p<0.01], reaction time (F(1,99)=31.2; p<0.01) and variability of reaction time (F(1,99)=46.8; p<0.01). The examination of Working Memory Test showed significant improvements regarding the number of mistakes (F(1,99)=34.1; p<0.01) and omissions (F(1,99)=39.8; p<0.01) between baseline and the “MPH test”. Finally, we noticed significant differences regarding the number of errors (F (1, 99) =33.7; p<0.01) and omissions (F(1,99)=155.3; p<0.01) as well as decreased variability of the reaction times (F(1,99)=103.8; p<0.01) on the Sustained Attention Test.
Chronic effects of MPH on cognition: effects of MPH after 6 months of treatment
These improvements on cognitive functions were still observed after 6 months of MPH treatment. At follow-up after 6 months (T3), test performances were comparable to those observed during the “MPH test” (T2). Neurocognitive tasks revealed no significant differences between T2 and T3 on errors and omissions, reaction times and variability of reaction times (Table 1).
| T1 vs. T2 | T2 vs. T3 | T3 vs. T1 | |
|---|---|---|---|
| Phasic Arousal | |||
| Reaction time without signal mean | 0.59 | 0.3 | 0.74 |
| Variability of reaction time without signal | 0.66 | 0.27 | 0.85 |
| Variability of reaction time with signal | 0.72 | 0.15 | 0.83 |
| Anticipations | 0.56 | 0.16 | 0.66 |
| Divided attention | |||
| Variability of reaction time visual modality | 0.58 | 0.05 | 0.59 |
| Variability of reaction time auditory modality mean | 0.73 | 0.07 | 0.75 |
| Omissions | 0.77 | 0.08 | 0.81 |
| Flexibility | |||
| Reaction time mean | 0.87 | 0.06 | 0.83 |
| Variability of reaction time | 0.8 | 0 | 0.83 |
| Number of errors | 0.56 | 0.25 | 0.87 |
| Incompatibility | |||
| Reaction time compatible condition mean | 0.64 | 0.04 | 0.65 |
| Reaction time incompatible condition mean | 0.74 | 0.01 | 0.71 |
| Variability reaction time compatible condition | 0.8 | 0.14 | 0.87 |
| Variability reaction time incompatible condition | 0.8 | 0.09 | 0.72 |
| Number of errors compatible condition | 0.57 | 0 | 0.56 |
| Number of errors incompatible condition | 0.82 | 0 | 0.78 |
| Visual scanning | |||
| Reaction time mean | 0.26 | 0.11 | 0.34 |
| Variability of reaction time | 0.62 | 0.03 | 0.62 |
| Number of errors | 0.38 | 0.28 | 0.19 |
| Number of omissions | 1.14 | 0.14 | 1.02 |
| Go/No Go | |||
| Reaction time mean | 0.6 | 0.04 | 0.56 |
| Variability of reaction time | 0.73 | 0.17 | 0.62 |
| Number of errors | 0.49 | 0.15 | 0.41 |
| Working memory | |||
| Number of errors | 0.65 | 0.17 | 0.85 |
| Number of omissions | 0.8 | 0 | 0.75 |
| Sustained attention | |||
| Variability of reaction time | 1.08 | 0.01 | 0.85 |
| Number of errors | 0.63 | 0.08 | 0.67 |
| Number of omissions | 1.42 | 0.17 | 1.59 |
Table 1: Neurocognitive tasks effect sizes (effect size index d): T1 vs T2, T2 vs .T3 and T3 vs. T1.
Significant differences observed on reaction time on Phasic arousal test without acoustic warning (F(1,99)=10.4; p=0.001) and variability of reaction times (F(1,99)=8; p=0.005) were linked to an improvement of the performances between T2 and T3. However, no significant differences were evidenced concerning the speed of performances (F(1,99)=2.07; p=0.152), variability of the reaction times (F(1,99)=2.17; p=0.143) with an acoustic warning and the number of anticipated responses (F(1,99)=3.08; p=0.082). Results on Divided Attention test were comparable between T2 and T3 for the variability of reaction time both for auditory (F(1,99)=0.30; p=0.580) and visual stimuli (F(1,99)=0.19; p=0.659) as well as the number of omissions errors (F(1,99)=0.30; p=0.583). Comparisons between T2 and T3 showed no significant differences regarding the Flexibility test (reaction time: F(1,99)=0.65; p=0.420; variability of reaction time: F(1,99)=0.00; p=0.977; number of errors: F(1,99)=4.11; p=0.045). Similar results were observed on Incompatibility test for both compatible condition (reaction time: F(1,99)=0.32; p=0.570; variability of reaction time: F(1,99)=2.22; p=0.138; number of errors: F(1,99)=0.01; p=0.914) and incompatible condition (reaction time: F(1,99)=0.00; p=0.975; variability of reaction time: F(1,99)=0.79; p=0.374; number of errors: F(1,99)=0.02; p=0.903). Between T2 and T3, performances on Visual Scanning did not differ significantly (reaction time: F(1,99)=1.75; p=0.188, variability of reaction time: F(1,99)=0.10; p=0.742, number of errors: F(1,99)=3.95 p=0.049, number of omissions: F(1,99)=1.47; p=0.227). No significant differences were evidenced concerning the speed of performances (F(1,99)=0.24; p=0.619), variability of the reaction times (F(1,99)=2.45; p=0.120) and the number of errors (F(1,99)=2.43; p=0.121) on Go No Go test. The number of errors (F(1,99)=2.05; p=0.154) and omissions (F(1,99)=0.09; p=0.757) of stimuli on Working Memory test were comparable between T2 and T3. Comparison of Sustained Attention between T2 and T3 revealed no significant differences regarding the variability of reaction time (F(1,99)=0.00; p=0.925), the number of errors (F(1,99)=0.64; p=0.425) and the number of omissions (F(1,99)=2.99; p=0.086).
Comparisons between baseline and chronic effects after 6 months of adapted MPH treatment on cognition tasks
Significant differences between baseline (T1) and follow-up after 6 months (T3) were evidenced on errors and omissions, reaction times and variability of reaction times (Table 1).
For the Phasic Arousal test, a significant enhancement in reaction times was observed whatever the condition, with (F (1,99)=33.2; p<0.01) or without (F(1,99)=34.3; p<0.01) an acoustic warning. A better reaction time variability with (F (1,99)=40.9; p<0.01) or without (F(1,99)=48.8; p<0.01) an acoustic warning as well as less anticipatory responses (F(1,99)=34.6; p<0.01) were evidenced. Patients realized significantly less omissions errors (F (1,99)=40; p<0.01) at T3 compared to T1 and showed decreased variability of reaction time both for auditory (F(1,99)=31.4; p<0.01) and visual (F(1,99)=24.5; p<0.01) stimuli. Reaction times (F(1,99)=84.2; p<0.01) were significantly improved and stabilized (F(1,99)=60.1; p<0.01) on Flexibility test and patients committed less errors (F(1,99)=53.5; p<0.01). They also made fewer errors, both for compatibility condition (F(1,99)=15.6; p<0.01) and incompatibility condition (F(1,99)=34.7; p<0.01). Speed of performance was also enhanced in both conditions (respectively, F(1,99)=32.3; p<0.01 and F(1,99)=43.4; p<0.01) and standard deviations were more homogeneous (respectively, F(1,99)=61.8; p<0.01 and F(1,99)=45.8; p<0.01) at T3 compared to T1. Results on Visual Scanning Test indicated significant improvements comparing reaction times (F(1,99)=6.84; p=0.01), variability of reaction times (F(1,99)=35.2; p<0.01), the number of omissions (F(1,99)=80.6; p<0.01) and the number of mistakes (F(1,99)=3.54; p=0.062). Comparison between T1 and T3 on Go no Go Test showed significant differences concerning the number of errors (F(1,99)=16.4; p<0.01), reaction time (F(1,99)=34.1; p<0.01) and variability of reaction time (F(1,99)=25.2; p<0.01). Patients committed significantly less mistakes (F(1,99)=42.1; p<0.01) and omissions (F(1,99)=41.9; p<0.01) on Working Memory test. Performances on Sustained Attention test evidenced marked differences regarding the number of errors (F(1,99)=37.8; p<0.01) and omissions (F(1,99)=167.5; p<0.01) as well as decreased variability of the reaction times (F(1,99)=39.5; p<0.01).
Acute and Chronic effects of MPH on clinical evaluations
Significant effects were noticed regarding the subjective assessments. Patients felt more alert (F (1, 99)=170.8; p<0.01) more satisfied (F (1, 99)=40.7; p<0.01) and calmer (F (1, 99)=11.7; p<0.01) comparing T1 vs. T2. These differences were still observed 6 months after adapted MPH treatment compared to T1 baseline for each factor (respectively, F(1,99)=95.4; p<0.01; F(1,99)=55.3; p<0.01; F(1,99)=19.1; p<0.01). No statistical significant differences were evidenced on alertness (F(1,99)=4.12; p=0.145), contentedness (F(1,99)=1.33; p=0.251) and calmness (F(1,99)=0.61; p=0.436) when comparing T2 and T3. The ASRS administered at T1 and T3 was statistically improved 6 months after MPH medication (F(1,99)=159.4; p<0.01). Positive impacts on everyday life were also noticed regarding the CAARS scales. Patients felt less inattentive (F(1,99)=74.8; p<0.01), hyperactive (F(1,99)=33.6; p<0.01), impulsive (F(1,99)=61.1; p<0.01) and had fewer problems with self-concept (F(1,99)=54.9; p<0.01). These improvements were noticed according to the DSM-V (Inattention: F(1,99)=80.4; p<0.01); hyperactivity/impulsivity: F(1,99)=57.7; p<0.01 and Total symptoms: F(1,99)=101.8; p<0.01) and the ADHD Index (F(1,99)=65.5; p<0.01) (Table 2).
| T1 vs. T2 | T2 vs. T3 | T3 vs. T1 | |
|---|---|---|---|
| Visual Analogic Scales (VAS) | |||
| Alertness | 1.63 | 0.18 | 1.28 |
| Contentedness (well-being) | 0.81 | 0.09 | 0.86 |
| Calmness | 0.47 | 0.07 | 0.52 |
| Adult Self-Report Scale (ASRS) | - | - | 1.67 |
| Conner’s Self Report Scale (CAARS) | |||
| A- Inattention/Memory Problems | - | - | 1.22 |
| B- Hyperactivity/Restlessness | - | - | 0.74 |
| C- Impulsivity/Emotional Lability | - | - | 1.03 |
| D- Problems with Self-Concept | - | - | 0.98 |
| E- DSM-V Inattentive Symptoms | - | - | 1.54 |
| F- DSM-V Hyperactive-Impulsive Symptoms | - | - | 0.89 |
| G- DSM-V ADHD Symptoms Total | - | - | 1.48 |
| H- ADHD Index | - | - | 1.24 |
Table 2: Clinical evaluations effect sizes (effect size index d): T1 vs T2, T2 vs T3 and T3 vs T1.
Linear regressions were calculated in order to predict the differences (Δ) observed on neurocognitive performances and clinical evaluations between baseline and after 6 months of MPH treatment (ΔT1-T3) based on the differences evidenced between baseline and the “MHP test” (ΔT1-T2) (Figure 1).
Figure 1: Scatterplots of neurocognitive tests (Phasic Arousal, Sustained Attention and Visual Scanning) and Alertness Scale with fitted regression lines and lower and upper values of a reliable prediction interval (95%): baseline vs. “MPH Test” (ΔT1-T2) against baseline vs. 6 months MPH medication (ΔT1-T3)
High significant regression equations were found for the Phasic Arousal test regarding the reaction times without (F(1,99)=789.78, p<0.01; R2=0.88) and with acoustic warning (F(1,99)=590.74, p<0.01; R2=0.85) and the variability of the reaction times without (F(1,99)=367.40, p<0.01; R2=0.78) and with (F(1,99)=789.78, p<0.01; R2=0.85) acoustic warning. We also evidenced high regression equation for reaction time variability for the Divided Attention test for auditory stimuli (F(1,99)=512.68, p<0.01; R2=0.83) and moderate regression for visual stimuli (F(1,99)=62.02, p<0.01; R2=0.37). Significant regression equations were observed concerning the Flexibility test (reaction time: F(1,99)=138.51, p<0.01; R2=0.57; variability of reaction time: F(1,99)=210.09, p<0.01; R2=0.67; number of errors: F(1,99)=70.68, p<0.01; R2=0.41). Similar results were observed on Incompatibility test for both compatible condition (reaction time: F(1,99)=232.69, p<0.01; R2=0.69; variability of reaction time: F(1,99)=191.68, p<0.01; R2=0.65; number of errors: F(1,99)=252.40, p<0.01; R2=0.71) and incompatible condition (reaction time: F(1,99)=234.98, p<0.01; R2=0.70; variability of reaction time: F(1,99)=197.22, p<0.01; R2=0.66; number of errors: F(1,99)=343.05, p<0.01; R2=0.77). We noticed significant regressions for Visual Scanning test (reaction time: F(1,99)=84.67, p<0.01; R2=0.45; variability of reaction time: F(1,99)=117.03, p<0.01; R2=0.53; number of errors: F(1,99)=155.85, p<0.01; R2=0.60 and number of omissions: F(1,99)=478.12, p<0.01; R2=0.85) as well as for the Go No Go test (reaction time: F(1,99)=265.08, p<0.01; R2=0.72; variability of reaction time: F(1,99)=147.06, p<0.01; R2=0.59; number of errors: F(1,99)=394.18, p<0.01; R2=0.79) and the Working Memory test (number of omissions: F(1,99)=152.38, p<0.01; R2=0.60 and number of errors: F(1,99)=129.02,p<0.01;R2=0.56].We evidenced comparable results regarding the number of omissions [F(1,99)=619.88, p<0.01; R2=0.86] and errors [F(1,99)=530.02, p<0.01; R2=0.84] on Sustained Attention test whereas a weak regression equation was observed regarding the reaction time variability [F(1,99)=39.89, p<0.01; R2=0.27] on Sustained Attention.
Finally, moderate significant regression equations was evidenced for the Alertness scale [F(1,99)=35.98, p<0.01; R2=0.35], the Contentedness scale [F(1,99)=32.86, p<0.01; R2=0.34] and the Calmness scale [F(1,99)=34.80, p<0.01; R2=0.35].
Discussion
The purpose of this study was first to examine the effect of a single 10 mg dose of methylphenidate on cognitive disturbances in adult ADHD patients by the mean of a computerized tests battery. We attempted to assess a large sample of drug naive adult patients without major psychiatric comorbidity. Computerized TAP battery was more appropriate to investigate neurocognitive effects because of the lack of significant learning effects. Attentional functioning in the present study was assessed from a multi componential perspective [26]. Our results show that adult ADHD patients were impaired in various assessments of selectivity and intensity of attention. A significant impairment of alertness, divided attention, selective attention and sustained attention was observed. Besides, our patients performed significantly worse in executive functions investigations. Impairments on flexibility, working memory and inhibition control were evidenced. This study confirmed prior reports of neurocognitive deficits in adult ADHD. Former studies have shown disturbances on vigilance [8,24], divided attention, selective attention and flexibility [27-29].
In the present study, the “MPH Test” has shown significant improvements on reaction times, variability of the performances and response accuracy in tasks measuring alertness, divided attention, selective attention and sustained attention. We also noticed a significant improvement concerning the executive functions such as the flexibility/shifting abilities, the inhibition and interference control and the working memory. In addition, the “MPH Test” had a positive effect on subjective feelings. Indeed, patients felt more alert and calmer compared to baseline according to the subjective visual analogical scales. We observed large effect sizes more particularly for variability of reaction times on Flexibility, Incompatibility and Sustained Attention tasks as well as for the number of omissions in Working Memory, Visual Scanning and Sustained Attention tests. Moreover, large effect sizes were evidenced on clinical evaluations such as alertness and contentedness scales.
Besides, these improvements were not correlated to the severity of the cognitive disturbances observed on baseline at least at the most sensitive parameters according to the clinical observations. Therefore, the initial cognitive level did not influence the response to the “MPH Test”. This observation suggested that a dose of 10 mg of MPH is effective and sensitive whatever the neuropsychological profile. A high intra and inter-individual variability of the cognitive symptoms in ADHD adults was underlined. Mostert et al. have demonstrated that ADHD adult patients were not distinctively impaired and confirmed that adult ADHD was neuropsychologically heterogeneous.[30]
The MPH-related benefits on attentional and executive functions were still observed after 6 months of MPH treatment. Our patients were medicated with an adequate dose of MPH (46.7 ± 23.8 mg/day) over a period of at least 6 months and assessed. A positive effect on cognitive performances and subjective feeling was still observed after 6 months of MPH medication. Patients reported not only effective improvements in neuropsychological functioning but also in everyday life, activities and social functioning. An undeniable positive clinical impact was highlighted regarding the large effect sizes observed on ARSRS and CAARS scales.
In our study the MPH dose used for the acute test was low (10 mg) but was sufficient to improve executive and attentional functions in ADHD adult patients. After a titration phase, an optimal adapted MPH treatment (immediate and /or extended release) was prescribed to our patients. According to several studies, the response to MPH is dependent of the dose administered [31]. However, our results suggested that a high MPH dose does not necessarily lead to better cognitive performances. Besides, a low cognitive initial level does not necessarily suppose a high MPH dose. In addition to the therapeutic response, other variables had obviously to be taken into account in the MPH dose prescription such as side effects and other adverse consequences [32-34].
The most striking finding in our study is that the neurocognitive and clinical difference observed between the baseline and the “MPH Test” was correlated to the difference noticed between the baseline and the assessment 6 months after MPH treatment. We noticed strong positive linear relationship concerning neurocognitive tasks, which evaluate more precisely vigilance and sustained attention. The differences evidenced between baseline and the “MPH Test” on these neurocognitive tests predicted the neurocognitive profile observed 6 months after MPH medication. On the other hand, moderate significant positive linear relationship was evidenced regarding the differences between baseline and the “MPH Test” on clinical evaluations 6 months after MPH medication. These effects concerned the alertness, contentedness and calmness scales [35-37].
We noticed high significant regression equations for the reaction times and the performance homogeneity for Phasic Arousal test, the number of omissions and errors in Sustained Attention and the number of omissions in Visual Scanning test. These neurocognitive parameters seemed to constitute the most sensitive and predictive markers. These results highlighted the predictive therapeutic response of the “MPH Test”. This latter seemed to constitute an accurate predictor of the long-term MPH treatment outcome on cognitive abilities but also an adapted tool in order to predict the clinical response.
Some limitations of this study can be noticed. First, we did not compare the effects of MPH against placebo in ADHD adults, neither the effects of MPH on neurocognitive sub-tests performances in healthy subjects. An analysis taking into consideration the different subtypes of ADHD (inattentive hyperactive-impulsive and combined) could also be carried out. Our results demonstrated that adult patients with ADHD are impaired in a variety of attention components and executive functions and stimulant drug therapy allowed to enhance all these functions. Further research will attempt to evidence more specific and sensitive neurocognitive indicators liable to predict long-term outcomes with MPH medication. Specific and sensitive indicators could be defined from these parameters in further studies in order to create clinical tools in the MPH treatment care in adult ADHD.
Conclusion
In summary, MPH does not only effectively improve clinical symptoms but also has a positive effect on several neurocognitive processes essentials in everyday life. This study shows the benefits of including a “MPH Test” in adult ADHD patient’s investigation. Our results suggest that the “MPH Test” would be useful in predicting subsequent responses to MPH in ADHD adultpatients
References
- Wender PH, Wolf LE, Wasserstein J (2001) Adults with ADHD. An overview. Ann N Y Acad Sci 931: 1-16.
- Barkley RA, Murphy KR (1998) Attention deficit hyperactivity disorder: A clinical workbook (2nd edn). Guilford Press, New York
- Murphy KR, Adler LA (2004) Assessing attention-deficit/hyperactivity disorder in adults: focus on rating scales 65: 12- 7.
- Adler LA, Chua HC (2002) Management of ADHD in adults. J Clin Psychiatry 63: 29-35,
- Ossmann JM, Mulligan NW (2003) Inhibition and attention deficit hyperactivity disorder in adults. Am J Psychol 116: 35-50.
- Tucha L, Tucha O, Laufkötter R, Walitza S, Klein HE (2008) Neuropsychological assessment of attention in adults with different sub-types of attention-deficit/hyperactivity disorder. Journal of Neural Transmission 115: 269-78.
- Dinn WM, Robbins NC, Harris CL (2001) Adult attention-deficit/hyperactivity disorder: psychological test profiles in a clinical population. Brain Cogn 46: 114-121.
- Epstein JN, Johnson DE, Varia IM, Conners CK (2001) Neuropsychological assessment of response inhibition in adults with ADHD. J Clin Exp Neuropsychol 23: 362-371.
- Gallagher R, Blader J (2001) The diagnosis and neuropsychological assessment of adult attention deficit/hyperactivity disorder. Scientific study and practical guidelines. Ann N Y Acad Sci 931: 148-71.
- Nigg JT, Butler KM, Huang-Pollock CL, Henderson JM (2002) Inhibitory processes in adults with persistent childhood onset ADHD. J Consult Clin Psychol 70: 153-7.
- Aron AR, Dowson JH, Sahakian BJ, Robbins TW (2003) Methylphenidate improves response inhibition in adults with attention-deficit/hyperactivity disorder. Biological Psychiatry 54: 1465-1468.
- Wodushek TR, Neumann CS (2003) Inhibitory capacity in adults with symptoms of Attention Deficit/Hyperactivity Disorder (ADHD). Arch Clin Neuropsychol 3: 317-30.
- Turner DC, Blackwell AD, Dowson JH, Mc Lean A, Sahakian BJ (2005) Neurocognitive effects of methylphenidate in adult attention-deficit/hyperactivity disorder. Psychopharmacology 178: 286-295.
- Emond V, Joyal C, Poissant H (2009) Structural and functional neuroanatomy of attention-deficit hyperactivity disorder (ADHD). Encephale 35: 107-14.
- Boonstra AM, Kooij JS, Oosterlaan J, Sergeant JA, Buitelaar JK (2005) Does methylphenidate improve inhibition and other cognitive abilities in adults with childhood-onset ADHD? Journal of Clin and Exp Neuropsychology 27: 278-298.
- Kurscheidt JC, Peiler P, Behnken A, Abel S, Pedersen A et al. (2008) Acute effects of methylphenidate on neuropsychological parameters in adults with ADHD: possible relevance for therapy. Journal of Neural Transmission 115: 375-62.
- Kooij JJS, Francken MH (2010) DIVA Foundation, The Netherlands.
- Conners CK (1998) Rating scales in attention-deficit/hyperactivity disorder; use in assessment and treatment monitoring. J Clin psychiatry 59: 24-30.
- Ward MF, Wender PH, Reimherr FW (1993) The Wender Utah Rating Scale: an aid in the retrospective diagnosis of childhood attention deficit hyperactivity disorder. Am J Psychiatry 150: 885-90.
- Retz-Junginger P, Retz W, Blocher D, Weijers HG, Trott GE, et al(2003) Wender Utah rating scale. The short-version for the assessment of the attention-deficit hyperactivity disorder in adults. Nervenarzt 74: 296-297.
- Biedermann J, Faraone SV, Spencer T, Wilens T, Norman D et al. (1993) Patterns of psychiatric comorbidity, cognition and psychosocial functioning in adults with attention deficit hyperactivity disorder. The American Journal of Psychiatry 150: 1792-1798.
- Barkley RA (1998) Attention-deficit/hyperactivity disorder: a handbook for diagnosis and treatment, (2nd edn.) Guilford, New York.
- Schweitzer JB, Cummins TK, Kant CA (2001) Attention-deficit/hyperactivity disorder. Med Clin North Am 85: 757-7.
- Epstein JN, Conners CK, Sitarenios G, Erhardt D (1998) Continuous performance test results in adult with attention deficit hyperactivity disorder (ADHD). The clinical Psychologist 12: 155-168.
- Zimmermann P, Fimm B (2010) Testbatterie zur Aufmerksamkeitsprüfung (TAP) Version 2.3 Handbuch 1. Vera Fimm Psychologische Test-systeme, Herzogenrath.
- Van Zomeren AH, Brouwer WH (1994) Clinical neuropsychology of attention. Oxford University Press, New York.
- Schoechlin C, Engel RR (2005) Neuropsychological performance in adult attention-deficit hyperactivity disorder: Meta-analysis of empirical date. Archives of Clinical Neuropsychology 20: 727-744.
- Woods SP, Lovejoy DW, Ball JD (2002) Neuropsychological characteristics of adults with ADHD: a comprehensive review of initial studies. Clin Neuropsychol, 16: 12-34.
- Taylor CJ, Miller DC (1997) Neuropsychological assessment of attention in ADHD adults. J Atten Disord 2: 77–88.
- Bond A, Lader M (1974) The use of analogue scales in rating subjective feelings. Br J Med Psychol 47: 211-8.
- Castells X, Ramos-Quiroga JA, Rigau D, Bosch R, Nogueira M, et al. (2011) Efficacy of methylphenidate for adults with attention-deficit hyperactivity disorder: a meta-regression analysis. CNS Drugs 25: 157-169.
- Cohen J (1988) Statistical power analysis for the behavioural sciences. (2nd edn.) Erlbaum, Hillsdale.
- Faraone SV, Biedermann J (2005) What is the prevalence of adult ADHD? Results of a population screen of 966 adults. J Atten Disord 9:384–391
- Kooij JJS (2012) Adult ADHD. Diagnostic assessment and treatment, (3rd edn.) Springer.
- Mackinnon A, Mulligan R (2005) The estimation of premorbid intelligence levels in French speakers. Encephale 31: 31-43.
- McLean A, Dowson J, Toone B, Young S (2004) Characteristic neurocognitive profile associated with adult attention deficit/hyperactivity disorder. Psychological Medicine 34: 681-692.
- Mostert JC, Onnink AMH, Klein M, Dammers J, Harneit A, et al. (2015) Cognitive heterogeneity in adult attention deficit/hyperactivity disorder: A systematic analysis of neuropsychological measurements. Eur Neuropsychopharmacol 25: 2062-2074.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi