Research Article, J Biochem Eng Bioprocess Technol Vol: 0 Issue: 0
The Microorganism Acidithiobacillus ferrooxidans can use Electrical Current as a Direct and Sole Energy Source
Dimitre G Karamanev*
Department of Chemical and Biochemical Engineering, Western University, Ontario, Canada
*Corresponding Author : Dimitre G Karamanev
Department of Chemical and Biochemical Engineering, Western University, Ontario, Canada
Tel: +1 (519) 661 2111-88230
Fax: +1 (519) 661 3498
E-mail: dkaraman@uwo.ca
Received: August 14, 2018 Accepted: August 27, 2018 Published: September 03, 2018
Citation: Karamanev DG (2018) The Microorganism Acidithiobacillus ferrooxidans can Use Electrical Current as a Direct and Sole Energy Source. J Biochem Eng Bioprocess Technol 1:2.
Abstract
Objective: It is well known that there are two main groups of living organisms in terms of their energy source: chemotrophs and phototrophs. Recently it has been shown that some living organisms, and in particular, microorganisms, could use electrical current as both a sole energy source and as an electron donor. This, third group of organisms, was named electro trophs.The main goal of this work is to prove that the microorganism Acidithiobacillus ferrooxidans can live in electro trophic mode.
Methods: A novel electro bioreactor was designed and built in order to supply microorganisms with electrical current (electrons) as an electron donor.
Results and Conclusion: The experimental results reported here show for the first time that the aerobic microorganism Acidothiobacillus ferrooxidans, can live in an electro trophic mode.
Keywords: Electrotrophy; Acidithiobacillus ferrooxidans; Electrical current; Electrodes
Introduction
All known living organisms such as animals, plants and microorganisms, require energy for their life. The energy is usually supplied by redox reactions, in which an electron acceptor is reduced by consuming electrons, while an electron donor is oxidized, releasing electrons. The process of transfer of electrons, downhill the energy curve, from the electron donor to the electron acceptor, releases energy which is used by the living organisms. The electron donors release electrons due to the energy of either chemical reaction or a photochemical one. Therefore, from the point of view of the nature of the electron donor reaction, all known living organisms are classified into two groups:
• Chemotrophs – These organisms use either organic or inorganic substances as electron donors, and use their chemical energy as energy source. Humans and animals are typical examples of chemoorganotrophs; some microorganisms such as Acidithiobacillus ferrooxidans are typical chemolithotrophs;
• Phototrophs – This type of organisms use photochemical reactions as a source of electrons, and their ultimate energy source is light. Typical examples are algae.
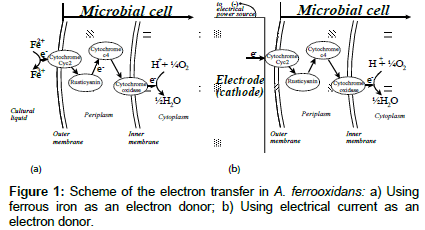
The bacterium A. ferrooxidans has been discovered more than 60 years ago in acid mine drainage [1]. As mentioned above, it is a typical chemolithotroph, obtaining energy from the oxidation of different inorganic electron donors such as sulfides, sulfur, sulfites and ferrous ions [2-4]. In the latter case, the overall energy-supplying reaction is as follows:
Fe2++¼O2+H+=Fe3++½H2O (1)
Where the ferrous ions are the electron donor and oxygen is the electron acceptor. Reaction (1) is actually the sum of two halfreactions separated in space: the oxidation of the electron donor:
Fe2+=Fe3++e- (2)
Takes place outside of the cell membrane (Figure 1a), while the reduction of the electron acceptor:
¼O2+H++e-=½H2O (3)
It is carried out in the cytoplasmic membrane (Figure 1a). The electrons are transported from the electron donor, through the periplasm, to the electron acceptor via a chain of several redox proteins (Figure 1a). While the exact mechanism of the electron transport in A. ferrooxiadans has not been completely characterized yet, a general scheme was originally proposed by Ingledew [5], and later updated by several other authors. Appia-Ayme et al. [6] proposed the electron transfer pathway showed in Figure 1a. The outer-membrane c-type cytochrome, Cyc2, acquires electrons from ferrous iron oxidation (reaction 2) and transfers them through the chain of other cytochromes and rusticianin to the oxygen reduction site. Regardless of the specific mechanism proposed, it has been established that ferrous iron oxidation takes place outside of the microbial cell [7], since the outer-membrane cytochrome is exposed to the external environment (Figure 1a). In addition, it has been shown that the cellfree isolate of A. ferrooxidans cytochromes has an electron affinity in the absence of iron ions [5].
A. ferrooxidans obtains carbon for cell growth from the fixation of carbon dioxide. The energy for carbon dioxide fixation comes from reaction (1).
Several researchers studied the coupled electrochemical reduction of Fe3+ with the biological oxidation of Fe2+ [8] in order to regenerate the substrate for the A. ferrooxidans growth (Fe2+) electrochemically. The electrochemical reaction taking place on the surface of the cathode is the reduction of ferric ions:
Fe3++e-=Fe2+ (4)
While in the liquid surrounding the cathode, free suspended cells of A. ferrooxidans oxidize the ferrous ions (Reaction 2), which actually is the reversal of the cathode reaction. Thus, electrical energy has been used for the regeneration of microbial substrate (Fe2+) according to Reaction 4, and therefore, the electricity was used indirectly as an energy source for microbial metabolism and growth. However, these microorganisms used directly a chemical electron donor (ferrous ions) for obtaining energy. While this mode of indirect use of electrical current was referred to as “electro trophy” [9], the microorganisms used ferrous iron (not electricity) as a direct electron donor.
For the first time the use of electrical current as a direct and sole energy source for living organisms (and more specifically, aerobic microorganisms) was reported a decade ago [10,11]. The term “electro trophy”, and alternatively, “electron trophy” was proposed in these works to describe the process. Gregory et al. [12] showed “for the first time that electrodes may serve as a direct electron donor for anaerobic respiration”. Later, other anaerobic microorganisms were reported to grow in electro trophic mode [13].
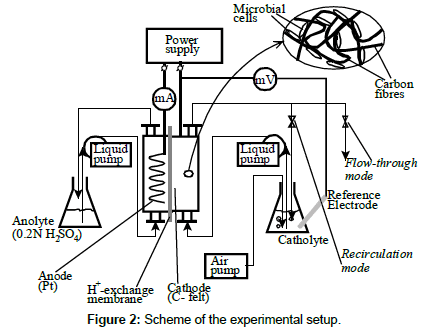
Since the electron generation reaction by the electron donor in A. ferrooxidans (2) is carried out at the outer cell surface, and since the redox proteins responsible for ferrous iron oxidation have electron affinity even in the absence of iron, the main question behind this work was: is it possible to supply the outer membrane redox proteins of the microbial cell with electrons from an electrode (replacing Reaction 2), which physically contacts the microorganism, according to Figure 1b? In order to answer this question, an electro biochemical experimental setup was designed (Figure 2).
Materials and Methods
The experimental setup used in this work is shown in Figure 2. Its main part was an electro biochemical cell, in which microbial cells of A. ferrooxidans were immobilized on the surface of the cathode. The cell had a cylindrical shape. The cathodic compartment was 2.7 cm in diameter and 2 cm deep, while the anodic compartment was 2.7 cm in diameter and 7 mm deep. The cathode was made of carbon felt, containing 0.5 g SiO2 powder, used to improve the immobilization of A. ferrooxidans cells. The current collector was a graphite rod, so that microorganisms and the catholyte contacted only carbon as an electrically-conductive material. The cathode potential was measured by using a reference Ag/AgCl electrode, submerged in the catholyte, and was controlled, when required, by varying the voltage of the power supply (Figure 2).
The catholyte, which contained the microbial nutrients, was an aqueous solution of 9K salts of Silverman and Lundgren [14]. During the electro trophic experiments, the liquid phase surrounding the microbial cells contained no chemical or photochemical electron donors. Therefore, the cathode itself was the only source of electrons to microorganisms, delivered by electrical current. The electron acceptor for the microbial cells was molecular oxygen, dissolved in the liquid; it was reduced according to reaction (3). The anode material was platinum wire, and the anolyte was 0.2 M aqueous solution of sulfuric acid. The cathodic space was separated from the anodic one by means of Nafion proton-exchange membrane (DuPont, USA). The microbial culture of A. ferrooxidans, JCM3863, was obtained from the Japan Collection of Microorganisms.
The oxygen concentration in liquid medium was measured using an Orion 850 dissolved oxygen meter equipped with a polarographic probe.
The electrical current and voltage were measured using a Fluke digital multimeter.
The microscopic study was performed using an optical microscope (Carl Zeiss 455043) with 1000X magnification having a phase contrast objective.
The ferric and total iron concentrations were measured spectrophotometrically using sulfosalicilic acid as an indicator and a UV-Vis spectrometer Cary 50 (Varian Inc., Sydney, Australia) [15].
Results and Discussion
The microbial culture was initially grown in a shake flask in an aqueous solution, containing 9 g/L Fe2+ in the form of sulfate, as well as a nutrient salts composition, according to the 9K medium. The pH of the medium was 2.2, obtained by adding sulphuric acid. Once 99% of ferrous iron was oxidized, liquid was pumped from the shake flask into the cathode space of the electro biochemical cell and returned back to the shake flask by means of continuously operating peristaltic pump (Figure 2). At this stage, the flask was aerated by air bubbling instead of shaking. The cathode potential was kept at 640 mV (vs. standard hydrogen electrode) by varying the voltage of the external power source. All potentials reported in this work are given vs. a standard hydrogen electrode. The initial value of the electrical current flowing through the cell was 103 mA, which was slowly decreasing until a steady-state condition, characterized by the equality between the rate of electrochemical reduction of ferric ions and biological oxidation of ferrous ions, was reached. The steady-state current was 45 mA. The microbial growth on the cathode surface was encouraged by the fact that the concentration of substrate (Fe2+) in the vicinity of the cathode surface was higher than anywhere else in the recirculation system, due to the electrochemical reduction of Fe3+ (reaction 4) on the cathode. The iron ions, present in the system, did not allow studying the possibility for electrotrophic growth of A. ferrooxidans, because iron acted as an intermediate electron carrier between the cathode and microbial cells. Therefore, our next goal was to remove the iron ions from the system in order to check the possibility of growing A. ferrooxidans directly on electrical current. Once a stable electrical current of 45 mA was obtained, the catholyte, containing iron ions, was replaced with a 9K solution, containing no iron ions. The system contained some residual iron in the form of both soluble and insoluble compounds. The latter was represented by jarosite [16] formed on the surface of the cathode. The slow dissolution of jarosite caused an increase in the iron concentration in the catholyte in time. In order to remove this residual iron, the catholyte was replaced by iron-free 9K solution 7 times. The concentration of iron ions in the catholyte was measured by the sulfosalycillic acid method [17]. In all the experiments described above, the liquid was recirculated between the cathode space and the aerated flask. In order to ensure complete removal of iron from the system, the iron-free 9K solution was next introduced to the cathode in a flow-through mode (retention time of 4.3 min) for a period of six days. At that point, the total iron ions concentration in the liquid effluent of the cathode fell below the detection level (approx. 50 μg/L) of the analytical method used, and no iron was detected in the liquid until the end of the experiments.
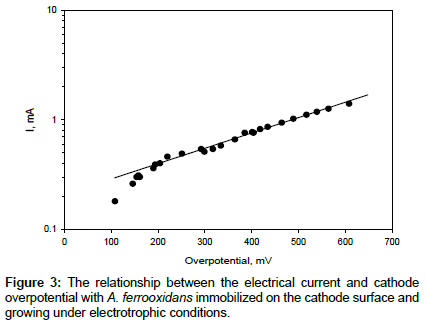
The effect of the cathode over potential on the current was studied (Figure 3). It can be seen that the curve is linear in a semi-logarithmic plot, and therefore, it follows the Tafel equation. In a separate, sterile experiment, no electrical current was detected within the same range of over potential values, and therefore, there was no electrochemical reduction of oxygen on the cathode. In the biological experiment, the open-circuit potential of the cathode was found to be 606 mV. It is interesting to note that this over potential is quite close to the formal potential of the Fe3+/Fe2+ electrochemical couple in 9K solution (676 mV) [18] which is the usual substrate for A. ferrooxidans. The oxygen consumption in the cathodic space was studied in continuous regime; when the liquid flowed through the cathodic space with different flow rates without recycle. The oxygen consumption rate in the cathodic space was relatively constant with an average value of 7.0 μg/min. The theoretical oxygen consumption rate, calculated from the current of the cell using the Faraday’s law, was 2.9 μg/min, or 41% of the actual value. The difference between the two values (59%) can be explained by the oxygen use for biomass formation. This value is somehow higher than that reported for A. ferrooxidans growing on ferrous iron, where between 3% and 30% of oxygen consumed is used for biomass formation [2]. The efficiency reported here (41%) is somehow lower than that reported by Lovley, at>90% [13].
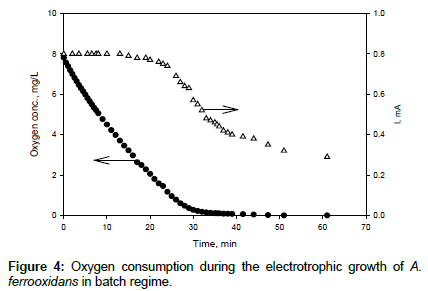
The oxygen consumption by microorganisms was also measured in batch regime (Figure 4). At oxygen concentrations higher than 2.6 mg/L, the oxygen consumption rate was approximately constant at 8.6 μg/min and the electrical current remained constant. At lower oxygen concentrations, the oxygen consumption rate and the current decreased proportionally to each other. The decrease in the oxygen consumption rate at low oxygen concentrations can be explained by the Monod-type relationship between these two parameters.
In order to make sure that the inorganic salts in the 9K medium are not electron donors for the microorganisms, the cathodic space was next fed with water containing only sulfuric acid (for pH control at 2.8). The liquid was recirculated between the cathodic space and the aerated vessel. Immediately after the replacement of the salts medium with H2SO4 solution, the current dropped from 0.74 mA to 0.28 mA, but within the next 30 hours it rose back to 0.72 mA, and remained at that value for the length of the experiment (four days).
The viability of microorganisms in the cathodic space was determined by both microscopic observation using a phase-contrast microscopy, and by determination of the ferrous iron oxidation activity. This study was performed after 10 days of electro trophic growth (i.e. in the absence of chemical electron donors such as dissolved iron in the catholyte). The microscopic study of a sample of the cathode (carbon fibre) showed a large amount of viable microbial cells having the shape and size of A. ferrooxidans. Most of the cells were attached to the carbon fibre surface. In order to check the iron oxidation activity of the microbial cells, immobilized on the cathode surface, a catholyte containing 1.2 g/L ferrous ions and nutrient salts (9K) was used. The power supply was detached from the electro biochemical cell. After a lag period of 30 minutes, the ferrous iron was oxidized with a rate of 650 mg/L.h. This is a typical value for the iron oxidation by immobilized A. ferrooxidans [19].
Electrotrophy can potentially have significant practical importance. Some potential applications of electro trophs can include:
• Production of biomass (single-cell protein) using electricity as a substrate. It has already been shown that A. ferrooxidans contains 44% protein, 26% lipids and 15% carbohydrates [20]. The single-cell protein produced by electrotrophy is expected to be free from residual toxic substrates (such as methanol in some of the conventional processes) and pathogenic microorganisms [21], since the latter are not expected to grow under electrotrophic conditions. It should be noted that in the case of electro trophic single-cell protein production, carbon dioxide is used as a carbon source, and therefore, the production of biomass is coupled with the consumption of CO2 from atmosphere;
• Production of energy in biofuel cells [11]. The electrotrophic microorganisms can replace platinum and other highly expensive cathode catalysts in fuel cells;
• Microbial electronics: Some of the electrical features of electro trophs, such as one-way electron transport, could be used in electronic devices.
Since A. ferrooxidans, as a chemolithotroph, is considered to be one of the simplest forms of life, according to some theories of the origin of life, it played a significant role in the life’s early evolution [22]. Now, with the discovery of electro trophy, even a simpler life form than lithotrophs has been found. Thus, electro trophy potentially could find its place in life evolution theories.
Conclusion
The results of this work show that the aerobic chemolithoautotrophic microorganism Acidothiobacillus ferrooxidans, can use electrical current as a sole energy source, i.e. as an electron donor. Therefore, these microorganisms are electrotrophs. This finding can find practical applications in various fields such as electrical protein production and bioelectricity generation.
Acknowledgement
This work was supported financially by the Natural Sciences and Engineering Research Council of Canada.
References
- Colmer AR, Hinkle ME (1947) The role of microorganisms in acid mine drainage: A preliminary report. Sci 106: 253-256.
- Nemati M, Harrison STL, Hansford GS, Webb C (1998) Biological oxidation of ferrous sulphate by Thiobacillus ferrooxidans: A review on the kinetic aspects. Biochem Eng J 1: 171-190.
- Suzuki I (2001) Microbial leaching of metals from sulfide minerals. Biotechnol Adv 19: 119-132.
- Taylor BE, Wheeler MC, Nordstrom DK (1984) Isotope composition of sulphate in acid mine drainage as measure of bacterial oxidation. Nature 308: 538-541.
- Ingledew WJ (1982) Thiobacillus ferrooxidans. The bioenergetics of an acidophilic chemolithotroph. Biochim Biophys Acta 683: 89-117.
- Appia-Ayme C, Guiliani N, Ratouchniak J, Bonnefoy V (1999) Characterization of an operon encoding two c-type cytochromes, an aa(3)-type cytochrome oxidase, and rusticyanin in Thiobacillus ferrooxidans ATCC 33020. Appl Environ Microbiol 65: 4781-4787.
- Yarzábal A, Brasseur G, Ratouchniak J, Lund K, Lemesle-Meunier D, et al. (2002) The high-molecular-weight cytochrome c Cyc2 of Acidithiobacillus ferrooxidans is an outer membrane protein. J Bacteriol 184: 313-317.
- Matsumoto N, Nakasono S, Ohmura N, Saiki H (1999) Extension of logarithmic growth of Thiobacillus ferrooxidans by potential controlled electrochemical reduction of Fe(III). Biotechnol Bioeng 64: 716-721.
- Yamanaka T (2008) Chemolithoautotrophic bacteria: Biochemistry and environmental biology. Springer.
- Karamanev D (2003) Production of microbial biomass using electrical current. Provisional US Patent.
- Karamanev D (2006) Electrotrophs – microorganisms that transform chemical energy into electricity. World Congr Ind Biotechnol.
- Gregory KB, Bond DR, Lovley DR (2004) Graphite electrodes as electron donors for anaerobic respiration. Environ Microbiol 6: 596-604.
- Lovley DR (2011) Powering microbes with electricity: Direct electron transfer from electrodes to microbes. Environ Microbiol 3: 27-35.
- Silverman MP, Lundgren DG, (1959) Studies on the chemoautotrophic iron bacterium Ferrobacillus ferrooxidans. I. An improved medium and a harvesting procedure for securing high cell yields. J Bacteriol 77: 642-647.
- Karamanev DG, Nikolov LN, Mamatarkova V (2002) Rapid simultaneous quantitative determination of ferric and ferrous ions in drainage waters and similar solutions. Miner Eng 15: 341-346.
- Daoud J, Karamanev D (2006) Formation of jarosite during Fe2+ oxidation by Acidithiobacillus ferrooxidans. Miner Eng 19: 960-967.
- Karamanev DG, Nikolov LN, Mamatarkova V (2002) Rapid simultaneous quantitative determination of ferric and ferrous ions in drainage waters and similar solutions. Miner Eng 15: 341-346.
- Penev K, Karamanev D (2010) Batch kinetics of ferrous iron oxidation by Leptospirillum ferriphilum at moderate to high total iron concentration. Biochem Eng J 50: 54-62.
- Karamanev DG, Nikolov LN (1988) Influence of some physicochemical parameters on bacterial activity of biofilm: Ferrous iron oxidation by Thiobacillus ferrooxidans. Biotechnol Bioeng 31: 295-299.
- Tributsch H (1979) Solar bacterial biomass bypasses efficiency limits of photosynthesis. Nature 281: 555-556.
- Anupama, Ravindra P (2000) Value-added food: Single cell protein. Biotechnol Adv 18: 459-479.
- Russell AJH (1997) The emergence of life from iron monosulphide bubbles at a submarine hydrothermal redox and pH front. J Geol Soc London 154: 377-402.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi