Research Article, J Plant Physiol Pathol Vol: 10 Issue: 5
Physiological and Biochemical Studies in Black Gram as Affected By Iron
Rashmi Upadhyay* and Yogesh Kumar Sharma
Department of Botany, University of Lucknow, Lucknow, India
*Corresponding Author: Rashmi Upadhyay, Department of Botany, University of Lucknow, Lucknow, India, E-mail: rashmilk11@gmail.com
Received Date: 31 March, 2022, Manuscript No. JPPP-22-54318;
Editor assigned Date: 04 May, 2022; Pre QC No. JPPP-22-54318 (PQ);
Reviewed Date: 13 May, 2022, QC No. JPPP-22-54318;
Revised Date: 22 May, 2022, Manuscript No: JPPP-22-54318 (R);
Published Date: 29 May, 2022, DOI: 10.4172/ 2329-955X.1000322.
Citation: Upadhyay R, Sharma YK (2022) Physiological and Biochemical Studies in Black Gram as Affected By Iron. J Plant Physiol Pathol 10:4.
Abstract
The response of black gram (Vigna mungo L.) at different levels i.e. control (5.6 ppm), deficient (1.4 ppm) and toxic (56 ppm) of iron was investigated. Deficient and toxic levels of iron suppressed the growth of black gram seedlings as compared to control. A significant reduction was seen in radicle and plumule length, and biomass of plant at deficient level of iron as compared to toxic. The photosynthetic pigments were also found to be reduced at deficient level of iron than toxic and control. Iron deficiency and toxicity were associated with an increase in the activities of catalase and peroxidase. Both iron deficiency and toxicity decreased the concentration of DNA, RNA and protein contents in young seedlings of black gram. Iron deficiency proved to be more deleterious for black gram than iron toxicity.
Keywords: Iron, Black gram ,Catalase, Peroxidase, Photosynthetic pigments, Growth
Introduction
Almost half of the world’s population suffers from mineral deficiencies, primarily of iron. Alleviation of iron deficiency was ranked as a top priority in Copenhagen consensus (2008). The World Health Organization (WHO) has estimated that approximately 3.7 billion people are iron deficient, with 2 billion of these being anemic [1]. In many crop plants Iron deficiency has been reported resulting in different nutritional disorder like poor growth and yield. Cakmak in 2002 reported that Fe deficiency in people might be alleviated by Increasing Fe in staple food crops [2]. Haas reported that to mitigate Fe deficiency there is need to promote food fortification [3]. Globally, an important economical approach to rescue Fe deficiency is bio fortification of staple crops with iron [4,5]. In edible parts of crop plants bio fortification could be achieved by management of soil and crop [6]. Clemens and his co-workers in 2002 have reported that Fe bio fortification has a great impact on human health to combat Fe deficiency [7].
Iron is third among all important micronutrients for plant growth [6,8]. It is involved in different biochemical processes like photorespiration, glycolate pathway and transport of energy in the plant [9]. It is an essential element for many enzymes, including cytochrome that is involved in electron transport chain, chlorophyll synthesis, involved in the structure of chloroplasts, and much enzyme activity [10-13]. Iron as an essential metal, its acquisition by plant roots from the soil enables its entry into the food chain. Therefore, in this way, essential nutrients reach to the animal and human diets.
Iron is involved in the formation of chloroplast protein hence due to deficiency of iron chloroplast protein is reduced and cell division stops, therefore, growth of leaves has been reported to be decreased [14,15]. Kumar and Soll in 2000 reported that iron is required in the active site of glutamyl-tRNA reductase, an enzyme needed for the formation of 5-Aminolevulinic acid which is a precursor of chlorophyll biosynthesis [16]. As iron is not trans located easily in the plant, the deficiency symptoms first appear in young leaves hence its deficiency causes chlorosis as reported in corn and sorghum. In flooding soils due to low redox potential large amounts of iron may be available for plant which could be toxic for the plants. Visual symptoms of iron toxicity include root flaccidity, reduced root branching and mottling of leaves [17,18]. Catalase and Peroxidase play a role in the protection against any type of environmental stress [19]. The levels of both the enzymes which belong to oxido-reductase group have been usually increased by catalyzing the reduction of H2O2 in preventing oxidative stress [20].
In present study, we have described the changes in growth, pigment content and activities of antioxidant enzymes (catalase and peroxidase) in response to deficient (1.4 ppm) and toxic (56 ppm) levels of zinc as compared to control (5.6 ppm) in the seedlings of black gram.
Materials and Methods
Black gram seeds (Vigna mungo CV.T9) were surface sterilized with mercuric chloride (0.1%, w/v) and thoroughly washed thrice with distilled water. 20 seeds for each treatment were spread over in petridish (of diameter 10 cm) lined with filter papers containing nutrient solution replaced every alternate day. Plants were given a daily supply of Hewitt nutrient solution containing: 4mM KNO3 ; 4mM Ca(NO3)2; 1.5mM NaH2 PO4; 2mM MgSO4; 0.1 mM NaCl; 0.1 mM Fe-EDTA; 30 μM H3BO3; 10 μM MnSO4; 1μM CuSO4; 1 μM ZnSO4; 0.2 μM
Na2MoO4; 0.1 μM CoSO4 and 0.1 μM NiSO4. The stock solutions of all nutrients were prepared with analytical reagent grade salts. Seeds were initially kept in dark and humid conditions for 48 hours. Seven days after the emergence, seedlings were subjected to different concentration of iron i.e. control (5.6 ppm), deficient (1.4 ppm), toxic (56 ppm). The seedlings were harvested after 14 days for the following studies.
The root and shoot lengths of black gram seedlings were measured. Fresh and dry weights of seedlings were also recorded after drying at 75℃ to constant mass. Pigment contents like chlorophyll a, chlorophyll b and total chlorophyll were measured according to Arnon [21]. Absorbance was estimated spectrophotometric ally at 645 nm and 663 nm by using Toshniwal TSUV 75 spectrophotometer. Chlorophyll contents were expressed in terms of mg/g fresh weight of tissue.
Catalase (EC 1.11.1.6; CAT) and peroxidase (EC 1.11.1.7; POD) activity were estimated according to Euler and Josephson & Luck method respectively, and were expressed as EU/mg protein and Δ in OD/mg protein respectively [22]. Protein content was measured according to Lowry [23]. Using bovine serum albumin as a calibration standard. DNA and RNA contents were assayed according to Nieman and Poulsen method [24].
The experiment was conducted in a Completely Randomized Design (CRD) with 3 replications. The data were analysed by one way ANOVA using software program Sigma stats 3.5. It was followed by comparison of mean values using Holm Sidak method.
Results and Discussion
Growth
Iron deficiency and toxicity has been reported in this plant species grown under laboratory conditions. The results observed are shown in Table 1. There is alteration in growth which is characterized by different parameters such as fresh and dry weight/biomass accumulation, shoot and root length. Snowden and Wheeler; Schmidt and Fühne; de la Guardia and Alcántara, 2002a have reported that at inadequate supply of Fe there is gradually growth depression showing reduction in shoot length and dry biomass [25,26,18]. In our results there is reduction in plumule length and radicle length which became more evident in deficient level, and it was 41.46% and 28.81% respectively (Table 1). During, Fe deficiency stress biomass allocation towards the roots might be explained by increased phosphoenolpyruvate carboxylase activity in roots [27]. Batty and Younger in 2003 reported that above a threshold Fe concentration in the medium there is reduction in growth due to toxic level of Fe. They observed changes in root growth of common reed. In the findings of Snowden and Wheeler (1993) root growth of most investigated dicotyledonous species was more susceptible than shoot growth [25].
| Parameters | Control (5.6ppm) | Iron deficient (1.4 ppm) | Iron toxic (56ppm) |
|---|---|---|---|
| Radiclelength(cm) | 5.9 ±0.203 | 4.2 ±0.203* (-28.81%) | 5.1 ±0.219* (-13.56%) |
| Plumulelength(cm) | 16.4 ±0.176 | 9.6 ±0.318* (-41.46%) | 14.3 ±0.208* (-12.80%) |
| Freshweight (g) | 1.124± 0.0356 | 0.632± 0.0147* (-43.77%) | 0.945± 0.0275* (-15.93%) |
| Dryweight (g) | 0.456± 0.0263 | 0.114± 0.00173* (-75%) | 0.323± 0.00722* (-29.17%) |
Table 1: Effects of Iron applied at deficient and toxic levels on growth parameters in black gram seedlings.
Means +-SE, n=3, * statistically significant difference (p<0.001), multiple comparisons Vs Control group (Holm Sidak method). Overall significance level= 0.05. Parenthesis ( ) shows the % depreciation in growth.
Photosynthetic Pigments
When there is Fe deficiency then there is loss of chlorophyll as Fe is involved in several steps of chlorophyll biosynthesis, and there are many changes in the assembly and expression of accessory components of the photosynthetic apparatus [28].
The first visual sign of iron deficiency in young leaves is Chlorosis. Gogorcena and co-workers in 2001 have reported that during Fe deficiency chlorosis might be developed without having any effect on growth while Kosegarten; Gruber and Kosegarten have reported that depreciation in growth have been seen at the beginning of yellowing of leaves or without yellowing [29-31].
Suh and co-workers in 2002 reported that toxic level of Fe increases cytochrome b6/f complex, a photosynthetic component of photosystem II (PSII) in thylakoids [32].
It has been studied that excessive dissipation of light might cause photo inhibitory damage [33-36].
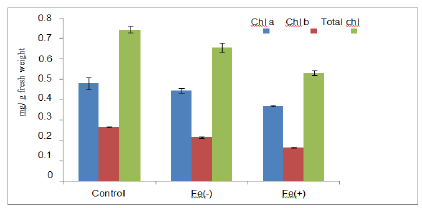
In our study toxic level of Fe resulted in increase in pigment concentration than deficient level (figure 1). Similar results were observed by Kampfenkel and co-workers (1995) in root cutting of Nicotiana plumbaginifolia plants.
They also gave evidences for photo inhibition. Suh and co-workers (2002) also supported this photo inhibition in pea. A high chl. a/chl. b ratio was observed in our experiment which indicated reduction in size of antenna related to reaction centre[28].
Oxidative stress
In the present study, two indicators of oxidative stress i.e. catalase and peroxidase were examined in the 14 days old seedlings of black gram. Fe deficiency impaired the electron transport chain both in the chloroplast and mitochondrion which resulted in the formation of Reactive Oxygen Species (ROS) leading to the induction of oxidative stress [37]. Mittler (2002) reported that against ROS formation plants use a series of antioxidant molecules represented by non-enzymatic antioxidant activity [38].
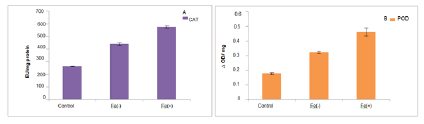
Catalase is a tetrameric iron (heme) containing enzyme which rapidly degrades H2O2 and exerts a dual function at low concentrations. It acts peroxidatically and shows catalytic function at high concentrations [39]. It has been proposed that catalase may be uniquely suited to regulate the homeostasis of H2O2 in the cell. In the present study catalase activity was increased at deficient and toxic level as compared to control (figure 2A). Baskaran and co-workers in 2009 found similar results in Vigna radiate [40]. As cells are in stress both at deficient and toxic level due to catabolic processes rapidly generating H2O2 which was degraded by catalase in an energy efficient manner. Peroxidase (POD) enzyme is involved in reducing H2O2 to H2O. In the present study, our results showed that there was increase in POD activity which was explained by the fact that POD contains Fe (figure 2B). Increased peroxidase activity is directly correlated to the Fe content of the plant. Fang and Kao in 2000 also reported that there was an increase in peroxidase activity in leaves of rice. De Santiago recorded almost 1.5 times increase in peroxidase activity in wheat [41].
DNA, RNA and protein
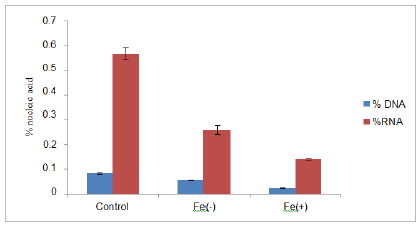
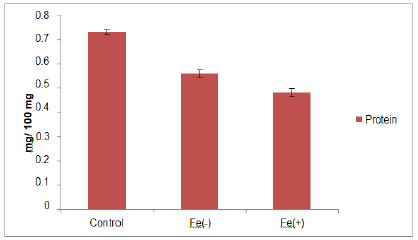
The nucleic acid contents were decreased at deficient and toxic level as compared to control (figure 3). Measurement of nucleic acid (DNA, RNA) contents has been showed the level of oxidative stress. One of the mechanisms of decrease in nucleic acid content involves the OH- radical, which is known to cause strand breakage, deoxyribose fragmentation, and extensive base modification [42]. In the present study black gram seedlings were subjected to iron treatment (56 ppm), showed reduction in the level of protein (figure 4). There was significant decrease at the toxic level of iron as compared to control. Similar results were observed by Kumar and coworkers in 2015 in Vigna mungo. According to Tripathi and Gautam in 2007 there was reduction in protein content which might be due to increased rate of denaturation of protein. Slatni and co-worker in 2011 reported in that breakdown of existing protein into amino acid is the main cause of decrease in protein content in bean.
Conclusion
When plants are not supplied with an optimum amount of Fe, reduction in growth and many physiological and biochemical changes were observed depending on the deficient and toxic levels. Chlorophyll concentration is also very sensitive to Fe supply as iron plays an indispensable role in photosynthetic pigments. A decrease in photosynthetic pigments under Fe deficiency has followed the same trend as growth depreciation. The impact of Fe deficiency on photosynthetic pigments mainly impairs electron transport chain both in mitochondria and chloroplasts leading to the formation of Reactive Oxygen Species (ROS). Though Fe is essential yet excess level of Fe is toxic results in the induction of oxidative stress in plant cells. Catalase and peroxidase activity was increased in both the treatments as compared to control. In the present study significant changes have been observed to understand the role of Fe in healthy or stressed condition, to quantify the antioxidants of plants and scope of ameliorating them by providing additional supply of essential nutrients may be worked out for better plant growth.
Acknowledgements
Authors are grateful to DAV Degree College, Department of Botany, University of Lucknow, for providing necessary laboratory facilities for experimental work and Library support.
References
- WHO (2007) Micronutrient deficiency: Iron deficiency anaemia. Geneva.
- Cakmak I (2002) Plant nutrition research: Priorities to meet human needs for food in sustainable ways. Plant and Soil 247: 3–24.
- Haas JD, Beard JL, Murray-Kolb LE, del Mundo AM, Felix A, et al. (2005) Iron biofortified rice improves the iron stores of non-anemic Filipino women. Journal of Nutrition 135:2823–2830.
[Crossref] [Google Scholar] [Indexed]
- Jeong J, Guerinot ML (2008) Biofortified and bioavailable: The gold standard for plant- based diets. Proc Natl Acad Sci USA 105:1777–1778.
- Nagesh, Ravindrababu V, Usharani G, Dayakar Reddy T (2012) Heterosis studies for grain iron and zinc content in rice (Oryza sativa L.). Ann Biol Res 3: 179–184.
- Zuo Y, Zhang F (2011) Soil and crop management strategies to prevent iron deficiency in crops. Plant and Soil 339: 83–95.
- Clemens S, Palmgren MG, Krämer U (2002) A long way ahead: Understanding and engineering plant metal accumulation. Trends Plant Sci 7: 309–315.
- Samaranayake P, Peiris BD, Dssanayake S (2012) Effect of excessive ferrous (Fe2+) on growth and iron content in rice (Oryza sativa). Int J Agric Biol 14: 296–298.
- Marschner H (1995) Mineral Nutrition of Higher Plants. Second ed. Academic Press, London: 405-434.
- Mamatha N (2015) Effect of sulphur and micronutrients (iron and zinc) on yield and quality of cotton in a vertisol. An Asian Journal of Soil Science 10:63-67.
- Ziaeian AH, Malakouti MJ (2006) Effects of Fe, Mn, Zn and Cu fertilization on the yield and grain quality of wheat in the calcareous soils of Iran. J Plant Nutr 92: 840-84.
- Zaharieva TB, Abadia J (2003) Iron deficiency enhances the level of ascorbate, glutathione, and related enzymes in sugar beet roots. Protoplasma 221: 269-75.
[Crossref] [Google Scholar] [Indexed]
- Welch RM (2002) The impact of mineral nutrients in food crops on global human health. Plant and Soil 247: 83-90.
- Mohamed AA, Aly AA (2004) Iron deficiency stimulated some enzymes activity, lipid peroxidation and free radicals production in borage officinalis induced in vitro. Int J Agric Biol 6:179-184.
- Manthey JA, Crowley DE (1997) Leaf and root responses to iron deficiency in avocado. Journal of Plant Nutrition 20: 683-93.
- Kumar AM, Soll D (2000) Antisense HEMA1 RNA expression inhibits heme and chlorophyll biosynthesis in Arabidopsis. Plant Physiol 122: 49-56.
- Batty LC, Younger PL (2003) Effects of external iron concentration upon seedling growth and uptake of Fe and phosphate by the common reed, Phragmites australis (Cav.) Trin ex. Steudel. Ann Bot 92:801-806.
[Crossref] [Google Scholar] [Indexed]
- Schmidt W (1994) Root-mediated ferric reduction-responses to iron deficiency, exogenously induced changes in hormonal balance and inhibition of protein synthesis. J Exp Bot 45: 725-741.
- Taiz L, Zeiger, E (2002) Plant Physiology. 2nd edition. Sinaurer Associates Inc., Publisher. Sunderland. Massachusets. 198-225.
- Weckx JEJ, Clijsters H (1996) Oxidative damage and defense mechanisms in primary leaves of Phaseolus vulgaris as a result of root assimilation of toxic amounts of copper. Physiol Plant 96: 506-512.
- Arnon D (1949) Copper enzymes in isolated chloroplasts: Polyphenoloxydase in Beta vulgaris, Plant Physiol 24:1-15.
- Euler H, Voa W, Josephson K (1927) Uberkatalase I. Leibigs Ann 452: 158181
- Lowry OH, Rosebrough NJ, Farr AL, Randall RJ (1951) Protein measurement with the Folin Phenol reagent. J Biol Chem 193:265-75.
- Nieman RH, Poulsen LL (1963). Spectrophotometric Estimation of Nucleic Acid of Plant Leaves. Plant Physiol 38: 31-35.
- Snowden R, Wheeler BD (1993) Iron toxicity to fen plant-species. J of Ecology 81:35-46.
- De la Guardia MD, Alcántara E (2002) A comparison of ferric-chelate reductase and chlorophyll and growth ratios as indices of selection of quince, pear and olive genotypes under iron deficiency stress. Plant and Soil 241:49-56.
- De la Guardia MD, Alcántara E (2002) Bicarbonate and low iron level increase root to total plant weight ratio in olive and peach rootstock. J Plant Nutr 25:1021-1032.
- Terry N, Abadía J (1986) Function of iron in chloroplasts. J Plant Nutr 9: 609-646.
- Gogorcena Y, Molias N, Larbi A, Abadía J, Abadía A (2001) Characterization of the responses of cork oak (Quercus suber) to iron deficiency. Tree Physiology 21:1335-1340.
- Kosegarten H, Wilsona GH, Escha A (1998) The effect of nitrate nutrition on iron chlorosis and leaf growth in sunflower (Helianthus annuus L.). European J Agronomy 8:283-292.
- Gruber B, Kosegarten H (2002) Depressed growth of non-chlorotic vine grown in calcareous soil is an iron deficiency symptom prior to leaf chlorosis. J Plant Nutr and Soil Sci 165:11- 117.
- Suh HJ, Kim CS, Lee JY, Jung J (2002) Photodynamic effect of iron excess on photosystem II function in pea plants. Photochem Photobiol 75: 513-518.
[Crossref] [Google Scholar] [Indexed]
- Kampfenkel K, Montagu MV, Inzé D (1995) Effects of iron excess on Nicotiana plumbaginifoli plants. Implications to oxidative stress. Plant Physiol 107:725-735.
- Abadía J, Morales F, Abadía A (1999) Photosystem II efficiency in low chlorophyll, iron deficient leaves. Plant and Soil 215:183-192.
- Asada K, Takahashi M (1987) Photo inhibition production and scavenging of activated oxygen. Elsevier Science Publishers. Amsterdam: 227-287.
- Donnini S, Castagna A, Guidi L, Zoechi G, Ranieri A (2003) Leaf responses to reduced iron availability in two tomato genotypes:T3238FER (Iron efficient) and T3238fer (iron inefficient). J Plant Nutr 26:2137-2148.
- Pascal N, Douce R. (1993) Effect of Iron Deficiency on the Respiration of Sycamore (Acer pseudoplatanus L.) Cells. Plant Physiology 103:1329-1338.
[Crossref] [Google Scholar] [Indexed]
- Mittler R (2002) Oxidative stress, antioxidants and stress tolerance. Trends Plant Sci 7:405-410.
[Crossref] [Google Scholar] [Indexed]
- Deisseroth A, Dounce A (1970) Catalase: Physical and chemical properties, mechanism of catalysis and physiological role. Physiol Rev 50: 319-375.
[Crossref] [Google Scholar] [Indexed]
- Baskaran P, Jayabalan N (2009) Psoralen production in hairy roots and adventitious roots cultures of Psoralea coryfolia. Biotechnol Lett 31:1073-7.
[Crossref] [Google Scholar] [Indexed]
- De Santiago A, Quintero JM, Avilés M, Delgado A (2011) Effect of Trichoderma asperellum strain T34 on iron, copper, manganese, and zinc uptake by wheat grown on a calcareous medium. Plant and Soil 342: 97–104.
- Halliwell B, Aruoma IO (1991) DNA damage by oxygen-derived species. Its mechanism and measurement in mammalian systems. FEBS Lett 281:9-19.
[Crossref] [Google Scholar] [Indexed]
- Fang WC, Kao CH (2000) Enhanced peroxidase activity in rice leaves in response to excess iron, copper and zinc. Plant Science 158: 71–76.
[Crossref] [Google Scholar] [Indexed]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi