Research Article, Int J Cardiovasc Res Vol: 9 Issue: 3
Routinely Performed Echocardiography and its Impact on Everyday Clinical Practice in Patients admitted to an Internal Medicine Ward
Ewa Kwiecien1, Leszek Drabi1,2, Aleksandra Matuszyk3, Anna Tyrka1, Barbara Widlinska1, Tomasz Luberda1, Barbara Biernacka1 and Wojciech Plazak1*
11Department of Cardiac and Vascular Diseases, Jagiellonian University Medical College, Krakow, Poland
22Department of Pharmacology, Jagiellonian University Medical College, Krakow, Poland
3Department of Anatomy, Faculty of Medicine, Jagiellonian University Medical College, Krakow, Poland
*Corresponding Author: Dr. Wojciech Plazak
Department of Cardiac and Vascular Diseases
Institute of Cardiology, Jagiellonian University School of Medicine
The John Paul II Hospital, Pradnicka 80 Street, 31-202 Krakow, Poland
Mobile: +48126142287
E-mail: w.plazak@szpitaljp2.krakow.pl
Received: April 24, 2020 Accepted: May 13, 2020 Published: May 20, 2020
Citation: Kwiecien E, Drabi L, Matuszyk A, Tyrka A, Widlinska B, et al. (2020) Routinely Performed Echocardiography and its Impact on Everyday Clinical Practice in Patients admitted to an Internal Medicine Ward. Int J Cardiovasc Res 9:3.
Abstract
Objectives: The aim of this study was to evaluate the frequency of significant heart abnormalities detected by routinely applied echocardiography in patients hospitalized in the internal medicine ward due to any medical disease and assess its impact on everyday clinical practice. Methods: Transthoracic Echocardiography (TTE) examinations were performed routinely in a cohort of 2756 patients admitted to the internal medicine ward. Demographic and clinical variables were collected retrospectively. Results: A total of 2756 patients (48.1%; n=1201 females) with a mean age of 76.3 ± 4.3 years were enrolled in the study. Almost half of the patients (41.8%; n=1153) showed significant heart pathology: Left Ventricular Ejection Fraction (LVEF) <50%, at least moderate valvular defect, Right Ventricular Systolic Pressure (RVSP) >36 mmHg, or pericardial effusion, which required further diagnosis and/or therapy. LVEF<50% was observed in 15.5% (n=428) of patients, including 31 (1.1%) patients with extremely low LVEF (<20%). A total of 10.7% of patients (n=295) had severe valvular pathology. Elevated estimated RVSP was observed in 1 out of 6 patients (15.9%, n=446) including 6.9% (n=189) with RVSP >50 mmHg. RVSP >50 mmHg was associated with lung disease (49.2%, n =87), left heart disease (29.6%, n=56), and pulmonary embolism (7.3%, n=13). In the examined group, 28.3% (n=781) of patients required specialist consultation and 10.7% (n=295) were considered as candidates for surgery. Conclusion: TTE is an essential non-invasive tool to evaluate heart structure and function. Routinely performed echocardiography has a significant impact on the decision-making process or treatment in almost half of the patients admitted to the internal medicine ward.
Keywords: Echocardiography; Significant heart pathology; Older adults; Internal medicine ward.
Keywords
Echocardiography; Significant heart pathology; Older adults; Internal medicine ward.
Introduction
Cardiovascular Diseases (CVD) are a leading cause of death in developed countries [1,2] and are the most common cause for hospitalization (13.5% patients) in Poland [3]. Globally, acute myocardial infarction, coronary artery disease, and atherosclerosis are major causes for patient admissions to hospitals [3]. Heart Failure (HF) and valvular heart diseases are common findings in clinical practice in internal medicine and geriatric departments. Older adults with structural heart disorders generally have multiple comorbidities such as HF and chronic obstructive pulmonary disease, which worsen their prognosis [4-7].
Transthoracic Echocardiography (TTE) is a non-invasive, easily accessible examination that evaluates structure and function of the left Ventricle (LV), determines Ejection Fraction (EF), can reveal wall motion abnormalities, and evaluate Stroke Volume (SV) and diastolic function. Taken together, this can provide data to diagnose acute or previous Myocardial Infarction (MI) and HF. TTE is also an important tool in the assessment of valvular heart diseases, cardiomyopathies, and can be used to predict the probability of pulmonary embolism, pulmonary hypertension, and many other cardiovascular disorders occurring in adults such as congenital heart defects, cardiac tumors, pericardial effusion, and aortic aneurysm.
To the best of our knowledge, there have been no studies investigating the predictive value of echocardiography performed in each patient admitted to the internal medicine ward.
The aim of this study was to evaluate the frequency of significant heart abnormalities detected by routinely applied echocardiography in patients admitted to the internal medicine ward of the Ministry of Internal Affairs and Administration Hospital in Krakow due to any internal medicine disease and to assess its impact on clinical diagnosis and therapeutic procedures in everyday practice.
Patients and Methods
Study design and data collection
Our study was a single-center, observational study. We evaluated 2756 consecutive patients admitted to the internal medicine ward of a district hospital in Krakow between January 2008 and June 2011. Data regarding demographic and clinical variables were collected retrospectively. Comorbidities were classified according to ICD- 10.
Echocardiographic evaluation
All patients underwent TTE with Doppler using an ultrasound scanner (Logic 5). Standard 2D and M-mode images were recorded from standard views: parasternal long axis, parasternal short axis, apical 4-chamber, apical 2-chamber, and apical 3-chamber views. We measured LV end-diastolic and end-systolic diameter, LV wall thickness, right ventricular end-diastolic diameter, left and right atrial dimensions, Right Ventricular Systolic Pressure (RVSP, estimated based on tricuspid regurgitation velocity), and evaluated for presence of pericardial fluid. Left Ventricular Ejection Fraction (LVEF) was measured using the biplane Simpson method. Valve function was assessed using both spectral Doppler and color Doppler and evaluated according to 2017 ESC/EACTS Guidelines for the management of valvular heart disease [8].
LVEF was defined as preserved (LVEF ≥ 50%), middle-range reduced (40‑49%), or reduced (<40%) [9].
Mitral Regurgitation (MR), Aortic Regurgitation (AR), and Tricuspid Regurgitation (TR) were defined as mild, moderate, or severe on the basis of jet extension, vena contracta, Proximal Isovelocity Surface Area (PISA) radius, and regurgitation wave maximal velocity [8,10,11]. Aortic Valve Stenosis (AS) was defined as mild, moderate, or severe when the mean gradient was 10-20 mmHg, 20-40 mmHg, or >40 mmHg respectively and the valve area was >1.5 cm2, 1.0-1.5 cm2, or <1.0 cm2 respectively. Mitral Valve Stenosis (MS) was defined as moderate when the mean gradient was 5-10 mmHg accompanied by reduction of valve area to 1.0-1.5 cm2. MS was defined as severe if the mean gradient was >10 mmHg and valve area <1.0 cm2. RVS >36 mmHg was considered elevated, while significantly elevated RVSP was defined as >50 mmHg.
LV thickening (thickness of Interventricular Septum (IVS) in diastole >11 mm) or LV hypertrophy (IVS ≥ 15 mm in diastole) were defined according to Elliot et al. [12].
Statistical analysis
Data were presented as mean ± Standard Deviation (SD) or median and interquartile range, as appropriate. Categorical data were verified using the chi-squared test or Fisher's exact test. Normality was verified using the Shapiro-Wilk test and compared by the Student’s t-test or by the Mann-Whitney U test, as appropriate (for 2 groups). Continuous data in >2 groups were compared using one-way ANOVA followed by the Tukey post-hoc test or Kruskal–Wallis test, followed by a test for multiple comparisons of mean rank, as appropriate. To identify independent predictors of LVEF <40% and RVSP >50 mmHg, multiple logistic regression with backward elimination was performed. Models were adjusted for severe valvular defect (AR, MR, TR), RVSP, IVS (for EF <40%) LVEF, LA and RV (for RVSP >50 mmHg). The significance level was set at p<0.05. Statistical analyses were performed using a standard software package, STATISTICA version 13 (Statsoft Inc, Tulsa, OK).
Results
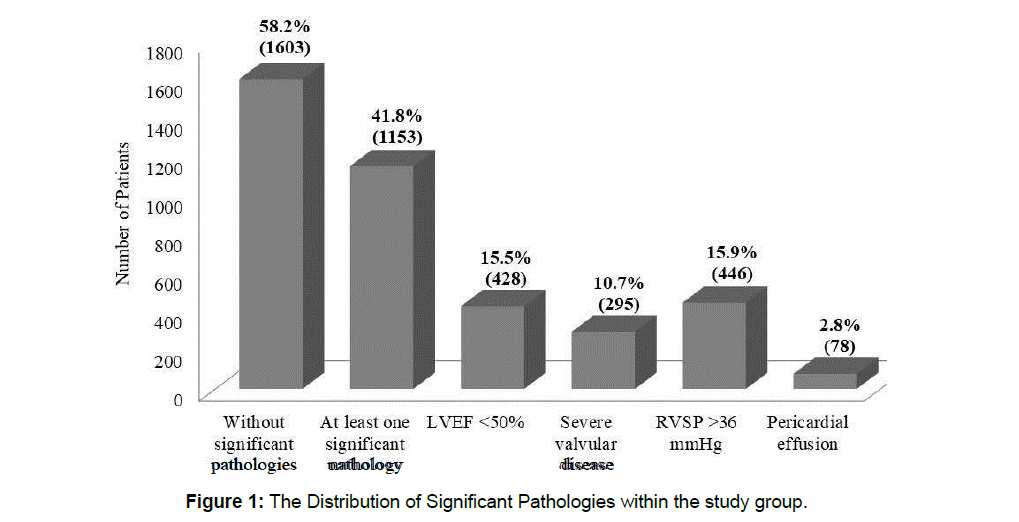
The main findings of the study are as follows (Figure 1)
1) In our study group, 41.8% (n=1153) of patients had at least 1 significant pathology defined as LVEF <50% or at least moderate valvular defect or RVSP >36 mmHg or pericardial effusion.
2) Reduced LVEF was observed in 15.5% (n=428) of patients. Approximately 1 out of 90 (1.1%, n=31) patients had extremely low LVEF, defined as LVEF <20%.
3) A total of 10.7% of patients (n=295) had severe valvular pathology.
4) Elevated estimated RVSP was observed in 1 out of 6 patients (15.9%, n=446), including 6.9% (n=189) with RVSP >50 mmHg.
5) RVSP >50 mmHg was associated with lung disease (49.15%, n =87), left heart disease (29.63%, n=56), and pulmonary embolism (7.3%, n=13).
6) Coexistence of 2 different pathologies was observed in 9.8% (n=268) of patients.
Patients with >3 pathologies (n=187) constituted 6.8% of the study group.
A total of 28.3% (n=781) of patients required specialist consultation and 10.7% (n=295) were candidates for surgical intervention.
Patient characteristics
A total of 2756 patients (48.1%; n=1201 females) with a mean age of 76.3±4.3 years were included in the study (Table 1-3)
| Variables | |
|---|---|
| Age (years) | 76.3 ± 4.3 |
| Women | 1201 (48.1) |
| BMI (kg/m2) | 28 (25-32) |
| Arterial hypertension | 2254 (83.1) |
| Hypercholesterolemia | 1738 (65.7) |
| Current smoking | 472 (18.2) |
| Diabetes mellitus | 594 (23.6) |
| Coronary artery disease | 753 (28.9) |
| Previous myocardial infarction | 415 (16.2) |
| Heart failure | 772 (29.1) |
| Atrial fibrillation | 849 (32.9) |
| Previous stroke/TIA | 659 (24.8) |
| Chronic kidney disease stage 3 | 754 (28.7) |
| COPD/asthma | 471 (19.9) |
| SBP (mmHg) | 143 (131-156) |
| DBP (mmHg) | 81 (73-91) |
| Heart rate (beats/min) | 76 (67-82) |
| GFR (mL/min) | 57 (41-74) |
| Hb (g/dL) | 12.9 (11.8–14.2) |
| ALT (U/L) | 23 (16.0-43) |
| LDL (mmol/l) | 3.3 (2.8-4.1) |
Values are n (%), mean (standard deviation), or median and interquartile range.
ALT – Alanine Amino Transferase, BMI – Body Mass Index, COPD – Chronic Obstructive Pulmonary Disease, DBP – Diastolic Blood Pressure, GFR – Glomerular Filtration rate, Hb – Hemoglobin, LDL – Low Density Lipoprotein, SBP – Systolic Blood Pressure, TIA – Transient Ischemic Attack.
Table 1: Baseline characteristics.
| LVEF <40% n=298 |
LVEF 40-49% n=130 | LVEF >50% n=2328 |
P-value | |
|---|---|---|---|---|
| Age, years | 79 (68-84) | 82 (65-87) | 81 (70-85) | 0.52 |
| Female n, % | 89 (29.8)* | 33 (25.3)* | 1015 (43.6) | <0.001 |
| LVEF, % | 28 (20-30)* | 45 (40-45)* | 65 (60-66) | <0.001 |
| LVEDD, mm | 57 (52-63)* | 52 (46-63)* | 47 (44-51) | <0.001 |
| Right ventricle, mm | 28 (25-31)* | 26.5 (24.5-29.5)* | 25 (23-28) | <0.001 |
| Left atrium, mm | 46 (41-50)* | 43 (38-47)* | 38 (35-42) | <0.001 |
| RVSP, mmHg | 45 (38-50)* | 40.5 (35-52.5)* | 35 (30-45) | <0.001 |
| RVSP | ||||
| ≤36 mmHg; n (%) | 163 (54.7)* | 85 (63.6)* | 2062 (88.7) | <0.0001 |
| 36-50 mmHg; n (%) | 75 (25.2)* | 24 (18.5)* | 158 (6.8) | |
| >50 mmHg; n (%) | 60 (20.1)* | 21 (16.2)* | 108 (4.6) | |
| Aortic stenosis | ||||
| Mild; n (%) | 25 (8.3) | 14 (10.7) | 182 (7.8) | 0.392 |
| Moderate; n (%) | 7 (2.3) | 3 (2.3) | 38 (1.6) | |
| Severe; n (%) | 4 (1.3) | 3 (2.3) | 13 (83.3) | |
| Aortic regurgitation | ||||
| Mild; n (%) | 56 (18.8) | 24 (18.5) | 228 (9.8) | <0.001 |
| Moderate; n (%) | 19 (6.4) | 7 (5.4) | 71 (3.1) | |
| Severe; n (%) | 3 (1.0) | 1 (0.8) | 7 (0.3) | |
| Mitral regurgitation | ||||
| Mild; n (%) | 60 (20.1)* | 38 (29.2) | 641 (27.5) | <0.0001 |
| Moderate; n (%) | 93 (31.2)* | 33 (25.4)* | 333 (14.3) | |
| Severe; n (%) | 106 (35.6)* | 24(18.5)* | 120 (5.1) | |
| Mitral stenosis | ||||
| Moderate; n (%) | 5 (1.7) | 5 (3.8) | 24 (1.0) | 0.357 |
| Severe; n (%) | 2 (0.7) | 6 (4.6) | 11 (0.5) | |
| Tricuspid regurgitation | ||||
| Mild; n (%) | 59 (19.9) | 32 (24.6)* | 429 (18.4) | <0.0001 |
| Moderate; n (%) | 56 (18.9)* | 21 (16.2)* | 227 (9.8) | |
| Severe; n (%) | 84 (25.0)* | 26 (20.0)* | 94 (5.3) | |
| Pericardial fluid; n (%) | 11 (3.7)* | 3 (2.3) | 25 (1.1) | <0.001 |
*P<0.05 vs EF >50%. Values are n (%) or median and interquartile range. LVEF – Left Ventricular Ejection Fraction, LVEDD – Left Ventricular End-Diastolic Diameter, RVSP – Right Ventricular Systolic Pressure
Table 2: Baseline characteristics according to Left Ventricular Ejection Fraction (LVEF).
| RVSP ≤36 mmHg n=2310 |
RVSP 36-50 mmHg n=257 |
RVSP >50 mmHg n=189 |
P-value | |
|---|---|---|---|---|
| Age, years | 78 (69-84) | 80 (71-86) | 82 (71-86) | 0.39 |
| Female n, % | 862 (37.3) | 36 (45.0)* | 102 (53.9)* | <0.001 |
| LVEF, % | 65 (60-66) | 60 (35-65)* | 50 (30-60)* | <0.001 |
| LVEDD, mm | 2062 (89.3) | 158 (61.5)* | 108 (57.2)* | |
| 85 (3.7) | 24 (9.3)* | 21 (11.1)* | <0.001 | |
| 163 (7.1) | 75 (29.2)* | 60 (31.8)* | ||
| Right ventricle, mm | 48 (44-52) | 49 (45-55)* | 50 (44-56)* | 0.001 |
| Left atrium, mm | 25 (23-28) | 27 (24.5-30)* | 28 (25-31)* | <0.001 |
| RVSP, mmHg | 38 (35-42) | 43 (38-47)* | 45 (40-50)* | <0.001 |
| RVSP | 30 (30-35) | 40 (40-45) | 55 (50-60) | <0.001 |
| Aortic stenosis | ||||
| Mild; n (%) | 166 (7.1) | 32 (12.4) | 23 (12.3) | 0.159 |
| Moderate; n (%) | 32 (1.3) | 9 (3.5) | 7 (3.7) | |
| Severe; n (%) | 7 (0.3) | 7 (0.3) | 4 (0.2) | |
| Aortic regurgitation | ||||
| Mild; n (%) | 224 (9.7) | 43 (16.7) | 41 (21.7) | <0.001 |
| Moderate; n (%) | 65 (2.8) | 65 (2.8)* | 10 (5.3)* | |
| Severe; n (%) | 5 (0.2) | 1 (0.4) | 5 (2.7)* | |
| Mitral regurgitation | ||||
| Mild; n (%) | 647 (28.0) | 63 (24.5) | 29 (15.3) | <0.001 |
| Moderate; n (%) | 311 (13.5) | 94 (36.6)* | 54 (28.6)* | |
| Severe; n (%) | 96 (4.2) | 68 (26.5)* | 86 (45.6)* | |
| Mitral stenosis | ||||
| Moderate; n (%) | 21 (0.1) | 8 (3.1)* | 5 (2.6)* | 0.017 |
| Severe; n (%) | 5 (<0.1) | 5 (1.9)* | 9 (4.7)* | |
| Tricuspid regurgitation | ||||
| Mild; n (%) | 412 (17.8) | 78 (30.4) | 30 (15.9) | <0.001 |
| Moderate; n (%) | 144 (6.2) | 89 (34.4)* | 71 (37.6)* | |
| Severe; n (%) | 50 (2.6) | 81 (31.6)* | 82 (43.3)* | |
| Pericardial fluid; n (%) | 23 (1.0) | 5 (1.9) | 11 (5.9)* | <0.001 |
*p<0.05 vs RVSP <36 mmHg. Values are n (%) or median and interquartile range. LVEF – Left Ventricular Ejection Fraction, LVEDD - Left Ventricular End-Diastolic Diameter, RVSP - Right Ventricular Systolic Pressure.
Table 3: Baseline characteristics according to Right Ventricular Systolic Pressure (RVSP).
LV hypertrophy
Mild hypertrophy of the LV wall was found in 885 (32.1%) patients, while significant segmental hypertrophy (IVS in diastole ≥ 15 mm) occurred in 133 (4.8%) patients.
Left ventricular ejection fraction
LVEF 40-49% and <40% were observed in 4.7% (n=130) and 10.8% (n=298) of patients, respectively. Patients with LVEF <40%, compared to those with preserved LVEF, had thicker IVS and larger heart chambers (p<0.001 for all), 7-fold higher prevalence of severe MR (35.6% versus 5.1% of patients; p<0.001), and 5-fold higher prevalence of severe TR (30.7% vs. 5.9%; p<0.001). Individuals in this group had 28% greater RVSP (p<0.001). Significantly elevated RVSP >50 mmHg was observed over 4 times more frequently in this group (20.1% vs. 4.6%; p<0.001). LVEF <40% was independently associated with severe MR (odds ratio, OR 2.39, 95% CI, 1.36-4.19, p=0.002) and RVSP (odds ratio 1.08 per 1 mmHg, 95% CI, 1.05- 1.10, p<0.001).
Valvular heart disease
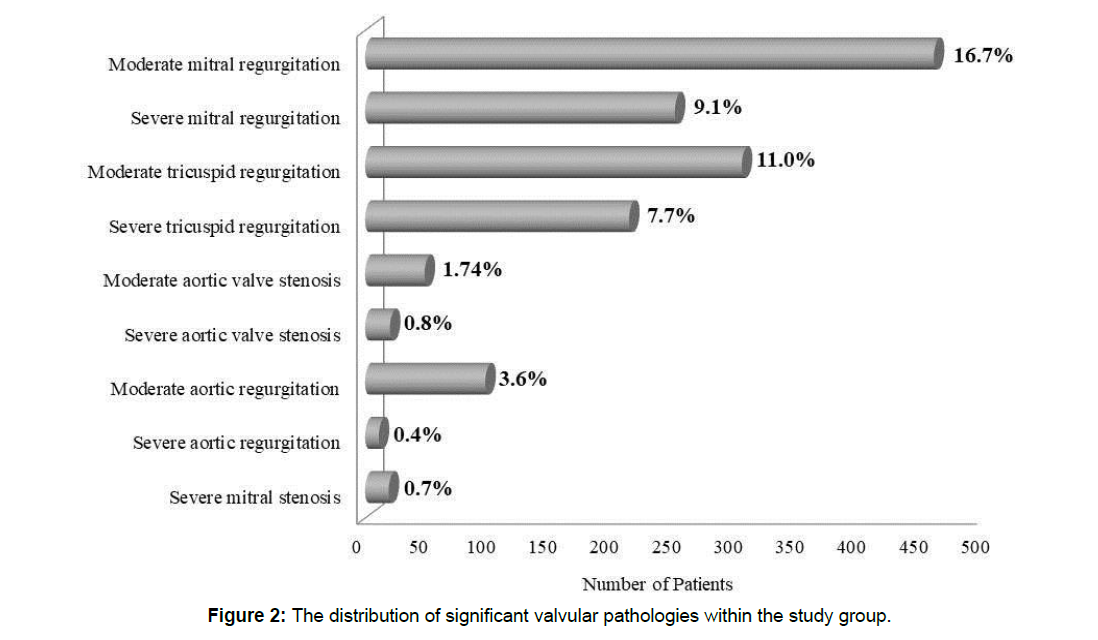
Coexistence of 2 valvular defects or presence of a single severe valvular defect was found in 96 (3.5%) cases. Moderate or severe MR was found in 16.7% (n=459) or 9.1% (n=250) of patients, respectively. Severe MS was diagnosed in 0.7% (n=19) of patients. In our study group, 3.6% of individuals (n=97) had moderate AR, while 0.4% (n=11) had severe aortic insufficiency. Moderate or severe AS was diagnosed in 1.7% (n=48) or 0.8% (n=20) of patients, respectively. Moderate TR constituted 11.0% (n=304) of the study group, whereas severe defect comprised 7.7% (n=213) (Figure 2). 5.6 Patent foramen ovale and congenital heart defects Patent foramen ovale/Atrial Septal Defect (ASD) was found in 36 (1.3%) cases. One post-myocardial infarct Ventricular Septal Defect (VSD) was diagnosed. The majority of ASD (93.1%) were hemodynamically non-significant. Two patients (6.9%) were referred for further evaluation. The patient with VSD was qualified for conservative treatment due to the high risk associated with surgery.
Elevated RVSP
Patients with significantly elevated RVSP >50 mmHg had a higher prevalence of mid-range LVEF (11.1% vs. 3.7%; p<0.001) and LVEF <40% (31.8% vs. 7.1%; p<0.001), as well as a larger LV (p=0.003), right ventricle and left atrium (p<0.001 for both), and thicker IVS (p=0.002). The prevalence of severe valvular defects was significantly higher in the group with RVSP >50 mmHg. These valvular defects included severe MR (45.6% vs. 4.2 %), severe TR (43.3% vs. 2.6%), severe AS (5.3% vs. 0.3%), and severe MS (5.3% vs. 0.2%; p<0.001 for all). Furthermore, patients in this group presented more frequently with pericardial effusion (5.9% vs. 1%; p<0.001). RVS >50 mmHg was independently predicted by LVEF (OR 0.95 per 1%, 95% CI, 0.94-0.98, p<0.001), severe TR (odds ratio 33.3, 95% CI, 17.9-61.9, p<0.001), right ventricular diameter (OR 1.08 per 1 mm, 95% CI, 1.03-1.20, p=0.003), and left atrial diameter (OR 1.08 per 1 mm, 95% CI, 1.03-1.20, p=0.001). A group of 33 patients (18.6%) was referred for diagnosis and treatment of suspected pulmonary arterial hypertension. Four (2.3%) patients with elevated RVSP died during hospitalization before a diagnosis was obtained.
Discussion
To the best of our knowledge, this is the first study showing that nearly half of patients admitted to the internal medicine ward have some form of significant heart pathology. This observation was highly influenced by age, as most of our patients were in their seventh or eighth decade of life. Older adults present with the highest incidence of CVD and have the largest potential to benefit from treatment [13]. The prevalence of cardiovascular diseases is increasing in Europe due to an aging society and there is a rising number of patients with chronic complications of ischemic heart disease and stroke [14]. The priority is in the prevention, early diagnosis, and proper treatment of these diseases. It has been reported that routinely performed echocardiographic examination may have an impact on long-term prognosis [5].
This study shows that echocardiography, applied in consecutive patients, may influence the decision-making process and should be considered as a supplementary procedure for patients admitted to geriatric and internal medicine departments. In our study group, 41.8% (n=1153) of patients had at least 1 significant pathology requiring intervention such as LVEF <50%, at least moderate valvular defect, RVSP >36 mmHg, or pericardial effusion. We believe this to be more than the number of abnormalities found in any other routinely performed test (such as morphology, electrolytes, C-reactive protein).
Left ventricular ejection fraction
LVEF was reduced in 15.5% of our patients. The first line treatment for LVEF <50% (15.5%) is pharmacological. In our study group, 1.1% (n=31) of patients had extremely low LVEF, defined as LVEF less than 20%, which corresponds to approximately 1 out of 90 cases. According to recent epidemiological reports, HF affects approximately 1–2% of the adult population and ≥ 10% among people>70 years of age in developed countries [9,15-18]. Echocardiography is essential in diagnosing and monitoring patients with HF. According to current guidelines, HF patients are divided into 3 groups: 1) those with normal LVEF ≥ 50%; 2) those with reduced LVEF <40%; 3) and those with LVEF in the range of 40–49% who represent a ‘grey area’, which is now defined as mid-range [9]. Assessment of LV function is crucial in evaluating the etiology of HF and response to therapies [19]. A diagnosis of heart failure worsens the patient’s prognosis and requires implementation of adequate treatment and monitoring [20]. The most recent European data (ESC-HF pilot study) show that 12-month all-cause mortality rates for hospitalized and stable/ambulatory HF patients were 17% and 7%, respectively, and the 12-month hospitalization rates were 44% and 32%, respectively [9, 21].
Valvular heart disease
Severe valvular heart diseases are less frequent than coronary artery disease, HF, or hypertension, but due to their negative impact on survival and quality of life, frequently require surgical intervention [4]. In our study 10.7% of patients had severe valvular pathology which should be considered as candidates for surgery or qualified to an observational group and intensively treated.
AS is the most frequent (33.9%) native valvular heart disease in Europ. Native valvular regurgitation accounts for 35% to 45% of all valvular heart disease [4,22,23]. MR comprises 24.8% of cases. The most frequent valvular heart diseases requiring surgery in adults are AS and MR [4]. In spite of the reduced frequency of rheumatic fever, MS is currently rare, while cases of MR are increasing in Europe [24]. Systematic monitoring by echocardiography is essential in the assessment and management of patients with MR [10].
Patients diagnosed with valvular heart disease are usually elderly and have a greater prevalence of comorbidities and cardiovascular risk factors, all of which have implications for therapeutic management [25].
Elevated RVSP
Patients with elevated RVSP should be evaluated for the presence of left heart disease, lung disease, thrombo-embolic pulmonary hypertension, and primary pulmonary arterial hypertension.
In our study group, 6.9% of patients presented with RVS >50 mmHg. This corresponds to more than 1 out of 15 patients who require further specialized investigation, diagnosis, and treatment due to the high probability of pulmonary hypertension.
Left heart disease (which is defined as HF, MR, or AS) is the most common cause of pulmonary hypertension [26, 27]. In patients with chronic HF, the occurrence of pulmonary hypertension rises with the progression of functional class impairment. Recent data show that up to 60% of patients with severe LV systolic dysfunction and up to 70% of HF patients with preserved ejection fraction may present with pulmonary hypertension [26]. Furthermore, pulmonary hypertension may be diagnosed in almost every patient presenting with severe symptomatic mitral valve disease and in up to 65% of those with symptomatic AS [28-30]. The prevalence of pulmonary hypertension increases with the severity of left-sided valvular defects and symptoms [28-30].
In patients with lung disease, it has been reported that systolic LV function is usually well preserved, but LV diastolic function is significantly reduced and is related to an increase in pulmonary artery pressure [7].
Pericardial effusion
Pericardial effusion due to any cause (such as inflammation, malignancy, MI, immunologic connective tissue diseases, hypothyroidism, etc.) requires prompt diagnosis and treatment.
Echocardiographic examination is useful as it allows quick evaluation of the patient’s status and is crucial in arriving at the correct diagnosis. Because of this, we recommend that echocardiographic examination should be considered for each patient admitted to the internal medicine ward.
Conclusion
Almost half of the patients admitted to an internal medicine ward due to any internal medicine disease presented with significant heart pathology (LVEF <50%, at least moderate valvular defect, RVSP>36 mmHg, or pericardial effusion) which required further diagnosis and/or therapy.
LVEF <50% was observed in 15.5% of patients, including extremely low LVEF (< 20%) in 1 out of 89 patients. Severe valvular pathology was seen in 1 out of 9 patients. More than 25% of the patients required specialist consultation, and more than 10% were candidates for surgical intervention. Elevated estimated RVSP was observed in 1 out of 6 patients including 6.9% of patients with RVSP >50 mmHg. RVSP elevation is associated with lung disease, left heart disease, and pulmonary embolism.
We showed that routinely performed echocardiography allowed detection of severe heart abnormalities in almost half of the patients admitted to the internal medicine ward due to any internal medicine pathology, which may have a significant impact on the decisionmaking process or treatment of these patients.
References
- Benjamin EJ, Virani SS, Callaway CW, Chamberlain AM, Chang AR, et al. (2018) Heart disease and stroke statistics-2018 update: a report from the american heart association. Circulation 137: e67-e492
- Nkomo VT, Gardin JM, Shelton TN, Gottdiener JS, Scott CG, et al. (2006) Burden of valvular heart diseases: a population-based-study. Lancet 368: 1005-1011.
- Wojtyniak B, Gorynski P (2018) Health status of polish population and its determinants – in short. National Institute of Public Health, National Institute of Hygiene, Warsaw.
- Iung B, Baron G, Butchart EG, Delahaye F, Gohlke-Bärwolf C, et al. (2003) A prospective survey of patients with valvular heart disease in Europe: The euro heart survey on valvular heart disease. Eur Heart J 24: 1231-1243.
- Song F, Liu FZ, Liang YF, Tse G, Li X, et al. (2019) Clinical, sonographic characteristics and long-term prognosis of valvular heart disease in elderly patients. J Geriatr Cardiol 16: 33-41.
- Douglas PS, Garcia MJ, Haines DE, Lai WW, Manning WJ, et al. (2011) Appropriate use criteria for echocardiography. J Am Soc Echocardiogr 24: 229–267.
- Suchoń E, Tracz W, Podolec P, Pieculewicz M, Płazak W, et al. (2007) Evaluation of left ventricular function in patients with chronic obstructive pulmonary disease. Pol Arch Med Wewn 117: 26-30.
- Baumgartner H, Falk V, Bax JJ, De Bonis M, Hamm C, et al. (2017) 2017 ESC/EACTS Guidelines for the management of valvular heart disease. Eur Heart J 38: 2739-2791.
- Ponikowski P, Voors AA, Anker SD, Bueno H, Cleland JGF, et al. (2016) 2016 ESC Guidelines for the diagnosis and treatment of acute and chronic heart failure: The task force for the diagnosis and treatment of acute and chronic heart failure of the European Society of Cardiology (ESC) Developed with the special contribution of the Heart Failure Association (HFA) of the ESC. Eur Heart J 37: 2129-2200.
- Lancellotti P, Tribouilloy C, Hagendorff A, Popescu BA, Edvardsen T, et al. (2013) Recommendations for the echocardiographic assessment of native valvular regurgitation: an executive summary from the european association of cardiovascular imaging. scientific document committee of the european association of cardiovascular imaging. Eur Heart J Cardiovasc Imaging 14: 611-644.
- Baumgartner H, Hung J, Bermejo J, Chambers JB, Edvardsen T, et al. (2017). Recommendations on the echocardiographic assessment of aortic valve stenosis: a focused update from the european association of cardiovascular imaging and the american society of echocardiography. J Am Soc Echocardiogr 30: 372-392.
- Elliott PM, Anastasakis A, Borger MA, Borggrefe M, Cecchi F, et al. (2014) 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: the task force for the diagnosis and management of hypertrophic cardiomyopathy of the European Society of Cardiology (ESC). Eur Heart J 35: 2733-2779.
- Orkaby AR, Rich MW (2018) Cardiovascular screening and primary prevention in older adults. Clin Geriatr Med 34: 81-93.
- The ERA-CVD Strategic Research Agenda (SRA-CVD): challenges and opportunities for cardiovascular disease research (2019) german aerospace center, Linder Hoehe, 51147 Koeln, Germany on behalf of the ERA-CVD consortium.
- Mosterd A, Hoes AW (2007) Clinical epidemiology of heart failure. Heart 93: 1137–1146.
- Redfield MM, Jacobsen SJ, Burnett JC Jr, Mahoney DW, Bailey KR et al. (2003) Burden of systolic and diastolic ventricular dysfunction in the community: appreciating the scope of the heart failure epidemic. JAMA 289: 194–202.
- Bleumink GS, Knetsch AM, Sturkenboom MC, Straus SM, Hofman A, et al. (2004) Quantifying the heart failure epidemic: prevalence, incidence rate, lifetime risk and prognosis of heart failure The Rotterdam Study. Eur Heart J 25: 1614–1619.
- Ceia F, Fonseca C, Mota T, Morais H, Matias F, et al. (2002) Prevalence of chronic heart failure in Southwestern Europe: the EPICA study. Eur J Heart Fail 4: 531–539.
- Butler J, Fonarow GC, Zile MR, Lam CS, Roessig L, et al. (2014). Developing therapies for heart failure with preserved ejection fraction: current state and future directions. JACC Heart Fail 2: 97–112.
- Rubiś P, Drabik L, Kopeć G, Olszowska M, Płazak W, et al. (2011) The prognostic role of exercise echocardiography in heart failure. Kardiol Pol 69: 656–663.
- Maggioni AP, Dahlström U, Filippatos G, Chioncel O, Crespo Leiro M, et al. (2013) EURObservational research programme: regional differences and 1-year follow-up results of the Heart Failure pilot survey (ESC-HF Pilot). Eur J Heart Fail 15: 808–817.
- Andell P, Li X, Martinsson A, Andersson C, Stagmo M, et al. (2017) Epidemiology of valvular heart disease in a Swedish nationwide hospital-based register study. Heart 103: 1696–1703.
- Cherry SV, Jain P, Rodriguez-Blanco YF, Fabbro M 2nd (2018) Noninvasive evaluation of native valvular regurgitation: a review of the 2017 american society of echocardiography guidelines for the perioperative echocardiographer. J Cardiothorac Vasc Anesth 32: 811-822.
- Carpentier A, Chauvaud S, Fabiani JN, Deloche A, Relland J, et al. (1980) Reconstructive surgery of mitral valve incompetence: ten-year appraisal. J Thorac Cardiovasc Surg 79: 338–348.
- Stewart BF, Siscovick D, Lind BK, Gardin JM, Gottdiener JS, et al. (1997). Clinical factors associated with calcific aortic valve disease. Cardiovascular Health Study. J Am Coll Cardiol 29: 630–634.
- Galiè N, Humbert M, Vachiery JL, Gibbs S, Lang I, et al. (2016) 2015 ESC/ERS Guidelines for the diagnosis and treatment of pulmonary hypertension: The joint task force for the diagnosis and treatment of pulmonary hypertension of the European Society of Cardiology (ESC) and the European Respiratory Society (ERS): Endorsed by: Association for European Paediatric and Congenital Cardiology (AEPC), International Society for Heart and Lung Transplantation (ISHLT). Eur Heart J 37: 67-119.
- Drabik L, Kopeć G, Podolec P (2013) Rapid progressive idiopathic pulmonary arterial hypertension (RCD cod: II-1A.1). J Rare Cardiovasc Dis 1: 33–38.
- Badesch BD, Champion HC, Gomez-Sanchez MA, Hoeper MM, Loyd JE, et al. (2009). Diagnosis and assessment of pulmonary arterial hypertension. J Am Coll Cardiol 54(1 Suppl): S55– S66.
- Oudiz RJ (2007) Pulmonary hypertension associated with left-sided heart disease. Clin Chest Med 28: 233–241.
- Vahanian A, Alfieri O, Andreotti F, Antunes MJ, Barón-Esquivias G, et al. (2012) Guidelines on the management of valvular heart disease (version 2012). Eur Heart J 33: 2451–2496.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi