Research Article, J Mar Biol Oceanogr Vol: 9 Issue: 1
Soft Bottom Macrobenthic Communities in Sandy Enclaves from the North Cotentin Peninsula (Central English Channel)
Salomé Andres1, Jean-Philippe Pezy1, Marine Martinez1, Alexandrine Baffreau2, Noémie Baux1,3, Yann Méar3 and Jean- Claude Dauvin1*
1Normandy University, UNICAEN, UNIROUEN, CNRS The Coastal and Continental Morphodynamics Laboratory, Caen, France
2GEMEL Normandy, Luc-sur-mer, France
3National Conservatory of Arts and Crafts, INTECHMER, Cherbourg en Cotentin, France and Caen-Normandy University, Cherbourg-en-Cotentin, France
*Corresponding Author : Jean-Claude Dauvin, Normandy University
UNICAEN, UNIROUEN, CNRS Coastal and Continental Morphodynamics
Laboratory, Cean, France
E-mail: jean-claude.dauvin@unicaen.fr
Received date: December 20, 2019; Accepted date: December 20, 2019; Published date: January 13, 2020
Citation: Andres S, Pezy JP, Martinez M, Baffreau A, Baux N, et al. (2020) Soft Bottom Macrobenthic Communities in Sandy Enclaves from the North Cotentin Peninsula (Central English Channel). J Mar Biol Oceanogr 9:1.
Abstract
The North Cotentin Peninsula (Normandy, France) presents a mosaic of substrates (from gravel to fine sand) favorable to the development of diversified marine benthic communities, so far, never described outside the Rade of Cherbourg. A total of 28 stations was sampled in winter 2017 and 2018, along a North Cotentin coasts, to complete the 30 stations already studied into the Rade de Cherbourg from 2012 to 2015 for a total number of 61 stations. Hierarchical Ascendant Classification and Multidimensional Scaling ordination on the 61 stations allows the identification of eight benthic assemblages corresponding to six EUNIS habitats: 1) Amphipholis squamata and Apseudopsis latreillii in mixed sediment; 2) Spio decorata with or without Apseudopsis latreillii in fine sand; 3) Melinna palmata in muddy sand; 4) Ampelisca spp., Photis longicaudata and other amphipods and tubicolous polychaetes in muddy sand; 5) Crepidula fornicata in coarse mixed sediment, and 6) Mediomastus fragilis, Lumbrineris spp. and venerid bivalves in circalittoral coarse sand or gravel. The area shows a high taxonomic diversity and remarkable benthic habitats with high natural heritage value, including the new Spio decorata fine sand habitat not previously identified in the EUNIS classification. Moreover, very high abundances of the tanaidacean crustacean Apseudopsis latreillii (>1,000 ind. 0.1 m²) are recorded in mixed sediment, whereas the species is absent in sand.
Keywords: EUNIS habitat; Soft-bottom communities; Macrobenthic habitats; Apseudopsis latreillii; Hydrodynanism; Taxonomic diversity
Introduction
The study of benthic habitats began at the end of the 19th century in the English Channel (EC). After a long period of qualitative sampling mainly by grab surveys at the scale of the whole EC sea [1-4], quantitative studies were subsequently carried out often at a local or regional scale [5-8]. Most of these more recent studies were designed to investigate the effects of human activities on benthic communities, in relation to the implementation of Nuclear Power Stations, aggregate extraction or the dredging and deposition of spoil sediment [8,9]. Even more recently, a new activity is being developed in the EC with the implementation of Offshore Wind Farms which requires the assessment of the state of benthic habitats before the deployment of wind turbines [10].
Therefore, the EC is now known as one of the more anthropized coastal seas of the Worldwide Ocean [11], the eastern part of the EC showing more intense human activities than the western part [9,12-16]. The shallow coastal zones of the EC have been more often investigated than the offshore (deeper) zones along the English and French coasts [7,8] especially in the eastern Bay of Seine where numerous benthic studies have been carried out [8,17-20]. By contrast, the distribution and characteristics of shallow benthic communities from coastal areas of the North Cotentin (NC) appear poorly known probably due to the very limited coverage of sediments in an area dominated by hard subtidal bottoms, and the absence of human coastal projects in the Normand-Breton Gulf [21] and in the Bay of Seine [2].
Mapping benthic habitats appear to be an essential tool not only for managing Marine Protected Areas, mainly the Natura 2000 sites, but also for a wide range of continental shelf habitats [22-24]. Thus, there is a current increase in studies of the distribution of marine habitats in European waters [25-28]. Nevertheless, most of the existing maps of the subtidal benthic communities of the EC were established in the 1970s [29] digitalized in the years 2000 and then classified under the EUNIS (European Nature Information Service) code [7,25,30]. Maps using the EUNIS classification at levels 3 or 4 were available for the whole EC [7] and for the Normand-Breton Gulf [30]. Only a few maps are established at levels 5 or 6, as in the case of the Bay of Seine [24] or in the RdC (Rade de Cherbourg) [31]. For the intertidal zone of the French Opal Coast (eastern part of the EC and southern part of the North Sea),[32] have mapped the soft-bottom habitats following the EUNIS habitat classification. Furthermore, some areas in the EC such as the NC shallow subtidal zones remain poorly explored and the benthic habitats are poorly known and described at precise levels of the EUNIS classification. Mapping distribution of benthic communities with the acquirement of baseline data are central to the applications of European Habitat, Water Framework, and Marine Strategy Framework Directives in the context of the management of these high anthropized coastal marine zone with an increase of human activities [27], including the future zones of implantation of the Marine Renewable Energy such as in Normandy and in the NC [31].
Recent studies conducted in the NC were focused on the description of benthic habitat in the semi-enclosed area [31] and on macrofauna of the Raz Blanchard area [33]. These studies highlighted the remarkable Natural Heritage in these areas and allow to map five EUNIS habitats in the semi-enclosed bay (Rade de Cherbourg), reflecting the high diversity of benthic communities. These two studies encourage continuing the investigation of benthic macrofauna communities to describe unexplored areas of the NC peninsula.
As the NC, remains an area with low knowledge on the structure of the soft-bottom benthic habitats, the objectives of the present study are to 1) to give an overview of marine benthic habitats from the NC completing previous data obtained only in the semi-enclosed RdC and 2) to characterize the marine benthic habitat of the NC according to the EUNIS habitat classification.
Materials and Methods
Study site
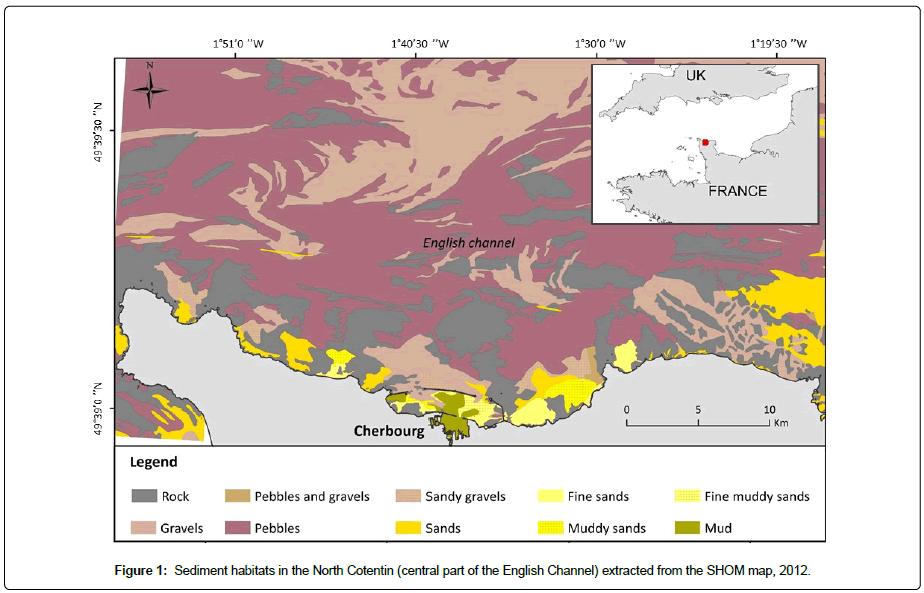
The Cotentin Peninsula is located in Normandy, France, in the central part of the EC, situated between the western and the eastern basins of the EC. The coastal zone is shallow with water depths of less than 30 m. The NC zone is characterized by semi-diurnal tides linked to strong tidal currents, but the tidal range at Cherbourg does not exceed 6.5 m. In this area with high energy hydrodynamics and rocky substratum, the soft bottom is mainly distributed in enclaves and covered by pebbles, gravels and coarse sand [34,35]. Moreover, fine sediments are only located near the coast in shallow bays and within the artificial semi-enclosed area of the RdC (Figure 1).
Sampling
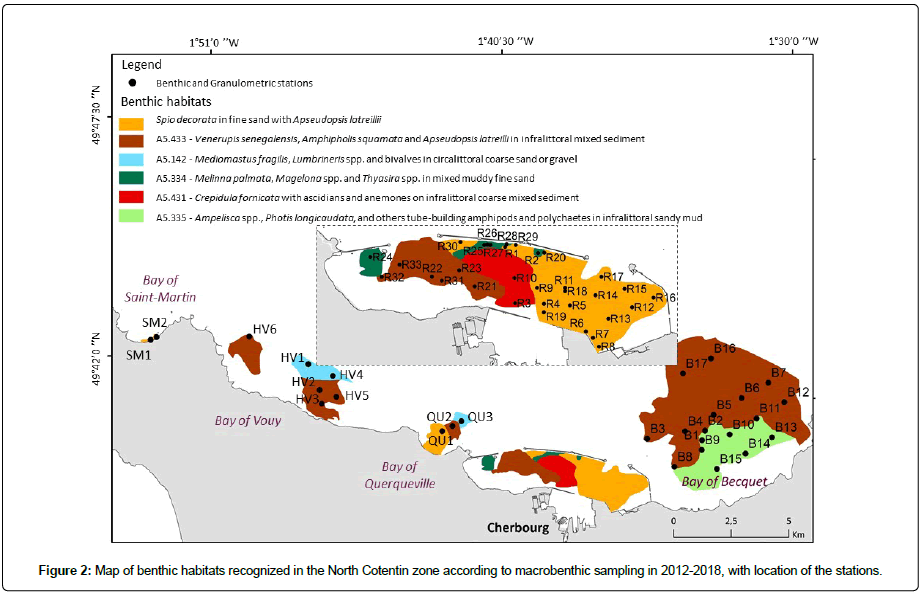
The macrofauna data used here come from three studies, all on the soft-bottom communities of the NC (Figure 2). All surveys were conducted using the same methods and were carried out in winter (December to early April).
The first study covers the Rade de Cherbourg, which pooled together the results of three surveys: March 2012 (15 stations), February 2014 (6 stations) and February 2015 (9 stations), making a total of 30 stations denoted as R stations [31] (Figure 2). The second study was focused on the benthic communities of the Bay of Becquet located in the eastern part of the RdC with 17 stations sampled in December 2016 (B stations) [27] (Table 1). The third study was carried out in the western part of the NC (Figure 2), 11 stations sampled from December 2017 to April 2018 (SM, HV and QU stations; Table 1) plus three stations (R31 to R33; Table 1) located in the western part of the RdC to provide supplementary information on this sector of the RdC [36] (Figure 2). The total number of benthic stations was 61 (Appendix 1). The sampling depth varied between 0 for both stations of the Bay of Saint Martin to 33-34 m in two offshore stations of the Bay of Becquet and 35 m in one offshore station of the Bay of Querqueville (Appendix 1). Most of the stations (31) had a depth included between 11 m and 20 m.
| Stations | S | A (Ind.0,1m2) ± SD | H' | J' | Lat | Long |
|---|---|---|---|---|---|---|
| SM1 | 21 | 134 ± 32 | 2.2 | 0.49 | -1.87586 | 49.70799 |
| SM2 | 19 | 435 ± 94 | 2.1 | 0.49 | -1.87238 | 49.70856 |
| HV1 | 33 | 26 ± 14 | 4.6 | 0.9 | -1.78033 | 49.7016 |
| HV2 | 41 | 602 ± 319 | 1.7 | 0.32 | -1.77266 | 49.6919 |
| HV3 | 63 | 1,223 ± 228 | 2.2 | 0.37 | -1.77083 | 49.68666 |
| HV4 | 21 | 14 ± 6 | 3.9 | 0.88 | -1.76516 | 49.69763 |
| HV5 | 58 | 806 ± 754 | 2.2 | 0.38 | -1.76233 | 49.68968 |
| HV6 | 61 | 370 ± 280 | 2.8 | 0.48 | -1.81675 | 49.71076 |
| QU1 | 35 | 71 ± 38 | 4 | 0.79 | -1.69763 | 49.679 |
| QU2 | 53 | 399 ± 152 | 3.6 | 0.63 | -1.69161 | 49.68118 |
| QU3 | 33 | 36 ± 8 | 4.1 | 0.82 | -1.68643 | 49.68325 |
| R31 | 82 | 2,025 ± 414 | 2.5 | 0.39 | -1.64561 | 49.66485 |
| R32 | 80 | 1,753 ± 377 | 2.3 | 0.36 | -1.66525 | 49.66493 |
| R33 | 78 | 1,796 ± 457 | 2.9 | 0.46 | -1.6597 | 49.66767 |
| B1 | 56 | 857 ± 312 | 2.9 | 0.73 | -1.54131 | 49.68143 |
| B2 | 66 | 676 ± 182 | 3.7 | 0.88 | -1.53996 | 49.6852 |
| B3 | 54 | 340 ± 120 | 3.5 | 0.84 | -1.57431 | 49.68077 |
| B4 | 54 | 515 ± 317 | 2.8 | 0.73 | -1.55187 | 49.68451 |
| B5 | 58 | 1,571 ± 537 | 1.8 | 0.44 | -1.53528 | 49.6914 |
| B6 | 55 | 2,726 ± 212 | 1.2 | 0.3 | -1.51905 | 49.69851 |
| B7 | 45 | 862 ± 523 | 1.4 | 0.35 | -1.50349 | 49.70494 |
| B8 | 69 | 967 ± 756 | 2.7 | 0.68 | -1.55699 | 49.67063 |
| B9 | 56 | 332 ± 22 | 4.1 | 0.9 | -1.54121 | 49.67771 |
| B10 | 51 | 1,772 ± 290 | 1.6 | 0.41 | -1.52501 | 49.68425 |
| B11 | 42 | 1,665 ± 99 | 1.3 | 0.35 | -1.5093 | 49.69102 |
| B12 | 52 | 313 ± 210 | 2.7 | 0.64 | -1.49318 | 49.6979 |
| B13 | 66 | 2,942 ± 241 | 2.9 | 0.8 | -1.49942 | 49.68415 |
| B14 | 62 | 1,058 ± 680 | 3 | 0.78 | -1.51481 | 49.67727 |
| B15 | 50 | 924 ± 192 | 2.5 | 0.66 | -1.53144 | 49.67075 |
| B16 | 50 | 1,131 ± 871 | 1.1 | 0.28 | -1.53873 | 49.713 |
| B17 | 60 | 1,037 ± 1,026 | 2 | 0.5 | -1.55509 | 49.70663 |
S: Taxonomic richness per 0.3 m2; A: mean abundance per 0.1 m2; H': Shannon Wiener diversity index (bits.ind-1) and J’ Pielou evenness index; SD: Standard Deviation
Table 1: Location and structural indices of the 31 sampling stations in the North Cotentin sampled in 2016-2018 (see Baux et al., 2017 for the data of the Rade de Cherbourg stations).
During these three surveys, three replicates were collected using a Van Veen grab (unit sampling surface area: 0.1 m2, for a total of 0.3 m2) at each station for macrofauna analysis plus one supplementary sample for grain-size analysis.
The sampled faunal material was sieved on board on a 1-mm circular mesh. Fauna was preserved in 10% buffered formaldehyde prior to sorting, identified to species level and counted in the laboratory. The taxonomic diversity (species richness) (SR, number of taxa per 0.3 m2.), abundance (number of individuals per 0.1 m2) and diversity indices per 0.3 m2 are calculated for each station. Species (or taxa) names were checked with the World Register of Marine Species (http://www.marinespecies.org) on 1st July 2018.
The grain size distribution of a sediment sample was determined firstly by the estimation of the fine fraction (<63 μm) which was obtained by wet sieving and rinsed with fresh water to remove the salt. Then the coarser sediment fractions (>63 μm) were sieved on a sieve shaker using an eight-sieve column (8, 4, 2,1, 0.5, 0.250, 0.125 and 0.063 mm). The sediment was then classified according to Wentworth’s grain-size scale [37] <63 μm, silt clay; (63 μm-125 μm), very fine sand; (125 μm-250 μm), fine sand; (250 μm-500 μm), medium sand; (500 μm-1,000 μm), coarse sand; (1,000 μm-2,000 μm), very coarse sand; (2,000 μm-4,000 μm), gravel; and >8,000 μm, pebbles. The results are expressed in percentage relative to the total dry weight sediment of the sample (Appendix 1). These grain-size analyses allow us to determine the different sediment according to Wentworth’s sedimentary nomenclature.
Data analysis
Data analyses were performed by non-metric Multidimensional Scaling Ordination (MDS), and a Hierarchical Ascendant Classification (HAC) was created by means of group-average linking using the Bray-Curtis similarity measure using the PRIMER-6 software package (Plymouth Routines In Multivariate Ecological Research. Log10(X+1) transformed abundance data were used to down-weight the effect of the highly abundant species. All the taxa were taken into account in the analyses. To identify the taxa within different groups which account for the observed community differences, SIMPER (SIMilarity PERcentage) and one-way ANOSIM (Analysis of Similarities) routines were also performed [38]. By considering the overall percentage contribution of each taxon to the average dissimilarity between sample groups, species are listed in decreasing order of their effect on discriminating the sets of samples. The main benthic assemblages are reported on a map created using ArcGis 10.2.2, and their species compositions are compared against the available EUNIS classification [26]. The values of Shannon-Weaver H’ diversity expressed in bits. ind-1 and Pielou’s evenness J’ are also calculated and the abundance distributions are compared between stations using the Kruskal-Wallis test and Kruskalmc post-hoc (pgirmess package with R).
Results
General patterns of the macrofauna
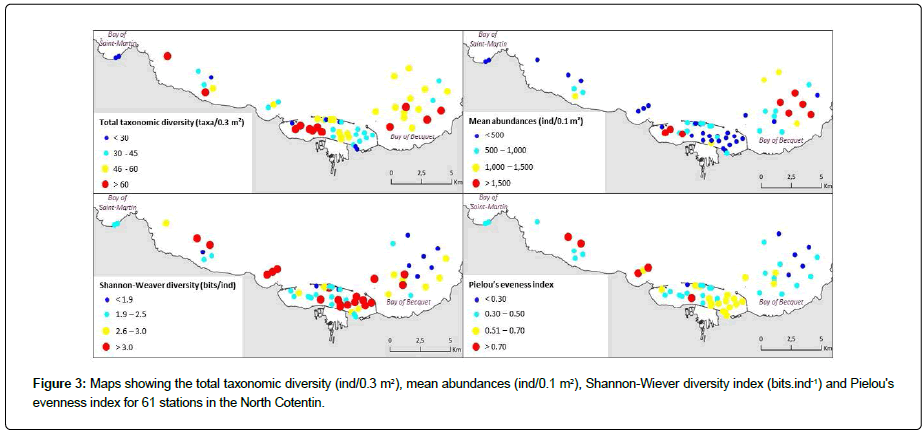
The 61 samples analyzed yielded a taxonomic diversity of 336 taxa (Appendix 2). The station with the highest number of taxa was localized within the RdC (82 taxa), while the lowest number of taxa was found in the Bay of Saint Martin (19 taxa) (Figure 3). The stations with the highest number of taxa were located in the Bay of Becquet (BB), the western part of the RdC, and in the western part of the NC (two stations) (Figure 3). The stations with the highest abundances were located in the BB (five stations) and in the western part of the RdC (three stations). Conversely, the highest values of Shannon- Weaver H’ diversity and Pielou’s evenness J’ were found in the central and eastern parts of the RdC and at stations located in the western part of the NC.
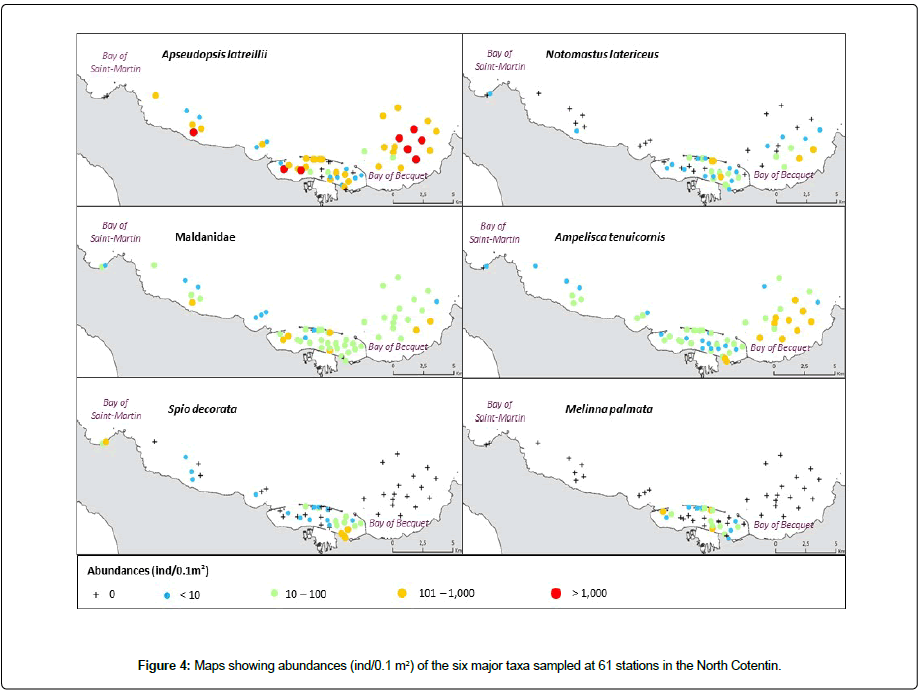
The malacostraceans dominated in terms of a number of taxa, being represented by 134 taxa, followed by the polychaetes (115 taxa), molluscs (63 taxa) and other groups making up 24 taxa. Abundances varied from 14 to 2,942 ind. 0.1m2. According to their mean abundances at the 61 stations, Polychaeta and Malacostraca were the dominant groups, with Maldanidae (abundance ± Standard Deviation: 49 ind.0.1 m2 ± 57:) (Figure 4), Spio decorata (16 ind.0.1 m2 ± 42) (Figure 4), Notomastus latericeus (52 ind.0.1 m2 ± 67) (Figure 4), Melinna palmata (14 ind.0.1 m2 ± 40) (Figure 4), Dipolydora spp. (12 ind.0.1 m2 ± 29) and Chaetozone gibber (8 ind.0.1 m2 ± 13) among the polychaetes, and Apseudopsis latreillii (343 ind.0.1 m2 ± 469) (Figure 4), Ampelisca tenuicornis (38 ind.0.1 m2 ± 137) (Figure 4) and Apseudes talpa (15 ind.0.1 m2 ± 38), among the Malacostraca.
Benthic assemblages
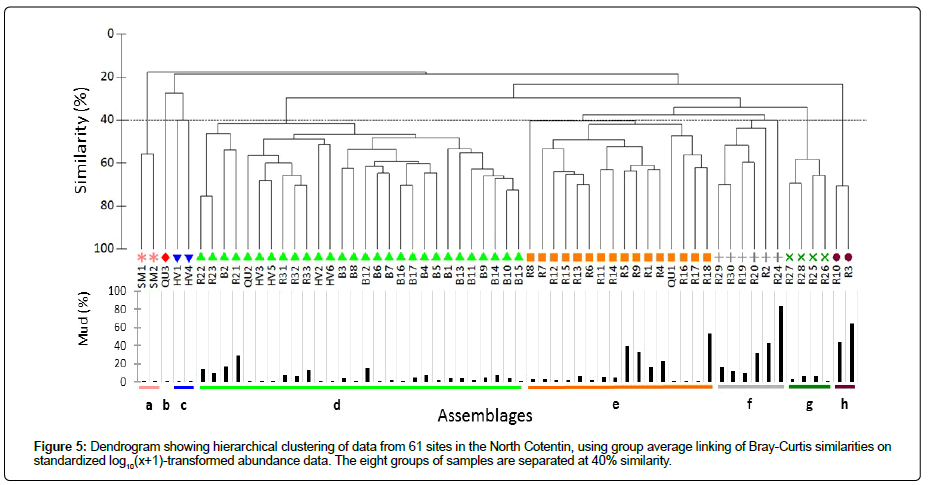
The HAC allowed us to identify eight assemblages (a to h) at a level of 40% similarity (ANOSIM; R=0.87; p<0.001) (Figure 5). Assemblage a grouped together two stations from the Bay of Saint Martin, assemblage b was made up of a single station QU3 located in the Bay of Querqueville and assemblage c was found at two stations in the Bay of Vouy, Among the five other assemblages, two main assemblages could be identified: assemblage d with 28 stations (from the Bay of Vouy to BB) and assemblage e with 16 stations (from the Bay of Querqueville to RdC). The three remaining assemblages (f, g, and h) accounted for six, four and two stations, respectively, which were all situated in the RdC. Apseudopsis latreillii, Spio decorata, Scoloplos armiger, Crepidula fornicata, Melinna palmata and Dipolydora spp., as well as the polychaetes Maldanidae and Syllidae, were the main taxa contributing to the similarities of the assemblages.
The abundances were not evenly distributed between the eight assemblages (Kruskal-Wallis test: H=79; p-value<0.001). Assemblage d showed higher abundances than assemblages b, c, e, and h, while assemblage c abundances were lower than in assemblages d, f and g (Table 2). Mean taxonomic diversity varied from 13 ± 5 (assemblage b) to 60 ± 4 (assemblage d) (Table 2). The highest mean values of H’ and J’ were observed in assemblages b and c, where mean abundances were the lowest (Table 2).
| Assemblages | S ± SD | A ± SD | H'± SD | J'± SD |
|---|---|---|---|---|
| a | 20 ± 1 | 285 ± 176 | 2.1 ± 0.1 | 0.5 ± 0.0 |
| b | 13 ± 5 | 36 ± 8 | 4.1 | 0.8 |
| c | 27 ± 8 | 20 ± 12 | 4.2 ± 0.5 | 0.9 ± 0.0 |
| d | 60 ± 4 | 1,045 ± 776 | 2.5 ± 0.9 | 0.4 ± 0.1 |
| e | 39 ± 4 | 368 ± 219 | 3.1 ± 0.6 | 0.6 ± 0.1 |
| f | 52 ± 12 | 685 ± 419 | 2.4 ± 0.6 | 0.4 ± 0.1 |
| g | 39 ± 13 | 504 ± 222 | 2.5 ± 0.2 | 0.5 ± 0.1 |
| h | 35 ± 4 | 192 ± 21 | 2.3 ± 0.3 | 0.5 ± 0.0 |
S: mean taxonomic richness per 0.1 m2; A: mean abundance per 0.1 m2; H’: Shannon Wiener diversity index in bits.ind-1; J’: Pielou’s evenness index calculated from the number of taxa and individuals counted per 0.3 m2 at each station; SD: Standard deviation.
Table 2: Main characteristics of the eight assemblages (see Figure 5).
The four assemblages d, e, f, and g were dominated by Apseudopsis latreillii (Table 3) with very high mean abundances observed in d: 630 ± 576 ind.0.1 m2. Apart from assemblage f, where the second most abundant species was the amphipod Ampelisca tenuicornis, the three other assemblages were dominated by polychaetes (Table 3). The assemblage was dominated by the polychaetes Scoloplos armiger and Spio decorata, assemblage b by the amphipod Orchomene humilis, c by the Syllidae (very low abundances) and h by the gastropod Crepidula fornicata, an introduced species in European waters.
| Assemblages | Taxons | A (ind.0.1 m2) ± SD |
|---|---|---|
| a | Scoloplos armiger | 86 ± 85 |
| Spio decorata | 85 ± 35 | |
| Urothoe poseidonis | 69 ± 79 | |
| Maldanidae | 17 ± 15 | |
| Ampelisca brevicornis | 11 ± 10 | |
| b | Orchomene humilis | 6 ± 10 |
| Syllidae | 6 ± 7 | |
| Caulleriella alata | 4 ± 6 | |
| Spirobranchus sp. | 4 ± 6 | |
| Glycera lapidum | 2 ± 2 | |
| c | Syllidae | 3 ± 3 |
| Apseudopsis latreillii | 2 ± 1 | |
| Notomastus latericeus | 2 ± 4 | |
| Maldanidae | 1 ± 1 | |
| Nemertea | 1 ± 1 | |
| d | Apseudopsis latreillii | 630 ± 576 |
| Notomastus latericeus | 85 ± 84 | |
| Maldanidae | 53 ± 58 | |
| Nucula hanleyi | 39 ± 32 | |
| Ampelisca tenuicornis | 38 ± 167 | |
| e | Apseudopsis latreillii | 96 ± 179 |
| Spio decoratus | 54 ± 64 | |
| Maldanidae | 45 ± 33 | |
| Notomastus latericeus | 30 ± 38 | |
| Ampelisca tenuicornis | 24 ± 48 | |
| f | Apseudopsis latreillii | 225 ± 240 |
| Ampelisca tenuicornis | 114 ± 200 | |
| Maldanidae | 96 ± 81 | |
| Melinna palmata | 73 ± 74 | |
| Apseudes talpa | 37 ± 65 | |
| g | Apseudopsis latreillii | 257 ± 170 |
| Dipolydora spp. | 71 ± 57 | |
| Notomastus latericeus | 57 ± 39 | |
| Capitella minima | 36 ± 39 | |
| Chaetozone gibber | 14 ± 18 | |
| h | Crepidula fornicata | 114 ± 7 |
| Maldanidae | 21 ± 13 | |
| Cirriformia tentaculata | 18 ± 11 | |
| Chaetozone gibber | 13 ± 9 | |
| Notomastus latericeus | 4 ± 2 |
Table 3: List of the five dominant species in the eight assemblages and their mean abundances with SD (Standard Deviation).
Sediment characteristics
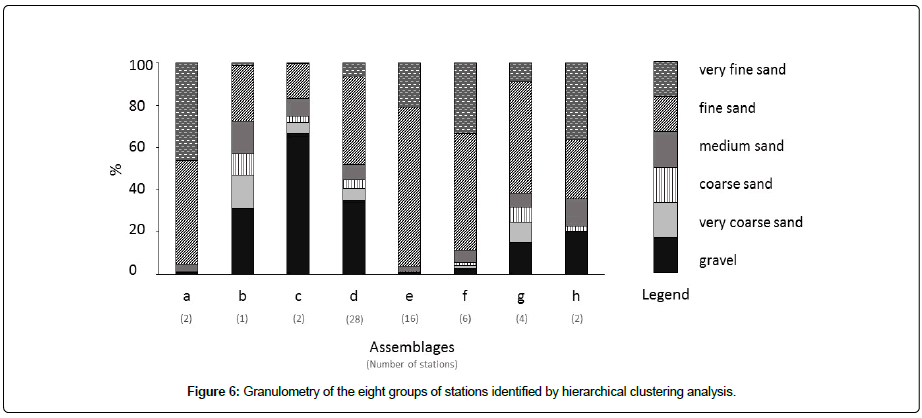
The sediments of assemblages a and e (from 2 and 16 stations, respectively) were both mainly composed of sand (99 ± 1 and 87 ± 17%, respectively). Nevertheless, the five stations of assemblage e (R1, R4, R5, R9, and R18) were all located in the RdC (Figure 6), showing a noticeable percentage (>10%) of fine particles (<63 μm). The assemblage b (found at a single station) was associated with mixed sediment having a high proportion of biogenic fragments (Figure 6). Assemblage c was found at station HV4, composed of gravelly sand (62% sand and 38% gravel) and at station HV1 with a higher proportion of gravel (95%) (Figure 6). The 28 stations associated with assemblage d showed two main granulometric classes (61 ± 20% sand and 33 ± 22% gravel). The assemblage f (six stations) was composed of 75 ± 13% sand and 23 ± 14% fine particles (<63 μm), except for station R24 which was mainly composed of mud (84%) with 15% sand (Figure 6). For assemblage g two stations were composed of 93 ± 3% of sand (R27 and R28) and two other (R25 and R26) contained a large proportion of gravel (27 ± 11%). The last assemblage h was composed of muddy sand (54 ± 14% fine particles (<63 μm) and 37 ± 12% sand) (Figure 6).
Distribution of main benthic species in the NC
The three taxa Apseudopsis latreillii, Notomastus latericeus, and the Maldanidae were collected at a large number of sampling stations from the Bay of Saint Martin in the western BB in the eastern part of the studied area (Figure 4). While A. latreillii was the more abundant species, it was dominant in the sandy gravel and gravelly sand in three main patches of the Bay of Vouy, in the western part of the RdC and in the BB. This species showed low abundance or was absent at sandy stations. Both the polychaete taxa Notomastus latericeus and Maldanidae were widely largely distributed across the sampling stations without displaying any apparent patterns (Figure 4).
The amphipod Ampelisca tenuicornis was abundant only at muddy sand stations of the RdC and BB, with three stations showing abundances >1,000 ind.0.1 m2. It is rare or absent in the western part of NC (Figure 4). The polychaete Spio decorata is present only at sandy stations in the eastern part of the RdC and at both stations of the Bay of Saint Martin (Figure 4). The polychaete Melinna palmata is only present in abundance at muddy stations of the RdC (Figure 4).
Discussion
For the first time, in this study, we present an overview of the softbottom benthic macrofauna identified in the NC up to the beginning of 2012. The map so produced for the NC provides a tool for a better understanding of the ecological status of soft-bottom benthic macrofauna, in the context of the application of European Directives involving the Natura 2000 Networks and the descriptors referring to the Marine Strategy Framework. The habitat mapping proposed here mainly concerns the descriptors 1 (Biodiversity is maintained), 2 (Non-Indigenous species do not adversely alter the ecosystem) and 6 (Sea-floor integrity ensures that the structure and functions of the ecosystems are safeguarded) [22].
Taxonomic diversity
The macrobenthos of the NC was composed of 336 taxa collected from 61 stations covering a total sampling area of 18.3 m2 (Appendix 2). By comparison, a sampling campaign in the winter of 2016 in the eastern Bay of Seine (72 stations with 0.5 m2 per station for a total sampling area of 36 m2; 1 mm circular mesh sieve) led to the recording of only 191 taxa (unpublished data), yielding a 44% lower taxonomic diversity for twice the sampling area.
The total abundances of macrofauna varied from 8 to 3,118 ind.0.1 m2. These maximal winter abundances were among the highest recorded on the coasts of the English Channel, with a high abundance of the amphipod Ampelisca tenuicornis (757 ind.0.1 m2 in the RdC; 21- 304 ind.0.1 m2 in the Rance and 127 ind.0.1 m2 in the Bay of Morlaix) [31]. The mean abundances of macrofauna in the assemblages d, e, f and g (Table 3) were among the highest found anywhere in the EC [31]. Only the amphipod Ampelisca from the Pierre Noire station on a fine sand community in the Bay of Morlaix showed an abundance that exceeded the highest values observed in the NC. High abundances of the tanaidacean Apseudopsis latreillii were found in the BB (2,500 ind.0.1 m2 in B6) [25].
EUNIS benthic habitats
The assignment of EUNIS benthic habitats was established on a combination of the results obtained by multivariate analyses to identify the benthic assemblages, (Figure 5), the sediment characteristics (Figure 6) and the distribution of dominant taxa (Figure 4), combined with the results from previous studies [25,31].
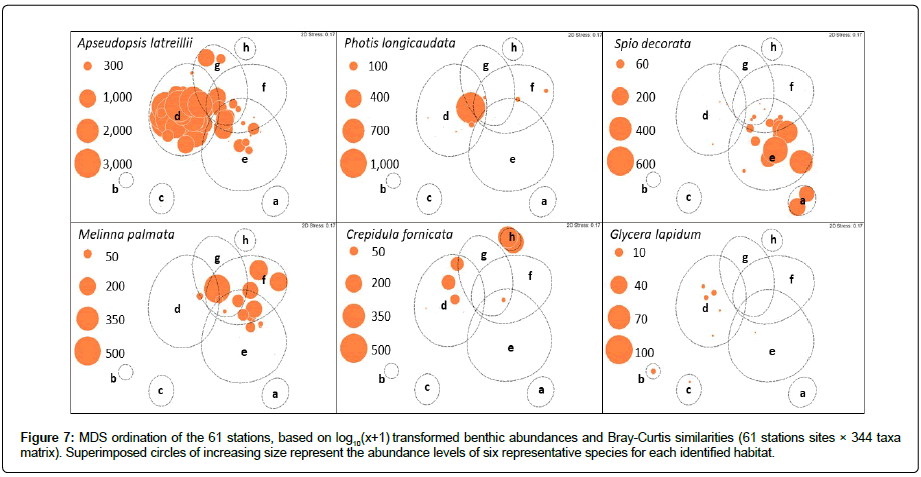
Habitats EUNIS A5.433 and A5.335: The sediment and faunal characteristics of assemblage d (28 stations, ~ 50% of the number of stations) corresponded to the EUNIS code A5.433 Venerupis senegalensis, Amphipholis squamata and Apseudopsis latreillii in infralittoral mixed sediment. The sediments were composed of gravelly sand and sand gravel, except for seven stations located in the shallow part of the BB (B1, B9, B10, B11, B13, B14, and B15) which consisted of sandy or muddy sand [25]. Apseudopsis latreillii showed very high abundances in this habitat, where the ophiurid Amphipholis squamata was also present at low abundance. This habitat was present in the western part of the RdC, mainly in the Bay of Vouy, and in the BB (Figure 2). Apseudopsis latreillii was highly dominant in this habitat (Figure 7). Finally, the A5.433 habitat previously recognized in the RdC [31], was composed of 21 stations of the NC.
Furthermore, in her study of benthic habitats, Martinez [25] showed the existence of a particular habitat, corresponding to the EUNIS habitat code A5.335, which was represented by the seven shallowest stations of the BB associated with sand or muddy sand. This habitat comprised Ampelisca spp., Photis longicaudata and other tube-building amphipods and polychaetes in infralittoral sandy mud. At certain stations, the amphipods Photis longicaudata (up to 464 ind.0.1m2 at station B13) and Ampelisca tenuicornis (up 1,036 ind. 0.1m2, station B13) showed high abundances; while this habitat had been maintained in the BB, it was not recognized elsewhere in the NC. Photis longicaudata was representative of this habitat (Figure 7), which could be distinguished from A5.433.
Finally, the A5.335 habitat was composed of seven stations. This habitat was no described before along the French coast [39].
Fine sand habitat and habitat EUNIS A5.334: The sediment characteristics of both stations sampled in the Bay of Saint Martin (assemblage a) and those of assemblage e (16 stations, one in the Bay of Querqueville and the other in the eastern part of the RdC) corresponded to fine to very fine sand. Both assemblages were characterized by the polychaete Spio decorata. Apseudopsis latreilli was present in abundance only in some stations of the assemblage e (Figure 7). This Spio decorata habitat was not recognized in the EUNIS classification and its species composition was previously discussed [31]. It consisted of a mixture between EUNIS A5.244 (Spisula subtruncata and Nephtys hombergii shallow muddy sand) and the Abra alba community in muddy fine sand. As such, it was close to the Abra alba-Hyalinoecia bilineata fine sand community in the Bay of Morlaix (western English Channel [40], which itself corresponded to a EUNIS habitat intermediate between A5.252 (Abra prismatica, Bathyporeia elegans and polychaetes in circalittoral fine sand) and A5.231 infralittoral mobile clean sand with sparse fauna [41].
Assemblage f (six stations in the RdC) was, in fact, a mixture of habitats, with four stations R19, R20, R29 and R30 showing fine sand sediment (53 ± 14% of fine sand and 18 ± 10% of fine particles <63 μm) and high abundance of A. latreillii being attributed to the Spio decorata habitat. The two last stations R2 and R24 were composed of 47% and 10% of very fine sand and 43% and 83% fine particles <63 μm, respectively. Samples from R2 and R24 showed a very low abundance of A. latreillii (only one individual) and were dominated by the polychaete Melinna palmata (20% of the total number of individuals collected at both stations) and the bivalve Thyasira flexuosa (10%). Both A. latreillii and M. palmata were characteristic of the EUNIS code A5.334 habitat (Melinna palmata with Magelona spp. and Thyasira spp. in infralittoral sandy mud).
In the case of assemblage g, the four stations were dominated by tolerant species or which lived in zones enriched in organic matter (opportunistic species) such as Dipolydora spp. (14%), Notomastus latericeus (11%), Capitella minima (7%) and Chaetozone gibber (3%). The presence of these species was associated with the proximity of salmon fish cages in the north of the RdC [42,43] This assemblage corresponded to the EUNIS code A5.334 (Melinna palmata with Magelona spp. and Thyasira spp. in infralittoral sandy mud) with the dominance of Maladanidae and Melinna palmata (Figure 7) [31].
Finally, 20 stations were recognized as belonging to the Spio decorata habitat and six stations were attributed to A5. 334.
Two sub-habitats of the Spio decorata habitat were recognized in the eastern part of the RdC, one consisting of fine sand without abundant A. latreillii population and the other in fine sand and very fine sand sediment with a high abundance of A. latreillii [31]. Analyses at the scale of the soft-bottom enclaves in the NC (Figures 2 and 5) failed to distinguish these two assemblages, and only one habitat was finally described here (Figure 2). It was present in three localities, in the Bay of Saint Martin, in the Bay of Querqueville and in the eastern part of the RdC.
The EUNIS A5.334 habitat (Melinna palmata with Magelona spp. and Thyasira spp. in infralittoral sandy mud) was present in the Bay of Morlaix [43,44] and in the Rance estuary [21,47]. It had been recently described in the eastern part of the Bay of Seine following the establishment of Melinna palmata in this area at the beginning of the years 2000 [24,45,46]. It was previously present near the fish cage in the RdC since 1995 [48].
Habitat EUNIS A5. 431: Both stations of the assemblage h were dominated by the introduced gastropod Crepidula fornicata, which formed 60% of the total individuals sampled at these stations. Polychaetes belonging to the Maldanidae (Cirriformia tentaculata, Chaetozone gibber, and Notomastus latericeus) also showed high abundances. This assemblage corresponded to the EUNIS benthic habitat A5.431 (Crepidula fornicata, with ascidians and anemones on infralittoral coarse mixed sediment). It was recognized only in the central part of the RdC where C. fornicata characterized this habitat (Figure 7). This invasive species was present in most of the shallow habitats of the EC [49], but the A5.431 habitat had only been described in the Bay of Saint Brieuc.
Habitat EUNIS A5.142: Offshore stations in the Bay of Querqueville (QU3, assemblage b) and the Bay of Vouy (HV1 and HV4, assemblage c) corresponded to the EUNIS A5.142 habitat in circalittoral coarse sand or gravel, and could be characterized by the polychaete Glycera lapidum present in abundance only in one station of the assemblage b (Figure 7); G. lapidum was associated with the urchin Echinocyamus pusillus. This habitat extended widely over the central part of the Bay of Seine [24]. Moreover, as suggested in the EUNIS habitat classification, it was possible that A5.142 was not a true biotope and might be quite variable over time. In fact, it might be closer to a biotope complex within which a number of biotopes or sub-biotopes remained to be defined [35].
This habitat neighbour the benthic habitat (A5.14) was found in the Raz Blanchard offshore Cap de la Hague [33]. In this area with strong tidal currents, habitat A5.142 corresponded to the very mobile coarse infralittoral sediments with scattered fauna (Sédiments grossiers très mobiles infralittoraux à faune éparse) described in the French Marine Benthic Habitats classification issued by the National Museum of Natural History (Paris) by Michez et al. [50].
Originality of the NC benthic habitats
Mosaic of EUNIS habitats. The NC was an area lacking large fluvial inputs, with high-energy hydrodynamics and delimited by two zones with strong tidal currents in the west (Raz Blanchard) and east (Raz de Barfleur). It was dominated by a hard bottom and pebbles, while the soft-bottom habitats were distributed in enclaves covering relatively small areas, apart from in the RdC and the BB. Two benthic habitats dominated the seabed in the prospected zone: A5.433, Venerupis senegalensis, Amphipholis squamata and Apseudopsis latreilli in infralittoral mixed sediment, and a new habitat ‘Spio decorata in fine sand’, neither described before in the EC. The four other habitats covered small patches sampled at two to six of the stations. This pattern was typical of shallow waters along the western part of the EC, especially in Brittany, such as in the Bay of Morlaix [40], or in the eastern Bay of Seine [24]. Moreover, the macrobenthic species and communities found in the RdC were more closely similar to those found in the western rather than the eastern part of the EC [31]. There was little equivalence between the macrobenthos in neighbouring parts of the Bay of Seine, such as between the Bay of Veys [5] and the eastern part of the bay near the mouth of the Seine estuary [2,17,18]. Conversely, the muddy sand and sand habitats resembled those found in the western part of the EC along the Brittany and English coasts [31]. As suggested previously, the NC represented the eastern limit of several macrobenthic species in response to the higher thermic amplitude of the seawater in the eastern part of the EC, which was not favorable for stenothermal species under such climatic conditions [7,27] and natural heritage. The NC zone had been recognized for its high natural-heritage value and several ZNIEFF-Marine (Natural Areas of Ecological, Faunal and Floral Interest) had been delimited in the NC [24]. The ZNIEFF inventory aimed to identify marine areas based on remarkable habitats, integrating a notion of functionality (food, reproduction) as well as the presence of species or combinations of species (benthos, fish, birds and mammals) of high natural-heritage value [48]. The fine sand communities in the eastern part of the RdC and the Melinna palmata muddy fine sand in the RdC were accepted as natural heritage habitats [31,51]. Two small patches of Zostera marina eelgrass beds had also been recognized as a ZNIEFF in the eastern part of the RdC [51]. The present study proposed to extend the Spio decorata habitat (with or without Apseudopsis latreillii), even though it not yet recognized in the EUNIS classification, to two new areas: the western part of the RdC in the Bay of Querqueville, and the Bay of Saint Martin (Figure 2) [50,51].
The high abundance of Apseudopsis latreillii was noteworthy. This species was widely distributed in the coastal waters of NC and showed very high abundances in the ‘Bay of Becquet’ in the eastern part of the RdC and in some sand and gravel habitats in the western part of the RdC (Figure 4). The abundances observed in the RdC and BB (20,000 ind.m-2-23,000 ind.m-2) were among the highest recorded values; higher abundances were only observed in the Persian Gulf, in the Dubai creek, with values of 43,000 ind.m-2 under the impact of organic pollution [52]. A. latreilli was a rare species in the benthic communities of the EC, except in the Aber Wrach, in the western part of the EC along the Brittany coast, where the abundance exceeded 3,000 ind.m-2 in the subtidal zone or in the intertidal Zostera marina beds [53,54]. It was very rarely recorded in the eastern part of the EC and is never present as a dominant species or in dense populations such as observed in the NC.
Characterized by high-energy hydrodynamics and a mainly rocky bottom, the NC nevertheless contained soft-bottom areas within the RdC and natural enclaves near the coast. Substratum and sediment types were the main environmental factors influencing the macrobenthic community distribution and their biodiversity [27,55,56].
Conclusion
Overall, the hierarchical classification of EUNIS (EUNIS, 2012) was sufficient, allowing us to identify six out of the seven different benthic habitats from the Bay of St-Martin to the BB. However, the EUNIS typology needed to be revised and updated, in particular, to include sub-littoral fine sand habitats missing from the descriptions. The two dominant habitats, A5.433 and the new habitat Spio decorata in fine sand were described here for the first time in the EC. The particularity of habitats on the NC coast, along with the presence of ZNIEFF-marine areas, the high taxonomic diversity and the occurrence of remarkable species, all reflected the special interest of NC and the importance of monitoring and preserving its natural heritage.
Acknowledgments
We gratefully acknowledge the salmon farming company, the crew of its support vessel, the GEDANOR project financed by the Conseil Régional de Basse-Normandie (CRBN) and the Ports of Normandy Authority (PNA) for the RdC study. We also thank the crew of the R.V. Côtes de la Manche operated by INSU-CNRS for sampling outside the RdC. We would like to extend our thanks to the staff of the M2C laboratory for their technical support, particularly M. Legrain. Finally, we thank M. Carpenter for post-editing the English style and grammar.
References
- Cabioch, L (1968) Contribution à la connaissance des peuplements benthiques de la Manche occidentale. Cah Biol Mar 9:493-720.
- Cabioch L, Gentil F (1975) Distribution des peuplements benthiques dans la partie orientale de la baie de Seine. C. R. Séances Acad. Sci. Paris 280, 571-574.
- Holme NA (1961) The bottom fauna of the English Channel. J Mar Biol Assoc UK. 41:397-461.
- Holme NA (1966) The bottom fauna of the English Channel. Part II. J Mar Biol Assoc UK. 46:397-461.
- Dauvin JC, Thiébaut E, Gesteira JLG, Ghertsos K, Gentil F, et al (2004) Spatial structure of a subtidal macrobenthic community in the Bay of Veys (western Bay of Seine, English Channel). J Exp Mar Biol Ecol. 307:217-235.
- Capasso E, Jenkins S, Frost M, Hinz H (2010) Investigation of benthic community change over a century-wide scale in the western English Channel. J. Mar. Biol. Assoc. UK, 90:1161-1172.
- Coggan R, Diesing M (2011) The seabed habitats of the central English Channel: A generation on from Holme and Cabioch, how do their interpretations match-up to modern mapping techniques. Cont. Shelf Res. 31:132-150.
- Dauvin JC (2015) History of benthic research in the English Channel: from general patterns of communities to habitat mosaic description. J Sea Res. 100:32-45.
- Dauvin JC (2012) Are the Eastern and Western basins of the English Channel two separate ecosystems? Mar Pollut Bull. 64:463-471.
- Pezy JP (2017). Approche écosystémique d’un futur parc éolien en Manche orientale: exemple du site de Dieppe-Le Tréport. PhD, Caen Normandy University, France. 324.
- Halpern BS, Walbridge S, Selkoe KA, Kappel CV, Micheli F, et al (2008) A global map of human impact on marine ecosystems. Science 319:948-952.
- Carpentier A, Vaz S, Martin CS, Coppin F, Dauvin JC, et al. (2005) Eastern Channel Habitat Atlas for Marine Resource Management (CHARM). INTERREG IIIa. Ifremer, Brest, France, 225.
- Carpentier A, Martin CS, Vaz S (2009) Channel Habitat Atlas for Marine Resource Management, final report (CHARM phase II). Aquat. Living Resour. 22, 499-508.
- Martin CS, Carpentier A, Vaz S, Coppin F, Curet L, et al (2009) The Channel habitat atlas for marine resource management (CHARM): an aid for planning and decision-making in an area under strong anthropogenic pressure. Aquat Liv Resour 22:499-508.
- Dauvin JC, Lozachmeur O (2006) Mer côtière à forte pression anthropique propice au développement d’une gestion intégrée: Exemple du bassin oriental de la Manche (Atlantique Nord-est). Vertigo 7:1-14.
- Dauvin JC (2019) The English Channel: La Manche. World Seas: An Environmental Evaluation. 1:153-188.
- Thiébaut E, Cabioch L, Dauvin JC, Retière C, Gentil F (1997) Spatio-temporal persistence of the Abra alba -Pectinaria koreni muddy-fine sand community of the Estern Bay of Seine. J Mar Biol Assoc UK. 1165-1185.
- Dauvin JC, Lucas S, Navon M, Lesourd S, Mear Y, et al (2017) Does the hydrodynamic, morphometric and sedimentary environment explain the structure of the soft-bottom benthic assemblages in the Eastern Bay of Seine (English Channel)? Estuar Coast Shelf Sci. 189:156-172.
- Pezy JP, Raoux A, Marmin S, Balay P, Dauvin JC (2018) What are the most suitable indices to detect the structural and functional changes of benthic community after a local short-term disturbances? Ecol Ind. 91:232-240.
- Baux N (2018) Dynamique d’habitats benthiques sous contraintes anthropiques : Le cas du site de dépôt de dragage d’Octeville. PhD, Caen Normandy University, France
- Retière C (1979) Contribution à la connaissance des peuplements benthiques du golfe Normanno-Breton. thèse doctorat d'Etat de l'Université de Rennes, 1-431.
- Buhl-Mortensen L, Buhl-Mortensen M, Dolan MJF, Gonzalez-Mirelis G (2015) Habitat mapping as a tool for conservation and sustainable use of marine resources: Some perspectives from the MAREANO Programme, Norway. J Sea Res. 100:46-61.
- Henriques V, Tuaty Guerra M, Mendes B, Gaudêncio MJ, Fonseca P (2015) Benthic habitat mapping in a Portuguese Marine Protected Area using EUNIS: An integrated approach. J Sea Res. 100:77-90.
- Baffreau A, Chouquet B, Dancié C, Duhamel S, Foveau F, et al. (2017) Mapping benthic communities: an indispensable tool for the preservation and the management of the Bay of Seine eco-socio-system. Reg Stud Mar Sci 9:162-173.
- Martinez M (2017) Caractérisation des habitats benthiques de la baie du Becquet, Report of Master two stage. Université Pierre et Marie-Curie. pp. 1-58. MESH, 2008.Mapping European seabed habitats.
- Galparsoro I, Connor DW, Borja A, Aish A, Amorim P, et al (2012) Using EUNIS habitat classification for benthic mapping in European seas: Present concerns and future needs. Mar Poll Bull. 64:2630-2638.
- Dutertre M, Hamon D, Chevalier C, Ehrhold A (2013). The use of the relationships between environmental factors and benthic macrofaunal distribution in the establishment of a baseline for coastal management. ICES J Mar Sci. 70:294-308
- Vasquez M, Mata Chacon D, Tempera F, O’Keeffe E, Galparsoro I, et al. (2015) Broad-scale mapping of seafloor habitats in the north-east Atlantic using existing environmental data. J Sea Res 100:120-132.
- Cabioch L, Gentil F, Glaçon R, Retière C (1978) Carte des peuplements macrobenthiques en Manche orientale. Produit numérique REBENT Ifremer-Université-CNRS-2007.
- Cabral P, Levrel H, Schoenn J, Thiébaut E, Le Mao P, et al. (2015) Marine habitats ecosystem service potential: A vulnerability approach in the Normand-Breton (Saint Malo) Gulf, France. Ecosyst Serv 16:306-318.
- Baux N, Bachelet Q, Baffreau A, Pez, JP, Mear Y, et al. (2017) An exceptional rich soft-bottom macrobenthic habitats in a semi-enclosed Bay of the English Channel: The Rade of Cherbourg. Reg Stud Mar Sci 9:106-116.
- Rolet C, Spilmont N, Dewarumez JM, Luczak C (2015) Linking macrobenthic communities structure and zonation patterns on sandy shores: Mapping tool toward management and conservation perspectives in Northern France Cont Shelf Res. 99:12-25.
- Foveau A, Dauvin JC (2017) Surprisingly diversified macrofauna in mobile gravels and pebbles from high-energy hydrodynamic environment of the ‘Raz Blanchard’ (English Channel). Reg Stud Mar Sci. 16:188-197.
- Larsonneur C, Vaslet D, Auffret JP (1979). Les Sédiments Superficiels de la Manche. Orléans, France Ministère de l'Industrie, Service Géologique National Carte Géologique de la Marge Continentale Française. Bureau des Recherches Géologiques et Minières
- Larsonneur C, Bouysse P, Auffret JP (1982) The superficial sediments of the English Channel and its western approaches. Sedimentology 29:851-864.
- Andres S (2018) Habitats benthiques des enclaves de sédiments meubles du Nord Cotentin (du Cap de la Hague à la rade de Cherbourg). Report Master 2, Paris Sorbonne University. pp. 1-55.
- Wentworth CK (1922) A scale of grade and class terms for clastic sediments. J Geol 30:377-392.
- Clarke KR, Gorley RN (2006) PRIMER V6: User Mannual/Tutorial. Dans: PRIMER-E. Plymouth.
- Croguennec C, Guillaumont B, Bajjouk T, Hily C, Gentil F (2011) REBENT - Atlas de cartes d’habitats historiques - Réseau Benthique. Edition 2011.
- Dauvin JC (1998) The fine sand Abra alba community in the Bay of Morlaix twenty years after the Amoco Cadiz oil spill. Mar Poll Bull. 36:669-676.
- Ehrhold A, Hamon D, Chevalier C (2011) Réseau de surveillance benthique (REBENT) – Région Bretagne. Approche sectorielle subtidale : Identification et caractérisation des habitats benthiques du secteur Morlaix
- Dauvin JC, Pezy JP, Baffreau A, Bachelet Q, Méar Y, et al. Effects of a salmon fish farm on the benthic habitats in a high hydrodynamic system: the case of the Rade de Cherbourg (English Channel). Aquaculture.
- Bachelet Q (2014). Etude de l’influence de l’élevage de Poissons en mer sur l’environnement benthique: Exemple de l’élevage de saumons en rade de Cherbourg. Report University Institut of Technology, Caen Normandy University. pp. 1-70.
- Dauvin JC (1982) Impact of Amoco Cadiz oil Spill on the muddy fine sand Abra alba and Melinna palmata community from the Bay of Morlaix. Estuar Coast Shelf Sci. 14:517-531.
- Dauvin JC (2000) The muddy fine sand Abra alba-Melinna palmata community of the Bay of Morlaix twenty years after the Amoco Cadiz oil spill. Mar Pollut Bull 40:528-536.
- Dauvin JC, Ruellet T, Thiébaut E, Gentil F, Desroy N, et al. (2007) The Presence of Melinna palmata (Annelid Polychaete) and Ensis directus (Mollusc Bivalve)as indicators of environmental changesin the Bay of Seine.Cah Biol Mar 48:391-401
- Desroy N, Retière C (2001) Long-term changes in muddy fine sand community of the Rance Basin: role of recruitment. J Mar Biol Assoc UK, 553-564.
- Kempf M, Merceron M, Cadour G, Jeanneret H, Mear Y, et al (2002) Environmental impact of a salmonid farm on a well flushed marine site: II. Biosedimentology. J Appl Ichthyol 18:51-60.
- Blanchard M (1997) Spread of the slipper limpet Crepidula fornitaca (L., 1758) in Europe. Current state and consequences. Scient Mar 61:109-118
- Michez N, Bajjouk T, Aish A, Andersen AC, Argall E, et al (2015) Typologie des habitats marins benthiques de la Manche, de la Mer du Nord et de l’Atlantique. (No. Rapport SPN 2015-45). Service du Patrimoine Naturel, 1-66.
- Baffreau, A., Dauvin, JC, Hacquebart P, Joncourt Y (2015) Proposition de nouveaux périmètres ZNIEFF-Marines et mise à jour des ZNIEFF existantes dans le nord Cotentin et la Manche centrale (Région Basse-Normandie). Report University of Caen Normandy, Laboratory ‘Morphodynamique Continentale et Cotière, M2C’) to the DREAL-Normandie, pp. 1-19
- Saunders JE, Al Zahed KM, Paterson DM (2007) The impact of orgnanic pollution on the macrobenthic fauna of Dubai Creek (UAE). Mar Poll Bull.54:1715-1723.
- Dauvin JC, Gentil F (1990) Conditions of the peracarids populations of subtidal communities in northern Brittany ten years after the Amoco Cadiz oil spill. Mar Poll Bull. 21:123-130
- Hily C, Bouteille M (1999) Modifications of the specific diversity and feeding guilds in an intertidal sediment colonized by an eelgrass meadow (Zostera marina) (Brittany, France). C.R. Acad. Sci. Paris, Sci. Vie 322:1121-1131.
- Nishijima W, Umehara A, Okuda T, Nakai S (2015) Variations in macrobenthic community structures in relation to environmental variables in the Seto Inland Sea, Japan. Mar Poll Bull. 92:90-98.
- Veiga P, Redondo W, Sousa-Pinto I, Rubal M (2017) Relationship between structure of macrobenthic assemblages and environmental variables in shallow sublittoral soft bottoms. Mar Envir Res 129:396-407.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi