Research Article, J Plant Physiol Pathol Vol: 6 Issue: 2
Fine-Tuning of PR Genes in Wheat Responding to Different Puccinia Rust Species
Hongtao Zhang1#, Yongchun Qiu1#, Congying Yuan2#, Xianming Chen2,3 and Li Huang1*
1Montana State University, Department of Plant Sciences and Plant Pathology, Bozeman, MT 59717-3150, USA
2Washington State University, Department of Plant Pathology, Pullman, WA 99164-6430, USA
3USDA-ARS, Wheat Health, Genetics, and Quality Research Unit, USDA-ARS, Pullman, WA 99164-6430, USA
#Those authors contributed equally to this work
*Corresponding Author : Li Huang
Montana State University, Department of Plant Sciences and Plant Pathology, Bozeman, MT 59717-3150, USA
Tel: +1 4069945058
Fax: +1 4069947600
E-mail: lhuang@montana.edu
Received: February 02, 2018 Accepted: March 24, 2018 Published: March 30, 2018
Citation: Zhang H, Qiu Y, Yuan C, Chen X, Huang L (2018) Fine-Tuning of PR Genes in Wheat Responding to Different Puccinia Rust Species. J Plant Physiol Pathol 6:2. doi: 10.4172/2329-955X.1000178
Abstract
Fine-Tuning of PR Genes in Wheat Responding to Different Puccinia Rust Species
Upregulation of pathogenesis-related genes (PRs) has been found to be associated with plant defense response. To better understand the role of PR genes in wheat defense response against rust pathogens, we studied the expression of five PR genes in six wheat lines during their interactions with three Puccinia species. The research revealed three PR gene expression patterns associated with resistance to the three rusts, implying different strategies from the host in response to different rusts. In addition, different PR gene expression patterns were found in the same genetic background when interacting with different races of the same Puccinia species, suggesting different counteractions from the pathogen during infection. Overall, our study revealed fine-tuning of PR genes in wheat responding to different Puccinia rust species, implicating the limitation of defense when only overexpressing a single PR gene in the host.
Keywords: PR genes; Triticum aestivum; defense response; qRT-PCR; Puccinia species
Introduction
The term “pathogenesis-related proteins (PRs)” was used to describe these proteins because they were first identified as additional proteins induced in the host plants under pathogen-inoculated conditions [1]. PR-1a, -1b and -1c were first purified in tobacco plants infected with tobacco mosaic virus (TMV) [2,3]. In TMV infected tobacco, the highest PR1 concentration was found at the hypersensitive lesion margins as well as non-infected leaves from resistant tobacco plants that previously inoculated with TMV, suggesting the role of PR1 in limiting the multiplication or spreading of the virus [4]. Later, PR1 proteins were also found in TMV inoculated susceptible tobacco [5]; healthy plants during flowering [6]; natural senescing plants [7,8] and plants treated by salicylic acid [9]. In 1980, PR proteins were defined as “proteins encoded by the host plant but induced only in pathological or related situations” [1].
Soon after the discovery of PR1, PR2 (β-1, 3-glucanases) [10] and PR3 (chitinase) [11], they were identified to have antifungal activity. So far, sixteen PR proteins have been described [12]. Most of them have antimicrobial functions including antifungal, antibacterial, and antiviral actions. Some have insecticidal or nematicidal activities [12]. These studies suggest that the function of PR proteins is positively associated with host plant defenses. During the past decades, many research efforts have been devoted to understanding of the regulation of PR proteins and their inducers. In Arabidopsis, PR1, PR2 and PR5 are associated with salicylic acid (SA)-regulated defense response [13,14]. Mutations that impaired SA biosynthesis strongly reduced PR1 expression and systemic acquired resistance (SAR) [15], suggesting that upregulation of PR1 was SA-dependent and a positive correlation between PR1 and SAR. However, the same mutations did not affect the expressions of PR2 and PR5, indicating their SA-independency [15]. In tobacco, both PR-1b and PR5 were found induced by the combination of ethylene and methyl jasmonate (MeJA) [16]. However, a protein that blocked ethylene induction of PR-1b was unable to block PR5 induction, suggesting a fine regulation of the two PR genes [16]. Desmond et al. [17] reported that either exogenous JA or benzo-(1, 2, 3)- thiadiazole-7-carbothioic acid S-methyl ester (BTH, a SA analog) treatment could result in upregulation of PR1, PR2, PR3, PR4, PR5 and PR10 in wheat cultivar Kennedy. In contrast, in a different wheat cultivar Sunco, only JA induced the expressions of those six PR genes, BTH suppressed the expressions of the same PR genes [17]. These reports revealed the complexity of PR gene regulation in different species, in different backgrounds of the same species or in response to different pathogens.
In our previous research, we noted that knocking out genes TaCSN5-2A or TaCSN5-2D in spring wheat cultivar Alpowa increased PR1 transcription and enhanced resistance to leaf rust [18]. However, enhanced PR1 level in the TaCSN5 mutants did not render resistance to stem rust. These findings draw our attention to the expression patterns of several antifungal PR genes of wheat, including PR1, PR2, PR3, PR5 and PR10, during the interactions with three rust pathogens, which are Puccinia triticina (Pt) causing leaf rust, P. graminis f. sp. tritici (Pgt) causing stem rust and P. striiformis f. sp. tritici (Pst) causing stripe rust. They are fungal pathogens having a similarl biotrophic life style when infecting wheat. In this study, we found different expression patterns of the five PR genes associated with wheat defense responses against different rust pathogens.
Materials and Methods
Plant materials
Six wheat lines were used in this study, including two mutants MNR180 and MNR220 and four cultivars containing known Lr, Sr or Yr genes for resistance to leaf rust, stem rust or stripe (yellow) rust, respectively. Alpowa (PI 566596), a soft white spring wheat cultivar, was obtained from the USDA National Plant Germplasm System (NPGS). MNR180 and MNR220 are EMS induced mutants of Alpowa generated by Dr. Michael Giroux at Montana State University, referred as MNR180 (Alpowa) and MNR220 (Alpowa) thereafter. Wheat cultivar Scholar with Lr47, was provided by Dr. Luther Talbert at Montana State University. The Sr33 line in the Chinese Spring background, Sr33 (CS), was provided by Dr. Evans Lagudah at CSIRO, Australia. The Yr5 line in the Avocet background, Yr5 (Avocet), was maintained in Dr. Xianming Chen’s lab at USDAARS, Pullman, Washington.
Pathogen
The Pt race PBJJG used for leaf rust assays was kindly provided by Dr. Robert Bowden, USDA-ARS Manhattan, KS. The Pgt races TMLKC and QFCSC were maintained at the Cereal Disease Laboratory, St. Paul, Minnesota. The Pst races PSTv-11 and PSTv-37 were maintained at the USDA-ARS Pullman, Washington.
Plant growth conditions and pathogen inoculation
Plant growth conditions: Before inoculation, all wheat seedlings were grown under the following conditions: 22°C/14°C day/night temperatures and a 16 h photoperiod. Plants were watered and fertilized with Peters General Purpose Plant Food (Scotts-Miracle- Gro Company, Marysville, OH) at a concentration of 150 ppm N-P-K every day.
Rust inoculations and hormone treatments: Leaf rust inoculations were performed as described in Campbell et al. [19]. Stem rust inoculations were conducted in a similar manner to leaf rust with the following exceptions: the dew chamber was pre-conditioned to an air temperature of 19–22°C. Inoculated plants were incubated for 24 h followed by at least 3 h under high humidity and light intensity conditions before being transferred to the greenhouse. Stripe rust inoculations were conducted following the methods described by Wan and Chen et al. [20]. Plants were uniformly inoculated with urediniospores mixed with talc in a ratio of 1:20, kept in a dew chamber without light for 24 h at 10°C, and then grown in a growth chamber with diurnal temperature cycles gradually changing from 20°C at 2:00 pm to 4°C at 2:00 am and 16 h light/8 h dark. Mock treatments were done the same as the corresponding rust inoculation except without urediniospores.
For hormone treatments, wheat seedlings were sprayed with 20 mM SA or 2 mM MeJA in 0.1% (v/v) ethanol, respectively at four different time points: 24 hours before rust inoculation (hbri), 0 hours post rust inoculation (hpri) in which, SA or MeJA were sprayed right before rust inoculation, 12 hpri and 24 hpri in which SA or MeJA were sprayed 12 hr or 24 hr post rust inoculation. The check (CK) was the plants without any chemical treatment and mock was the plants sprayed with 0.1% (v/v) ethanol prior to rust inoculation.
Gene expression analysis by qRT-PCR
Leaf tissues were collected at 0, 1, 2, 3, 5, 8 and 10 dpi, snap frozen in liquid nitrogen and stored at –80°C until RNA isolations were performed. Total RNA was isolated and treated with DNase I on a column using a Qiagen RNeasy Plant Mini Kit (Qiagen, Valencia, CA) following the manufacturer’s instructions. The quality and concentration of total RNA were assessed via agarose gel electrophoresis and 260/280ABS measurements on a NanoDrop 1000 spectrophotometer (Thermo Fisher Scientific Inc., Wilmington, DE).
Basal level of the PR genes and time course expression study post inoculation with different rust pathogens on different lines were quantified by quantitative real-time-PCR (qRT- PCR). The primers used to measure the PR gene transcripts were according to Desmond [17]. qRT-PCRs were performed using the iScript One- Step RT-PCR Kit with SYBR Green (Bio-Rad, Hercules, CA) on a CFX96 real-time PCR detection system (Bio-Rad, Hercules, CA) following the manufacturer’s protocol. Transcript abundance was normalized to the reference gene ACTB (β-actin) [21]. Expression measurement of each gene was conducted in triplets with three biological replicates. Data were used only if the Ct standard deviation among the triplets was ≤ 0.2, and the mean of the triplet’s Ct was used for the calculation. Relative expression was calculated using the ΔΔCt method as described in the CFX96 manual (Bio-Rad, Hercules, CA), where fold change = 2–ΔΔCt. Relative expression of the PR genes at different time points are presented as the relative expression to the 0 hr time point. Standard deviations were calculated among the three biological replicates. Student’s t-tests were performed to test whether the expression level at different time points are different. The p-values were calculated based on an unpaired two-tailed distribution.
Results
Basal expression levels of the PR genes
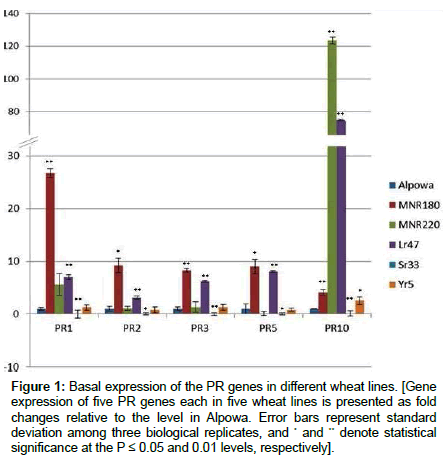
As the term PR gene implies, their expressions should be pathogenesis-related or stress- related. However, when the expression of five PR genes were investigated in the six wheat lines without pathogen inoculation or stresses via qRT-PCR, the basal levels of the PR genes were different in Alpowa, MNR180 (Alpowa), MNR220 (Alpowa), Lr47 (Scholar), Sr33 (CS) and Yr5 (Avocet) (Figure 1). Alpowa was susceptible to all the races of three rust species selected; therefore the level of each PR gene in Alpowa was used as a reference and normalized to 1 for calculating the relative expressions of PR genes in other lines. The expression levels of the others were calculated as fold changes relative to that in Alpowa. Among the six lines, Sr33 (CS) had the lowest basal levels of all five PR genes. The levels were almost undetectable, consistent with the term of “pathogenesis-related”. The remaining five lines, including Alpowa, PR genes have been elevated to some extent compared to the levels of Sr33 (CS). Mutant MNR180 (Alpowa) was resistant to all three rust species, and had the highest basal levels of PR1, PR2, PR3 and PR5 among all. Although the basal PR10 level of MNR180 was not the highest, it was still significantly higher than the level of Alpowa. Lr47 (Scholar) was highly resistant to leaf rust and had second highest basal levels of all five PR genes. MNR220 (Alpowa), which is resistant to leaf and stem but not to stripe rust races at the seedling stage, had the highest level of PR10. Yr5 (Avocet), resistant to stripe rust, had the second lowest expression levels of PR genes except PR10.
Figure 1: Basal expression of the PR genes in different wheat lines. [Gene expression of five PR genes each in five wheat lines is presented as fold changes relative to the level in Alpowa. Error bars represent standard deviation among three biological replicates, and * and ** denote statistical significance at the P ≤ 0.05 and 0.01 levels, respectively].
Contrast expression profiles of PR genes in MNR220 responding to leaf and stem rust pathogens
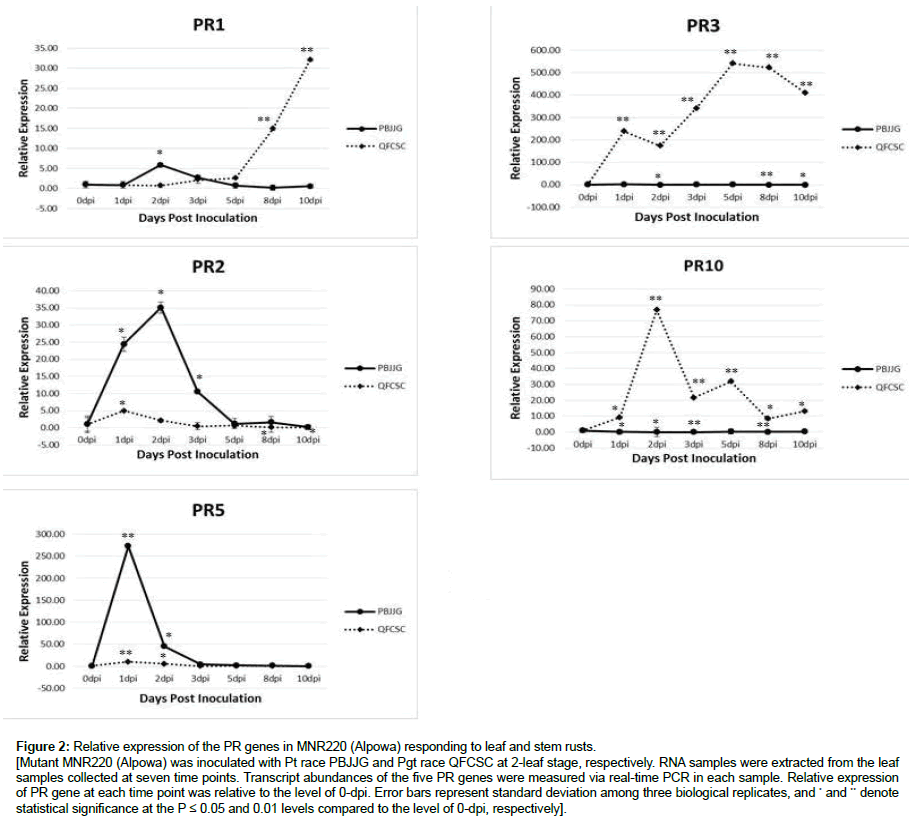
MNR220 (Alpowa) is a mutant that acquired new resistance to more than a dozen races of the leaf and stem rust pathogens. The new resistance to the two rusts is conferred by the same locus of MNR220 [19]. The two rust pathogens belong to the same genus and have a similar life style. To understand if the same strategy reflected by PR gene expression patterns was deployed to defend against the two rust species, the five PR genes were investigated in MNR220 (Alpowa) at seven time points after inoculation with Pt PBJJG and Pgt QFCSC, respectively. Unexpectedly, expression patterns of all five PR genes were quite different in MNR220 in response to leaf and stem rust pathogens (Figure 2). When challenged with Pt, PR1 expression was elevated and reached to the highest at 2 dpi with a 6-fold increase compared to the 0-dpi level. In contrast, there was little change in PR1 expression in MNR220 until 8 dpi when infected with Pgt. The level of PR2 was increased for 35 fold at 2 dpi and level of PR5 was up for 280 fold at 1 dpi after inoculated with Pt. Similarly, both PR2 and PR5 were upregulated at 1 dpi during Pgt infection, but the magnitude was insignificant when compared to the increased levels detected during Pt infection. Interestingly, and quite opposite, highly elevated PR3 and PR10 expressions were detected in the mutant at 1 dpi and 2 dpi after inoculated with Pgt, but not after Pt inoculation (Figure 2). These results suggested a different strategy was deployed in MNR220 (Alpowa) to defend against the two different rust species. PR1, PR2 and PR5 were more important in MNR220 (Alpowa) during defense against leaf rust pathogen, and PR3 and PR10 were more crucial during defense against stem rust pathogen.
Figure 2: Relative expression of the PR genes in MNR220 (Alpowa) responding to leaf and stem rusts.
[Mutant MNR220 (Alpowa) was inoculated with Pt race PBJJG and Pgt race QFCSC at 2-leaf stage, respectively. RNA samples were extracted from the leaf samples collected at seven time points. Transcript abundances of the five PR genes were measured via real-time PCR in each sample. Relative expression of PR gene at each time point was relative to the level of 0-dpi. Error bars represent standard deviation among three biological replicates, and * and ** denote statistical significance at the P ≤ 0.05 and 0.01 levels compared to the level of 0-dpi, respectively].
PR gene expression patterns in leaf rust resistant lines during infection
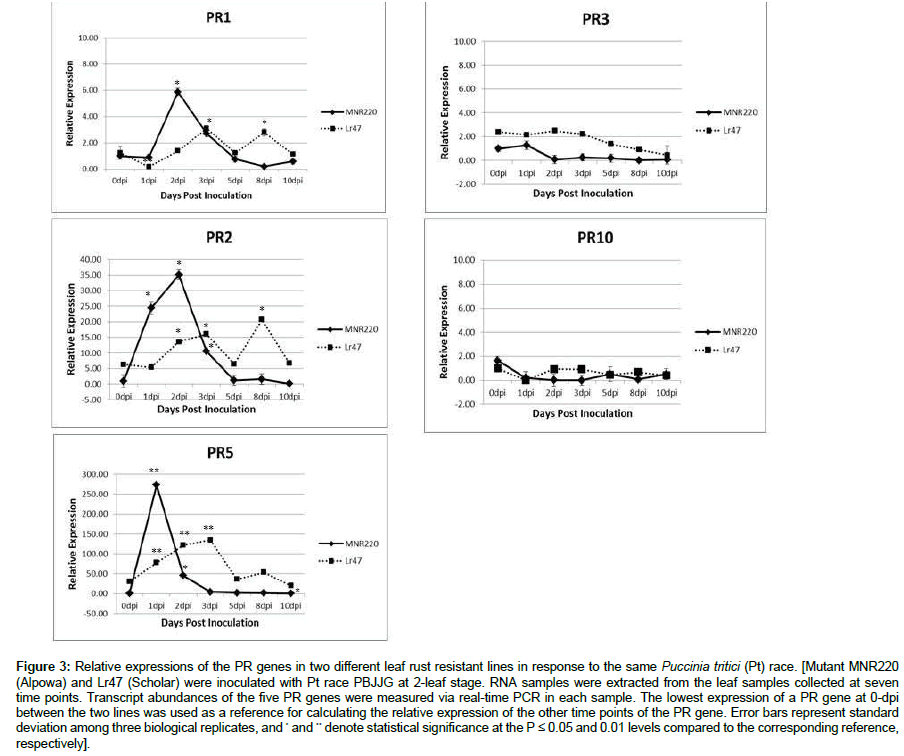
After seeing different PR gene expression patterns in the same genetic background MNR220 (Alpowa) against two different rust species, it was interesting to investigate the PR gene expression patterns among different lines with resistance to the same rust species. Lr47 (Scholar) and MNR220 (Alpowa) had a similar level of resistance to Pt race PBJJG. Expression patterns of the five PR genes were compared between the two lines at seven time points from 0~10 dpi (Figure 3). As shown in Figure1, the two lines had different PR basal expression levels. MNR220 (Alpowa) had a lower PR basal expression level compared to Lr47 (Scholar) except PR10, therefore, expression levels of PR1, PR2, PR3 and PR5 in MNR220 (Alpowa) at 0 dpi were normalized to 1 for calculating the relative expressions of other time points for the two lines. In Figure 3, expression pattern of each PR gene was presented by the curve plotted by using relative expressions to the level of 0-dpi MNR220 at seven time points. When the patterns were compared, PR1, PR2 and PR5 genes in MNR220 (Alpowa) were upregulated much earlier and higher than that in Lr47 (Scholar) (Figure 3). However, there was something in common between the two lines, both had significantly elevated PR1, PR2 and PR5 at early time points, and not much change in PR3 or PR10 expression was detected over time. It seemed that the significantly increased PR1, PR2 and PR5 expression levels at early time points were corresponding to resistance to leaf rust, suggesting that these three PR genes in wheat had a more critical role than PR3 and PR10 when defending against leaf rust pathogen.
Figure 3: Relative expressions of the PR genes in two different leaf rust resistant lines in response to the same Puccinia tritici (Pt) race. [Mutant MNR220 (Alpowa) and Lr47 (Scholar) were inoculated with Pt race PBJJG at 2-leaf stage. RNA samples were extracted from the leaf samples collected at seven time points. Transcript abundances of the five PR genes were measured via real-time PCR in each sample. The lowest expression of a PR gene at 0-dpi between the two lines was used as a reference for calculating the relative expression of the other time points of the PR gene. Error bars represent standard deviation among three biological replicates, and * and ** denote statistical significance at the P ≤ 0.05 and 0.01 levels compared to the corresponding reference, respectively].
Different PR gene expression patterns in the same genetic background to different races of stem rust pathogen
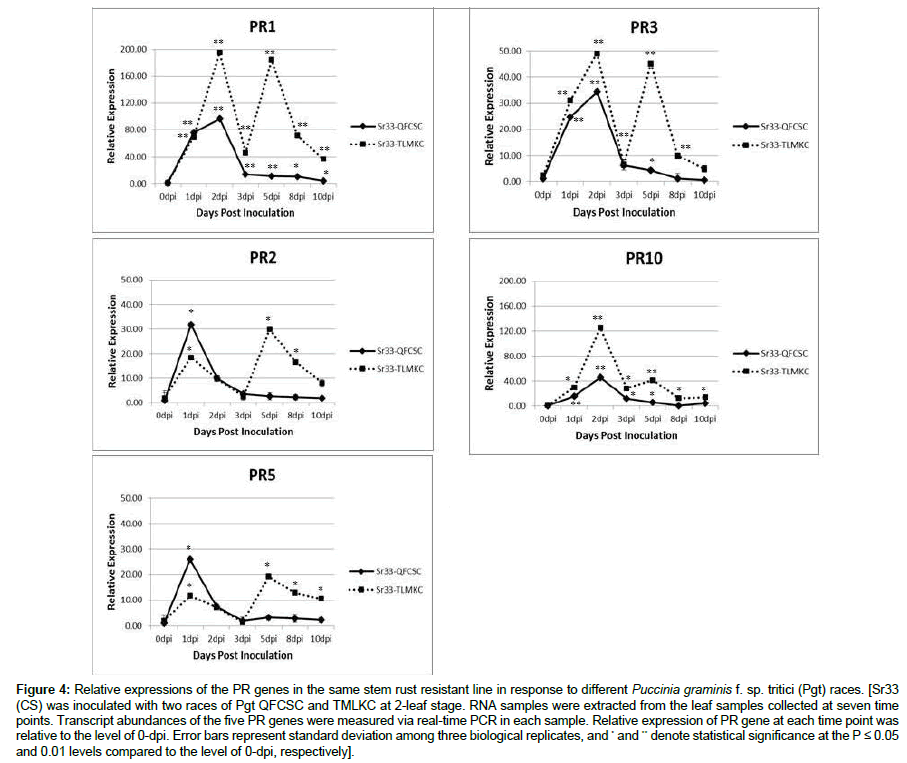
The stem rust resistance gene Sr33 encodes a protein containing a coiled-coil, nucleotide-binding site and leucine-rich repeat (CC-NBSLRR) domains [22]. The gene was discovered from Aegilops tauschii and introgressed into cultivar Chinese Spring (CS) through a single chromosome substitution [23]. Sr33 (CS) is resistant to diverse Pgt races including QFCSC and TLMKC. The infection types of Sr33 (CS) to the two Pgt races were similar. Five PR genes were monitored in Sr33 (CS) line after being inoculated with QFCSC and TLMKC, respectively. As shown in Figure 4, the five selected PR genes had very similar patterns at early time points in the host between the two interactions. The genes were all upregulated at 1 or 2 dpi although different patterns were seen after 3 dpi. Interestingly, all five PR genes showed a two-peak pattern in Sr33 (CS) when infected with TLMKC, but a one-peak pattern when infected with QFCSC (Figure 4).
Figure 4: Relative expressions of the PR genes in the same stem rust resistant line in response to different Puccinia graminis f. sp. tritici (Pgt) races. [Sr33 (CS) was inoculated with two races of Pgt QFCSC and TMLKC at 2-leaf stage. RNA samples were extracted from the leaf samples collected at seven time points. Transcript abundances of the five PR genes were measured via real-time PCR in each sample. Relative expression of PR gene at each time point was relative to the level of 0-dpi. Error bars represent standard deviation among three biological replicates, and * and ** denote statistical significance at the P ≤ 0.05 and 0.01 levels compared to the level of 0-dpi, respectively].
Notably, PR1 had the highest increased level, upregulated 200 folds compared to the 0-dpi level. The second highest was PR10, more than 120-fold increase responding to TLMKC. The rest of the three PR genes had similar levels of enhancement, around 20~40 folds increase compared to that at 0-dpi. PR2 and PR3 had a very similar expression pattern in Sr33 (CS) in response to the two races of stem rust pathogen, respectively.
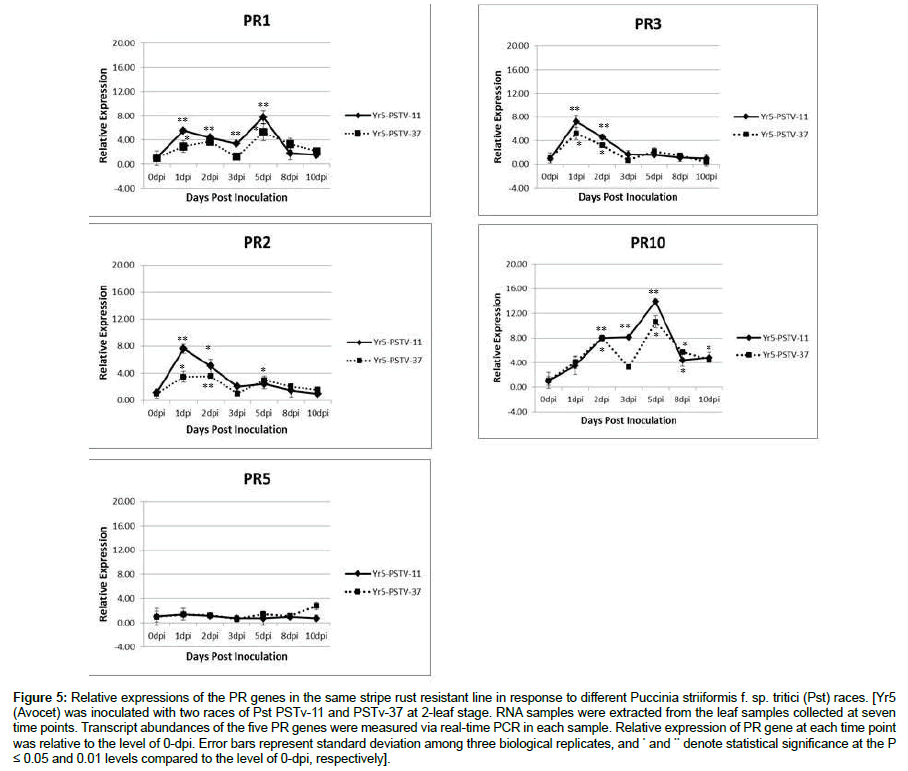
Similar PR gene expression patterns in Yr5 (Avocet) in response to two different races of stripe rust pathogen
The two Pst races PSTv-11 and PSTv-37 were collected in the state of Washington. Yr5 in the Avocet background had similar infection types, 1-2 in a 0-9 scale, when tested with the two races. All five PR genes had very similar expression patterns in the host when interacting with the two Pst races (Figure 5). PR5 was the only one that did not show a significant change at the selected time points during the infection process. PR1 expression showed two peaks. The first peak appeared at a slightly different time point between the two interactions, at 1 dpi in Yr5- PSTv-37 interaction and at 2 dpi in Yr5-PSTv-11 interaction. The second peak at 5 dpi was the same for both interactions. Again, PR2 and PR3 had very similar expression patterns, both were upregulated and reached the highest level at 1 dpi, declined rapidly at 3 dpi, and then a second weak upregulation at 5 dpi was detected in Yr5-PSTv-37 interaction. PR10 expression has a clear two-peak pattern in Yr5-PSTv-37 interaction. Although it seemed like only one peak in Yr5- PSTv-11 interaction, one common observation in the two interactions was that the level of PR10 at 3 dpi has either stopped rising or declined, and then a further increase at 5 dpi.
Figure 5: Relative expressions of the PR genes in the same stripe rust resistant line in response to different Puccinia striiformis f. sp. tritici (Pst) races. [Yr5 (Avocet) was inoculated with two races of Pst PSTv-11 and PSTv-37 at 2-leaf stage. RNA samples were extracted from the leaf samples collected at seven time points. Transcript abundances of the five PR genes were measured via real-time PCR in each sample. Relative expression of PR gene at each time point was relative to the level of 0-dpi. Error bars represent standard deviation among three biological replicates, and * and ** denote statistical significance at the P ≤ 0.05 and 0.01 levels compared to the level of 0-dpi, respectively].
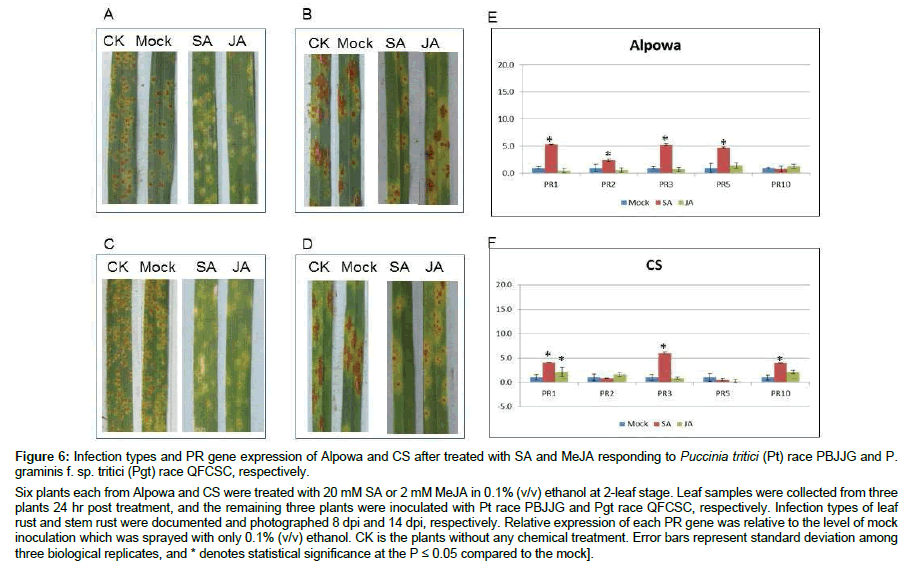
Exogenous hormone treatments enhanced resistance to rusts
The observations of more PR genes upregulated in resistance lines against Pgt than those against Pst or Pt invited a hypothesis that when a wheat plant is in the resistance status to the Pgt races tested then it would also be resistant to the Pt race tested. To test this hypothesis, we monitored the infection types of two susceptible wheat lines of Alpowa and CS to Pt and Pgt after exogenous SA and JA treatments. As shown in Figure 6, post SA treatment, both Alpowa and CS had enhanced resistance to Pt PBJJG (Figure 6A and 6C) and Pgt QFCSC (Figure 6B and 6D) compared with the CK and the mock treatment. MeJA treatment on two cultivars also enhanced their resistance to leaf rust (Figure 6A and 6C) and a little to stem rust (Figure 6B and 6D). Analysis of qRT-PCR results revealed elevated PR1, PR2, PR3 and PR5 in Alpowa (Figure 6E) and PR1, PR3 and PR10 in CS (Figure 6F) 24 hr post SA treatment. The results did support the aforementioned hypothesis.
Figure 6: Infection types and PR gene expression of Alpowa and CS after treated with SA and MeJA responding to Puccinia tritici (Pt) race PBJJG and P. graminis f. sp. tritici (Pgt) race QFCSC, respectively.
Six plants each from Alpowa and CS were treated with 20 mM SA or 2 mM MeJA in 0.1% (v/v) ethanol at 2-leaf stage. Leaf samples were collected from three plants 24 hr post treatment, and the remaining three plants were inoculated with Pt race PBJJG and Pgt race QFCSC, respectively. Infection types of leaf rust and stem rust were documented and photographed 8 dpi and 14 dpi, respectively. Relative expression of each PR gene was relative to the level of mock inoculation which was sprayed with only 0.1% (v/v) ethanol. CK is the plants without any chemical treatment. Error bars represent standard deviation among three biological replicates, and * denotes statistical significance at the P ≤ 0.05 compared to the mock].
Discussion
Breeding constraints in favor of lines with a high PR expression
Plant SAR plays an important role in defense against pathogens. Effective SAR is normally associated with higher expression levels of some PR genes, those genes were used as SAR marker genes [24]. However, inductions of PRs by endogenous and exogenous signaling compounds in the absence of pathogens suggest their functions besides defense against pathogens. High induction of PR genes in tobacco under drought and high salt condition [25], after exposure to UV light or wounding [26] indicated their important roles in maintaining cellular structure under various stresses. From our study, we found that all wheat lines except Sr33 (CS) had a high basal level expression of the five PR genes studied (Figure 1).
The six selected lines were derived from Alpowa, Scholar and CS three genetic backgrounds. Among them, Alpowa and Scholar were once popular cultivars in Washington and Montana states, respectively. The only low basal PR expression line was in the CS background, and CS is a landrace from China with little breeding efforts. This observation may imply that breeding practice in selecting good performance under various stresses resulted in the lines in favor of higher basal PR gene expressions.
Expression patterns of PR gene combinations associated with resistances to the three rusts
It has been noticed that different PR genes associated with the onset of SAR in different species, for example, PR1, PR2 and PR5 were upregulated when effective SAR was detected in Arabidopsis [14]. At least nine PR gene families were found associated with SAR in tobacco [3,27,5]. In our study, significantly upregulated PR1 and PR2 expressions were detected in each resistant line infected with the corresponding rust at early time points (Figure 2- 5). PR1 protein has been reported to inhibit the broad bean rust hyphae differentiation [28] although the function of the protein has not been clearly elucidated. In wheat, there are 23 PR1 genes identified [29], and some have been shown to play distinct roles in host-pathogen interactions, such as PR1.1 and PR1.3 [29]. In this study, the primers of PR1 used for qRT-PCR are specific to PR1.1 and PR1.3 transcript abundances. PR2 is β-1,3-endoglucanase, an enzyme that catalyzes endo-type β-1,3-glucans which is a basic cell wall compound of almost all higher plants and fungi. β-1,3-endoglucanase may directly degrade fungal cell walls or release cell-wall debris as elicitors of defense response [30] . The primers of PR2 will detect the three β-1,3-endoglucanase orthologs on the wheat chromosomes 3A, 3B and 3D. Inductions of PR1 and PR2 by exogenous SA or JA have been reported in some but not all wheat cultivars tested [17,31-33]. Elevated PR1 and PR2 were associated with enhanced resistance to Fusarium head blight caused by a necrotrophic fungal pathogen [32] or to leaf rust caused by a biotrophic fungal pathogen [33].
From our study, we found three expression patterns of the five PR gene combinations associated with resistance to different wheat rust. Pattern 1 represents the combination of greater than 2 fold upregulation of PR1, PR2 and PR5 before 3 dpi and less than 2 fold changes of PR3 and PR10 over the time course (Figure 3). This pattern is associated with resistance to leaf rust.
Pattern 2 represents the significant upregulation of all five PR genes at 1~2 dpi (Figure 4), this pattern is associated with resistance to stem rust. Pattern 3 represents the significant upregulation of four but PR5 genes at 1 dpi (Figure 5), and this pattern was associated with resistance to stripe rust. This observation implied the enhancement of only PR1 gene expression was not sufficient to render resistance to all three rust pathogens, and also explained why a mutant line with increased PR1 could be resistant to leaf rust but not to stem rust.
Different defense strategies of the hosts and the counteractions of the pathogens
Among the five rust resistance lines, host defense response in Lr47 (Scholar), Sr33 (CS) and Yr5 (Avocet) were mediated by a resistance (R) gene. Different PR gene expression patterns mediated by the three R genes in different genetic backgrounds suggested different defense strategies of the hosts to different rust pathogens. Sr33 and Yr5 are race-specific R genes that recognize the corresponding avirulence (Avr) gene in the pathogens although each confirms resistance to more than one race of the corresponding rust species. Sr33 (CS) is resistant to both Pgt QFCSC and TMLKV, it is believed that Sr33 recognizes the same Avr gene in the two races and mediates the same defense response in the host. Similarly, Yr5 should recognize the same Avr gene in Pst PSTv-11 and PSTv-37. It is reasonable to believe that the same defense response in the same genetic background should result in the same PR gene expression pattern. However, when Sr33 (CS) and Yr5 (Avocet) each was tested with two races of the corresponding Puccinia species, different PR gene expression patterns were found in response to different races of the same rust pathogen (Figure 4,5), suggesting different counteractions of the pathogen races in response to the same defense response.
Fine-tuning of PR gene regulation
The regulatory regions of the Arabidopsis PR1 [34-39] and PR2 genes [40,41] have been well studied. A region mainly comprised of binding sites for WRKY and TGA transcription factors relaying SA-dependent signals has been identified and characterized for PR1 [38]. This gene is regulated by both positive and negative cis-acting factors through NPR1. In contrast, the PR2 regulatory region showed no sequence similarity to PR1 [40] although the two genes were upregulated in response to the same pathogen [32,33] or negatively regulated by TGA2 in Arabidopsis [39]. A 125-bp fragment in the PR2 regulatory region is sufficient for the activation of the gene by a 61 amino acid-homeodomain protein [41]. To the best of our knowledge, there is no report on the regulatory regions of the five wheat PR genes we selected. Based on our observations, it seemed that the expression pattern of each PR gene was independent of the expression of the other PR genes. More often than not, PR2 expression was increased when PR1 gene was upregulated (Figures 1-5). PR1 increased its expression in CS after treated with MeJA, but PR2 remained unchanged (Figure 6F). In summary, the expression patterns of the five PR genes were the result of host-pathogen interactions, reflecting the host defense strategies and the pathogen counteractions to the defense responses.
Acknowledgments
The author HZ wishes to thank the funding of NSF BREAD program (Grant no IOS-096-5429), and the Montana Agricultural Experiment Station.
References
- Antoniw JF, Ritter CE, Pierpoint WS, Van loon LC (1980) Comparison of three pathogenesis-related proteins from plants of two cultivars of Tobacco infected with TMV. J Gen Virol 47: 79-87.
- Van Loon LC (1976) Systemic acquired-resistance, peroxidase-activity and lesion size in tobacco reacting hypersensitively to tobacco mosaic-virus. Physiol Plant Pathol 8: 231- 242.
- Gianinazzi S, Pratt HM, Shewry PR, Miflin BJ (1977) Partial-purification and preliminary characterization of soluble leaf proteins specific to virus-infected tobacco plants. J Gen Virol 34: 345-351.
- Wagih EE, Coutts RHA (1981) Similarities in the soluble-protein profiles of leaf tissue following either a hypersensitive reaction to virus-infection or plasmolysis. Plant Sci Lett 21: 61-69.
- Van loon LC (1985) Pathogenesis-related proteins. Plant Mol Biol 4: 111-116.
- Fraser RSS (1981) Evidence for the occurrence of the pathogenesis-related proteins in leaves of healthy tobacco plants during flowering. Physiol Plant Pathol 19: 69-76.
- Henriquez AC, Sanger HL (1982) Analysis of acid-extractable tomato leaf proteins after infection with a viroid, 2 viruses and a fungus and partial-purification of the pathogenesis-related protein-p14. Arch Virol 74: 181-196.
- Conejero V, Picazo I, Segado P (1979) Citrus exocortis viroid (cev) - protein alterations in different hosts following viroid infection. Virology 97: 454-456.
- Van Loon LC, Antoniw J F (1982) Comparison of the effects of salicylic-acid and ethephon with virus-induced hypersensitivity and acquired-resistance in tobacco. Neth J Plant Pathol 88: 237-256.
- Kauffmann S, Legrand M, Geoffroy P, Fritig B (1987) Biological function of pathogenesis-related proteins - four PR proteins of tobacco have 1,3-beta-glucanase activity. Embo J 6: 3209-3212.
- Legrand M, Kauffmann S, Geoffroy P, Fritig B (1987) Biological function of pathogenesis-related proteins-four tobacco pathogenesis-related proteins are chitinases. PNAS 84: 6750-6754.
- Edreva A (2005) Pathogenesis-related proteins: research progress in the last 15 years. Gen Appl Plant Physiology 31: 105-124.
- Ward ER, Uknes SJ, Williams SC, Dincher SS, Wiederhold DL, et al. (1991) Coordinate gene activity in response to agents that induce systemic acquired-resistance. Plant Cell 3: 1085-1094.
- Uknes S, Mauchmani B, Moyer M, Potter S, Williams S, et al. (1992) Acquired- resistance in Arabidopsis. Plant Cell 4: 645-656.
- Nawrath C, Métraux JP (1999) Salicylic acid induction-deficient mutants of Arabidopsis express PR-2 and PR-5 and accumulate high levels of camalexin after pathogen inoculation. Plant Cell 11: 1393-1404.
- Xu Y, Chang PFL, Liu D, Narasimhan ML, Raghothama KG, et al. (1994) Plant defense genes are synergistically induced by ethylene and methyl jasmonate. Plant Cell 6: 1077- 1085.
- Desmond OJ, Edgar CI, Manners JM, Maclean DJ, Schenk PM, et al. (2005) Methyl jasmonate induced gene expression in wheat delays symptom development by the crown rot pathogen Fusarium pseudograminearum. Physiol Mol Plant P 67: 171-179.
- Zhang H, Wang X, Giroux MJ, Huang L (2017) A wheat COP9 subunit 5-like gene is negatively involved in host response to leaf rust. Mol Plant Pathol 18: 125-133.
- Campbell J, Zhang H, Giroux MJ, Feiz L, Jin Y, et al. (2012) A mutagenesis-derived broad-spectrum disease resistance locus in wheat. Theor Appl Genet 125: 391-404.
- Wan A, Chen, X (2014) Virulence characterization of Puccinia striifornis f. sp. tritici using a new set of Yr single-gene line differentials in the United States in 2010. Plant Dis 98: 1534-1542.
- Kozera B, Rapacz M (2013) Reference genes in real-time PCR. J Appl Genet 54: 391-406.
- Periyannan S, Moore J, Ayliffe M, Bansa U, Wang X, et al. (2013) The Gene Sr33, an Ortholog of Barley Mla Genes, Encodes Resistance to Wheat Stem Rust Race Ug99. Science 341: 786-788.
- Kerber ER, Dyck PL (1978) Resistance to stem and leaf rust of wheat in Aegilops squarrosa and transfer of a gene for stem rust resistance to hexaploid wheat. Proc Int Wheat Genet Symp 5th 1978. New Delhi, India.
- Ryals JA, Neuenschwander UH, Willits MG, Molina A, Steiner HY, et al. (1996) Systemic acquired resistance. Plant Cell 8: 1809-1819.
- Singh NK, Nelson DE, Kuhn D, Hasegawa PM, Bressan RA (1989) Molecular-cloning of osmotin and regulation of its expression by aba and adaptation to low water potential. Plant Physiol 90: 1096-1101.
- Brederode FT, Linthorst HJM, Bol JF (1991) Differential induction of acquired- resistance and pr gene-expression in tobacco by virus-infection, ethephon treatment, uv- light and wounding. Plant Mol Biol 17: 1117-1125.
- Van Loon LC, Van Kammen A (1970) Polyacrylamide disc electrophoresis of the soluble leaf proteins from Nicotiana tabacum var. ‘Samsun’ and ‘Samsun NN’. Virology 40: 199- 211.
- Rauscher M, Adam AL, Wirtz S, Guggenheim R, Mendgen K, et al. (1999) PR-1 protein inhibits the differentiation of rust infection hyphae in leaves of acquired resistant broad bean. Plant J 19: 625-633.
- Lu SW, Friesen TL, Faris JD (2011) Molecular characterization and genomic mapping of the pathogenesis-related protein 1 (PR-1) gene family in hexaploid wheat (Triticum aestivum L.). Mol Genet Genomics 285: 485-503.
- Boller T (1995) Chemoperception of microbial signals in plant. Annu Rev Plant Physiol Plant Mol Biol 46: 189-214.
- Molina A, Gorlach J, Volrath S, Ryals J (1999) Wheat genes encoding two types of PR-1 proteins are pathogen inducible, but do not respond to activators of systemic acquired resistance. Mol Plant-Microbe Interact 12: 53-58.
- Makandar R, Nalam VJ, Lee H, Trick HN, Dong YH, et al. (2012) Salicylic acid regulates basal resistance to Fusarium head blight in wheat. Mol Plant-Microbe Interact 25: 431-439.
- Casassola A, Brammer SP, Chaves MS, Martinelli JA, Stefanato F, et al (2015) Changes in gene expression profiles as they relate to the adult plant leaf rust resistance in the wheat cv. Toropi. Physiol Mol Plant P 89: 49-54.
- Lebel E, Heifetz P, Thorne L, Uknes S, Ryals J, et al. (1998) Functional analysis of regulatory sequences controlling PR-1 gene expression in Arabidopsis. Plant Journal 16: 223-233.
- Despres C, DeLong C, Glaze S, Liu E, Fobert PR (2000) The arabidopsis NPR1/NIM1 protein enhances the DNA binding activity of a subgroup of the TGA family of bZIP transcription factors. Plant Cell 12: 279-290.
- Despres C, Chubak C, Rochon A, Clark R, Bethune T, et al. (2003) The Arabidopsis NPR1 disease resistance protein is a novel cofactor that confers redox regulation of DNA binding activity to the basic domain/leucine zipper transcription factor TGA1. Plant Cell 15: 2181-2191.
- Fan WH, Dong, XN (2002) In vivo interaction between NPR1 and transcription factor TGA2 leads to salicylic acid-mediated gene activation in Arabidopsis. Plant Cell 14: 1377-1389.
- Eulgem T (2005) Regulation of the Arabidopsis defense transcriptome. Trends Plant Sci 10: 71-78.
- Kesarwani M, Yoo JM, Dong XN (2007) Genetic interactions of TGA transcription factors in the regulation of pathogenesis-related genes and disease resistance in Arabidopsis. Plant Physiol 144: 336-346.
- Van de löcht U, Meier I, Hahlbrock K, Somssich IE (1990) A 125 bp promoter fragment is sufficient for strong elicitor-mediated gene activation in parsley. Embo J 9: 2945-2950.
- Korfhage U, Trezzini GF, Meier I, Hahlbrock K, Somssich IE (1994) Plant Homeodomain protein involved in transcriptional regulation of a pathogen defense- related gene. The Plant Cell 6: 695-708.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi