Research Article, Int J Cardiovasc Res Vol: 0 Issue: 0
Prevalence of Heart Disease among Asymptomatic Chronic Type 2 Diabetic Patients
Vinod Kumar Balakrishnan1*, Sangeetha Devendiran2, Anand NN2 and Rajendran SM2
1Sri Ramachandra University, Chennai, Tamil Nadu, India
2Sree Balaji Medical College and Hospital, Chennai, Tamil Nadu, India
*Corresponding Author : Vinod Kumar Balakrishnan
Assistant Professor, Cardiology Care Centre, Sri Ramachandra University, Chennai, India
Tel: 044 4592 8500
E-mail: ramangam@gmail.com, vinodkumar.b@sriramachandra.edu.in
Received: June 01, 2017 Accepted: July 31, 2017 Published: August 04, 2017
Citation: Balakrishnan VK, Devendiran S, Anand NN, Rajendran SM (2017) Prevalence of Heart Disease among Asymptomatic Chronic Type 2 Diabetic Patients. Int J Cardiovasc Res 6:4. doi: 10.4172/2324-8602.1000319
Abstract
Background: The burden of non-communicable diseases in India is rapidly increasing and two major diseases among them are Type 2 Diabetes Mellitus and Cardiovascular Diseases. The poor prognosis of diabetic patients has been explained by a greater incidence of heart failure and the adverse impact of diabetes on heart failure, perhaps reflecting an underlying diabetic cardiomyopathy exacerbated by left ventricular hypertrophy (LVH) and coronary artery disease (CAD). This could be avoided if screening techniques could be used to identify LV dysfunction in its preclinical phase. This study was done to evaluate the role of BNP as a screening tool.
Methodology: This study was done as a cross sectional study among 77 diabetic patients visiting a cardiac care center in a tertiary hospital in Chennai. BNP was immunoassayed using fluorescence detection technique. The results were compared with Treadmill test and Echocardiography. Validity of the screening test was assessed by estimating sensitivity, specificity, predictive values. ROC curve was plotted to assess the area under the curve.
Results: BNP levels >600 were found to have high sensitivity and specificity in detection of LV dysfunction, when compared with the gold standard investigations. The results were statistically significant (p<0.05).
Conclusion: BNP is a noninvasive and a feasible screening tool which may be used for early detection of LV dysfunction among the patients with Type 2 Diabetes Mellitus.
Keywords: BNP; Diabetes mellitus; LV dysfunction; Sensitivity
Introduction
Adults with diabetes have a two to fourfold greater risk for dying from cardiovascular diseases compared to those without diabetes [1]. The poor prognosis of these patients has been explained by a greater incidence of heart failure and the adverse impact of diabetes on heart failure, perhaps reflecting an underlying diabetic cardiomyopathy exacerbated by left ventricular hypertrophy (LVH) and coronary artery disease (CAD). Many patients with LV dysfunction remain undiagnosed and untreated until advanced disease causes disability [2]. This delay could be avoided if screening techniques could be used to identify LV dysfunction in its preclinical phase.
Standard echocardiography and stress echocardiographic testing may be used to screen for LVH and CAD. In addition, abnormal myocardial function in patients with diabetes mellitus but without overt heart disease has been identified by a number of groups [3-5] using sophisticated new echocardiographic techniques, including myocardial velocity, strain, and strain rate. Although these results have been shown to be abnormal across populations, the prevalence of abnormal findings has remained undefined. Moreover, the new tissue characterization parameters are not yet routine parts of the echocardiographic examination, giving them limited feasibility in the primary care setting. Measurement of brain natriuretic peptide (BNP) is a useful marker of both systolic and diastolic heart failure, and BNP levels are elevated in patients with LVH [6] and CAD [7]. We sought to determine the prevalence of LVH, CAD, and subclinical LV dysfunction in diabetic patients without known cardiac disease and whether a simpler approach on the basis of BNP measurements could be used to screen patients likely to have LVH, CAD, and abnormal myocardial function.
Fifty percent of all sudden cardiac deaths occur in people with no history of cardiac disease [8]. In fact, sudden cardiac death in people without prior cardiac disease accounts for nearly 10% of all adult deaths. This may be particularly pertinent to patients with diabetes, who have a high risk of coronary artery disease that is often silent. To prevent such events the first necessary step would be to develop a simple test (for example, a blood test) that can identify those patients with diabetes with silent coronary artery disease. Recent studies have led us to hypothesizes that B-type natriuretic peptide (BNP) may be able to do this. This is in addition to the well-known ability of BNP to identify left ventricular (LV) systolic dysfunction [9,10].
Our hypothesis is based on three main observations. Firstly, Goetze et al. [11] recently showed that ischemic myocardial tissue expresses more BNP than non-ischemic tissue. Secondly, in vitro cardiomyocytes degranulate and release BNP when they are made to be hypoxic [12]. Thirdly, in symptomatic angina BNP can identify the presence of coronary artery disease and myocardial ischaemia [13-17]. No study has yet addressed the key population of asymptomatic patients who are nevertheless at high risk of cardiac events, such as patients with type 2 diabetes.
Objective
The objective of the study is to evaluate the role of serum BNP value in early detection of LVD dysfunction & ischemia among diabetic patients as a screening tool.
Methodology
Study design
This cross sectional study was done to evaluate the role of serum BNP level in screening asymptomatic type 2 diabetic patients for silent myocardial infarction.
Study population
This study was conducted in the Department of Medicine and Department of cardiology Sree Balaji Medical College, Chennai, Tamil Nadu among the diabetic patients visiting the hospital.
Inclusion criteria
• Male and female patients of age > 30 years were selected.
• Subjects who are diabetic for >3 years with or without treatment.
• Subjects Blood Pressure should be between 110-140/70- 90mm hg with or without treatment.
• Subjects with no history of heart disease or complaints related to cardiac disease and no history of CAD, moderate to severe valvular disease, atrial fibrillation, or other severe arrhythmias and congenital heart disease.
• Subject with no history of End Stage Renal Failure.
• Subjects HbA1C >6%.
• Adult patients willing to give informed consent.
• Subject must be available for and willing to attend all evaluation visits.
Exclusion criteria
• Subjects with presence of pathological Q waves, LV hypertrophy on voltage criteria, or ST/T wave abnormalities in ECG.
• History of Congestive Heart Failure.
• History of End Stage Renal Failure.
• History of Smoking
• History of Alcohol
• Subject with uncontrolled hypertension
• Clinically significant abnormal laboratory results at screening
• Patient who is not willing to participate in the study
• Participating in a clinical research trial within 30 days of our study.
• Individuals who are cognitively impaired and/or who are unable to give Informed consent.
• Any other health or mental condition that in the Investigator’s opinion may adversely affect the subject’s ability to complete the study or its measures or that may pose significant risk to the subject.
Study duration
The study was carried out for one year between periods of August 2010 to August 2011.
Sample size and sampling
Based on the available literature, the prevalence of CAD among diabetic patients in Chennai was found to be 21.4% in the CUPS study [18]. Based on this the sample size was calculated as 67.3 at 95% confidence levels and 7% absolute precision. Accounting 10% for non-response, the final sample size was calculated as 74. A total of 77 patients with type 2 Diabetes Mellitus participated in the study. The subjects were selected by convenience sampling.
Ethical approval and informed consent
Ethical approval was obtained from the Institutional Ethics Committee. Informed consent was obtained from the study subjects prior to the data collection.
Data collection
Data collection comprised of obtaining background information, syptomatology and physical examination. The laboratory investigations included fasting blood glucose, HbA1C, Lipid profile, urea, creatinine, Liver function test and ECG. The study subjects then underwent next sets of investigation like BNP, Tread Mill Test and Resting echocardiography.
Operational definitions
BNP assay: The test measured BNP by immunoassay and utilized a fluorescence detection system. The assay is linear between 20 and 1300 pg/ml, with a lower limit of detection of 20 pg/ml. Results below and above the detection limits will be reported as <20 pg/ml and >1300 pg/ml, respectively. Samples >1300 pg/ml cannot be re-assayed after dilution. Whole blood was collected in a 4 ml lavender top tube. Moderately hemolyzed specimens were acceptable. Samples greater than 4 hours old were not acceptable for testing, based on stability studies. The rapid BNP test was available only on a STAT basis with a turn-around time of less than 60 minutes. No panic value levels were set [19].
Resting echocardiography: Subjects were examined in the left lateral decubitus position using a standard commercial ultrasound machine with a 2.5-MHz phased array probe. Three apical views (apical 4 chamber, 2-chamber, and long-axis views) were acquired using standard harmonic imaging. Mitral and pulmonary inflow velocities were recorded by using conventional pulsed-wave Doppler echocardiography. Left ventricular diameters and wall thicknesses were measured from 2-dimensional targeted M-mode echocardiography. Left ventricular mass was determined by Devereux’s formula [20]. Left ventricular hypertrophy was defined as LV mass index (g/m2) greater than 131 g/m2 in men and greater than 100 g/m2 in women [21]. Resting LV end-diastolic, end-systolic volumes and ejection fraction were computed using a modified Simpson’s biplane method. Regional wall motion analysis was scored as normal, mildly hypokinetic, severely hypokinetic, and akinetic by 2 observers blinded to the patient’s clinical data. Infarction was identified by resting wall motion abnormalities.
Stress testing: Exercise echocardiography was performed in patients who have fulfilled the inclusion and exclusion criteria. Blood pressure and cardiac status by 12-lead electrocardiogram were monitored during the exercise test. Regional wall motion was compared before and after stress, and patients with ischemia are identified by inducible wall motion abnormalities.
Results
There were 22 male and 55 female patients ranging from 30 years to 70 years. Of the 77 patients 28 had diastolic dysfunction and 23 had ischemia (inducible wall motion abnormalities and peak exercise), with 17 having both LVD and ischemia. The clinical characteristics of the study subjects are given in Table 1.
| Total Population n = 77 | Treadmill Positive n = 23 | Echocardiography Positive n = 28 | Both Treadmill & Echocardiography Positive n = 17 | Both Treadmill & Echocardiography Negative n = 43 | |
|---|---|---|---|---|---|
| Age | 51.32± 1.11 | 52.96 ± 2.24 | 52.21 ± 1.92 | 52.24 ± 2.63 | 50.23 ± 1.41 |
| Sex | 22/55 | 6/17 | 9/19 | 4/13 | 11/32 |
| Duration of Diabetes | 6.96 ± 0.55 | 10.65 ± 2.64 | 10.36 ± 2.28 | 11 ± 3.48 | 5.72 ± 0.53 |
| Urea | 27.67± 0.65 | 28.87 ± 1.34 | 28 ± 1.15 | 28.41 ± 1.71 | 27.12 ± 0.86 |
| Creatinine | 0.94 ± 0.02 | 0.97 ± 0.03 | 0.95 ± 0.03 | 0.97 ± 0.05 | 0.93 ± 0.02 |
| SGOT | 34.53 ± 1.73 | 33.91 ± 2.87 | 36.61 ± 3.51 | 35.82 ± 3.42 | 34.02 ± 1.98 |
| SGPT | 34.62 ± 1.81 | 34.35 ± 3.5 | 37.43 ± 3.66 | 36.18 ± 4.28 | 33.56 ± 2.06 |
| BAP | 222.89 ± 23.64 | 221.09 ± 24.68 | 204.71 ± 20.41 | 212.82 ± 32.48 | 231.72 ± 40.24 |
| Cholesterol | 168.77 ± 5.42 | 175.91 ± 10.11 | 163.61 ± 7.71 | 165.12 ± 10.83 | 166.88 ± 7.64 |
| Triglyceride | 185.33 ± 8.73 | 197.65 ± 19.52 | 181.11 ± 13.87 | 187.88 ± 21.14 | 177.30 ± 11.12 |
| HDL | 39.27 ± 0.42 | 38.70 ± 0.66 | 38.39 ± 0.47 | 28.35 ± 0.63 | 39.79 ± 0.63 |
| LDL | 92.45 ± 4.84 | 97.61 ± 9.31 | 87.43 ± 7.14 | 89.18 ± 10.23 | 91.67 ± 6.76 |
| VLDL | 37.06 ± 1.75 | 39.57 ± 3.89 | 37.79 ± 2.76 | 37.79 ± 2.76 | 35.47 ± 2.23 |
| BNP | 841.87 ± 39.39 | 834.57 ± 30.30 | 834.57 ± 30.30 | 440.53 ± 29.11 |
Table 1: Clinical characteristic of diabetic patients.
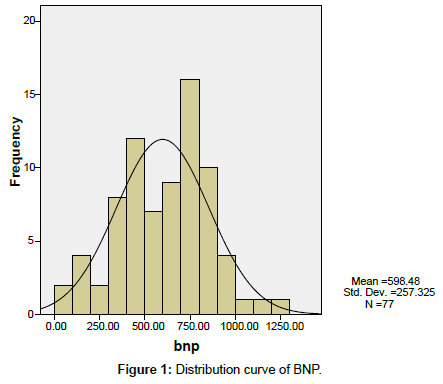
The distribution of BNP levels among the study subjects is given in Figure 1. The median NT pro BNP value of entire study population (n=77) were 598, age 51.32 ± 1.11, Male: Female ratio distribution was 22:55 and mean duration of diabetes was 6.96 ± 0.55 years.
The percentile distribution of BNP levels is given in Table 2. To identify the cut off value of NT pro BNP various range were considered using statistical analysis, the value was arived as >600
| Percentiles | |||||||
|---|---|---|---|---|---|---|---|
| 5 | 10 | 25 | 50 | 75 | 90 | 95 | |
| Weighted average BNP | 109.000 | 243.200 | 430.000 | 610.000 | 770.000 | 895.200 | 972.000 |
| Tukey’s Hinges BNP | 430.000 | 610.000 | 770.000 | ||||
Table 2: Percentile distribution of BNP.
The validity of BNP as a screening tool was assessed and is given in Table 3. It was observed that BNP levels beyond 600 and less than 800 had a statistical significant effect in terms of both the sensitivity and specificity (p<0.05).
| Parameters | BNP > 375 | BNP > 485 | BNP > 600 | BNP > 700 | BNP > 800 |
|---|---|---|---|---|---|
| Sensitivity (%) | 35 | 42 | 50 | 63 | 88.6 |
| Specificity (%) | 93 | 92 | 94 | 95 | 86 |
| Positive Predictive Value (%) | 95 | 91 | 91 | 91 | 65 |
| Negative Predictive value (%) | 25 | 46 | 61 | 77 | 96 |
| P value | 0.02 | 0.002 | .0001 | .0001 | .0001 |
| Odds Ratio | 7.70 | 9.05 | 16.50 | 36.7 | 48.7 |
| 95 % Confidence Interval | 0.94 – 167.24 | 1.77 – 62.05 | 3.20 – 113.86 | 6.74 – 265.7 | 1.11 – 384.15 |
Table 3: Validity of BNP as a screening tool against TMT gold standard test.
The validity of BNP with Echocardiography as the gold standard test was assessed and the results are given in Table 4. A higher sensitivity and specify was observed with statistical significance in BNP levels >700 and 800 (p<0.05).
| BNP > 375 | BNP > 485 | BNP > 600 | BNP > 700 | BNP > 800 | |
|---|---|---|---|---|---|
| Sensitivity | 45 | 54 | 64 | 72 | 100 |
| Specificity | 100 | 96 | 97 | 91 | 81 |
| Positive Predictive Value | 100 | 96 | 97 | 91 | 81 |
| Negative Predictive value | 15 | 53 | 73 | 81 | 100 |
| P value | 0.001 | .0001 | .0001 | .0001 | .0001 |
| Odds Ratio | - | 30.53 | 61.2 | 66.6 | - |
| 95 % Confidence Interval | - | 3.85 – 650.76 | 7.53 – 1323.4 | 6.49 – 121.4 | - |
Table 4: Validity of BNP as a screening tool against Echocardiography as gold standard test.
The validity of BNP as a screening test for BNP ≥ 600 is given in Table 5. Among 77 patients 28 had subclinical left ventricular diastolic dysfunction identified by a 2D echo. In these patients NT pro BNP was compared. NT pro BNP (>600) could predict diastolic dysfunction at sensitivity of 64% and negative predictive value was 73%, P = 0.0001. Suggestive BNP could be used effectively as screening tool to identify diastolic dysfunction.
| LVD | Silent Ischemia | Both Positive | |
|---|---|---|---|
| Sensitivity | 64 | 50 | 100 |
| Specificity | 97 | 94 | 58 |
| Positive Predictive Value | 96 | 91 | 40 |
| Negative Predictive Value | 73 | 61 | 100 |
| P value | .0001 | .0001 | .0001 |
| AUC | .829 | .762 | .792 |
Table 5: Validity of BNP as a screening tool for BNP ≥ 600.
Treadmill
Among the 77 patients 23 patients had developed silent ischemic changes on performing ETT. When BNP (>600) of this patients were observed was found BNP could predict silent ischemia at sensitivity of 50%, negative predictive value of 61%, P=0.0001.
Both treadmill and echocardiography
Out of the 77 subjects enrolled in the study, 17 of them had features of both diastolic dysfunction and silent ischemic changes. When compared with BNP cut off value it had sensitivity and negative predictive value was 100%.
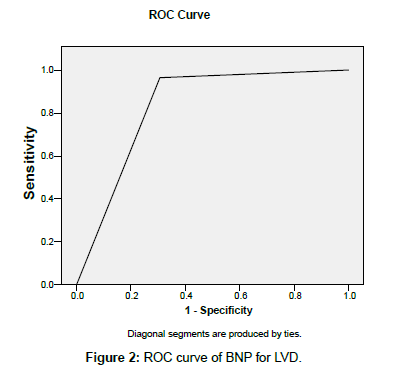
ROC curve for LVD is given in Figure 2. The Area Under the Curve (AUC) was found to be 82.9%.
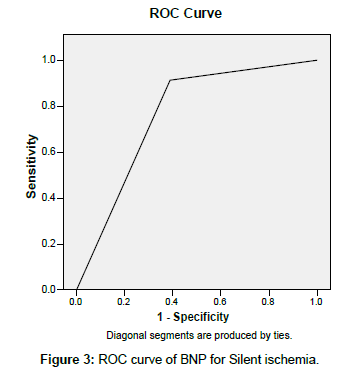
The ROC curve for Silent Ischemia is given in Figure 3. The AUC was found to be 76.2%
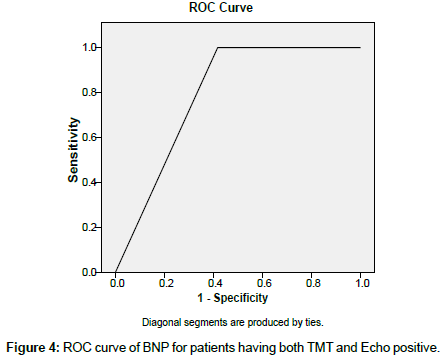
The ROC Curve for Both TMT and Echocardiography positive subjects is given in Figure 4. The AUC was found to be 79.2%
Discussion
Both echocardiography and BNP have clinically been used for screening for and monitoring LV dysfunction, particularly in those with high possibility of development of heart failure. The present study has demonstrated that echocardiographic screening of asymptomatic diabetic subjects with apparently normal cardiac function may identify significant numbers of patients with asymptomatic ischemia, LVH, and subclinical LV dysfunction. Measuring BNP is simple and rapid, and has been suggested as a screening test for LV dysfunction in patients with diabetes mellitus [22]. BNP levels in the present study were significantly different between patients with and without subclinical LV dysfunction.
Screening for LVD and CAD
Conventional echocardiography is routinely used for screening for LVD in clinical settings. The current study demonstrated that BNP was significantly increased in older or female patients, confirming previous findings [23]. However, although BNP was higher in patients with LVD than those without LVD, consistent with previous findings [24] an abnormal BNP level was found in 22 patients, suggesting usefulness of BNP as a screening tool for LVD [25,26].
Coronary artery disease is commonly silent in patients with diabetes mellitus, and screening with stress echocardiography may be of value in identifying patients with occult CAD. However, although the coronary natriuretic peptide system is involved in the pathobiology of intimal plaque formation in human beings [27] and plasma BNP concentration has been shown to be markedly increased in patients with CAD, even without concomitant LV dysfunction [7], the present study suggests that BNP levels can be used for CAD screening. Our findings are differs with the previous work, showing that BNP is elevated even in uncomplicated ischemic heart disease without increased LV wall stress or myocardial dysfunction [28].
Subclinical diabetic heart disease
Recent application of new sensitive techniques such as myocardial velocities, strain, and strain rate has enabled noninvasive detection of the abnormal LV function in diabetic hearts at an early stage [3,5]. However, age and sex are important factors associated with LV dysfunction [9] and have not individually been taken into account in the determination of cardiac functional status in these studies. By application of recently described age- and sex-adjusted normal myocardial velocity ranges to define the prevalence of subclinical diabetic heart disease in patients without known heart disease, the present study demonstrated that 36% patients had significant subclinical LV dysfunction in patients with diabetes but without LVD or CAD.
BNP in Diabetes
Brain natriuretic peptide has been found to be elevated in patients with heart failure and asymptomatic or minimally symptomatic LV dysfunction [29] and reliably predicts the presence of overt LV dysfunction by echocardiography [30]. However, BNP measurement seems to have usefulness in identifying patients with LVH and seems to have value in detection of CAD in diabetic patients without known heart disease.
Importantly, the balance of evidence suggests that BNP is uninfluenced by diabetes itself—BNP release has no relationship with acute hyperinsulinemic conditions [31] does not rise in response to acute hyperglycemia in type 1 diabetes [32] and diabetes does not influence the expression of BNP messenger RNA (mRNA) in the ventricular myocardium [33]. However, some data do suggest that BNP release is reduced in diabetes; after ligation of the coronary artery for 1 week, the ventricular BNP mRNA increased less in diabetic than nondiabetic rats [34]. The association of obesity and type 2 diabetes may be important, as obesity is associated with low BNP [35,36]. Overall, these data suggest that diabetes may inhibit or at least have no effect on LV BNP synthesis.
Since the study population group is very small it can’t be generalized for entire population. Further studies are required in larger groups to identify whether NT pro BNP can be used as a screening tool at primary health care center level. This will help in identifying asymptomatic cardiac disease among diabetic patients. The currently cost of NT pro BNP is high, when we use it in a larger scale we hope the cost might get reduced.
Conclusion
This study shows a clear correlation between occurrence of increase in diastolic dysfunction and ischemia with increase in BNP concentration. Furthermore, the results suggest that NT pro BNP value above 600 is a good indicator for referring a patient for echocardiography and exercise tolerance test. We conclude that a single measurement of NT pro BNP at the diabetic OPD can provide important information about the cardiac status of asymptomatic diabetes patients who might require a cardiac evaluation.
References
- CDC (2003) Self-reported heart disease and stroke among adults with and without diabetes. Morb Mortal Wkly Rep 52: 1065-1070.
- Fang ZY, Leano R, Marwick TH (2004) Relationship between longitudinal and radial contractility in subclinical diabetic heart disease. Clin Sci (Lond) 106: 53-60.
- Andersen NH, Poulsen SH, Eiskjaer H, Poulsen PL, Mogensen CE (2003) Decreased left ventricular longitudinal contraction in normotensive and normoalbuminuric patients with type II diabetes mellitus: a Doppler tissue tracking and strain rate echocardiography study. Clin Sci 105: 59 - 66.
- Fang ZY, Najos-Valencia O, Leano R, Marwick TH (2003) Patients with early diabetic heart disease demonstrate a normal myocardial response to dobutamine. J Am Coll Cardiol 42: 446 -453..
- Vinereanu D, Nicolaides E, Tweddel AC, Madler CF, Holst B, et al. (2003) Subclinical left ventricular dysfunction in asymptomatic patients with type II diabetes mellitus, related to serum lipids and glycated haemoglobin. Clin Sci 105: 591-599.
- Murakami Y, Shimada T, Inoue S, Shimizu H, Ohta Y, et al. (2002) New insights into the mechanism of the elevation of plasma brain natriuretic polypeptide levels in patients with left ventricular hypertrophy. Can J Cardiol 18: 1294-1300.
- Goetze JP, Christoffersen C, Perko M, Arendrup H, Rehfeld JF, et al. (2003) Increased cardiac BNP expression associated with myocardial ischemia. FASEB J 17: 1105-1107.
- Vreede-Swagemakers JJ, Gorgels AP, Dubois-Arbouw WI, van Ree JW, Daemen MJ, et al. (1997) Out-ofhospital cardiac arrest in the 1990’s: a population-based study in the Maastricht area on incidence, characteristics and survival. J Am Coll Cardiol 30: 1500-1505.
- Chen HH, Burnett JC Jr (1999) The natriuretic peptides in heart failure: diagnostic and therapeutic potentials. Proc Assoc Am Physicians 111: 406-416.
- Stanek B, Frey B, Hülsmann M, Berger R, Sturm B, et al. (2001) Prognostic evaluation of neurohumoral plasma levels before and during beta-blocker therapy in advanced left ventricular dysfunction. J Am Coll Cardiol 38: 436-442.
- Goetze JP, Christoffersen C, Perko M, Arendrup H, Rehfeld JF, et al. (2003) Increased cardiac BNP expression associated with myocardial ischemia. FASEB J 17: 1105-110.
- Hopkins WE, Chen Z, Fukagawa NK, Hall C, Knot HJ, et al. (2004) Increased atrial and brain natriuretic peptides in adults with cyanotic congenital heart disease: enhanced understanding of the relationship between hypoxia and natriuretic peptide secretion. Circulation 109: 2872-2877.
- Weber M, Dill T, Arnold R, Rau M, Ekinci O, et al. (2004) N-terminal B-type natriuretic peptide predicts extent of coronary artery disease and ischemia in patients with stable angina pectoris. Am Heart J 148: 612-620.
- Davidson NC, Pringle SD, Pringle TH, McNeill GP, Struthers AD (1997) Right coronary artery stenosis is associated with impaired cardiac endocrine function during exercise. Eur Heart J 18: 1749-54.
- Asada J, Tsuji H, Iwasaka T, Thomas JD, Lauer MS (2004) Usefulness of plasma brain natriuretic peptide levels in predicting dobutamine-induced myocardial ischemia. Am J Cardiol 93: 702-704.
- Sadanandan S, Cannon CP, Chekuri K, Murphy SA, Dibattiste PM, et al. (2004) Association of elevated B-type natriuretic peptide levels with angiographic findings among patients with unstable angina and non-ST-segment elevation myocardial infarction. J Am Coll Cardiol 44: 564-568.
- Bibbins-Domingo K, Ansari M, Schiller NB, Massie B, Whooley MA (2003) B-type natriuretic peptide and ischemia in patients with stable coronary disease: data from the Heart and Soul study. Circulation 108: 2987-2992.
- Mohan V, Venkatraman JV, Pradeepa R (2010) Epidemiology of cardiovascular disease in type 2 diabetes: the Indian scenario. J Diabetes Sci Technol 4: 158-170.
- UCLA Diagnostic Module-2001.
- Devereux RB, Reichek N (1977) Echocardiographic determination of left ventricular mass in man. Anatomic validation of the method. Circulation 55: 613-618.
- Levy D, Savage DD, Garrison RJ, Anderson KM, Kannel WB, et al. (1987) Echocardiographic criteria for left ventricular hypertrophy: the Framingham Heart Study. Am J Cardiol 59: 956-960.
- Redfield MM, Rodeheffer RJ, Jacobsen SJ, Mahoney DW, Bailey KR, et al. (2002) Plasma brain natriuretic peptide concentration: impact of age and gender. J Am Coll Cardiol 40: 976-982.
- Dawson A, Struthers AD (2003) Screening for treatable left ventricular abnormalities in diabetic patients. Expert Opin Biol Ther 3: 107-112.
- Almeida P, Azevedo A, Rodrigues R, Dias P, Friões F, et al. (2003) B-type natriuretic peptide and left ventricular hypertrophy in hypertensive patients. Rev Port Cardiol 22: 327-336.
- Vasan RS, Benjamin EJ, Larson MG (2002) Plasma natriuretic peptides for community screening for left ventricular hypertrophy and systolic dysfunction: the Framingham heart study. JAMA 288:1252 -1259.
- Nakamura M, Tanaka F, Yonezawa S, Satou K, Nagano M, et al. (2003) The limited value of plasma B-type natriuretic peptide for screening for left ventricular hypertrophy among hypertensive patients. Am J Hypertens 16: 1025-1029.
- Casco VH, Veinot JP, Kuroski de Bold ML, Masters RG, Stevenson MM, et al. (2002) Natriuretic peptide system gene expression in human coronary arteries. J Histochem Cytochem 50: 799-809.
- Arad M, Elazar E, Shotan A, Klein R, Rabinowitz B (1996) Brain and atrial natriuretic peptides in patients with ischemic heart disease with and without heart failure. Cardiology 87: 12-17.
- Tsutamoto T, Wada A, Maeda K, Hisanaga T, Mabuchi N, et al. (1999) Plasma brain natriuretic peptide level as a biochemical marker of morbidity and mortality in patients with asymptomatic or minimally symptomatic left ventricular dysfunction. Comparison with plasma angiotensin II and endothelin-1. Eur Heart J 20:1799 - 1807.
- Maisel AS, Koon J, Krishnaswamy P, Kazenegra R, Clopton P, et al. (2001) Utility of B-natriuretic peptide as a rapid, point-of-care test for screening patients undergoing echocardiography to determine left ventricular dysfunction. Am Heart J 141: 367-374.
- Tanabe A, Naruse M, Wasada T, Naruse K, Yoshimoto T, et al. (1995) Effects of acute hyperinsulinemia on plasma atrial and brain natriuretic peptide concentrations. Eur J Endocrinol 132: 693-698.
- McKenna K, Smith D, Tormey W, Thompson CJ (2000) Acute hyperglycaemia causes elevation in plasma atrial natriuretic peptide concentrations in type 1 diabetes mellitus. Diabet Med 17:512 -517.
- Christoffersen C, Goetze JP, Bartels ED, Larsen MO, Ribel U, et al. (2002) Chamber-dependent expression of brain natriuretic peptide and its mRNA in normal and diabetic pig heart. Hypertension 40: 54-60.
- Inoue M, Kanda T, Arai M, Suga T, Suzuki T, et al. (1998) Impaired expression of brain natriuretic peptide gene in diabetic rats with myocardial infarction. Exp Clin Endocrinol Diabetes 106: 484 - 488.
- Wang TJ, Larson MG, Levy D, Benjamin EJ, Leip EP, et al. (2004) Impact of obesity on plasma natriuretic peptide levels. Circulation 109: 594-600.
- Mehra MR, Uber PA, Park MH, Scott RL, Ventura HO, et al. (2004) Obesity and suppressed B-type natriuretic peptide levels in heart failure. J Am Coll Cardiol 43: 1590-1595.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi