Research Article, Int J Cardiovasc Res Vol: 8 Issue: 6
Physical Fitness Study in Children with Previous Surgery for Congenital Heart Disease
Amr M Kotb*, Salah-Eldin Amry, Khalid I Elsayh and Ahmed M Ghoneim
Pediatric Department, Faculty of Medicine, Assiut University, Egypt
*Corresponding Author: Amr Mohamed Kotb
Faculty of Medicine, Assiut University, 47 Al-azhar University Street, Assiut 71111, Egypt
Tel: +201009898678
E-mail: amrkotb336@aun.edu.eg; amrkotb336@yahoo.com
Received: April 30, 2019 Accepted: August 05, 2019 Published: August 12, 2019
Citation: Kotb AM, Amry S, Elsayh KI, Ghoneim AM (2019) Physical Fitness Study in Children with Previous Surgery for Congenital Heart Disease. Int J Cardiovasc Res 8:6.
Abstract
Introduction: The six minute walk test (6MWT) is generally used for evaluating the functioning of people with chronic diseases; the main result is the distance a person may walk for 6 minutes. The 6MWT was originally developed to measure the submaximal level of functioning in adult cardiac or chronic disease patients with moderate-to severe heart or lung disease and has been widely used in other patient subgroups. Because the test provides a level of training close to that of daily life, it is easy to use and increasingly is being used as a functional outcomes measurement for people with chronic disease, to include pediatric populations.
Patients and methods: Our study is cross sectional study included 100 cases (55 males and 45 females) who had congenital heart diseases surgically repaired in one stage (34 had Tetralogy of Fallot, 27 had ventricular septal defect, 15 had atrial septal defect, 10 had patent ductus arteriosus, 5 had congenital valvular aortic stenosis, 4 had sub aortic membrane, 3 had coarctation of aorta and 2 had d-transposition of great arteries). The study included pediatric patients who had been admitted to the pediatric cardiology department of Assiut Children’s Hospital from 5 to 18 years of age and for a one year period from 1st August 2016 to 31st July 2017. Control: One hundred (100) healthy children (55 males, 45 females).
Results and conclusion: The 6MWT is a simple, applicable test with a significant value in the demonstration of the effect of one stage surgically repaired congenital heart diseases on the functional capacity of pediatric cardiac patients
Keywords: Six-minute walk test; Pediatric; Exercise; Congenital heart diseases
Introduction
The ability to walk a distance is a quick and easy way to determine the physical function of an individual. It is also an important part of the QOL because it reflects the ability to exercise daily life [1-4]. Balke developed a simple test to check the functioning by measuring the distance travelled over a defined time period [5]. A 12-minute performance test was then developed to assess the physical fitness of healthy people [6]. This test was then modified for use in patients with chronic bronchitis [7]. To enable patients with respiratory diseases for whom 12-minute walking was too demanding, a shortened version of 6 minutes was found to provide comparable clinical information [8]. A recent review of the functional walking attempts concluded that the six-minute walking test (6MWT) is simple to perform, better accepted by individuals and provides better reflection of daily lifestyle activities than other walking tests [4].
The American Thoracic Society has also recently approved and published a guideline for the clinical use of 6MWT [9], 6MWT has often been used to measure pre- and post-treatment results in patients with moderate to severe heart and lung disease [10].
Aim of the Work
Evaluations of the functional capacity of children with surgically repaired congenital heart disease through assessment of 6MWT and compare the results to those of healthy children of the same age.
Patients and Methods
Patients
The study included 100 cases (55 males and 45 females) who had congenital heart diseases surgically repaired in one stage (34 had Tetralogy of Fallot, 27 had ventricular septal defect, 15 had atrial septal defect, 10 had patent ductus arteriosus, 5 had congenital valvar aortic stenosis, 4 had subaortic membrane, 3 had coarctation of aorta and 2 had d- transposition of great arteries.
Control group
One hundred (100) healthy children (55 males, 45 females). From primary, preparatory and secondary schools from the same age category of the patients.
Inclusion criteria
Children age between 5 and 18 years old with previous one-stage corrective surgery for congenital heart disease after full recovery from acute illness.
Exclusion criteria
Children with acute respiratory condition, children with chronic systemic illness like respiratory, hepatic, renal, neurologic, or GIT conditions, children with skeletal disorders, children with hemodynamic disorders like anemia or thyrotoxicosis and cases with cardiac arrhythmia as detected by ECG.
Tools of study
ALL cases (patients and control) included in the study were subjected to Anthropometric measures like (weight and height) according to methods described by Reiter and Rosenfield. 6MWT was performed according to the guidelines of the American Thoracic Society [9].
Patient preparation
All patients’ worn comfortable clothes, appropriate shoes for walking were worn, the patient’s usual medical regimen was continued, a light meal was taken before the test and patients were not exercised vigorously 2 hours before the test.
Safety issues: The test was performed in the pediatric cardiology unit of the Children’s Hospital of the University of Assiut, where a rapid emergency response was obtained. The oxygen source and the telephone were installed to enable the request for help.
Technical aspects of the 6MWT location
The 6MWT was performed in pediatric cardiology unit at Assiut University children hospital, where a long, flat, straight, enclosed corridor at a course of 30 m was assigned. The length of the corridor was marked every 3 m. The turnaround point was marked with a cone.
Results and Discussion
The six-minute walking test (6MWT) is a functioning test performed in a conceptional submaximal effort proposed as a reflective of the activities of daily life [11]. Since the development of the 6MWT in the initial phase, in 1970s [12], this test became increasingly significant in clinical practice and research. 6MWT has been gaining importance in clinical practice and in the scientific context because it is easy to apply, has low costs and the maximum distance travelled is a high diagnostic value for several cardiopulmonary diseases [13,14]. This test is also often used to assess exercise capacity both before and after an intervention, such as a training programme [12].
Surgical correction of congenital heart diseases (CHD) has contributed to a substantial increase in the survival rates of these patients, resulting in a quality of life close to normal [15]. However, reduced exercise capacity, may occur in patients, even after surgical correction of the disease [16].
This cross-sectional case-control study aimed at the evaluation of children with congenital cardiac defects that were corrected surgically in Assiut university hospitals to detect their anatomical and functional cardiac condition. In addition, the 6MWT was used to evaluate their exercise capacity. No significant difference could be detected between the studied patients and the control cases regarding age, sex and most of the subjects from rural area (Table 1). For every patient studied, an age and sex matched healthy control was studied.
| Variables | Cases (n=100) | Control (n=100) | p value | ||
|---|---|---|---|---|---|
| No. | % | No. | % | ||
| Age | |||||
| From 5 to 12 years | 67 | 67 | 67 | 67 | NS |
| From 12 to 18 years | 33 | 33 | 33 | 33 | |
| Mean ± SD | 10.04 ± 3.63 | 10.68 ± 4.03 | NS | ||
| Sex | |||||
| Male | 55 | 55 | 55 | 55 | NS |
| Female | 45 | 45 | 45 | 45 | |
| Residence | |||||
| Rural | 90 | 90 | 97 | 97 | 0.085 |
| Urban | 10 | 10 | 3 | 3 | |
Table 1: Demographic data of the studied cases with one-stage surgery corrected congenital cardiac defects and controls.
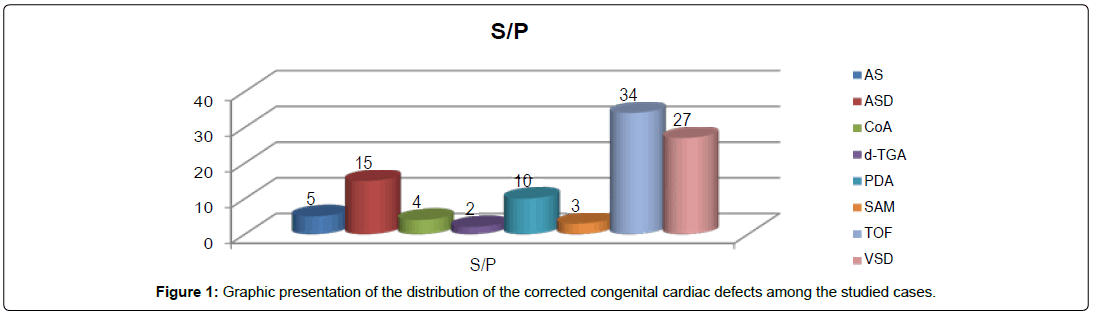
Regarding distribution of the corrected cardiac defects among our studied patients (Table 2), it was observed that tetralogy of Fallot followed by ventricular septal defect, and then atrial septal defects and patent ductus arteriosus were the common defects corrected. This represents the surgical state of art in our pediatric cardiac center during this period. All defects in this study were corrected in one stage and without using synthetic conduits or patches (Figures 1 and 2).
| Cardiac defects corrected surgically | Cases (n=100) | |
|---|---|---|
| No. | % | |
| Tetralogy of Fallot (TOF) | 34 | 34 |
| Ventricular septal defects (VSD) | 27 | 27 |
| Atrial septal defects (ASD) | 15 | 15 |
| Patent ductus arteriosus (PDA) | 10 | 10 |
| Valvar aortic stenosis | 5 | 5 |
| coarctation of the aorta (COA) | 4 | 4 |
| subaortic membrane (SAM) | 3 | 3 |
| Transposition of the great arteries (d-TGA) | 2 | 2 |
Table 2: Distribution of the corrected congenital cardiac defects among the studied cases.
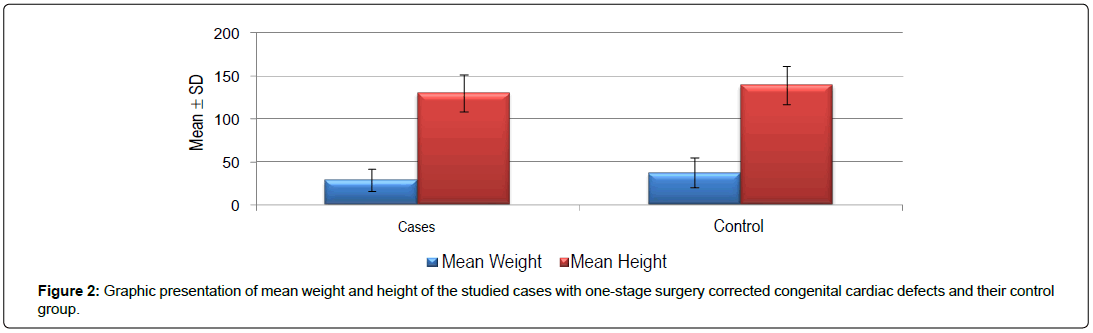
It was observed that the mean weight and height was significantly higher in the control group than in the patient group (Table 3). This is in agreement with the results found by Norozi et al. [16]. These finding could be explained by the fact that malnutrition is common in children with congenital heart defects [17]. The mechanisms for growth deficiency in CHD are multifactorial [7-10] and include associated chromosomal anomalies/genetic syndromes, inadequate nutritional intake due to feeding difficulties, and poor absorption of nutrients from the digestive tract in chronic congestive heart failure (CHF). Also, increased calories are required to sustain the increased myocardial, respiratory and neuro-humoral functions in CHDrelated heart failure. Chronic CHF and chronic hypoxaemia in CHD impair cellular metabolism and cell growth, while repeated chest infections are associated with increased metabolic demands [17].
| Variables | Cases (n=100) | Control (n=100) | p value |
|---|---|---|---|
| Mean ± SD | Mean ± SD | ||
| Mean Weight (kg) | 28.46 ± 12.892 | 37.05 ± 17.391 | <0.001** |
| Mean Height (cm) | 130.12 ± 21.720 | 139.45 ± 22.321 | 0.003** |
Table 3: Mean weight and height of the studied cases with one-stage surgery corrected congenital cardiac defects and their control group.
It was observed that the mean distance walked in the 6MWT by our cases with surgically corrected congenital cardiac defects was significantly lower than the control group (Figure 3 and Table 4). This applied to both males and females (Table 5). Our results in this regard are keeping with Feltz et al. [15]. Congenital heart diseases are associated with organic abnormalities, both previously as after surgical correction of the defect. Among these, changes in nutritional status, height and weight as well as reduced exercise capacity are included. The causes of reduced exercise capacity in CHD patients may be related to hemodynamic changes, to electrical conduction disturbances, ventricular pressure and volume overload, change in simpatovagal activity, increased neurohumoral activity, reduction in lung function and changes in muscle metabolism [18].
| Cases (n=100) | Control (n=100) | p value | |||
|---|---|---|---|---|---|
| Range | Mean ± SD | Range | Mean ± SD | ||
| 6MWD | 385-512 | 440.77 ± 38.13 | 466-700 | 600.22 ± 54.5 | <0.001 |
Table 4: The mean walked distance during the 6MWT by cases and controls.
| Sex | No. | The mean distance of 6MWD of the cases (Mean ± SD) | No. | The mean distance of 6MWD of the controls (Mean ± SD) | p value |
|---|---|---|---|---|---|
| Males | 55 | 446 ± 37.79 | 55 | 621.36 ± 54.88 | <0.001 |
| Females | 45 | 434.38 ± 37.99 | 45 | 579.08 ± 45.58 | <0.001 |
Table 5: The mean walked distance during the 6MWT by cases and controls in male and female children.
This is further supported our results in Table 3 that show statistically significant decrease in weight and height of our cases compared to the control group.
The physiologic response of our cases with surgically corrected congenital cardiac defects after exercise testing using the 6MWT was studied by measuring vital signs and oxygen saturation before and after the test in both cases and controls (Tables 6-9).
| Variables | Before | After | p value |
|---|---|---|---|
| Mean Heart Rate | 97.9 ± 8.21 | 99.97 ± 8.48 | NS |
| Mean Respiratory Rate | 22.3 ± 3.73 | 29.46 ± 3.41 | <0.001** |
| Mean Blood Pressure | |||
| Systolic | 88.11 ± 9.32 | 98.43 ± 9.59 | <0.001** |
| Diastolic | 60.55 ± 7.64 | 61.31 ± 7.74 | NS |
| S O2 | 95.2 ± 0.68 | 94.61 ± 1.81 | NS |
Table 6: Vital signs and pulse oxymetry results in the studied cases before and after the 6MWT.
| Variables | Before | After | p value |
|---|---|---|---|
| Mean Heart Rate | 89.18 ± 11.99 | 98.86 ± 9.71 | <0.001** |
| Mean Respiratory Rate | 18.1 ± 2.59 | 24.04 ± 3.38 | <0.001** |
| Mean Blood Pressure | |||
| Systolic | 90 ± 9.32 | 103.5 ± 8.92 | <0.001** |
| Diastolic | 60.5 ± 6.72 | 61 ± 6.44 | NS |
| S O2 | 96.03 ± 1.77 | 96.59 ± 0.93 | NS |
Table 7: Vital signs and pulse oxymetry results in controls before and after the 6MWT.
| Variables | Cases | Controls | p value |
|---|---|---|---|
| Mean Heart Rate | 97.9 ± 8.21 | 89.18 ± 11.99 | <0.001** |
| Mean Respiratory Rate | 22.3 ± 3.73 | 18.1 ± 2.59 | <0.001** |
| Mean Blood Pressure | |||
| Systolic | 88.11 ± 9.32 | 90 ± 9.32 | NS |
| Diastolic | 60.55 ± 7.64 | 60.5 ± 6.72 | NS |
| S O2 | 95.2 ± 0.68 | 96.03 ± 1.77 | NS |
Table 8: Vital signs and pulse oxymetry results in cases and controls before the 6MWT.
| Variables | Cases | Controls | p value |
|---|---|---|---|
| Mean Heart Rate | 99.97 ± 8.48 | 98.86 ± 9.71 | NS |
| Mean Respiratory Rate | 29.46 ± 3.41 | 24.04 ± 3.38 | <0.001** |
| Mean Blood Pressure | |||
| Systolic | 98.43 ± 9.59 | 103.5 ± 8.92 | <0.01** |
| Diastolic | 61.31 ± 7.74 | 61 ± 6.44 | NS |
| S O2 | 94.61 ± 1.81 | 96.59 ± 0.93 | NS |
Table 9: Vital signs and pulse oxymetry results in cases and controls after the 6MWT.
It was observed that there was no statistically significant difference in the mean heart rate in cases before and after the exercise test, while statistically significant difference occurred in the control group. The mean heart rate in cases was significantly higher the that of the control group before the exercise test while no statistically significant difference could be detected after the exercise test. This is in agreement with Norozi et al. [16], Massin et al. [19], and Heiberg et al. [20]. It could be explained by the permanent chronotropic impairment that occur after surgical correction of cardiac defects either simple like ASD and VSD or complex like tetralogy of Fallot.
Factors like sedentary lifestyle that may reflect both inherent physiological limitations in addition to overprotection of parents [21]. Residual defects after surgery may contribute to this chronotropic impairment [16,19]. The activity of the sympathetic and parasympathetic nervous system, which plays an important role in the modulation of HR during exercise, can be affected by ischemia and/or denervation resulting from surgical procedure or, in cases of cyanotic CHD, by chronic hypoxemia [19]. Massin et al. concluded that the chronotropic impairment is significantly less important after trans catheter closure of atrial septal defect than after surgical closure (Figure 4).
It was observed that the mean respiratory rate was significantly higher after exercise test both in cases and controls. It was also observed that the mean respiratory rate was significantly higher in cases that in controls before and after the exercise test. Increased respiratory rate is an indication of decreased (VO2 max). Maximal oxygen consumption (VO2 max) has been widely used as gold standard for evaluation of functional capacity in healthy or ill individuals. There is a difference in cardiorespiratory responses between adults and children. The anatomically smaller heart size in children results in lower venous return, and therefore lowers cardiac output, which in turn results in lower VO2 max when compared with adults. Therefore, the most important compensatory mechanism for children is through the increase in HR. During exercise, the systolic volume increases around 20% in a normal heart, and the further increase in the cardiac output is due to an increase in HR. Although expected, the information that children and adolescents with CHD in fact have lower functional capacity than their peers, even after corrective surgery, is first summarized in the meta-analysis of Schaan et al. [21].
Individuals with CHD have insufficient chronotropic response, which leads to a decreased maximum HR, consequently reducing the VO2 max in this population [22]. Our results in this regard are also in agreement with those concluded by Reybrouck et al. [23].
It was observed that there was statistically significant increase in the mean systolic blood pressure after the exercise test both in cases and controls while no significant increase in the mean diastolic pressure was detected. While no statistically significant difference could be detected between cases and controls in the mean systolic and diastolic blood pressure before the exercise test, the mean systolic blood pressure in controls was significantly higher than that in cases after the exercise test. Systolic blood pressure rises with increased dynamic work load as a result of increasing cardiac output. At each level of exercise there is a more consistent increase in systolic blood pressure during the first few minutes, and then a steady state is attained. Systolic blood pressure generally correlates with the maximal exercise level achieved.
You can define normal values for maximum systolic blood pressure. After maximum exercise there is a decrease in systolic blood pressure, which usually reaches a basal level in six to seven minutes and then often remains below the level before exercise for several hours. During exercise, there is immediate vasodilatation of the arteries and capillaries in active skeletal muscle tissue due to increased metabolic demand, compared to tissue that is not involved when peripheral vascular resistance increases. The overall result is a decrease in total systemic vascular resistance. While systolic blood pressure increases simultaneously, diastolic pressure usually remains more or less the same [24].
An inadequate increase or fall in systolic blood pressure during exercise can be caused by aortic outflow obstruction, severe mitral disease, left ventricular dysfunction or limited compliance of the systemic ventricle. Additionally, conditions of reduced preload can affect significantly the systolic blood pressure by a reduction of the stroke volume during exercise. Since changes of blood pressure also depend on peripheral resistance, they reflect more than the contractile function of the left ventricle [25].
The significant increase in systolic blood pressure in controls than in cases after exercise test could be explained by the significant difference in weight and height between the two groups (Table 3) and by the insufficient chronotropic response occurring in cases with congenital heart disease after surgical correction [19].
Conclusion
Exercise testing should be an essential part of the evaluation of children and adolescents with congenital heart disease before and after intervention.
The six-minute walk test is a simple noninvasive easy to perform and cheap exercise test that should be used for evaluation of children and adolescents to detect the ordinary effort performance and its change after intervention for congenital heart disease.
Obtaining normal body measurements and physical fitness of children with congenital heart disease should be the goal of all personnel caring for those children by early intervention, proper nutritional management, physical fitness rehabilitation and avoiding overprotection and activity limitation.
Recommendations
Researches should be directed to the factors improving physical fitness of children with congenital heart disease like preoperative preparation, time of intervention, mode of intervention, postoperative rehabilitation in each individual congenital cardiac defect.
References
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories (2002) ATS statement: guidelines for the Six Minute Walk Test. Am J Respir Crit Care Med 166: 111-117.
- Kierkegaard M, Harms-Ringdahl K, Edstrom L (2011) Feasibility and effects of a physical exercise programme in adults with myotonic dystrophy type 1: arandomized controlled pilot study. J Rehabil Med 43: 695-702.
- Bart B, de Groot JF, Terwee CB (2013) The Six-Minute Walk Test in Chronic Pediatric Conditions: A Systematic Review of Measurement Properties. Phys Ther 93: 529-541.
- Solway S, Brooks D, Lacasse Y, Thomas SA (2001) Qualitative systemic overview of the measurement properties of functional walk tests used in the cardiorespiratory domain. Chest 119: 256-270
- Balke B (1963) A simple field test for the assessment of physical fitness. Rep Civ Aeromed Res Inst US 53: 1-8.
- Cooper KH (1968) A means of assessing maximal oxygen intake: correlation between field and treadmill testing. JAMA 203: 201-204.
- Mcgavin CR, Gupta SP, Mchardy GJ (1976) Twelve-minute walking test for assessing disability in chronic bronchitis. Br Med J 1: 822-823.
- Butland RA, Pang J, Gross ER, Woodcock AA, Geddes DM, et al. (1982) Two-,six- and 12-minute walking tests in respiratory disease. BMJ 284: 1607- 1608.
- American Thoracic Society (2002) ATS statement: Guidelines for the six-minute walk test. Am J Respir Crit Care Med 166: 111- 117.
- Enright PL (2003) The six-minute walk test. Respir Care 48: 783-785.
- Nery RM, Martini MR, Vidor Cda R, Mahmud MI, Zanini M, et al. (2010) Changes in functional capacity of patients two years after coronary artery bypass grafting surgery. Rev Bras Cir Cardiovasc 25: 224-228.
- ATS Committee on Proficiency Standards for Clinical Pulmonary Function Laboratories (2002) ATS statement: guidelines for the six-minute walk test. Am J Respir Crit Care Med 166: 111-117.
- Maltais F, Decramer M, Casaburi R, Barreiro E, Burelle Y, et al. (2004) An official American Thoracic Society/European Respiratory Society statement: update on limb muscle dysfunction in chronic obstructive pulmonary disease. Am J Respir Crit Care Med 189: 15-62.
- Boxer R, Kleppinger A, Ahmad A, Annis K, Hager D, et al. (2010) The 6- minute walk is associated with frailty and predicts mortality in older adults with heart failure. Congest Heart Fail 16: 208-213.
- Feltez G, Coronel CC, Pellanda LC, Lukrafka JL (2015) Exercise capacity in children and adolescents with corrected congenital heart disease. Pediatr Cardiol 36: 1075-1082.
- Norozi K, Gravenhorst V, Hobbiebrunken E, Wessel A (2005) Normality of cardiopulmonary capacity in children operated on to correct congenital heart defects. Arch Pediatr Adolesc Med 159:1063-1068.
- Okoromah CAN, Ekure EN, Lesi FEA, Okunowo WO, Tijani BO (2011) Prevalence, profile and predictors of malnutrition in children with congenital heart defects: a case–control observational study. Arch Dis Child 96: 354-360.
- Ulrich S, Hildenbrand FF, Treder U, Fischler M, Keusch S, et al. (2013) Reference values for the 6-minute walk test in healthy children and adolescents in Switzerland. BMC Pulmonary Med 13: 49.
- Massin MM, Dessy H, Malekzadeh-Milani SG, Khaldi K, Topac B, et al. (2009) Chronotropic impairment after surgical or percutaneous closure of atrial septal defect. Catheter Cardiovasc Interv 73: 564-567.
- Heiberg J, Nyboe C, Hjortdal VE (2017) Permanent chronotropic impairment after closure of atrial or ventricular septal defect. Scand Cardiovasc J 51: 271-276.
- Schaan CW, Pinto de Macedo AC, Sbruzzi G, Umpierre D, Schaan BD, et al. (2017) Functional Capacity in Congenital Heart Disease: A Systematic Review and Meta-Analysis. Arq Bras Cardiol 109: 357-367.
- Amiard V, Jullien H, Nassif D, Maingourd Y, Ahmaidi S (2007) Relationship between dyspnea increase and ventilatory gas exchange thresholds during exercise in children with surgically corrected heart impairment. Int J Sports Med 28: 333-339.
- Reybrouck T, Boshoff D, Vanhees L, Defoor J, Gewillig M (2004) Ventilatory response to exercise in patients after correction of cyanotic congenital heart disease: relation with clinical outcome after surgery. Heart 90: 215-216.
- Hauser M (2003) Exercise blood pressure in congenital heart disease and in patients after coarctation repair. Heart 89: 125-126
- Gewillig MH, Lundström UR, Bull C, Wyse RKH, Deanfield JE (1990) Exercise responses in patients with congenital heart disease after fontan repair: Patterns and determinants of performance. J Am Coll Cardiol 15: 1424-1432.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi