Research Article, J Polym Sci Appl Vol: 1 Issue: 1
Synthesis and Characterization of Electroactive Fluorinated Poly (amic acid) Bearing Oligoaniline Pendants
| Ying Yan, Yanyan Li and Danming Chao* | |
| College of Chemistry, Jilin University, Changchun, 130012, P.R. China | |
| Corresponding author : Danming Chao College of Chemistry, Jilin University, Changchun, 130012, P.R. China Tel: +86-431-85168292; Fax: +86- 431-85168292; E-mail: chaodanming@jlu.edu.cn |
|
| Received: February 08, 2017 Accepted: March 15, 2017 Published: March 18, 2017 | |
| Citation: Yan Y, Li Y, Chao D (2017) Synthesis and Characterization of Electroactive Fluorinated Poly (Amic Acid) Bearing Oligoaniline Pendants. J Polym Sci Appl 1:1. |
Abstract
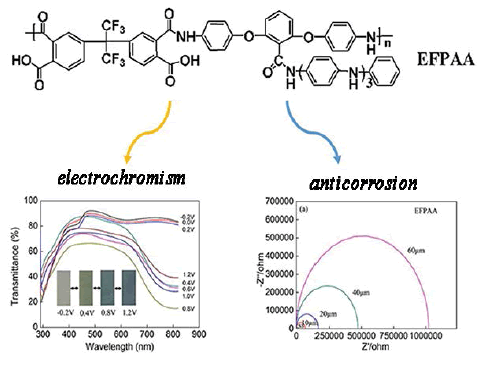
A novel electroactive fluorinated poly (amic acid) (EFPAA) was firstly synthesized in a single step from 4,4’-(Hexafluoroisopropylidene) diphthalic anhydride (HFDA) and electroactive diamine monomer (EDA). The structure of EFPAA was confirmed by Fourier-transform infrared spectra (FTIR), nuclear magnetic resonance (NMR) and gel permeation chromatography (GPC). The electroactivity of EFPAA was evaluated by performing electrochemical cyclic voltammetry in 0.5 M H2SO4 confirming a surface-controlled process. EFPAA/ITO electrode displayed good electrochromic behavior with high contrast value, moderate switching times, acceptable coloration efficiency. Moreover, anticorrosion performance of the EFPAA coatings on the stainless steel (SS) in 3.5 wt% NaCl solution was investigated by tafel plot analysis and electrochemical impedance spectroscopy. A comprehensive and detailed study of corrosive protection of EFPAA will provide an insightful tool for developing electrochromic displays and anticorrosive paint.
Keywords: Polyaniline; Fluorinated, Electrochromic; Anticorrosion
Graphical Abstract |
|
 |
|
| We report the synthesis of a novel electroactive fluorinated poly(amic acid) (EFPAA) containing tetraaniline pendants. The expected electrochromic behavior was observed through the electrochemical measurement of EFPAA/ITO electrode. Moreover, the as-prepared EFPAA also shows outstanding anticorrosive performance for stainless steel. | |
Keywords |
|
| Polyaniline; Fluorinated; Electrochromic; Anticorrosion | |
Introduction |
|
| As one of the most heat-resistant polymers, polyimides are widely used in high temperature plastics, adhesives, sealants, photoresists, separation membranes, dielectrics, coatings, and optical materials [1]. Due to their good thermal, chemical and mechanical properties, polyimides are utilized as stable coatings for anticorrosion [2-4]. Recently, researchers have intensely focused on the preparation of improved anticorrosive polyimide coatings to resolve the water penetration issue. First, some inorganic materials, such as silica [5], clay [6], boron nitride [3] and graphene [7] have been imported into the polymer matrix. The resulting polyimide/ inorganic composites coatings exhibit better water resistance and better anticorrosion behavior. Moreover, the fabrication of super hydrophobic nanostructured polyimide surfaces would be another effective strategy to improve corrosion protection [4,8] Improved anticorrosion should be resulted from super hydrophobicity and dual barrier protection. To expand practical applicability, more polyimide materials need to be explored, with emphasis on materials with good anticorrosive properties. | |
| Among the conducting polymers, polyaniline (PANI) is considered to be one of the most promising materials in anticorrosive coatings as an alternative to chromium-containing systems due to its high conductivity, good environmental stability, easy synthesis and relatively low cost [9-13]. PANI coatings can provide sacrificial cathodic protection as well as a physical barrier against any aggressive corrosion. During the anticorrosive process, a metal passivation phenomenon has been observed, which could be ascribed to the redox catalytic effect of PANI [14,15]. Recently, several kinds of polyanilinetype anticorrosive materials have been designed and prepared, such as polyaniline-salts with various dopant types [16,17], polyanilineinorganic nanocomposites with graphite, TiO2, ZnMoO4, ZnAl-LDH [18-21], polyaniline-polymers composites/blends [22-25]. However, the poor solubility and process ability of polyaniline will impede its development in anticorrosion applications. Although re-doping techniques, sulfonating methods, and other modified means could partially solve the above problem, more convenient and effective methods should be explored. | |
| To improve the solubility and process ability, a new approach incorporating the oligoanilines into other polymeric systems, has recently emerged. With the design and synthesis of various monomers, electroactive polymers containing oligoanilines have attracted much attention both in fundamental and applied research in fields of electroluminescent devices, electrochromic materials, sensors, etc. [26-30]. As a result, electroactive polymers with different topological structures, such as graft, alternating, hyperbranched and block-like polymers [26-33], have been synthesized and reported. Herein, considering the protective nature of polyimide and the versatility of PANI-based materials, a novel precursor of polyimide with oligoaniline segments was designed and synthesized. The structure, electroactivity and electrochromic properties of the obtained polymer were studied. Furthermore, the corrosive protection of EFPAA for SS was investigated in detail. | |
Experimental |
|
| Materials | |
| 4,4’-(Hexafluoroisopropylidene) diphthalic anhydride and 4,4'-(1,3-phenylenedioxy) dianiline were purchased from Sigma- Aldrich. Ammonium persulfate (APS), ferric chloride, and potassium carbonate were obtained from Tianjin Chemical Factory. Hydrochloric acid (37%) and ammonia water (25%) were purchased from Shenyang Chemical Factory. N,N’- Dimethylformamide (DMF), N,N'-dimethylacetamide (DMAc), tetrahydrofuran, toluene, dichloromethane and ethanol were obtained from commercial sources and used as received without further purification. EDA was prepared in our lab by an established synthetic route [30]. Optically transparent indium-tin oxide (ITO) glass substrates were obtained from Reintech electronic technologies Co. Ltd (Beijing) and used as working electrode substrates in the electrochemical measurements. The composition of stainless steel (SS, T301), used in the anticorrosion tests is as following (wt%): C 0.15%, Si 1.0%, Mn 2.0%, Cr 16.0~18.0%, Ni 6.0~8.0%, S 0.03%, P 0.045% and Fe bal. | |
| Structure characterization of EFPAA | |
| Mass spectra were performed on an AXIMA-CFR laser desorption ionization time of flight mass spectrometer (COMPACT). Fourier transform infrared (FTIR) spectra were recorded on a BRUKER VECTOR 22 Spectrometer by averaging 128 scans at a solution of 4 cm-1 in the range of 4000-400 cm-1. NMR spectra in deuterated dimethyl sulfoxide (DMSO) were acquired using a Bruker-500 spectrometer at room temperature. | |
| Molecular weight of EFPAA | |
| The molecular weight information of EFPAA was collected using Shimadzu gel permeation chromatography (GPC) unit equipped with a Shimadzu GPC-802D gel column and SPD-M10AVP detector. N,N’-Dimethylformamide (DMF) was used as the eluent at a flow rate of 1 mL/min. Before analysis, the sample was filtered through a 0.2 mm poly (tetrafluoroethylene) syringe filter. Calibration was accomplished with monodispersed polystyrene standards with a molecular weight range from 500 to 500000 g/mol. | |
| Thermal analysis of EFPAA | |
| The thermal stability of EFPAA was studied on Perkin-Elmer PYRIS 1 TGA in the temperature range of 100-680°C at a rate of 10°C/min under nitrogen atmosphere. Before testing, the EFPAA was kept under nitrogen atmosphere at 120oC for 0.5 h to remove any residual water and/or solvents.s | |
| Electrochemical properties of EFPAA | |
| The electrochemical properties of EFPAA, including electroactivity, electrochromic performance, polarization curve and electrochemical impedance spectroscopy, were investigated on a CHI 660A Electrochemical Workstation (CH Instruments, USA) with a conventional three electrode cell, using a saturated calomel electrode (SCE) as the reference electrode, a platinum wire electrode as the counter electrode, and the EFPAA-decorated working electrode. Moreover, the spectroscopic properties of EFPAA in the electrochromic measurements were studied using a UV-3101 PC spectrometer (SHIMADZU). | |
| Anticorrosive performance of EFPAA | |
| The anticorrosive performance of EFPAA and the referential polymer for the SS in 3.5 wt% NaCl corrosive solution was studied by polarization curve technique and electrochemical impedance spectroscopy. EFPAA/SS electrode was applied as the working electrode in the traditional three electrode system. | |
| In the polarization curve measurements, the electrochemical corrosion parameters were calculated using the following equations. First, the polarization resistances (Rp) were calculated from the Tafel curves according to the Stearn-Geary equation [34-36]: | |
| where Icorr is corrosion current (A/cm2), ba and bc are the anodic and cathodic Tafel slope (ΔE/Δlog I), respectively. Corrosion rate (Rcorr, in mm per year) was evaluated using the following equation [37,38]: | |
| Where Icorr is the corrosion current density (A/cm2), M is the molecular weight of the metal subjected to corrosion (g/mol), V is the valence, 3270 is the constant and D is the density of the corroding metal (g/cm3). The protection efficiency (PEF%) values were estimated using the equation [39,40]: | |
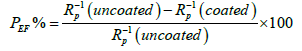 |
|
| Where Rp(uncoated) is the polarization resistances of bare SS substrate, Rp(coated) is the polarization resistances of SS substrate coated with polymers. | |
| Synthesis of EFPAA and NPAA | |
| EFPAA was synthesized in a single step as depicted in Figure 1. Firstly, 2.053 g EDA and 1.333 g HFDA were dissolved in 15 mL DMAc in a three-necked round-bottom flask. The mixed solution was stirred for 24 h under nitrogen atmosphere at room temperature. The resultant 20 wt% EFPAA DMAc solution was placed in the freezer. | |
| Figure 1: Synthetic route of EFPAA and the structure of NPAA. | |
| EFPAA (KBr, cm-1): 3411(υN-H), 2969(υC-H), 1659(υC=O), 1570(C=C of benzenoid rings), 1501(υC=C of benzenoid rings), 1316(υC−N), 1255(υC− O−C), 1100-1250(υC−F), 852(δC-H), 748(δC-H), 685(δC-H). 1H-NMR (d6- DMSO): δ=13.60 (m, -COOH), δ=10.60 (s, Ar-NH-CO-), δ=10.4(s, Ar-NH-CO-), δ=8.90(m, N-H), δ=7.29-6.84(m, Ar-H). GPC results: Mn: 55960, Mw: 108250, PDI: 1.93. | |
| Furthermore, a referential polymer was prepared through the same synthetic strategy using 4,4'-(1,3-phenylenedioxy)dianiline as the diamine monomer so as to clarify the effect of oligoaniline segments on the anticorrosion property. The resulting poly(amic acid) was named NPAA and also depicted in Figure 1. | |
| NPPA (KBr, cm-1): 3419(υN-H), 3000(υC-H), 1598(υC=C of benzenoid rings), 1504(υC=C of benzenoid rings), 1417(υC−N), 1265(υC−O-C), 1100- 1260(υC−F), 846(δC-H), 733(δC-H), 683(δC-H).1H-NMR (d6-DMSO): δ=13.58 (m, -COOH), δ=10.70(m, Ar-NH-CO-), δ=8.07 (m, N-H), δ=7.91-6.98(m, Ar-H). GPC results: Mn: 65460, MW: 114670, PDI: 1.75. | |
| Fabrication of EFPAA/ITO electrodes and NPAA/SS electrodes | |
| ITO substrates were washed ultrasonically with acetone and distilled water for 3 min each, followed by drying in the air. EFPAA DMAc solution was filtered through 0.2 μm syringe filter, and then spin-coated onto the ITO substrate for spectroelectrochemical measurements. The spin-coating process started at 500 rpm for 9 s and then 1000 rpm for 30 s. The EFPAA films, with thickness of 300 nm, were used in the electroactivity and electrochromism measurement. | |
| For the anticorrosion measurements, the SS substrates were prepared with 1.0 × 1.0 × 0.1 cm dimension and polished with emery papers. Then they were washed and degreased ultrasonically with acetone, distilled water and acetone again, followed by drying in a vacuum oven. The filtered polymer DMAc solution was cast onto treated SS substrates and then dried in the air to give the electrodes. The thickness of anticorrosive coatings was controlled by adjusting the concentration of polymer solution and/or increasing the times of dripping. | |
Results and Discussion |
|
| EFPAA structure | |
| EFPAA was synthesized using a one-step synthetic route as depicted in Figure 1. EFPAA, 1H NMR spectroscopy and GPC were applied to confirm the chemical structure of as-synthesized EFPAA. EFPAA spectrum displays the characteristic absorption bonds around 3411 cm-1 corresponding to the N-H stretching vibration, around 2969 cm-1 due to the C-H stretching vibration of aryl groups. The vibration around 1659 cm-1 is attributed to C=O stretching vibration. The chracteristic peaks at 1570 cm-1 and 1501 cm-1 were designated as the streching vibration of C=C groups in the benzene rings. Moreover, the bands at 1316 cm-1 (υC−N), 1255 cm-1 (υC−O-C), 1100-1250(υC−F), 852(δC-H), 748(δC-H), 683(δC-H), based on the structure of EFPAA are also observed. In the 1H NMR spectra of EFPAA, the characteristic proton signal of carboxylic acids appears at 13.6 ppm. The proton signals observed at 10.6 ppm and 10.4 ppm are attributed to the protons of amide groups. The signals in the range of δ=7.29-6.84 ppm are ascribed to the aromatic protons. All of the information from FTIR spectrum and 1H NMR spectrum support the expected molecular structure of EFPAA. Moreover, the number average molecular weight (Mn) and polydispersity index of EFPAA, obtained by GPC, are 55960 and 1.93. | |
| Thermal properties | |
| The thermal properties of EFPAA were evaluated by TGA. As shown in Figure 2, a clear three-step decomposition path is observed for the obtained EFPAA. The first weight loss began at 170°C with a mass loss of 8%, which was attributed to the progress of imidization and the evaporation of residual solvents. The second weight loss occurred in the temperature range of 330-430°C with a mass loss of 3% which corresponds to the achievement of imidization. The following weight loss after 500°C was assigned to the decomposition of the polymer main chain. When the temperature increases to 600°C, the weight loss is about 27%. When the temperature increases to 680°C, approximately 63% of the material remains. These results indicated that the imidized EFPAA exhibits good thermal stability and high degradation temperature, compared with other PANI materials [41]. | |
| Figure 2: TGA curve of EFPAA under nitrogen atmosphere. | |
| Electrochemical activity | |
| Cyclic voltammetry (CV) was performed to explore the electrochemical activity of EFPAA. In the traditional three-electrode system, the platinum wire electrode, saturated calomel electrode and EFPAA/ITO electrode were used as counter electrode, reference electrode and working electrode, respectively. The liquid electrolyte was 0.5 M H2SO4 solution. The CV curve of EFPAA/ITO is shown in Figure 3 and reveals two pairs of redox peaks at 310 mV/180 mV and 465 mV/385 mV. The first pair of redox peaks at 310 mV/180 mV should be attributed to the transition between the leucoemeraldine base (LEB) and the emeraldine base (EB), and the second pair of redox peaks should be assigned to the transition between EB and the pernigraniline base (PNB). Furthermore, CV of EFPAA was also carried out with the potential scan rate in the range of 10~100 mV/s. A linear dependence of the peak currents, as a function of scan rates, is seen in the inset of Figure 3, which confirmed a surface controlled process. | |
| Figure 3: 3: CV of EFPAA/ITO electrode in 0.5 M H2SO4 at different potential scan rates: 10-100 mV/s. Inset shows the relationships between the oxidation and reduction current vs. potential scan rate. | |
| Electrochromic behavior | |
| Polyaniline (PANI) is considered to be one of the most promising electrochromic materials due to its high optical contrast, good redox reversibility and electrochemical stability. Due to the introduction of oligoaniline, the synthesized EFPAA should be investigated as an electrochromic polymer. Firstly, the optical properties of EFPAA during the oxidation process were investigated by UV-vis spectroscopy Figure 4. When the oligoaniline segment is in its reduced state, only one strong absorption around 325 nm is observed in the UV-vis spectra, which should be associated with a π–π* transition of the conjugated ring system [42,43]. After the addition of ammonium persulfate, the absorption band of the π–π* transition continually decreased in intensity and underwent a blue shift. Meanwhile, a new absorption band appeared at 570 nm and continually increased in intensity, which is assigned to exciton-type transition between the HOMO orbital of the benzoid ring and the LUMO orbital of the quinoid ring in oligoaniline segment [42,43]. When the oligoaniline segment was oxidized to EB, the intensity of π-polaron transiting absorption reached a maximum. After that, the absorption peak of exciton-type transition decreased gradually with further oxidation. | |
| Figure 4: UV-Vis spectra monitoring the chemical oxidation of EFPAA, (a) from LEB to EB, and (b) from EB to PNB. | |
| The above spectroscopic properties of EFPAA are consistent with those of polyaniline, which indicate that the EFPAA shows great potential as an electrochromic material. Therefore, the electrochromic behavior of EFPAA was investigated in detail by sp ectroelectrochemical experiments, which were performed in 0.5 mol/L H2SO4 coupled with applied potentials (-0.2 V, 0.2 V, 0.4 V, 0.6 V, 0.8 V, 1.0 V and 1.2 V (vs. Ag/AgCl)). As shown in Figure 5, the EFPAA film shows different UV-Vis absorption spectra at various applied potentials, exhibiting the maximum optical contrast value (%ΔT) about 55% at 700 nm between its coloring (0.8 V) and bleaching (-0.2 V) states. Moreover, the color of EFPAA film changes drastically from transmissive gray (-0.2 V), to green (0.4 V), and finally to absorptive black blue (at 0.8 V) (inset of Figure 5). | |
| Figure 5: The spectral changes of the EFPAA/ITO electrode at different potentials, and the inset show photographs of EFPAA/ITO electrode at different potentials. | |
| Furthermore, the switching time and the electrochromic efficiency were also studied in detail by spectrochronoamperometry, which was conducted in the optical contrast at 700 nm during repeated potential stepping between reductive (-0.2 V) and oxidative state at 0.8 V with a residence time of 30 s. The results for the first 500 s were presented in the Figures 6a and 6b. From the electrochemical curves, the switching time of EFPAA film is about 5.2 s at 0.8 V for the coloring process at 700 nm. The EFPAA film requires 4.0 s at -0.2 V for the bleaching process, which is moderate for a polymeric electrochromic material. In addition, the electrochromic coloration efficiency (CE) is calculated for 45.4 cm2/C (at 700 nm) at the oxidation stage by monitoring the amount of ejected charge (Q) as a function of the change in optical density (ΔOD) of the EFPAA film. | |
| Figure 6: a) Current consumption, (b) absorbance changes (first 500 s) monitored at 700 nm of EFPAA/ITO electrode in 0.5 mol/L H2SO4 when the anodic potential was switched between -0.2 V and 0.8 V with a residence time of 30 s. | |
| Anticorrosion performance | |
| Considering the good electrochemical activity of EFPAA, the anticorrosive performance is investigated here in detail. To clarify the effect of oligoaniline segments on the anticorrosion properties, a referential polymer without electroactive segment (NPAA) was also studies. Firstly, the polarization technique was used to study the anticorrosion performance of EFPAA and NPAA for SS, where the large cathodic and anodic polarizations provide curves for the respective corrosion processes. The corrosive solution is 3.5 wt% NaCl solutions. The anticorrosive coatings were cast with thickness of 10 μm, 20 μm, 40 μm and 60 μm by dripping method. From the Tafel curves (Figure 7), both corrosion potential (Ecorr) and corrosion current density (Icorr) of all the samples are determined according to Tafel extrapolation method, and listed in Table 1. In case of the bare SS sample, the Ecorr and Icorr are -0.744 V and 6.277 μA/cm2, respectively. When NPAA is used as the anticorrosive coating with the thickness of 10 μm, 20 μm, 40 μm and 60 μm, the Ecorr shifts to 0.300 V, 0.453 V, 0.912 V, 1.446 V and the Icorr decreases drastically to 1.178 μA/cm2, 0.637 μA/cm2, 0.039 μA/cm2, 0.027 μA/cm2. All the above results demonstrate the corrosion protection of the NPAA coatings. After coated by EFPAA with thickness of 10 μm, 20 μm, 40 μm and 60 μm, the Ecorr shift significantly positively to 0.399 V, 0.584 V, 1.381 V and 2.169 V, which confirm an effective corrosive protection, is provided by the EFPAA coatings. The Icorr is also found to decrease drastically to 0.259 μA/cm2, 0.043 μA/cm2, 0.029 μA/cm2 and 0.011 μA/cm2 indicating that the overall corrosion process is also reduced significantly. | |
| Table 1: Electrochemical corrosion parameters of the EFPAA/SS and NPAA/ SS samples with various thicknesses (10 µm, 20 µm, 40 µm, 60 µm) in 3.5 wt% NaCl solution. | |
| Figure 7: Tafel plots for EFPAA/SS (a) and NPAA/SS (b) corrosive electrodes with various thicknesses (10 µm, 20 µm, 40 µm, 60 µm) in 3.5 wt% NaCl solutions. | |
| According to the related equations, other electrochemical corrosion parameters of the samples, such as Rp value, Rcorr and PEF, were calculated and presented in Table 1. The Rp value of EFPAA/ SS samples increase from 0.149 to 85.335 kΩ cm2 as the thickness of EFPAA increase from 0 to 60 μm in 3.5 wt % NaCl solution. In contrast, the Rp value of NPAA/SS increases to 8.934 kΩ cm2 when the thickness increases to 60 μm. As shown in Table 1, Rcorr is found to be 4.83 × 10-2 mm per year for the bare SS. The Rcorr of EFPAA coating with thickness of 60 μm decreases greatly to 8.54 × 10-5 mm per year, giving a high PEF (99.83%). In contrast, the Rcorr of NPAA is about 2.07 × 10-4 mm per year, which is bigger than that of EFPAA. Through the comparative and detailed study of the anticorrosive coatings, the effect of the oligoaniline segment on the anticorrosion has been illuminated, which can be attributed mainly to the presence of π-electron clouds coexisting with nitrogen atom [44,45]. | |
| Electrochemical impedance spectroscopy (EIS) was also used to investigate the anticorrosion behavior of EFPAA and NPAA coatings. All the Nyquist plots of the samples, fitted by equivalent circuit, are presented in Figure 8, which are close to a semicircle arc. The Randles type equivalent circuit model was chosen to simulate the experimental data at the initial state. The film resistance (Rf) for the samples is determined by the intersection of the low-frequency end of the semicircle arc with the real axis. For the bare SS samples, the Rf of the bare SS is 17742 Ω. The Rf of NPAA/SS samples increases to 69640 Ω, 162710 Ω, 472052 Ω and 1022856 Ω as the thickness of EFPAA film increased to 10 μm, 20 μm, 40 μm and 60 μm. For the EFPAA/SS samples, the Rf values are 75710 Ω, 12984 Ω, 161966 Ω and 181951 Ω as the thickness increased to 10 μm, 20 μm, 40 μm and 60 μm. Comparing all the Rf values of the samples, the higher Rf values of EFPAA would demonstrate the availability of the oligoaniline segments to prevent the corrosion of SS, which also implies that the prepared EFPAA coating offers excellent corrosion protection to SS in the brine. | |
| Figure 8: EIS for EFPAA/SS (a) and NPAA/SS (b) with various thicknesses (10 µm, 20 µm, 40 µm, 60 µm) in 3.5 wt% NaCl solution. | |
Conclusion |
|
| EFPAA, with pendant tetraaniline and fluorine groups, was synthesized by simple polycondensation chemistry. The novel polymer exhibits good thermal stability and reversible electrochemical properties. EFPAA film reveals good electrochromic performance with high contrast value, moderate switching times and acceptable coloration efficiency. The effective anticorrosion behaviors of the EFPAA coatings were confirmed by tafel plots analysis and electrochemical impedance spectroscopy, which indicated the promise as competitive anticorrosion materials. The detailed investigation of EFPAA not only paves the way for its practical application, but has also inspired us to explore new characteristics for polymer materials with various functional groups. | |
References |
|
|
|
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi