Case Report, Int J Cardiovasc Res Vol: 11 Issue: 2
Acute Occlusion of Whole Length of Inferior Vena Cava Presenting as Pain in Sacroiliac Region
Rajeev Bhardwaj*
Department of Cardiology, MM Institute of Medical Sciences and Research, Mullana, Ambala, India
*Corresponding Author: Rajeev Bhardwaj
Department of Cardiology, MM Institute of Medical Sciences and Research, Mullana, Ambala, India
Email: rajeevbhardwaj_dr@yahoo.com
Received date: 01 February, 2022; Manuscript No. ICRJ-21-25427;
Editor assigned date: 03 February, 2022; PreQC No. ICRJ-21-25427(PQ);
Reviewed date: 14 February, 2022; QC No ICRJ-21-25427;
Revised date: 24 February, 2022; Manuscript No. ICRJ-21-25427(R);
Published date: 03 March, 2022; DOI: 10.4172/2324-8602.1000455.
Citation: Bhardwaj R (2022) Acute Occlusion of Whole Length of Inferior Vena Cava Presenting as Pain in Sacroiliac Region. Int J Cardiovasc Res 11:2.
Abstract
Thrombosis of the Inferior Vena Cava (IVC) is an under recognized entity with a variety of clinical presentations. Thrombosis of the IVC in the absence of congenital abnormalities is rare; when it does occur, it is usually the result of a predisposing hypercoagulable state. We describe a rare presentation of acute thrombotic occlusion of IVC, which was diagnosed accidentally.
Keywords: Inferior vena cava; Occlusion; Acute; Filter
Introduction
50 years male presented with history of pain in bilateral sacro iliac region for 4 days. Pain was moderate in severity, and was more on walking. Patient took treatment from local hospital in the form of analgesics. There was no relief of pain. His X ray of lumbosacral spine was done, which was normal. His pain increased in severity over next 3-4 days and he was confined to bed. He then went to private hospitals, where his blood investigations were done, which were normal. His CT for lumbosacral was also done and was normal. They also did ultrasound of abdomen, which showed thrombotic occlusion of right common iliac vein and whole of Inferior Vena Cava (IVC). He was then referred to us. All other abdominal organs were normal. X ray of chest was also normal.
There was no history of swelling of lower limbs or pain in lower limbs. There was no history of fever, pain in abdomen or recent surgery or prolonged immobilization. Appetite was normal. Bowel and urinary habits were normal.
Case Study
On examination, there was no pallor, cyanosis, jaundice or pedal oedema. Jugular venous pressure was normal. Blood pressure was 130/80 mm HG, pulse 76/min, regular. Homans’ sign was negative on both sides.
The international normalized ratio was 1.1. Other laboratory findings were as follows: Leucocyte count 12,800/mm3, platelets 234,000/mm3, hemoglobin 14.9 mg/dL, fasting blood glucose 108 mg/dL, ESR 15 mm in first hour, D dimer 2000 mg/dl, lipoprotein (a) 28.3 mg/dL (normal range<30 mg/dl): C-reactive protein 74.3 mg/dL (normal range: 0-0.8 mg/dL), homocystein 7.46 μmol/L (normal range:<35) protein C 74% (normal range: 70%-130%), protein S 85% (normal range: 65%-130%), factor V 76% (normal range: 70%-120%), antithrombin III 94% (normal range: 75%-125%).
CT scan of abdomen showed Intraluminar filling defect in IVC, extending from hepatic veins to a tributary of right common iliac vein, thrombus extending into bilateral renal veins.
He was taken up to catheterization laboratory. Femoral venography was done on right side, which showed total occlusion of right common iliac vein. IVC was not visualized. Large collateral was there on the right side of IVC opening just below Right Atrium (RA) in to the IVC (Figure 1).
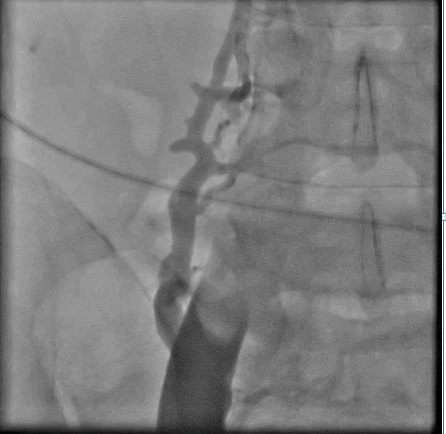
Figure 1: Femoral venography shows total occlusion of upper common iliac vein and IVC. There is large collateral on to the right of IVC.
Terumo guide wire easily crossed through IVC into RA, suggesting that it was occluded with recent thrombus. 60 mm balloon was taken and multiple holes were made in it with femoral puncture needle. It was kept in IVC and through it alteplase infusion 0.001 mg/kg/hour was given for 20 hours. Check angio was done after 20 hours. Common iliac and lower portion of IVC was reanalyzed but upper part of IVC was still occluded. Again balloon with holes was kept in upper part of IVC and 24 hours infusion of alteplase was given through it. Check angio showed recanalization of IVC, but still a lot of thrombus was there (Figures 2 and 3).
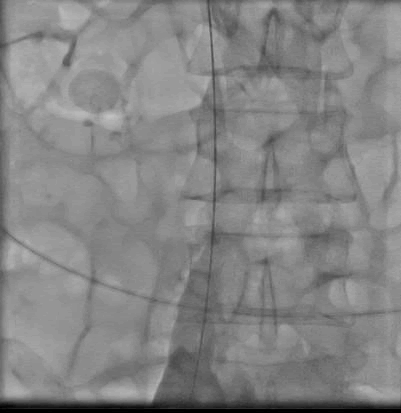
Figure 2: Recanalization of common iliac vein and lower part of IVC after catheter directed thrombolysis.
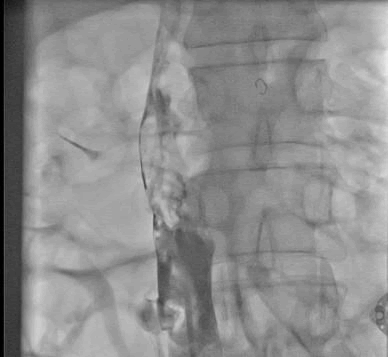
Figure 3: Recanalization of IVC after catheter directed thrombolysis for another 24 hours, bot thrombus still there in IVC.
9F long sheath was now inserted into IVC and manual thrombus extraction was done. There was good flow, but still thrombus was there and there was narrowing in upper part of IVC (Figure 4). IVC filter was inserted from internal jugular approach (Figure 5), and IVC was dilated with 14 mm balloon (Figure 6).
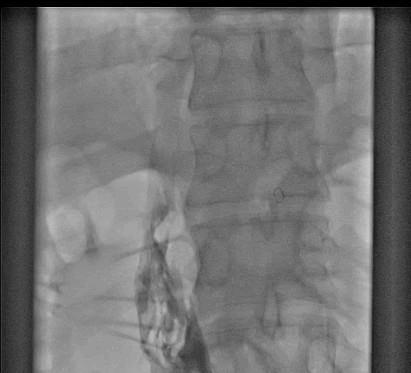
Figure 4: Result in IVC after Manual thrombus extraction.
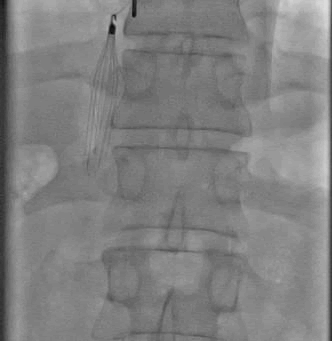
Figure 5: IVC filter implantation through internal jugular vein.
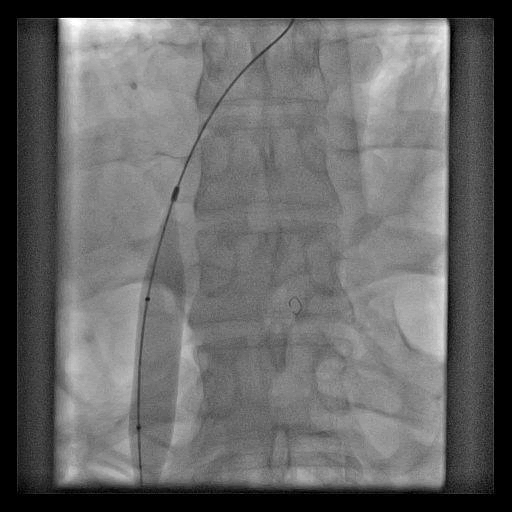
Figure 6: Balloon dilatation of IVC with 14 mm balloon.
There was good flow in IVC (Figure 7), with hardly any thrombus left. Patient had complete relief of has pain in sacro iliac region and was able to walk normally. He was discharged on tablet dabigatran 150 mg twice a day. Check angio after two months showed no evidence of thrombus in IVC. IVC filter was retrieved. Patient remained asymptomatic. Anticoagulants were stopped after one year. On follow up of two years, patients remained asymptomatic.
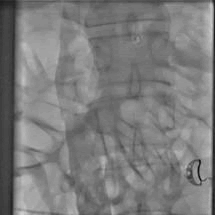
Figure 7: Final result in IVC.
Discussion
The clinical presentation of IVC thrombosis varies according to the anatomical level of thrombosis, the degree of occlusion and chronicity of the thrombus. Acute IVC Thrombosis (IVCT) is generally accompanied by a sudden onset of symptoms, while chronic IVCT, presents with a more gradual onset, secondary to thrombotic IVC occlusion with already established venous collateralization or gradual narrowing of the IVC [1]. As with DVT, the typical presentation of IVC thrombosis may present with pain, swelling, and cramping of the lower extremities. In addition, and more specific to IVC thrombosis, back and pelvic pain are generally the earliest symptoms. In cases that have renal vein involvement, symptoms may involve hematuria and flank pain whereas bilateral renal venous thrombosis can result in acute renal failure [2].
In addition, patients with chronic thrombosis can present with severe lumbar radicular pain, sciatica, orcauda-equina symptoms secondary to compression from Para spinal collaterals [3]. Thrombosis of the IVC in the absence of congenital abnormalities is rare; when it does occur, it is usually the result of a predisposing hypercoagulable state along with an acquired pathology in the IVC or one of its adjacent structures. Iatrogenic thrombotic occlusion of IVC filters is increasing, particularly in the United States. To a large degree, the etiology of IVCT mirrors that of DVT in general. However, specific situations relate to the IVC only, but the wide variety of these situations all relate in one or more ways to Virchow's classic description. Numerous malignancies have been associated with IVCT [4]. Perhaps the most familiar is Renal Cell Carcinoma (RCC). Other retroperitoneal organ systems that have been shown to cause IVCT include the pancreas and the kidneys. Polycystic disease of the right kidney has reportedly been clinically associated with thrombosis of the IVC [5]. Other retroperitoneal organ systems that have been shown to cause IVCT include the pancreas and the kidneys. Pancreatic pseudo cyst has also been observed to cause thrombosis of the IVC [6]. Acute pancreatitis has also been found to generate thrombosis of the IVC [7,8]. Patients with a recent history of medical care may present with iatrogenic IVCT. The expansion of endovascular technology has led to increased recognition of iatrogenic IVCT [9].
The cornerstone of treatment is systemic anticoagulation therapy to reduce thrombus propagation and minimize the risk of pulmonary embolization while alleviating lower extremity symptoms .The open vein hypothesis of DVT would seem to translate to patients with IVC thrombosis as well, in that long term outcomes would improve with more aggressive endovascular treatment involving thrombolysis or thrombectomy [10]. Although placement of IVC filter for endovascular treatment of iliofemoral vein thrombosis may not be indicated consideration for placing a filter should be made when treating IVC thrombus given the more extensive and possibly free floating clot [11].
Our patient was unusual in that he presented with pain in bilateral sacro iliac regions, had no predisposing conditions, and had no clinical features of IVC occlusion, and had a very short history.Paksyo and Gormus examined 9640 patients experiencing back pain or sciatica, using MRI over two years.
Conclusion
There were 13 (0.13%) patients who had radicular symptoms that clinically mimicked lumbar disc herniation or spinal stenosis. All of these patients had inferior vena caval obstruction or occlusion that caused engorgement in the epidural and paravertebral venous system, causing nerve root compression.
Patient was managed with catheter directed thrombolysis, and manual thrombus extraction and responded well.
References
- Marcy PY, Magné N, Frenay M, Bruneton JN (2001) Renal failure secondary to thrombotic complications of suprarenal inferior vena cava filter in cancer patients. Cardiovasc Intervent Radiol 24: 257-259. [Crossref][Google Scholar][Indexed]
- Go MR, Baril DT, Leers SA, Chaer RA (2009) Acute cauda equina syndrome secondary to iliocaval thrombosis successfully treated with thrombolysis and pharmacomechanical thrombectomy. J Endovasc Ther 16: 233-237. [Crossref][Google Scholar][Indexed]
- Kraft C, Schuettfort G, Weil Y, Tirneci V, Kasper A, et al. (2014) Thrombosis of the inferior vena cava and malignant disease. Thromb Res 134: 668-673. [Crossref][Google Scholar][Indexed]
- O'Sullivan DA, Torres VE, Heit JA, Liggett S, King BF (1998) Compression of the inferior vena cava by right renal cysts: an unusual cause of IVC and/or iliofemoral thrombosis with pulmonary embolism in autosomal kidney disease. Clin Nephrol 49: 332-334. [Crossref][Google Scholar][Indexed]
- Peillon C, Manouvrier JL, Testart J (1991) Inferior vena cava thrombosis secondary to chronic pancreatitis with pseudocyst. Am J Gastroenterol 86: 1854-1856. [Crossref][Google Scholar][Indexed]
- Jones AL, Ojar D, Redhead D, Proudfoot AT (1998) Case report: Use of an IVC filter in the management of IVC thrombosis occurring as a complication of acute pancreatitis. Clin Radiol 53: 462-464. [Crossref][Google Scholar] [Indexed]
- Stringer MD, Michell M, McIrvine AJ (1988) Inferior vena caval thrombosis complicating acute pancreatitis. case report. Acta Chir Scand 154: 161-163. [Crossref][Google Scholar][Indexed]
- Peck KE, Bonoan JT, Cunha BA (1998) Postlaparoscopic traumatic inferior vena caval thrombosis. Heart Lung 27: 279-282. [Crossref][Google Scholar][Indexed]
- Sista AK, Vedantham S, Kaufman JA, Madoff DC (2015) Endovascular interventions for acute and chronic lower extremity deep venous disease: State of the art. Radiology 276: 31-53. [Crossref][Google Scholar][Indexed]
- Protack CD, Bakken AM, Patel N, Saad WE, Waldman DL, et al. (2007) Long-term outcomes of catheter directed thrombolysis for lower extremity deep venous thrombosis without prophylactic inferior vena cava filter placement. J Vasc Surg 45: 992-997. [Crossref][Google Scholar][Indexed]
- Paksyo Y, Gormus N (2004) Epidural venous plexus enlargements presenting with radiculopathy and back pain in patients with inferior vena cava obstruction or occlusion. Spine 29: 2419-2424. [Crossref][Google Scholar][Indexed]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 



