Research Article, Int J Cardiovasc Res Vol: 8 Issue: 2
Preoperative Platelet Activation Markers as a Risk Predictor of Postoperative Atrial Fibrillation after Coronary Artery Bypass Grafting
Mahmoud Khairy1*, Eman NasrEldin2 and Ali Elsharkawi3
1Cardiothoracic Surgery Department, Assiut University, Assiut, Egypt
2Clinical Pathology Department, Assiut University, Assiut, Egypt
3Cardiology Department, National Heart Institute, Cairo, Egypt
*Corresponding Author : Mahmoud Khairy
Cardiothoracic Surgery Department, Assiut University, Assiut, Egypt
Tel: (002)01224579171
E-mail: mahkhairy02@yahoo.com
Received: December 05, 2018 Accepted: December 24, 2018 Published: January 09, 2018
Citation: Khairy M, NasrEldin E, Elsharkawi A (2019) Preoperative Platelet Activation Markers as a Risk Predictor of Postoperative Atrial Fibrillation after Coronary Artery Bypass Grafting. Int J Cardiovasc Res 8:2. doi: 10.4172/2324-8602.1000371
Abstract
Background: Atrial fibrillation (AF) is the most well-known arrhythmia following coronary artery bypass grafting (CABG) that in turn may lead to a longer hospital stay and a worse postoperative prognoses. AF is known to be a cause of platelet activation; we investigated the relationship between the platelet activation markers and the risk of AF after coronary bypass graft surgery.
Methods: Blood was drawn one day before the operation from fifty patients scheduled for isolated on pump CABG. The monocyte–platelet aggregates (MPAs) content were assessed by Flow cytometric quantification analysis. Moreover, soluble CD40 Ligand (sCD40L) level, soluble P-selectin (sP-selectin) and D-dimer levels were quantified by immunological ELISA technique. Post-operative AF (POAF) events were followed up during hospitalization.
Results: Post-operative AF (POAF) noticed in 22% of patients, in the first postoperative week. Preoperative levels of both sCD40L, sP-selectin and D-Dimer were significantly higher in those who developed POAF. CD41 expression on monocytescellular marker of platelet activation- and the content of MPAs were increased in those patients encountered POAF.
Conclusion: Preoperative platelet activation as affirmed by both soluble and cellular markers in addition to the content of MPAs markers seems to be an unprecedented predictor of the postoperative atrial fibrillation.
Keywords: Platelet activation markers; Atrial fibrillation; CABG
Introduction
Post-operative atrial fibrillation (POAF) is prevalent after coronary artery bypass grafting (CABG). POAF incidences range reported from 25% to 40% [1].The incidence rang increased in patients who underwent valve procedure with CABG [2,3]. Many proarrhythmic factors are attributed for the occurrence of POAF and the exact mechanism is still unclear, the systemic inflammatory response to surgery [4] furthermore, myocardial ischemia accompanied by dysfunctional grafts is of noteworthy significant [5]. Valvular or ischemic heart diseases, age-associated degenerative changes in the conductive system or inter atrial impulse conduction disorders could be also implicated [6-8].
The inflammatory processes and AF, in general, could be associated with high level of platelet activation markers. Monocyte–platelet aggregates (MPAs) are precise and sensitive markers to identify platelets activation [9,10]. The majority of the soluble CD4O Ligand (sCD40L) reflects platelet activation and its level is elevated in patients with AF [11] which will increase the risk of thrombosis and stroke during the arrhythmia particularly after CABG [12,13].
We tested the relation between preoperative platelet activation markers and the risk of POAF in patients in sinus rhythm undergoing on-pump CABG.
Methods
Study population
The study included 50 patients undergoing isolated on pump CABG in the Cardiothoracic Surgery Department, of Assiut university hospital. Detailed medical history and cardiac investigations data were collected including; Euroscore, left ventricular function, and left atrial diameter, history of medication, and any cardiac risk factors before the operation. Selected patients had no history of AF prior to CABG, where patients with history of paroxysmal AF or chronic AF were excluded from the study.
Other exclusion criteria included patients on medical treatment (amiodarone or digoxin), patients presenting with an acute coronary syndrome, concomitant valve or vascular surgery, infectious or inflammatory disorders, chronic obstructive pulmonary disease (COPD) and thyroid disease.
Selected patients were advised to stop antiplatelet medication (aspirin and clopidogrel) one week before surgery. Those who did not develop postoperative AF during the same period served as the control population (who remained in sinus rhythm until discharge).
AF was diagnosed on the basis of abnormalities on 12-lead electrocardiogram (ECG) recording, lasting for at least 30 seconds and was characterized by sustained arrhythmia, irregular RR intervals, absent P waves, and different intervals between atrial contractions (cycle<200 ms) According to the guidelines of the European Society of Cardiology, 2010 [8].
Clinical end points
Clinical data were collected during the hospital stay, including the length of hospital stay, any major cardiovascular events (like myocardial infarction, unstable angina, need for redo surgery, or death) during postoperative follow-up.
The clinical endpoint of the study was the development of AF of any duration, during hospitalization until discharge. Heart rhythm was monitored continuously for the first 48 hours postoperatively and at 4- hour intervals thereafter for 2-3 days. Then by daily ECG until discharge.
Angina was defined by Canadian Cardiovascular Society Classification (CCS) [14] and Heart failure classifications were based on the New York Heart Association (NYHA) guidelines [15]. Postoperative MI was characterized by the rise of myocardical markers above the normal levels, or appearance of new Q waves on electrocardiograms through follow-up.
Acute coronary syndrome was distinguished by ST-elevation myocardial infarction, non ST-elevation myocardial infarction, or unstable angina.
Left atrial (LA) diameter, evaluation of valvular heart diseases and LVEF were gotten by Echocardiography.
The study was approved by the institutional medical ethics committee of faculty of medicine-Assiut University, and all patients gave informed consent.
Data are presented as mean ± SD. *P value <0.05=significant. COPD, chronic obstructive pulmonary disease; MI, myocardial infarction; CCS, Canadian Cardiovascular Society score; LVEF left ventricular ejection fraction; ACEIs, angiotensin-converting enzyme inhibitors; IABP, intra-aortic balloon pulsation; HDL, high density lipoprotein; WBC, white blood cells; N/L, neutrophil/lymphocyte.
Blood samples
Venous blood samples were obtained preoperatively, from all of the study participants with minimal stasis into citrated vacutainers tubes. Samples for flow cytometric analysis were utilized instantly or within 20 minutes from the collection time to avoid artificially misleading increased levels secondary to in vitro platelet activation. Other samples were centrifuged; plasma aliquots were collected and stored at -80°C until assayed.
Assessment of the levels of soluble platelet activation markers sCD40L, sP-selectin, high-sensitivity C-reactive protein (CRP), and D-Dimer were determined by a Sandwich enzyme immunoassay (ELISA) (using ELISA kit by R&D Systems®, Germany). The lower limits of detection for the assay were 0.016 ng/mL0.8 ng/mL, 0.175 mg/Land 0.10 μg FEU/ml respectively.
sCD40-ligand was chosen to symbolize the interface of platelets, thrombosis, and inflammation. So also, soluble P-selectin was chosen to quantify platelet and even endothelial cell activation.
Flow cytometric analysis
Monocyte–Platelet Aggregates (MPA): Flow cytometric analysis was performed using a BD FACSCalibur flow cytometer, all used antibodies were purchased from Becton–Dickinson (BD), Oxford, UK.
In brief, 50 μl blood was labeled within 20 min with CD45-FITC, CD14-APC, and CD41-PE for 15 minutes at room temperature. At that point, RBCs were lysed using buffered ammonium chloride and examained by FACScan.
Monocytes were specified by gating based on CD45 expression and side scatter to select monocytes. The content of MPAs was determined by the co-expression of the platelet marker CD41 (glycoprotein IIb/ IIIa) and monocyte marker CD14 (lipopolysaccharide protein receptor) on monocytes and was expressed as mean fluorescence intensity (MFI), as relative count (represent the percentage of monocytes with coexpression CD41 andCD14 on all monocytes). The absolute count (cells per μl) was calculated from the relative number proportionally to the count in the standard blood test. A least of 3000 monocytes was calculated for each test.
Platelet surface CD40 ligand
Blood was diluted 1:10 in (phosphate-buffered saline solution (PBS) and labeled within 10 minutes of collection by incubation with specific antibodies. Fifteen microliters of diluted blood sample were incubated with 10 μl of CD42b-FITC (binds to “resting and activated” platelet glycoprotein1b) and 10 μl of CD154 (which binds to platelet CD40L). Samples were diluted with 200 mL PBS after incubation and instantly examined and interpreted by FACScan flow cytometer. The results were expressed as the mean fluorescence index (MFI) of CD40L [11]. A minimum of 10000 platelets was counted for each test.
Statistical analysis
Statistical analysis was performed using SPSS version18. Data are given as mean ± standard deviation error of the mean (SD). For comparison, the data with a normal distribution were analyzed utilizing an unpaired Student’s t-test and data with a non-Gaussian distribution were tested using the Mann-Whitney U test.
Multivariate test was used for logistic regression analysis, Odds ratio and 95% confidence interval were calculated, and Receiver-operating characteristic (ROC) curve analyses were used to evaluate the sensitivity and specificity of each parameter for predicting POAF. A p value<.05 was considered as statistically significant.
Results
Clinical characteristics and laboratory investigations of the study participants are outlined in Table 1. Fifty patients were included in this study all patients underwent isolated on-pump surgery. Postoperative new AF was detected in 22% during post-operative hospitalization. No significant differences in the clinical demographic characteristics between the patients who remained in sinus rhythm and those who developed AF with respect to patients sex, associated diabetes, hypertension, and levels of preoperative hematological and other laboratory parameters. However, the neutrophil/lymphocyte (N/L) ratio was significantly higher in patients who develop POAF although no difference was found between neutrophil and lymphocyte counts. Also, platelet count was similar in both groups while MPV was significantly higher in the in patients who develop POAF group (8.7 ± 1.3 vs. 7.8 ± 1.2, p=0.001). Furthermore, serum cholesterol level and serum triglyceride level were significantly higher in patients with AF group.
| Parameters | Patients with AF (n=11) | Patients without AF (n=39) |
|---|---|---|
| Age (yrs.) mean | 62 ± 8 | 54 ± 9* |
| Range | (40-74) | (38-71) |
| Male/Female | 43167 | 43373 |
| Comorbidities: | ||
| Hypertension, n (%) | 5(45%) | 18(46%) |
| Diabetes mellitus, n (%) | 6(55%) | 20(50%) |
| Obesity, n (%) | 3(27%) | 14(36%) |
| Smoking n (%) | 6 (56%) | 23(59%) |
| COPD n (%) | 2(22%) | 5(13%) |
| Previous MI n (%) | 44(63%) | 43(55%) |
| Euroscore | 3.20 ± 1.98 | 2.52 ± 1.69* |
| Preoperative CCS, n (%): | ||
| I or II | 2(18%) | 6(15%) |
| III or IV | 9(82%) | 33(85%) |
| Echo. Findings: | ||
| Left atrial diameter, cm | 4.2± 1.95 | 4.1 ± 1.67 |
| LVEF (%) | 54.21 ±10.47% | 53.81 ± 10.29% |
| Angio. Findings n (%): | ||
| <4 vessels | 5(45%) | 19(49%) |
| = 4 vessels | 6(55%) | 20(51%) |
| Medications, n (%): | ||
| Aspirin | 10(90%) | 38 (97%) |
| Clopidogrel | 2(18%) | 12(31%) |
| Statins | 10(90%) | 35(89%) |
| ß-Blockers | 5(46%) | 27(69%) |
| ACEIs | 5(46%) | 22(56%) |
| Operative Data: | ||
| Operative time, min | 234.4 ± 33.7 | 216.2 ± 44.1* |
| Use of IABP n (%) | 1 (9%) | 3 (8%) |
| Laboratory parameters: | ||
| Cholesterol, mg/dl | 195 ± 78.7 | 169.4 ± 58.9* |
| Triglycerides, mg/dl | 205.2 ± 95.5 | 156.7 ± 62.4* |
| HDL, mg/dl | 44.9 ± 12.8 | 38.3 ± 15.9 |
| WBC count, × l09//L | 6.6 ± 3.4 | 7.1 ± 1.7 |
| Monocytes, % | 6.2 ± 1.8 | 5.1 ± 1.4 |
| Neutrophils, % | 56.4 ± 11.9 | 54.1 ± 10.4 |
| Lymphocyte, % | 21.1 ± 3.9 | 24.6 ± 5.1 |
| N/L ratio | 2.9 ± 1.4 | 2.3 ± 0.9* |
| Hb, gldL | 11.9 ± 1.8 | 12.4 ± 1.6 |
| Platelets, x 109/L | 219 ± 66 | 199 ± 92 |
| Creatinine, µmol/L | 114.1 ±2.6 | 110.1 ± 4.4 |
Table 1: Demographic and clinical characteristics of the study participants.
Results are presented as mean ± SD. P value<0.05 is significant. MPV= mean platelet volume; fl=femtoliter; sCD40L=soluble CD40 Ligand; sP-selectin= soluble P selectin; CRP= c reactive protein; MPA= monocyte-platelet aggregates.
Additionally, there was no variation in the preoperative medication lines between groups. What’s more, all patients underwent on-pump CABG, and there were no preoperative remarkable differences in the left ventricular function or LA diameter between those who experienced postoperative AF and those patients remained in sinus rhythm. However, mean age and Euroscore were significantly higher in the AF group, with a trend for a longer total duration of operation (Table 1).
Patients who developed POAF had higher levels of sCD40L and sPselectin however only sP-selectin reached statistical significance (p=0.04) compared to patients remained in sinus rhythm (SR). In addition, the level of CRP and D-dimer were higher in POAF patients (6.1 ± 1.8 vs. 5.9 ± 2.1; 0.14 ± 0.05 vs. 0.12 ± 0.03 respectively) (Table 2).
| Parameters | Patients with AF (n=11) | Patients without AF (n=39) | p-value |
|---|---|---|---|
| Platelet count | 219 ± 66 | 199 ± 92 | 0.54 |
| MPV (fl) | 8.7 ± 1.3 | 7.8 ± 1.2 | 0.001 |
| sCD40L (pg/ml) | 36.1 ± 3.4 | 34.1 ± 3.5 | 0.65 |
| sP-selectin (ng/ml) | 65.2 ± 1.9 | 52.1 ± 3.1 | 0.04 |
| D-dimer (mg FEU/l) | 0.14 ± 0.05 | 0.12 ± 0.03 | 0.45 |
| CRP (mg/l) | 6.1 ± 1.8 | 5.9 ± 2.1 | 0.65 |
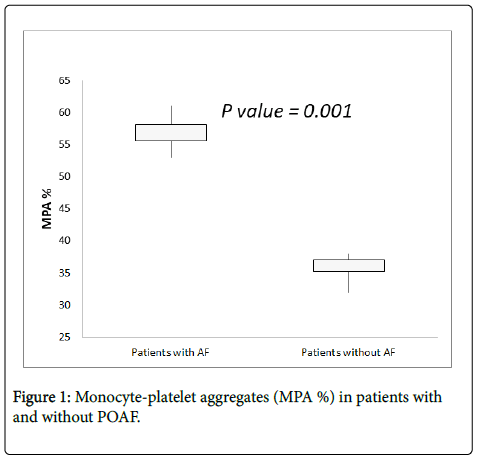
| MPA (%) | 57±4 | 35 ± 3 | 0.001 |
| Total MPA, per µl | 295 ± 31 | 137 ± 21 | 0.001 |
| CD4OL (MFI) | 12.6 ± 0.9 | 10.2 ± 1.4 | 0.04 |
| CD41 on monocytes (MFI) | 192.2 ± 12 | 119.2 ± 18 | 0.01 |
Table 2: Markers of platelet activation within the study patients
MPV=mean platelet volume; fl=femtoliter; sCD40L=soluble CD40 Ligand; sP-selectin= soluble P selectin; CRP=c reactive protein; MPA=monocyte-platelet aggregates.
Monocyte-platelet aggregates
The total Monocyte–platelet aggregates count were significantly increased in patients with POAF (295 ± 31 vs. 137 ± 21 cells/μl, p=0.001), and the relative proportion of MPAs on monocytes was also increased (57 ± 4 vs. 35 ± 3 %, p=0.001). In addition; the MFI of CD41 on monocytes were increased in patients with POAF and it was statistically significant (192.2 ± 12 vs. 119.2 ± 18, p=0.01) (Table 2 and Figure 1).
A multivariate logistic regression analysis was performed showed that MPV, MPAs% in addition to CRP were an independent predictors of postoperative AF after adjustment of the other parameters (odds ratio [OR] 1.97, 95% confidence interval [CI] 1.41-3.12, p=0.001; OR 2.09, 95% CI 1.89-3.22, p=0.005, and OR 1.18, 95% CI 1.09-1.25, p=0.05 respectively) (Table 3).
| Parameters | Odds ratio | 95% Confidence interval | p-value |
|---|---|---|---|
| Platelet count | 0.97 | 0.92-1.06 | 0.29 |
| MPV (fl) | 1.97 | 1.41-3.12 | 0.001 |
| sCD40L (pg/ml) | 1.87 | 1.38-2.98 | 0.004 |
| sP-selectin (ng/ml) | 1.07 | 0.97-1.34 | 0.23 |
| D-dimer (mg FEU/l) | 0.93 | 0.82-1.11 | 0.41 |
| CRP (mg/l) | 1.18 | 1.09-1.25 | 0.05 |
| MPA (%) | 2.09 | 1.89-3.22 | 0.005 |
| CD4OL (MFI) | 1.13 | 1.02-1.24 | 0.016 |
| CD41 on monocytes (MFI) | 1.15 | 1.06-1.32 | 0.03 |
Table 3: Multivariate logistic regression analyses of predictors for atrial fibrillation among AF patients.
CD 41 and CD40 L expression
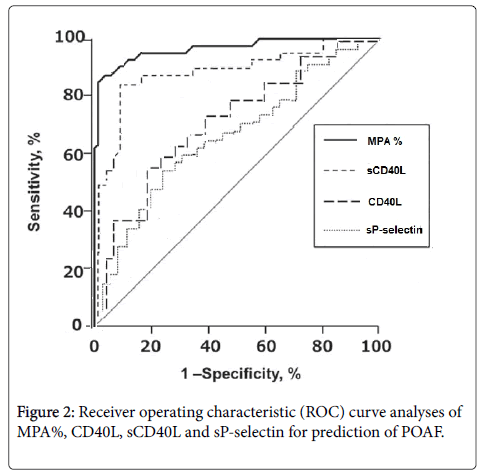
High expression of CD41 on monocyte was observed in patients with POAF compared to patients without POAF (192.2 ± 12 vs.119.2 ± 18, p=0.01). Similarly, The level of CD 40L expression on platelet surface was significantly higher in patients with POAF (p=0.04) (Table 2). Multivariate logistic regression analysis validated both, CD41 expression on monocytes and CD40L expression on platelets(p=0.01, p=0.04 respectively) as independent predictors for POAF after adjustment of the other parameters like age, diabetes, LVEF, LA diameter, leukocyte count, CRP and D-dimer (Table 3). In the Receiver operating characteristic (ROC) curve analysis, MPAs% in relation to the other markers demonstrated the highest sensitivity and specificity for prediction POAF (Figure 2).
Discussion
In the present study, POAF occurred in 22% of total 50 patients during hospitalization. The incidence of POAF has not decreased despite the growing advance in the surgical techniques and improved modality of post-operative care. Previous studies revealed different incidences of POAF which varies from 12% in a study by Sobczyk et al. [16] to 23% [8,17] and 26% [18] in other studies. The differences may be due to the age range of the studied patients, presence of coexisting diseases, the type of cardiac surgery and finally the criteria used for defining arrhythmia [8] inflammatory response induced during the extracorporeal circulation claimed to be the cause of increasing incidence of POAF [17].
In this study, the age of POAF patients were older than those without AF (average age 62 vs. 54 years, p<0.05), that may be due the changes occurred in the cardiac conduction system with aging. Similar results were reported by Banach et al [18], age was considered the most significant factor in increasing the risk of POAF [8,19]. There is an 8- fold increase in the risk of POAF with the age over 60 years (18), the risk increased by 100% with patients aged over 70 years [16,20]. The incidence rates of concurrent diseases like diabetes, hypertension, coexisting heart failure, or renal failure did not differ between those with and those without POAF patients in the present study as revealed previously [21].
High preoperative CRP was linked to the expanded risk of POAF [22,23]. Similarly, an elevated N/L ratio was related to the occurrence of post CABG AF [22,24]. Here, both CRP and N/L ratio were significantly higher in patients with POAF. Many methods are used to measure platelet activity either by direct assays (e.g. flow cytometry), a highly sensitive technique which can detect at least 1% of activated platelets or indirectly by detecting the activated platelets metabolites [25].
For the few last decades, MPV has been proved as an easy simple method of platelet activation in many diseases usually coexist with AF [25]. Previous studies found that MPV was an independent predictor of POAF and its level was significantly higher in those patients [22,26], we got the same finding in this study. MPV is an indicator for platelet activity where large platelets are commonly reactive with expanded inflammatory activity and thrombotic possibility [27].
Previous studies have proved the relationship between the leading role of platelets in inflammation and occurrence AF [28]. This could be explained by the release of some inflammatory markers like thromboxane A2 and beta thromboglobulin during platelet activation and even high expression of the adhesion molecules (22).
Elevated preoperative levels of sCD40L “a platelet activation marker” is associated with a higher risk of POAF after CABG surgery [5]. This platelet marker belongs to the tumor necrosis factor superfamily act as a proinflammatory and prothrombotic mediator [29].
sCD40L from the activated platelets promotes clot formation and consequently the risk of microthrombi. Together with it is role in the proinflammatory transcriptional pathways involved in the development of AF and postoperative arrhythmogenesis [30].
Studies denoted the role of sCD40L not only in the pathogenesis of AF but also in increasing the risk of thrombotic complications in POAF patients [12,31]. sP-selectin “ soluble product from platelet granule membrane protein P-selectin “ is another marker of platelet activation released mainly from the activated platelet after degranulation and a minimal amount from the endothelium [32]. Initial studies had identified associations between sP-selectin and many atherothrombotic risk factors like peripheral vascular disease and smoking [25]. Others proved that a higher level of sP-selectin is associated with an increased incidence of permanent AF than paroxysmal AF [11,33]. They had been suggested that sP-selectin gives background on chronic platelet activation [11], it can better detect activated platelets in clinical situations where chronic stimuli are existed [25]. Consistent with these studies, we found that the preoperative levels of the soluble platelet markers sCD40L together with sP-selectin were greater in patients with postoperative AF.
Postoperative AF can prompt hemodynamic instability, which in turn will increase the length of hospital stay and risk of stroke by twoto four fold [4,5]. While few studies described the association between postoperative AF and mortality risk. This association is independent of coexisting cardiovascular disease as CHF, left ventricular dysfunction, old age, and other comorbidities [1,34].
Platelet activation markers which are associated with AF, like CD40L and P-selectin remained in the blood for a short period after activation [10,35]. MPAs, on the other hand, believed to be more steady and stayed elevated for a while may be months after activation [36]. Previous studies evaluate the role of MPA as a marker of thrombotic risk in POAF patients [10]. The effect of blood stasis on platelets activation explained by Igarashi et al. as a negative correlation between A-ATIII complex and D-dimer level with the low blood flow founded [37].
Conclusion
Therefore, our results suggest that the increased level of platelet activation markers which indicates excess platelet activation usually connected with higher incidence of AF after coronary artery surgery. Preoperative platelet activation as affirmed by both soluble and cellular markers in addition to the content of MPAs markers seems to be an unprecedented predictor of the postoperative atrial fibrillation.
References
- Villareal RP, Hariharan R, Liu BC, Kar B, Lee VV (2004) Postoperative atrial fibrillation and mortality after coronary artery bypass surgery. J Am Coll Cardiol 43: 742-748.
- Mahoney EM, Thompson TD, Veledar E, Williams J, Weintraub WS, et al. (2002) Cost-effectiveness of targeting patients undergoing cardiac surgery for therapy with intravenous amiodarone to prevent atrial fibrillation. J Am Coll Cardiol 40: 737-745.
- Mariscalco G, Engstrom KG (2008) Atrial fibrillation after cardiac surgery: risk factors and their temporal relationship in prophylactic drug strategy decision. Int J Cardiol 129: 354-362.
- Mathew JP, Fontes ML, Tudor IC, Ramsay J, Duke P, et al. (2004) A multicenter risk index for atrial fibrillation after cardiac surgery. JAMA 291: 1720-1729.
- Antoniades C, Van-Assche T, Shirodaria C, Diesch J, Antonopoulos AS, et al. (2009) Preoperative sCD40L levels predict risk of atrial fibrillation after off-pump coronary artery bypass graft surgery. Circulation 120: S170-s176.
- Shen J, Lall S, Zheng V, Zheng V, Buckley P, Damiano RJ Jr, et al. (2011) The persistent problem of new-onset postoperative atrial fibrillation: a single-institution experience over two decades. J Thorac Cardiovasc Surg 141: 559-570.
- Echahidi N, Pibarot P, O’Hara G, Mathieu P (2008) Mechanisms, prevention, and treatment of atrial fibrillation after cardiac surgery. J Am Coll Cardiol 51: 793-801.
- Gorczyca I, Michta K, Pietrzyk E, Wozakowska-Kaplon B (2018) Predictors of post-operative atrial fibrillation in patients undergoing isolated coronary artery bypass grafting. Kardiol Pol 76: 195-201.
- Burdess A, Michelsen AE, Brosstad F, Fox KA, Newby DE, et al. (2012) Platelet activation in patients with peripheral vascular disease: reproducibility and comparability of platelet markers. Thromb Res 129: 50-55.
- Pfluecke C, Tarnowski D, Plichta L, Berndt K, Schumacher P, et al. (2016) Monocyte–platelet aggregates and CD11b expression as markers for thrombogenicity in atrial fibrillation. Clin Res Cardiol 105: 314-322.
- Choudhury A, Chung I, Panja N, Patel J, Lip GYH (2008) Soluble CD40 Ligand, Platelet Surface CD40 Ligand, and Total Platelet CD40 Ligand in Atrial Fibrillation Relationship to Soluble P-Selectin, Stroke RiskFactors, and Risk Factor Intervention. CHEST 134: 574-581.
- Choudhury A, Freestone B, Patel J, Lip GY (2007) Relationship of solubleCD40 ligand to vascular endothelial growth factor, angiopoietins, and tissue factor in atrial fibrillation: a link among platelet activation, angiogenesis, and thrombosis? Chest 132: 1913-1919.
- Ferroni P, Santilli F, Guadagni F, Basili S, Davi G (2007) Contribution of platelet-derived CD40 ligand to inflammation, thrombosis and neoangiogenesis. Curr Med Chem 14: 2170-2180.
- Canadian Cariovascular Society. CCS Mission, Canada.
- Dolgin M, Association NYH, Fox AC, Gorlin R, Levin RI (1994) Criteria Committee. Nomenclature and criteria for diagnosis of diseases of the heart and great vessels. 9th edn. Boston, MA: Lippincott Williams and Wilkins, New York Heart Association.
- Sobczyk D, Sadowski J, Sniezek-Maciejewska M (2005) Causes of atrial fibrillation early after coronary artery bypass grafting. Przegl Lek 62: 141-147.
- Wijeysundera DN, Beattie WS, Djaiani G, Rao V, Borger MA, et al. (2005) Off-pump coronary artery surgery for reducing mortality and morbidity: meta-analysis of randomized and observational studies. J Am Coll Cardiol 46: 872-882.
- Banach M, Rysz J, Drozdz JA, Okonski P, Misztal M, et al. (2006) Risk factors of atrial fibrillation following coronary artery bypass grafting. A Preliminary Report. Circ J 70: 438-441.
- Piccini JP, Zhao Y, Steinberg BA, He X, Mathew JP, et al. (2013) Comparative effectiveness of pharmacotherapies for prevention of atrial fibrillation following coronary artery bypass surgery. Am J Cardiol 112: 954-960.
- Dabrowski R, Sosnowski C, Jankowska A, Religa G, Kowalik I, et al. (2007) ACE inhibitor therapy: Possibleeffective prevention of new-onset atrial fibrillation following cardiac surgery. Cardiol J 14: 274-280.
- Filardo G, Hamilton C, Hebeler RF Jr (2009) New-onset postoperative atrial fibrillation after isolated coronary artery bypass graft surgery and long-term survival. Circ Cardiovasc Qual Outcomes 2: 164-169.
- Erdem K, Ayhan S, Ozturk S, Bugra O, Bozoglan O, et al. (2014) Usefulness of the mean platelet volume for predicting new-onset atrial fibrillation after isolated coronary artery bypass grafting. Platelets 25: 23-26.
- Lo B, Fijnheer R, Nierich AP, Bruins P, Kalkman CJ (2005) C-reactive protein is a risk indicator for atrial fibrillation after myocardial revascularization. Ann Thorac Surg 79: 1530-1535.
- Gibson PH, Cuthbertson BH, Croal BL, Rae D, El-Shafei H, et al. (2010) Usefulness of neutrophil/lymphocyte ratio as predictor of new-onset atrial fibrillation after coronary artery bypass grafting. Am J Cardiol 15: 186-191.
- Choudhury A, Chung I, Blann AD, Lip GY (2007) Platelet surface CD62P and CD63, mean platelet volume, and soluble/platelet P-selectin as indexes of platelet function in atrial fibrillation: a comparison of "healthy control subjects" and "disease control subjects" in sinus rhythm. J Am Coll Cardiol 49: 1957-64.
- Colkesen Y, Acil T, Abayli B, Yigit F, Katircibasi T, et al. (2008) Mean platelet volume is elevated during paroxysmal atrial fibrillation: A marker of increased platelet activation? Blood Coagul Fibrinol 19: 411-414.
- Gasparyan AY, Ayvazyan L, Mikhailidis DP, Kitas GD, et al. (2011) Mean platelet volume: A link between thrombosis and inflammation? Curr Pharm Des 17: 47-58.
- Aukrust P, Halvorsen B, Ueland T, Michelsen AE, Skjelland M, et al. Activated platelets and atherosclerosis. Expert Rev Cardiovasc Ther 8: 1297-1307.
- Henn V, Slupsky JR, Grafe M, Anagnostopoulos I, Forster R, et al. (1998) CD40 ligand on activated platelets triggers an inflammatory reaction of endothelial cells. Nature 391: 591-594.
- Kim YM, Kattach H, Ratnatunga C, Pillai R, Channon KM, et al. (2008) Association of atrial nicotinamide adenine dinucleotide phosphate oxidase activity with the development of atrial fibrillation after cardiac surgery. J Am Coll Cardiol 51: 68-74.
- Ferro D, Loffredo L, Polimeni L, Fimognari F, Villari P, et al. (2007) Soluble CD40 ligand predicts ischemic stroke and myocardial infarction in patients with non valvular atrial fibrillation. Arterioscler Thromb Vasc Biol 27: 2763-2768.
- Kamath S, Blann AD, Caine GJ, Gurney D, Chin BS, et al. (2002) Platelet P-selectin levels in relation to plasma soluble P-selectin and beta-thromboglobulin levels in atrial fibrillation. Stroke 33: 1237-1242.
- Li-Saw-Hee FL, Blann AD, Gurney D (2001) Plasma von Willebrand factor, fibrinogen and soluble P-selectin levels in paroxysmal, persistent and permanent atrial fibrillation: Effects of cardioversion and return of left atrial function. Eur Heart J 22: 1741-1747.
- Stamou SC, Dangas G, Hill PC (2000) Atrial fibrillation after beating heart surgery. Am J Cardiol 86: 64-67.
- Lim HS, Willoughby SR, Schultz C, Gan C, Alasady M, et al. (2013) Effect of atrial fibrillation on atrial thrombogenesis in humans: Impact of rate and rhythm. J Am Coll Cardiol 61: 852-860.
- Wrigley BJ, Shantsila E, Tapp LD, Lip GY (2013) Increased formation of monocyte-platelet aggregates in ischemic heart failure. Circ Heart Fail 6: 127-135.
- Igarashi Y, Kashimura K, Makiyama Y, Sato T, Ojima K, et al. (2001) Left atrial appendage dysfunction in chronic non valvular atrial fibrillation is significantly associated with an elevated level of brain natriuretic peptide and a prothrombotic state. Jpn Circ J 65: 788-792.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi