Case Report, Int J Cardiovasc Res Vol: 9 Issue: 8
Multiple Rheumatic Valvular Heart Disease Involving all Four Valves in a Young Patient: Case Report and Literature Review
Zeine El Abasse, Rime Benmalek, Ali Rida Bah and Rachida Habbal
Department of Cardiology, Hospital University Center Ibn Rochd, Casablanca, Morocco
*Corresponding Author: Dr. Zeine El Abasse
Department of Cardiology, Hospital University Center Ibn Rochd, Casablanca, Morocco
Tel: 0627781863
E-mail: abassedr@gmail.com
Received: October 30, 2020 Accepted: December 01, 2020 Published: December 08, 2020
Citation: Abasse ZE, Benmalek R, Bah AR, Habbal R (2020) Multiple Rheumatic Valvular Heart Disease Involving all Four Valves in a Young Patient: Case Report and Literature Review. Int J Cardiovasc Res 9:8. doi: 10.37532/icrj.2020.9(8).425
Abstract
Multivalvular Heart Disease (MVD) is still a highly prevalent condition in developing countries such as Morocco causing significant morbidity and mortality. The complex hemodynamic interactions between coexisting valve lesions may alter the clinical expression of each singular lesion, and the clinician should be aware of these interactions that make the diagnosis challenging and the management of these patients often empirical given the absence of current guidelines to guide the strategy. In the available literature, quadrivalvular stenosis is exceptional, mostly diagnosed at autopsy. We here described a very rare case of rheumatic involvement of all four cardiac valves discovered in a 38-year-old female with a history of recurrent attacks of rheumatic fever in her childhood, who presented with gradually progressive exertional dyspnea, palpitations and fatigue of 14-years duration. TTE revealed severe left valves’ disease in addition to a severe Tricuspid disease and moderate Pulmonary Stenosis (PS), with signs of right heart congestion. The case was discussed by the heart-team who decided to try a transcatheter approach given the patient’s high surgical risk, by dilating the aortic valve first, then the mitral and finally the tricuspid valves, in order to avoid sudden increase in cardiac output that may worsen the transaortic valve gradient. Our case highlights the diagnosis and management challenges encountered in the context of a developing country where treatment options are limited. Therefore, the expertise of a multidisciplinary Heart Valve Team is of crucial to properly discuss treatment indications based on several factors such as symptoms, severity and hemodynamic significance of valvular lesions, the risk of morbidity and mortality associated with multiple valve prostheses and the risk of reintervention. The introduction of transcatheter therapy into clinical practice has provided new treatment options for patients with MVD, and new decision-making algorithms are evolving quickly.
Keywords: Quadruple valve stenosis; Rheumatic heart disease; Transthoracic echocardiography; Management, Challenge
Introduction
Although Acute Rheumatic Fever (ARF) has considerably decreased in developed countries, it still remains a public health issue in countries like Morocco, with a reported incidence of 18.3 per 100.000 inhabitants in 2010 [1]. Carditis, which occurs in almost half of the patients, can affect all cardiac valves and is associated with an important morbidity and mortality in young patients. According to the 2015 Global Disease Burden study, Rheumatic Heart Disease (RHD) was associated with 319,000 deaths and 33.4 million cases globally [2]. MVD, defined by the combination of stenotic and/ or regurgitant lesions, occurring on ≥ 2 cardiac valves, is a highly prevalent clinical condition among patients with RHD, concerning 20% of the patients with native valve disease and 15% of those undergoing valvular surgery, in the EuroHeart Survey [3].
In the literature, a limited data on MVD is available, which is emphasized both by the European and the American Guidelines, that include a limited number of evidence-based recommendations, mainly based on small studies or a consensus [4,5], which can be explained by the heterogenicity of MVD combinations, pathogenesis, severity and surgical outcomes, leading to a wide spectrum of clinical scenarios. In the available literature, quadrivalvular stenosis is exceptional [6], mostly diagnosed at autopsy [7]. We here describe a very rare case of rheumatic involvement of all four cardiac valves in a young patient, highlighting the diagnosis and management challenges encountered in the context of a developing country.
Case Presentation
A 38-year-old female presented with gradually progressive exertional dyspnea, palpitations and fatigue of 14-years duration. She had had recurrent attacks of rheumatic fever in her childhood. Initially she had New York Heart Association (NYHA) class II symptoms, which progressively deteriorated to class IV at the time of presentation. She reported having recurrent attacks of rheumatic fever in her childhood.
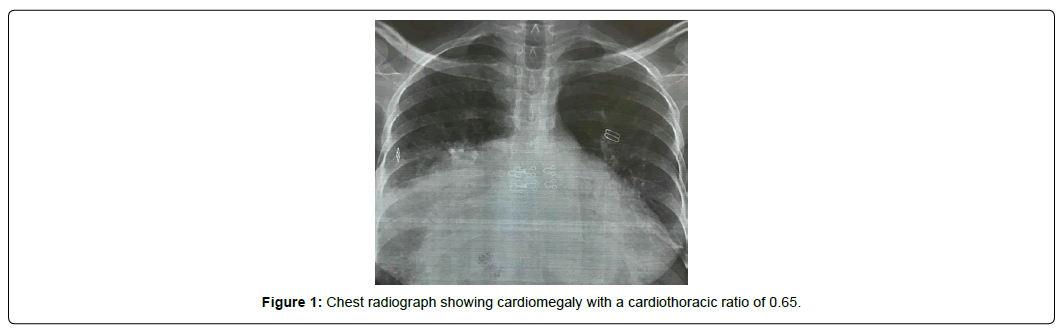
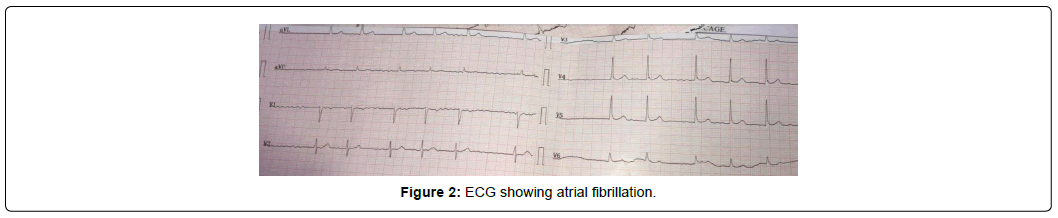
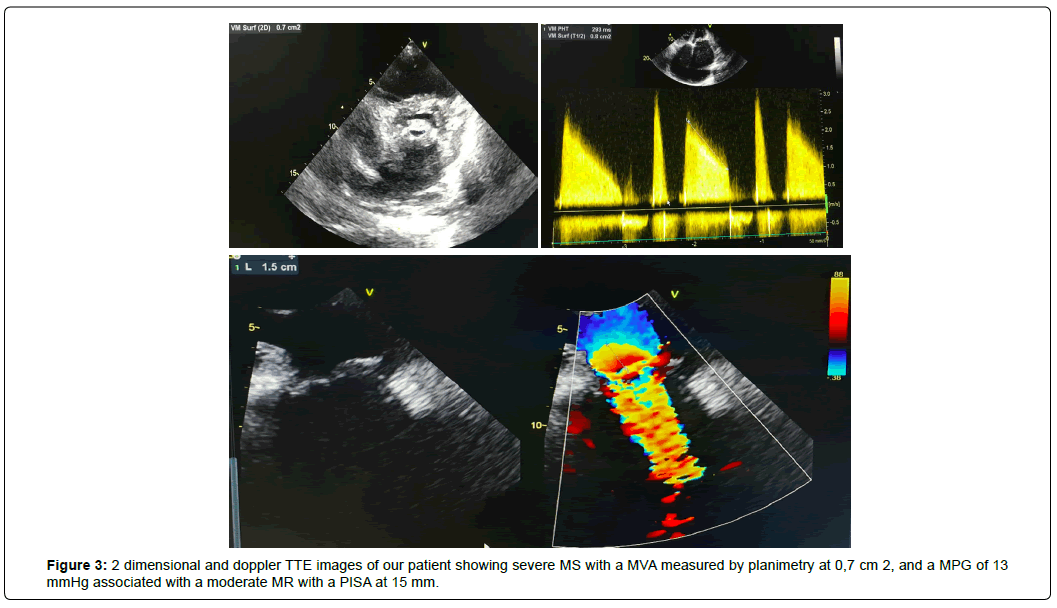
On examination, blood pressure was 106/50 mmHg. Pulse rate was 67/min irregular, she had bilateral pedal oedema, distended neck veins, with no abdominal distention. At auscultation, the first heart sound was loud with a increased intensity of the second sound. Moreover, we heard a grade 5/6 diastolic rumble increasing with expiration in the apex, a grade 3/6 systolic murmur in the aortic area with a systolic thrill in the carotid arteries, as well as separate systolic and diastolic murmurs in the tricuspid area. Electrocardiogram (ECG) showed Atrial Fibrillation (AF) with signs of left and right atrial overload (Figure 1). Chest radiograph showed cardiomegaly with a cardiothoracic ratio of 0.65 (Figure 2). TTE revealed calcified and thickened valve leaflets with severe Mitral Stenosis (MS) with a Mitral Valve Area (MVA) of 0,7 cm2 by planimetry and a Mean Pressure Gradient (MPG) of 13 mmHg associated with a moderate to severe Mitral Regurgitation (MR) with a Proximal Isovelocity Surface Area (PISA) of 15 mm (Figure 3).
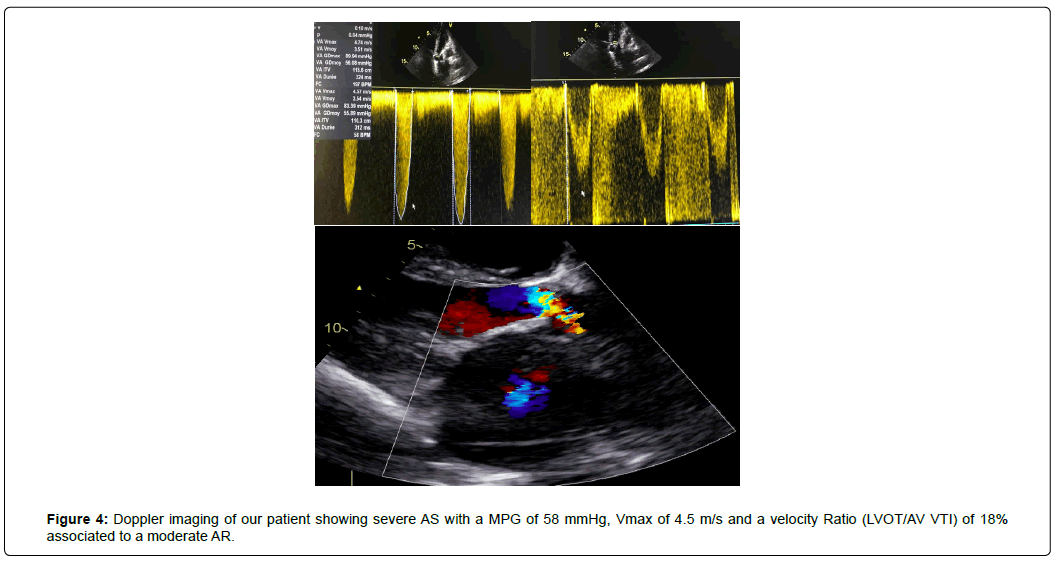
In addition to this, a severe Aortic Stenosis (AS) with a MPG of 58 mmHg, peak aortic valve velocity (Vmax) of 4.5 m/s and a velocity Ratio of 18% associated to a moderate Aortic Regurgitation (AR), were assessed (Figure 4).
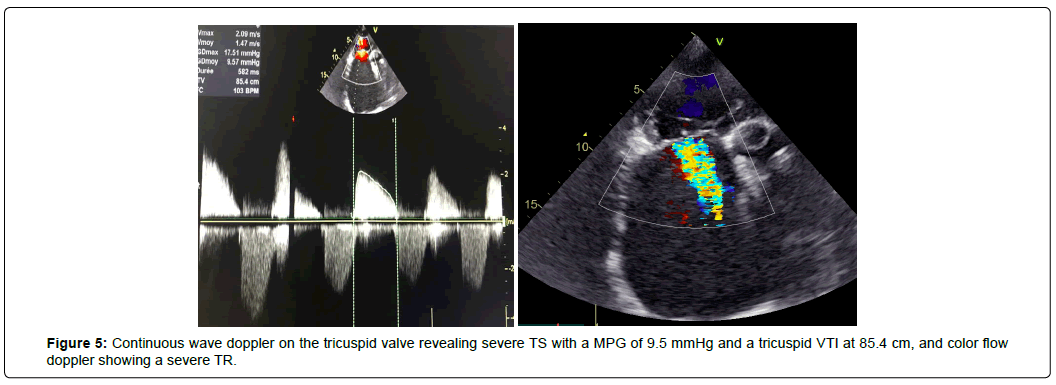
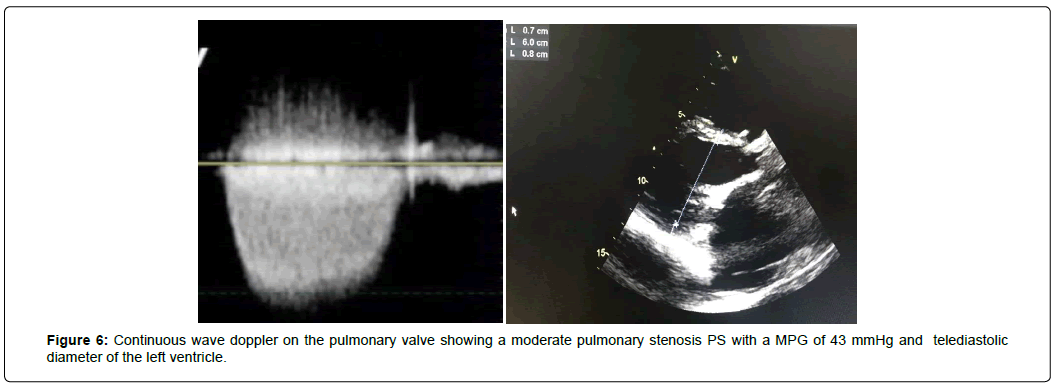
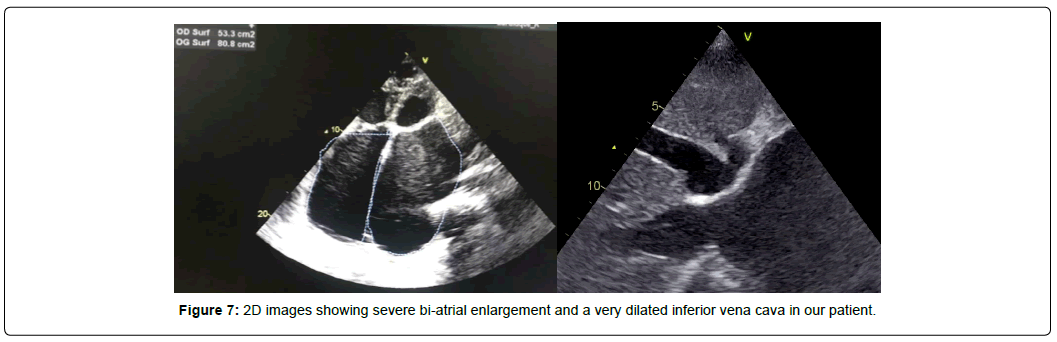
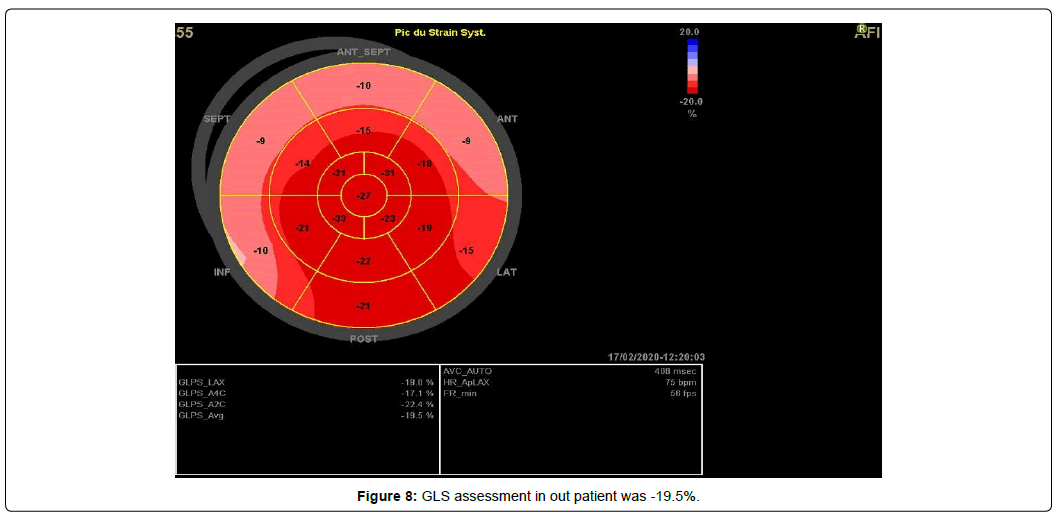
We also found a severe Tricuspid Stenosis (TS) with a MPG of 9.5 mmHg and a tricuspid veloctiy time integral (VTI) of 85.4 cm and a severe organic Tricuspid Regurgitation (TR) (Figure 5), and restricted mobility of pulmonary cusps with moderate PS with a MPG of 43 mmHg (Figure 6) and mild Pulmonary Regurgitation (PR). The patient had bi-atrial enlargement and a very dilated inferior vena cava (figure 7). Biventricular function was normal, with a Left Ventricular Ejection Fraction (LVEF) of 55% and a Global Longitudinal Strain (GLS) of -19.5% (figure 8). LV dimension during systole (LVDs = 24cm/m2) and diastole (LVDd = 30cm/m2) were normal, Interventricular Septal Thickness (IVST) and Posterior Wall Thickness (PWT) were also normal (10 and 9 mm respectively). Pulmonary artery pressure couldn’t be assessed using conventional echocardiographic methods.
All the valvular pathologies’ quantification was made according to the 2017 ESC/EACTS Guidelines for the management of valvular heart diseases [4].
We initiated medical therapy in our patient with low dose furosemide (40 mg/Day), Acenocoumarol 4mg and Digoxin 0.25 mg/Day for her AF. The case was then discussed by the heart-team who decided to try a transcatheter approach given the patient’s high surgical risk, by dilating the aortic valve first, then the mitral and finally the tricuspid valves, in order to avoid sudden increase in cardiac output that may worsen the transaortic valve gradient in case the mitral valve was dilated first.
In time of submission, the patient was getting stabilized under medical treatment and was still awaiting intervention.
Discussion
Overview
MVD (defined by the association of stenotic or regurgitant lesions occurring on more than two cardiac valves) and mixed valvular disease (both stenotic and regurgitant lesions combined on the same valve) are prevalent conditions especially in developing countries such as Morocco where rheumatic etiology is predominant, but also in developed countries where degenerative etiology is increasingly frequent.
However, despite MVD prevalence, few data is available in the literature, and many areas related to the topic are not covered by the current guidelines [4,5], with recommandations mostly graded level C, based on evidence from limited populations or on expert consensus opinion.
This paradox can be explained by the heterogeneity of physio pathological combinations and the fact that hemodynamic interactions between valve lesions can either exacerbate, or blunt the clinical expression of each singular lesion, which can make the diagnosis, quantification and management of MVD, tricky for the heart team.
In the available literature, rheumatic involvement of all four valves is very rare and quadrivalvular stenosis exceptional [6], it is mostly diagnosed on autopsy or cardiac catheterization [7] and rarely by echocardiography [8].
The rarity of studies with echocardiographic diagnosis of this pathology can be imputed to the large decline of rheumatic valvular disease’s prevalence and severity by the time echocardiography became a common diagnosis tool, even in developing countries.
One case described preoperative echocardiographic diagnosis of quadrivalvular rheumatic stenosis with successful surgical treatment [9]. In another case, the same author reported late diagnosis of pulmonary valve stenosis that was diagnosed only at surgery [10], which emphasizes the challenge of this disease and the importance of operator awareness regarding possible quadrivalve damage when MVD is suspected in order to make appropriate therapeutic decisions in collaboration with the heart valve team, considering the severity of MVD, the patient’s comorbidities, in addition to the risks of multiple prostheses and eventual reoperation.
In our work, we reported a rare case of quadrivalvular rheumatic disease in a young patient, diagnosed by echocardiography, highlighting the diagnosis and management challenges encountered in the context of a developing country.
Physiopathology
In patients with MVD, the hemodynamic derangements associated with any given valvular lesion can be modulated by the concomitant presence of another valvulopathy. The clinical consequences depend on the complex interference of several pathophysiological factors such as the specific combination of valves involved, the severity and timing of onset of each individual lesion, the loading conditions and ventricular compensation, making the quantification by echocardiography difficult. Usually, the hemodynamically more severe lesion is responsible of the predominant clinical features; however, if all valvular diseases have similar degrees of severity, the more proximal valve disease may mask the full expression of the more distal valve lesion.
The physiopathology of this phenomenon can be easily explained when two valves are involved ; like it is the case for functional TR secondary to Right Ventricular (RV) dilatation and Pulmonary Artery Hypertension (PAH) in the setting of significant MS, however, in advanced MVD, let alone when all four valves are involved, the pathophysiology of valvular lesions and their evaluation remains very challenging. Such patients often have symptoms of Heart Failure (HF) and valvular lesions are usually accompanied by secondary hemodynamic effects on the cardiac chambers [11,12]. In our patient, fatigue and dyspnea were the only symptoms of left HF but no episodes of acute HF were noted, the LV being most likely protected by the diminished pulmonary blood flow resulting from the rightsided valve stenosis that provided relative compensation of more or less long duration, however, a rapid deterioration can be expected after right heart failure appearance.
Besides, some hemodynamic interactions may impact the diagnosis of such patients mainly depending on flow and gradient [3,13] and eventually, any severe valvular lesion may increase upstream secondary MR or TR [14].
The case of our patient was very difficult to evaluate since she associated severe stenosis of all four cardiac valves with mild to moderate regurgitation.
In mixed aortic/mitral valve disease, when aortic or mitral regurgitation is the dominant lesion, the LV is dilated, however, when stenosis predominates, LV size is normal. In our patient’s case, since valvular stenosis were predominant over regurgitations, we focused only on stenoses. The association of severe mitral and aortic stenosis is responsible of a greater reduction in Cardiac Output (CO) decreasing the flow rate and pressure gradients across both valves, which can falsely underestimate the severity of both AS and MS [15]. Tricuspid valve disease is also highly sensitive to changes in loading conditions and can decrease CO, thus underestimating, the severity of both mitral and aortic disease. Usually, TR is functional and secondary to left-side lesions, but in our case, it was organic secondary to severely uncoated tricuspid valves, and associated to a calcified tricuspid valve responsible of severe TS. Finally, significant acquired rheumatic PS is distinctly unusual – PS being usually congenital- and its physio pathological mechanisms are hardly described since its occurrence is usually in patients with quadrivalvular involvement. In our case, the patient had moderate PS with a peak systolic gradient of 42 mmHg, and we couldn’t precisely assess the pathophysiological interaction with other valvular diseases that were dominant over pulmonary valve disease.
Role of imaging modalities for assessment of MVD
Doppler Echocardiography
According to the recent recommendations on the multimodality imaging in the diagnosis and management of Valvular Heart Disease (VHD) [16], TTE is the cornerstone for assessment of MVD. It’s the most cost-effective way to evaluate valve diseases’ etiology and mechanisms, as well as its consequences on both LV and RV function and on pulmonary circulation, and thus, guiding the clinical and therapeutic decision. However, hemodynamic interactions between valve lesions including changes in stroke volume and in intracardiac pressure, may interfere with several Doppler-echocardiographic measures that have mainly been validated in single-valve disease and might not be valid in MVD [17].
The main hemodynamic interactions that may impact on the diagnosis of MVD are [18]:
Flow and thus gradient reduction by any severe valve disease across another severely stenotic valve.
The increase of anterograde flow and pressure gradient across a stenotic valve in the presence of concomitant regurgitation, with possible overestimation of stenosis severity.
Invalidity of continuity equation to assess valve area in case flows are unequal.
Aortic or pulmonary valve disease may induce or increase MR or TR.
The co-dependance between Left-sided and right-sided valvular lesions.
Inaccuracy of methods derived from pressure half-time assessment in the presence of altered LV compliance/relaxation.
Given the inaccuracy of methods routinely used to assess VHD that may be misleading if not correctly interpreted in the context of MVD and the lack of published data specific to MVD quantification, echocardiography experts preferred measurements less dependent on loading conditions. Direct planimetry of the stenotic valve [4] or, for regurgitant lesions, the assessment of PISA or the vena contracta were the measurements we used in our case to assess mitral, aortic and tricuspid diseases. Concerning the pulmonary valve, we used peak transpulmonary velocity measured with continuous wave Doppler to calculate the transvalvular pressure gradients and found a moderate PS with a peak gradient of 43 mmHg.
For MVD echocardiographic assessment, an integrategrative approche is hence critical in determining the indication and timing for surgery and the likelihood of successful valve repair or of transcatheter valve intervention.
Other imagined modalities
TTE is the main imaging modality used to establish a diagnosis and monitor patients with MVD, but other imaging modalities can be helpful when the information obtained from echocardiography is not sufficient to determine the severity of each individual valvular lesion.
Transesophageal echocardiography plays an important role to precise anatomic before and during percutaneous and surgical procedures [16].
Real-time 3D Transesophageal Echocardiography (TEE) is also useful to accurately measure mitral valve area in rheumatic MS when it is difficult to assess by planimetry due to extensive calcifications and even more if combined with aortic valve disease since anatomical measurements are more reliable than Doppler methods in such cases [19], it may also be used to measure aortic valve area in rheumatic AS allowing direct measurement of LV outflow tract area. Stress echocardiography (SE) using preferably bicycle is indicated when symptoms are disproportionate to the resting hemodynamics [4,5], but can’t be used in our patient since all lesions are severe.
If the echocardiographic evaluations are inconclusive or discordant with clinical assessment, the current guidelines recommend the use of Cardiac Magnetic Resonance (CMR) or invasive hemodynamic assessment for further clarification [4,5]. In fact, CMR provides additional anatomic and physiologic information by quantifying changes in ventricular volumes, mass and function and, may hence contribute to determine the optimal timing for intervention [20]. Finally, right and left heart catheterization may be required to characterize more completely the individual involvement of each lesion, by enabling precise CO measurement in addition to pulmonary artery pressures and calculation of pulmonary vascular resistance, which can guide clinical decision-making in certain patients, such as those with advanced MVD [21].
Management
The limited evidence on medical, surgical, and interventional management of patients with MVD is emphasized by both European [4] and American [5] guidelines given the poor level of evidence for this pathology. Because of the large number of possible combinations of valve lesions, a standardized approach cannot be proposed. However, despite the paucity of data, these guidelines suggest following two general principles : First, the management of MVD should be dictated by the predominant valve lesion and secondly, when valve lesions are of at least moderate severity, the overall impact of the multivalve pathology should be considered severe.
In case ≥ 2 severe lesions with ≥ 2 severely stenotic or regurgitant lesions, and who are symptomatic with ventricular dysfunction or dilatation, severe functional intolerance may occur if one of the lesions is left untreated, therefore, surgical correction of all lesions during a single procedure has been given a class I recommendation in current guidelines [4,5].
However, these recommendations never focused on quadrivalvular lesions, which makes our case much more complicated. In the literature, we only found one case reporting successful quadruple valve replacement in a patient with severe RHD in South Africa [22].
In most studies, despite improvements in surgical techniques, combined valve replacement is associated with significant perioperative risk and poorer survival [23]. In the EuroHeart Survey, the operative risk rose to 6.5% in cases of MVD versus 0.9% to 3.9% for single-valve interventions [3].
Despite a substantial decline in short- and long-term mortality [23], quadruple valve replacement remains a challenging procedure, therefore, mitral and tricuspid valves repair is preferred whenever possible. However, valve repair procedure may be tricky in RHD due to the leaflets’ thickening and calcification, and Trans-catheter treatments have several contraindications. Percutaneous Mitral Balloon Valvuloplasty (PMBV) for MS is contraindicated in presence of moderate or severe MR, as well as Transcatheter Aortic Valve Replacement (TAVR) is not performed in the presence of severe, coexistent AR. Though, combining both TAVR for AS and MitraClip® for functional MR has been reported in addition to transcatheter approach for tricuspid disease [4,5].
In a propensity-matched analysis of patients with aortic and mitral valve disease, Aortic Valve Replacement (AVR) and Mitral Valve Repair (MVR) improved late survival compared with double valvular replacement [24]. Similarly, in another study of patients with RHD, MVR associated with AVR improved eventfree survival compared with the replacement of both valves [25], thus suggesting that efforts should be directed towards improving rates of mitral valve repair.
If valve repair is impossible and multiple prosthetic valves must be considered, no data are available on which type of valve prosthesis is preferable.
However, it is recommended to use the same type of prothesis (bio prosthesis or mechanical) for the left side lesions in order to avoid superimposing the risk of anticoagulation and the risk of bio prosthesis deterioration [5]. For tricuspid valve disease, tricuspid annuloplasty is the preferred technique since it has a lower rate of complications than tricuspid valve replacement [26]. In case of combined pulmonic and tricuspid valve disease, pulmonary valve replacement in addition to tricuspid valve replacement has been advocated even in some patients having relatively mild pulmonary valve disease [27].
In practice and particularly in the context of a developing country such Morocco, in view of the higher risks, a higher threshold is required for MVD versus single-valve surgery and the surgical decision-making process should take into account several factors. First, the increased surgical risk of combined surgeries and the longterm increase in morbidity associated with thrombotic and bleeding complications of multiple prosthetic valves. Second, the likelihood of spontaneous changes in MR or TR after surgery on a downstream valvular lesion. Third, the risk of eventual reoperation and the prognostic effect of not correcting the non-severe lesions. Fourth, the choice between surgery or percutaneous intervention and eventually the choice of the appropriate surgical technique whether it is valve repair or replacement. A collaborative approach between a heart team with expertise in cardiac surgery, transcatheter interventions, cardiac imaging, hemodynamics and anesthesia is critical in the management of patients with MVD, which is also highlighted in the current guidelines [4,5].
Conclusion
Despite the prevalence of Rheumatic multiple and mixed VHDs especially in developing countries, quadrivalvular disease remains an exceptional entity in which paucity of data does not allow to follow a standardized, evidence-based management strategy, thus the management of these patients remains empirical and most often challenging. Due to the complex hemodynamic interactions between coexistent valve lesions, a careful quantification and a comprehensive assessment of the consequences of the valve lesions are required to propose an optimal therapeutic strategy for each patient.
A case-by-case therapeutic management strategy should be made by a Heart Valve team, based on symptoms, severity and hemodynamic significance of valvular lesions, the individual risk profile, the risks of combined valve surgery and the risk of reintervention. Current and future advances in transcatheter valve therapies are likely to change the therapeutic approach of MVD, but further studies are needed to guide therapeutic decision-making and to appreciate the real benefits of these emerging treatments.
References
- Ghanem, Rabat. MAR (2012) Epidemiology of rheumatic fever in Morocco: Description of surveillance data collected between 2000 and 2010. Ann Ref Bibl. 24.
- David AW, Catherine OJ, Samantha MC, Ganesan K, Andrea B, et al. (2017) Global, regional, and national burden of rheumatic heart disease, 1990–2015. N Eng J Med. 377:713–722.
- Iung, B, Baron G, Butchart EG, Delahaye F, Gohlke-Bärwolf C, et al. (2013) A prospective survey of patients with valvular heart disease in europe: the euro heart survey on valvular heart disease. Eur Heart J. 24:1231–1243.
- Baumgartner H, Falku V, Bax JJ, Bonis M, Hamm C, et al. (2017) 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J. 38:2739–2791.
- Nishimura RA, Otto CM, Bonow RO, Carabello BA, Erwin JP, et al. (2017) 2017 AHA/ACC Focused update of the 2014 AHA/ACC guideline for the management of patients with valvular heart disease a report of the american college of cardiology/american heart association task force on clinical practice guidelines. Circulation. 135:e1159–e1195
- Gialloreto O, Aerichide N, Allard PP (1961) Stenotic involvement of all four heart valves. Report of three cases. Am J Cardiol. 7:865-873.
- Hardin BL, Daniels WB (1942) Tricuspid stenosis: report of a case with involvement of all four valves of the heart. Ann Jut Med. 17:536.
- Bandin MA, Vargas-Barron J, Keirns C, Romero-Cardenas A, Villegas M, et al. (1990) Echocardiographic diagnosis of rheumatic cardiopathy affecting all four cardiac valves. Am Heart J. 120:1004–1007.
- Kumar N, Rasheed K, Gallo R, Al-Halees Z, Duran CM, et al. (1995) Rheumatic involvement of all four heart valves: preoperative echocardiographic diagnosis and successful surgical management. Eur J Cardiothorac Surg. 9:713–714.
- Kumar AS, Iyer KS, Chopra P (1985) Quadrivalvular heart disease. Int J Cardiol. 7:66–69.
- Patricia A, Pellikka (2018) Tricuspid, pulmonic, and multivalvular disease. in: braunwald's heart disease: a textbook of cardiovascular medicine. (11th edn), Disease of the heart valves.
- Unger P, Clavel MA, Lindman BR, Mathieu P, Pibarot P, et al. (2016) Pathophysiology and management of multivalvular disease. Nat Rev Cardiol. 13:429-440.
- Andell P, Li X, Martinsson A, Andersson C, Stagmo M, et al. (2017) Epidemiology of valvular heart disease in a Swedish nationwide hospital-based register study. Heart. 103:1696–1703.
- Lung B, Baron G, Tornos P, Gohlke-Bärwolf C, Butchart EG, et al. (2007) Valvular heart disease in the community: a European experience. Curr Probl Cardiol. 32:609–661
- Honey M (1961) Clinical and haemodynamic observations on combined mitral and aortic stenosis. Br Heart J. 23:545–555.
- Doherty JU, Kort S, Mehran R, Schoenhagen P, Soman P, et al. (2018) ACC/AATS/AHA/ASE/ASNC/HRS/SCAI/SCCT/SCMR/STS 2017 Appropriate use criteria for multimodality imaging in valvular heart disease: a report of the american college of cardiology appropriate use criteria task force, american association for thoracic surgery, american heart association,american society of echocardiography, american society of nuclear cardiology, heart rhythm society, society for cardiovascular angiography and interventions, society of cardiovascular computed tomography, society for cardiovascular magnetic resonance, and society of thoracic surgeons. J Am Soc Echocardiogr. 31:381–404.
- Unger P, Lancellotti P, de Canniere D (2016) The clinical challenge of concomitant aortic and mitral valve stenosis. Acta Cardiol. 71:3–6
- Unger P, Maurer G (2016) Heart valve disease: mixed valve disease, multiple valve disease, and others. (1st Edi) Oxford University Press. Oxford Medicine Onlie.
- Schlosshan D, Aggarwal G, Mathur G, Allan R, Cranney G, et al. (2011) Real-time 3D transesophageal echocardiography forthe evaluation ofrheumatic mitral stenosis. JACC Cardiovasc Imaging. 4:580–588.
- Hundley WG, Bluemke DA, Finn JP, Flamm SD, Fogel MA, et al. (2010) ACCF/ACR/AHA/NASCI/SCMR 2010 expert consensus document on cardiovascular magnetic resonance: a report of the american college of cardiology foundation task force on expert consensus documents. Circulation. 121:2462-2508.
- Zoghbi WA, Adams D, Bonow RO, Enriquez-Sarano M, Foster E, et al. (2017) Recommendations for noninvasive evaluation of native valvular regurgitation: A report from the american society of echocardiography developed in collaboration with the society for cardiovascular magnetic resonance. J Am Soc Echocardiogr. 30:303-371.
- Adembesa I, Myburgh A, Swanevelder J (2018) Quadruple valve replacement in a patient with severe rheumatic heart disease. Echo Res Pract. 5:K47–K51.
- Alsoufi B, Rao V, Borger MA, Feindel CM, Scully HE, et al. (2006) Short- and long-term results of triple valve surgery in the modern era. Ann Thorac Surg. 81:2172-2178.
- Gillinov AM, Blackstone EH, Cosgrove DM, White J, Kerr P, et al. (2003) Mitral valve repair with aortic valve replacement is superior to double valve replacement. J Thorac Cardiovasc Surg. 125:1372–1387.
- Talwar S, Mathur A, Choudhary SK, Singh R, Kumar AS, et al. (2007) Aortic valve replacement with mitral valve repair compared with combined aortic and mitral valve replacement. Ann Thorac Surg. 84:1219–1225.
- Chikwe J, Itagaki S, Anyanwu A, Adams DH (2015) Impact of concomitant tricuspid annuloplasty on tricuspid regurgitation, right ventricular function, and pulmonary artery hypertension after repair of mitral valve prolapse. J Am Coll Cardiol. 65:1931–1938.
- Bhattacharyya S, Davar J, Dreyfus G, Caplin ME (2007) Carcinoid heart disease. Circulation. 116:2860-2865.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi