Research Article, J Biodivers Manage Forestry Vol: 9 Issue: 6
Phytogeographical Assessment of Family Poaceae Barnhart in Western Himalaya, India
Shailja Tripathi1,2, Priyanka Agnihotri1, Shubham Jaiswal1, Rekha Yadav1, Dileshwar Prasad1, Vivek Vaishnav3 and Tariq Husain1*1 Plant Diversity, Systematics and Herbarium Division., CSIR-National Botanical Research Institute, Lucknow, Uttar Pradesh, India
2 Department of Botany, University of Lucknow, Lucknow, Uttar Pradesh, India
3 Department of Forestry & Environmental Sciences, School of Human & Environmental Science, Manipur University, Canchipur, Imphal, Manipur, India
*Corresponding Author : Tariq Husain
Systematics and Herbarium Division, CSIR-
National Botanical Research Institute, Lucknow, 226022, Uttar Pradesh, India
Tel: +91-8004920968
E-Mail: hustar_2000@yahoo.co.uk
Received: November 23, 2020; Accepted: December 08, 2020 Published: December 14, 2020;
Citation: Tripathi S, Agnihotri P , Jaiswal S , Yadav R , Prasad D. etal., (2020) Phytogeographical Assessment of Family Poaceae Barnhart in Western Himalaya, India. J Biodivers Manage Forestry 9:6.
Abstract
A phytogeographical assessment of grass family Poaceae in three states namely Himachal Pradesh (HP), Jammu and Kashmir (J&K) and Uttarakhand (UK) of Western Himalaya found that 5.06% of the taxa found globally have been abundant in the region. Overall, 582 species belonging to 143 genera and 29 tribes of the family were found distributed among the three states. UK was found exhibiting maximum number of taxa with the highest level of Shannon’s diversity index (4.13) based diversity among them.Maximum number of the species of the family was distributed over the altitudinal range of 1500−2000 m showing a hump shaped curve. Poa L. was found as the most diverse genus, and Andropogoneae was found with maximum number of the species dominantly distributed in the region. Among the overall species found in the region, 61 taxa of the family were enlisted in IUCN red list hence prioritized for conservational purposes.
Keywords: Conservation; Diversity; Gramineae; Phytogeography; Taxonomy
Introduction
Poaceae, the grass-family, is the fifth largest plant family in terms of number of species. It comprises approximately 750−770 genera and 11,500 species [1] with cosmopolitan distribution [2]. The varied breeding systems that maintain the variability in the genepool through apomixis, local adaptation and resilience of the grasses altogether led them to be one of the most successful terrestrial life forms on the Earth in turbulent bio-climatic variables [3]. From the pre-historic times, man has been dependent on grass species for food, fodder, shelter and medicines. Seeds of grasses comprise grains, which amplify nutritional profile of the group by supplying ample amounts of calories for human and animal consumption. With long back history of domestication, some of the grass species form a major portion of our staple crops viz. rice, wheat, oats, rye, barley, maize, sorghum, sugarcane and other millets. Approximately 70% of the world’s farmlands are cultivated with domesticated crop species from the family Poaceae [3]. Nevertheless, major number of the species of the family still occurs in wild and the final number of the species is yet to be finalized for documentation through revision of different genera of the family describing the species from different biogeographical regions of the world.
India maintains one of the most diverse life forms in the world in ten biogeographical regions of varied geoclimatic conditions. These biogeographical regions support the distribution of many species from the family Poaceae with diverse phyto-socio-structure of abundance. Documentation of their abundance in such a diverse condition always needs intensive taxonomical assessment. The phytotaxonomical work on Indian grasses was first reported from places of the Himalayan region by Griffith [4]. Later, Duthie et al. [5-8] also surveyed the grasses from different regions of British Indian sub-continent. Hooker [9] included 146 genera and 838 species in his “Flora of British India”. A detailed account of grasses was provided by Bor [10] with taxonomical identification of 242 genera and 1243 species in India. Meanwhile, revision of the tribes of the family viz. Garnotiae [11-13], and a genus Heteropogon Pers. Deshpande [14] was also incorporated. Among the major works on Indian grasses, Karthikeyan et al. [15] documented 260 genera with 1300 species of grasses from India. Sreekumar and Nair [16] reported 103 genera and 296 species from a coastal region state Kerala. Shukla [17] included 148 genera and 482 taxa from North Eastern India and Potdar et al. [18] gave detailed description of grasses of state Maharashtra from Deccan plateau region and reported 125 genera and 458 taxa. These studies dealt mainly with the taxonomical characterization and distribution of the grass family in different but only few biogeographical regions of the country. Moreover, no study has been conducted reporting the species abundance and diversity within a specific biogeographical region of the country.
Among the ten biogeographical regions, Himalayan region has the youngest geological structures ranged over the highest elevation supporting various unique life forms. It has been enlisted as biodiversity hotspot due to endemism of enormous life forms in threat of loss. Himalayan region broadly forms the northern boundary of the country occupying an area of about 5,31,000 km2. The eastern Himalaya which includes the states Sikkim, Arunachal Pradesh, Meghalaya, Nagaland, Manipur, Tripura & Mizoram completely and hill districts of Assam and West Bengal, is considered as the “Cradle of flowering plants” harbouring about 8000 species of Angiosperms. The Western Himalaya of India spreads over three states namely Himachal Pradesh (HP), Jammu and Kashmir (J&K) and Uttarakhand (UK) occupying an area of about 3,31,000 km2. Representing approximately 10% of the total geographical area of the country, it supports about 5000 species of flowering plants. In comparison to the Eastern Himalaya, Western Himalayan region has not been explored for phytogeographical status of the species of Poaceae. Since the region is in threat of habitat loss towards the projected changing climatic regimes, the assessment of abundance, distribution and diversity of grasses in the region may help to take early action for conservation and management of the available genetic resources from Poaceae. Therefore, in present investigation, we surveyed the species from the family Poaceae distributed over the Western Himalaya along the ranges of altitude and documented their diversity after taxonomical authentication.
Materials and Methods
To assess the phytogeography of the Western Himalayan region and to obtain the abundance and distribution related information of the taxa, the literature was surveyd before sampling, followed by the consultation of some Indian herbaria viz. BSD, CAL, CDRI, CIMAP, DD, KASH, LWG & RRLH, published floras and several plant database websites (The Plant list, IPNI, Tropicos) etc [19-21]. Following the information, for the ground-truthing of abundance and distribution of the taxa, field visits and sampling were carried out.
Plant sampling
Field expedition was done during the months of March 2018-November 2019. Extensive sampling of plant specimens was carried out in the regions of HP, J&K and UK (Figure 1). Mean annual temperature of the region ranges from 21.7°C to 24.2°C and annual rainfall ranges from 1238 mm to 1631 mm (Table 1).
| States | Area(km2) | Latitude (N) | Longitude (E) | Altitude (m) | Annual rainfall (mm) | Mean annual temperature (°C) |
|---|---|---|---|---|---|---|
| Himachal Pradesh (HP) | 55,673 | 32°29' | 75°10' | 450-6826 | 2909–3800 | 21.7 |
| Jammu & Kashmir (J&K) | 2,22,236 | 32°44' | 74°54' | 300-7672 | 693–1238 | 24.2 |
| Uttarakhand (UK) | 53,483 | 30° 15' | 79°15' | 187-7816 | 1631 | 21.8 |
Table 1: Biogeographical and climatic condition of states of Western Himalaya.
Species identification
The identification of the specimens was done using literature from different sources. The taxonomical keys as described by Bor et al. [10,16-18] were used for the identification of the grass specimens. For validation, the herbaria specimens were also referred. The systematic treatment of Indian grasses as described by Bor [10] was followed for taxonomical classification of identified specimens.
Data preparation and analysis
Abundance data was prepared for hierarchical taxa (sub-family, tribe, genus, species, sub-species, variety) of the plants distributed over the region. Shannon’s diversity index (H) was measured applying formula ‘H=-Σs i=1pi lnpi for overall, rare and common plants distributed over different states of the region.
Results
Abundance
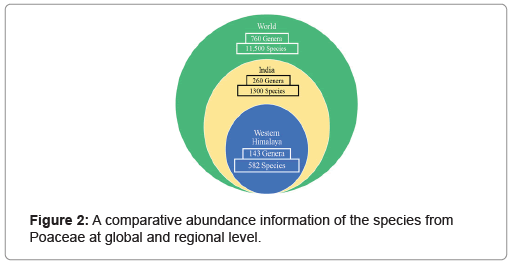
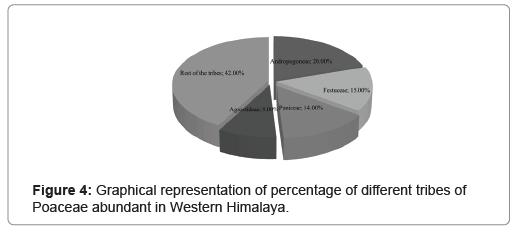
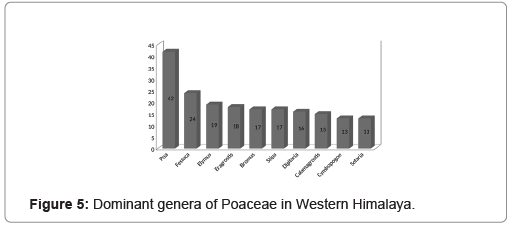
Overall, plants of 582 species belonging to 143 genera of Poaceae were found in the Western Himalayan region. In terms of number, the region was found to share 44.45% of the total species of grasses found in India and 5.06% of those distributed globally. In the same manner, the region exhibited 55% of grass genera from the family distributed over India and 19% of those found globally (Figure 2). A higher number of grass species was found belonging to the subfamily Pooideae as compared to Panicoideae. Among the 29 tribes of the species found in the region, three tribes consisting 203 taxa were from the subfamily Panicoideae while the rest 26 tribes of 379 taxa were from Pooideae (Figure 3). In terms of the number of the genera and species, the tribe Andropogoneae was found with the highest number followed by Festuceae, Paniceae and Agrostideae (Figure 4). Among the genera, Poa L. was the most dominant (42 species) followed by Festuca L. (24 species) and Eragrostis Wolf (18 species) (Figure 5).
Distribution
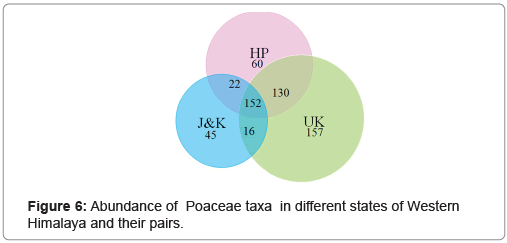
The maximum number of the taxa was found in UK (157) followed by HP (60) and J&K (45). Total 152 taxa were found commonly distributed over all the three states. Apart from this, 130 taxa were common among HP and UK, 22 taxa among HP and J&K and 16 taxa among J&K and UK (Figure 6). The maximum number of taxa was abundant between altitudinal ranges of 1500 m-2000 m, which declined further up to 6000 m (Figure 7). Species Pennisetum lanatum Klotzsch (2000 m-6500 m) and Stipa caucasica Schmalh (1000 m-5500 m) were found to have widest distribution along the altitudinal classes. Among the taxa occuring in the study area, about nine species have been found to be restricted to their locality viz. Calamagrostis gamblei Paszko, Dendrocalamus somdevae Naithani, Festuca nandadevica Hajra, Festuca sanjappae Chandra S. & Srivast., Helictotrichon uniyalii Kandwal & Gupta, Muhlenbergia rakchamensis Arum., Murthy & Nair Pseudodanthonia himalaica (Hook) Bor & Hubb., Stipa klimesii M.Nobis and Trisetum micans (Hook) Bor.
Diversity
The maximum diversity for genera (4.13) and minimum diversity for tribe (2.39) were recorded from UK. HP was found with minimum diversity for genera (3.22). UK and HP both were found the most diverse for sub-families (Table 2).
| States | Genera | Tribes | Sub-families |
|---|---|---|---|
| -143 | -29 | -2 | |
| HP | 3.22 | 2.48 | 0.41 |
| J&K | 3.41 | 2.59 | 0.36 |
| UK | 4.13 | 2.39 | 0.41 |
| HP+J&K | 2.75 | 2.34 | 0.47 |
| J&K+UK | 2.65 | 1.56 | 0.67 |
| HP+UK | 3.88 | 2.72 | 0.39 |
| HP+J&K+UK | 4.05 | 2.46 | 0.41 |
Table 2: Shannon’s diversity index (H) for different taxa of Poaceae distributed in Western Himalaya.
Discussion
A multiplicity of factors such as vegetation type, slope, temperature and altitude regulates the structure and composition of a community and distribution pattern and diversity of its species in a mountainous ecosystem [23]. Western Himalaya experiences homogenous but extreme conditions of temperature range, rainfall and other climatic factors. Apart from these, the region also displays wide altitudinal variations. A wide range of geo-climatic conditions support varied phytogeographical diversity and distribution of life forms. The diversity and distribution of the species from Poaceae have not been documented in the Western Himalaya. Therefore, according to the best of our knowledge, the present survey is the first attempt of its kind reporting the phytogeographical status of Poaceae species in the region.
Abundance of Poaceae taxa in the region
A remarkable diversity of grasses with wide distribution has been observed in the study area. A total of 582 taxa represent an appreciable contribution of grasses in maintaining biodiversity of the region. Higher percentage of Pooideae occurring in Western Himalaya may be attributed to the fact that individual clades of grasses have distinct ecological preferences. On their onset, grasses used to occupy warm and moist habitats, but with the passage of time, there have been independent radiations into cold temperate regions. Increased number of genes responsible for tolerance of low temperature has been found in Pooideae whereas Panicoideae prefers warm and mesic conditions, particularly the tribe Andropogoneae is the most diverse in areas with high fire frequency [24]. Abundance of Poa and Festuca in the study area reflects their pronounced adaptive potential to the cold conditions found in the region.
Distribution pattern of Poaceae taxa among the states in the region
Grasses found in Western Himalaya fall under seven types of distribution patterns. Nearly 45% of total grass species have been found to be confined to one of the three centres of distribution: HP, JK and UK. About 26% grasses show widest distribution pattern occurring in all the three states. It has been observed that about 22% taxa show continuous distribution between HP and UK, about 4% between HP and JK and the least 3% taxa display discontinuous distribution between JK and UK. UK supports aout 78% of the total diversity of grasses found in the region. Festuceae is the most common tribe occurring in HP while JK supports maximum taxa of Triticeae which include commercially important cereals. UK harbours Andropogoneae to a greater extent as compared to other tribes found in the study area. As revealed by observed distribution patterns of different tribes in the study region, it may be inferred that these tribes show heterogenous geographical distribution pertaining to the optimum conditions for their survival.
Diversity among Poaceae taxa in the region
To characterize species diversity, Shannon’s diversity index is one of the commonly used indices. The value of Shannon’s index ranges between 0.0-5.0. In the present study, values of Shannon’s diversity index were found to be varying for different ranks. For genera, it was maximum ranging between 2.75-4.13, moderate for tribes (1.56-2.59) and minimum for sub families (0.39-0.67). These values indicate that on moving down in the hierarchal order variations increase and stability decrease for the subsequent ranks. As observed from the results, maximum diversity of grass species occurs in Uttarakhand (H=4.13) which may be ascribed to the optimum survival conditions for grasses prevailing there. Uttarakhand represents a craggy and mountainous state with a broad class interval of altitude, ranging from 187 m-7816 m asl, rendering the region highly variable in terms of topography and meteorology which enables the state to sustain enormous biodiversity influenced by copious variations in geological, biological and geographical factors. This region possesses highly diversified climate across the transverse zones varying from the subtropical humid climate of the Terai to the tundra-like climate of the Great Himalayan ridges [25].
Effect of altitude on the species distribution and abundance
The present study revealed expected pattern of relationship between altitude and species richness. The number of species plotted against the altitudinal gradient displayed a hump-shaped curve. The mid altitudinal ranges constituted higher species richness which dwindled towards extremes of the gradient. A general trend of alleviated species richness with increase in altitude has been evinced long back which may be attributed to depauperate ecosystem observed in moving from the Equator towards the poles [26]. Maximum number of species was found in the range between 1500 m-2000 m (279 species). The least number of species (one species) was found in the highest range (6000 m-6500 m). A slightly higher number was found in the lowest range (0 m-500 m) (30 species). This reflects a unimodal relationship between species richness and altitude. The decline in number of species at higher altitudinal gradient could be attributed to eco-physiological impediments, including low temperature, shortened growing season and other geographical barriers. Further, being affected by frequent anthropogenic disturbances, such as road construction, habitat destruction, deforestation and agricultural practices, the lower altitudes harbour lesser number of species. Pattern of diversity observed at the mid-altitudes could be explained by Intermediate Disturbance Hypothesis which stated that intermediate intensities of disturbance maximize species diversity [27].
Conservation status and implications
Owing to the high economic and ecological importance, Poaceae is one of the most explorable groups in Angiosperms but selection and domestication of only fewer over all existing species and exploitation due to negligence resulted in decline of grass species habitat and inclusion of 61 taxa of the grasses from Western Himalaya in IUCN red list which urges them to be prioritized for conservational purposes. These grasses with their present status in IUCN red list have been enlisted later (Table 3). Therefore, it is the need of the hour to study population trends so that research can be conducted to conserve biodiversity of grasses. Change in the climatic conditions rendered the biodiversity to get confined. Lot of plants and animals are losing their habitat which may be a threat to the ecological balance. Climate change negatively affects the existence of biotic components of ecosystem. Loss in diversity and distribution of grasses can be easily observed by their occurrence in IUCN Red list. Several environmental parameters play significant role in maintaining grasslands which mainly focus on water use efficiency of grasses. Moreover, anthropogenic activities like herbivory, urbanization, etc tend to alleviate the grass cover of earth by converting grasslands to human-dominated landscapes. Grasslands act as attractive sites for agriculture as they possess excellent soil. They also provide buffering action against burgeoning CO2 concentration as they can store almost equal amount of carbon as being stored by forests [28]. Previous studies suggest the fate of grasslands hard to be predictable towards changing climate, attributed to complex set of factors maintaining them. Variations in content and season of precipitation also influence the occurrence of some grassland as suggested by Gibson [29] who concluded that long lasting drought resulted in altered structure and dominance relationship in Prairies grassland. On the other hand, certain grasslands are resistant to perturbations in climatic conditions as exemplified by the reports of Grime et al. [30] who despite substantial alterations in temperature and precipitation, observed no significant changes in community composition of a low-productivity grassland in England [24].
| S.no. | Taxa | IUCN status |
|---|---|---|
| 1 | Agrostis canina L. | LC |
| 2 | Agrostis nervosa Nees ex Trin. | LC |
| 3 | Agrostis stolonifera L. | LC |
| 4 | Alopecurus aequalis Sobol. | LC |
| 5 | Alopecurus arundinaceus Poir. | LC |
| 6 | Arundo donax L. | LC |
| 7 | Avena fatua L. | LC |
| 8 | Avena sterilis L. | LC |
| 9 | Brachiaria eruciformis (Sm.) Griseb. | LC |
| 10 | Brachiaria ramosa (L.) Stapf | LC |
| 11 | Brachiaria reptans (L.) C.A.Gardner & C.E.Hubb. | LC |
| 12 | Calamagrostis canadensis (Michx.) P.Beauv. | LC |
| 13 | Calamagrostis pseudophragmites (Haller f.) Koeler | LC |
| 14 | Catabrosa aquatica (L.) P.Beauv. | LC |
| 15 | Cenchrus ciliaris L. | LC |
| 16 | Chionachne gigantea (J. Koenig) Veldkamp | LC |
| 17 | Crypsis schoenoides (L.) Lam. | LC |
| 18 | Dendrocalamus giganteus Munro | LC |
| 19 | Desmostachya bipinnata (L.) Stapf | LC |
| 20 | Digitaria duthieana Henrard ex Bor | DD |
| 21 | Dimeria ornithopoda Trin. | LC |
| 22 | Echinochloa colona (L.) Link | LC |
| 23 | Echinochloa crus-galli (L.)P.Beauv. | LC |
| 24 | Echinochloa frumentacea Link | LC |
| 25 | Eleusine indica (L.) Gaertn. | LC |
| 26 | Elytrophorus spicatus (Willd.) A.Camus | LC |
| 27 | Eragrostis unioloides (Retz.) Nees ex Steud. | LC |
| 28 | Eriochloa procera (Retz.) C.E.Hubb. | LC |
| 29 | Eulalia contorta (Brongn.) Kuntze | LC |
| 30 | Hemarthria altissima (Poir.) Stapf & C.E.Hubb. | LC |
| 31 | Hemarthria compressa (L.f.) R.Br. | LC |
| 32 | Hordeum murinum L. | LC |
| 33 | Imperata cylindrica (L.) P.Beauv. | LC |
| 34 | Isachne albens Trin. | LC |
| 35 | Isachne globosa (Thunb.) Kuntze | LC |
| 36 | Isachne pulchella Roth | LC |
| 37 | Leersia oryzoides (L.) Swartz | LC |
| 38 | Leptochloa panicea (Retz.) Ohwi | LC |
| 39 | Panicum repens L. | LC |
| 40 | Panicum sumatrense Roth | LC |
| 41 | Paspalidium flavidum (Retz.) A.Camus | LC |
| 42 | Paspalidium geminatum (Forssk.) Stapf | LC |
| 43 | Paspalidium punctatum (Burm.) A.Camus | LC |
| 44 | Paspalum conjugatum P.J.Bergius | LC |
| 45 | Paspalum distichum L. | LC |
| 46 | Paspalum scrobiculatum L. | LC |
| 47 | Phalaris arundinacea L. | LC |
| 48 | Phleum alpinum L. | LC |
| 49 | Phragmites australis (Cav.) Trin. ex Steud. | LC |
| 50 | Phragmites karka (Retz.) Trin. ex Steud. | LC |
| 51 | Poa angustifolia L. | LC |
| 52 | Poa annua L. | LC |
| 53 | Poa pratensis L. | LC |
| 54 | Pogonatherum paniceum (Lam.) Hack. | LC |
| 55 | Polypogon monspeliensis (L.) Desf. | LC |
| 56 | Polypogon viridis (Gouan) Breister | LC |
| 57 | Saaciolepis myosuroides (R.Br.) A.Camus | LC |
| 58 | Schizachyrium brevifolium (Sw.) Buse | LC |
| 59 | Setaria parviflora (Poir.) M.Kerguelen | LC |
| 60 | Urochloa setigera (Retz.) Stapf | LC |
| 61 | Vulpia octoflora (Walter) Rydb. | LC |
Table 3: Conservation status (IUCN Red list) of taxa from Poaceae found in Western Himalaya.
Acknowledgement
The authors are thankful to the Director, CSIR-NBRI, Lucknow for providing a platform to conduct the research. We are also grateful to the concerned forest authorities for their consent to provide access to the collection of specimens. We acknowledge the curators of herbaria consulted for the study of specimens. The valuable suggestions from Prof. S. R. Yadav, Department of Botany, Shivaji University, Kolhapur, India are also gratefully acknowledged. The research fellowship support by CSIR, New Delhi, India to first, fourth and fifth author is dully acknowledged. The third author acknowledges UGC, New Delhi for providing research fellowship.
References
- Hodkinson TR (2018) Evolution and taxonomy of the grasses (poaceae): A model family for the study of species- rich groups. Annual Plant Reviews 1: 1-39.
- Kiran Raj MS, Sivadasan M, Ravi N (2003) Grass diversity of Kerala- Endemism and its phytogeographical significance. In: Janarthanam MK, Narasimhan D (Eds.), Plant diversity, Human welfare and Conservation, Goa University, Goa.
- Yadav SR (2010) Know your grass genera through hand lens. Shivaji University, Kolhapur, India.
- Griffith W (1834) Grasses of Jheels of Sylhet. J Asiat Soc Bengal 5: 570.
- Duthie JF (1883) A list of the grasses of North West India, indigenous and cultivated. Roorkee, India.
- Symonds TJ (1886) Indian grasses. (2nd edition), Higginbotham, Madras, India.
- Coldstream (1889) Illustrations of grasses of South Punjab. W. Thacker & Co, London.
- Lisboa JC (1896) List of Bombay grasses and their uses. Government Central Press, Bombay, India.
- Hooker JD (1896) Flora of British India. L Reeve & Co, London.
- Bor NL (1960) The Grasses of Burma, Ceylon, India and Pakistan. Pergamon Press, London.
- Prakash V, Jain SK (1979) Poaceae: Tribe Garnotiae. Fasc Fl Ind, BSI, Calcutta, India. pp: 1-16.
- Prakash V, Jain SK (1984) Poaceae: Tribe Isachneae. Fasc Fl Ind, BSI, Calcutta, India. pp: 1-42.
- Deshpande UR (1984) Poaceae: Tribe Andropogoneae. Fasc Fl Ind 5: 1-30.
- Deshpande UR (1990) The genus Heteropogon Pers. Poaceae in India. Bulletin BSI 30: 120-125.
- Karthikeyan S, Jain SK, Nayer MP, Sanjappa M (1989) Florae Indicae Enumeratio: Monocotyledonae. BSI, Calcutta.
- Sreekumar PV, Nair VJ (1991) Flora of Kerala- grasses. Fl Ind, BSI, India.
- Shukla U (1996) Grasses of North-Eastern India. Scientific publishers, Jodhpur, India.
- Potdar GG, Salunkhe CB, Yadav SR (2012) Grasses of Maharashtra. Shivaji university, Kolhapur, India.
- The Plant List (2010) A working list of all plant species.
- The International Plant Names Index (2012).
- http://legacy.tropicos.org/Home.aspx
- Shannon CE (1948) A mathematical theory of communication. Bell Labs Tech J 27: 379-423.
- Dar JA, Sundarapandian S (2016) Patterns of plant diversity in seven temperate forest types of Western Himalaya, India. J Asia Pac Biodivers 9: 280-292.
- Kellogg EA (2015) The families and genera of vascular plants. (1 edition) Springer, Berlin.
- Malik DS, Tyagi D (2017) Biodiversity and ecosystem services of ramganga reservoir (Uttarakhand). Essence 8: 214-223.
- Bruun HH, Moen J, Virtanen R, Grytnes JA, Oksanen L, et al. (2006) Effects of altitude and topography on species richness of vascular plants, bryophytes and lichens in alpine communities. J Veg Sci 17: 37-46.
- Connell JH (1978) Diversity in tropical rain forests and coral reefs. Science 199: 1302-1310.
- White R, Murray S, Rohweder M. (2000) Pilot analysis of global ecosystems (PAGE): Grassland ecosystems.World Resources Institute.
- Gibson DJ (2009) Grasses and grassland ecology. Oxford university press, New York.
- Grime JP, Fridley JD, Askew AP, Thomson K, Hodgson JG, et al. (2008) Long term resistant to simulated climate change in an infertile grassland. Proc Natl Acad Sci 105: 10028-10032.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi