Research Article, Int J Cardiovasc Res Vol: 10 Issue: 11
Valvuloarterial Impedance in Patients over 75 Years with Aortic Stenosis Undergoing SAVR and TAVI
Glib Yemets*, Telehuzova O. V., Maksymenko A. V., Mankovsky G. B., Marushko Y. Y., Dovgalyuk A. A., Sokol A. A., Yemets I.M.
Department of Cardiology, Ukrainian Children’s Cardiac Center, Kyiv, Ukraine
*Corresponding author:
Glib Yemets, Department of Cardiology, Ukrainian Children’s Cardiac Center, Kyiv, Ukraine
Tel: 687044822,
E-mail: oleksandra.telehuzova-2020@ppcr.org
Received date: October 06, 2021; Accepted date: October 20, 2021; Published date: October 27, 2021
Citation: Yemets G, Telehuzova OV, Maksymenko AV, Mankovsky GB, Marushko YY, et al. (2021) Valvuloarterial Impedance in Patients over 75 Years with Aortic Stenosis Undergoing SAVR and TAVI. Int J Cardiovasc Res 10:10.
Abstract
Aortic stenosis (AS) is a common cardiovascular disease with prevalence in an aging population. There is a growing body of literature that recognizes the variety of treatment techniques based on the morphological and clinical features. Currently, two major approaches in managing patients with AS are widely performed, including Surgical Aortic Valve Replacement (SAVR) and Transcatheter Aortic Valve Implantation (TAVI). Data from several studies suggest that AS is characterized with both genetic and nongenetic causes (the latter include risk factors, namely, age, diabetes, hypertension, obesity, dyslipidemia, smoking, and male gender). Our attention to the correlation between age and aetiology underlying AS. The prevalence of aortic stenosis is age dependent estimated at 1% of those aged over 65 years, 2.5% of those aged 75 years, and 8% of those aged 85 years. Central to the entire concept of AS is the role of key hemodynamic effects which are 1) increased left ventricular (LV) afterload, 2) reduced myocardial compliance, and 3) increased myocardial workload.
Keywords: Aortic stenosis; Surgical aortic valve replacement; Transcatheter aortic valve implantation
Introduction
Recently, a considerable literature has grown up around the theme of the valvuloarterial impedance (Zva). In the current article we are going to use the definition of an impedance as a measure of how much a structure resists motion when subjected to a given force. Detailed examination of Zva by Patrizio Lancellotti showed that it could provide an estimation of the global left ventricle (LV) hemodynamic load that results from the summation of the valvular and vascular loads, and the concept is very useful because it incorporates stenosis severity, volume flow rate, body size, and systemic vascular resistance. The indicator shows the interaction or counteraction on the one hand between the arterial and valvular factors, LV ejection and mechanical energy produced by the LV on the other hand. The lower rates of Zva the less load on the LV. Increased valvuloarterial impedance indicates the development of LV dysfunction, impaired longitudinal, radial and circular deformity, especially in patients with low ejection fraction. As previously described, increased Zva could be a strong, significant predictor and prognostic factor of mortality in the postoperative period. Moreover, Zva could be easily calculated with the Doppler echocardiography mentioning the systemic arterial compliance (SAC) calculated as the ratio of stroke volume index to arterial pulse pressure, the transvalvular mean gradient, and systolic arterial pressure. However, Zva difference in elderly patients after SAVR and TAVI has remained unclear and poor established. This study set out to investigate the usefulness of Zva as the remodelling and the hemodynamic load parameter, to evaluate the risk stratification significance of elderly patients with AS and to find out the difference between the Zva’s in SAVR and TAVI groups [1].
Materials and Methods
Study was performed according to the Good Clinical Practice (GCP) standards and World Medical Association (WMA) Helsinki Declaration. Prior to participation and data collection, all subjects received an explanation of the project and signed the inform consent. An observational and longitudinal study was carried out at Ukrainian Children’s Cardiac Center. Adult’s Clinic (UCCC) located in Kyiv. We include all patients over 75 with severe, symptomatic AoS, submitted to TAVI or SAVR, from January 2016 to February 2021. The sample was representative with respect to gender and age. In order to achieve valid results, we analysed anamnesis of patients, treatment protocols and direct postprocedural results of 93 aortic valve replacement operations. Preprocedural data included the clinical examination and complementary tests (resting electrocardiography, chest X-ray, transthoracic Doppler echocardiography with protocol for aortic complex measurements; computed tomography angiography of the heart and total aorta, and cardiac catheterization with coronary angiography). Transesophageal echocardiography was used to guide the procedure and detect possible complications due to prosthesis implantation. In some cases, additional tests were performed to rule out or confirm comorbidity (MRI of the abdominal cavity, etc.). Patients, undergoing TAVI were additionally examined by MRI with contrast to assess femoral and superficial arteries. Among the analyzed cases, 83 procedures (89.24%) were SAVR and 10 (10, 75%)-TAVI [2].
Eligibility criteria
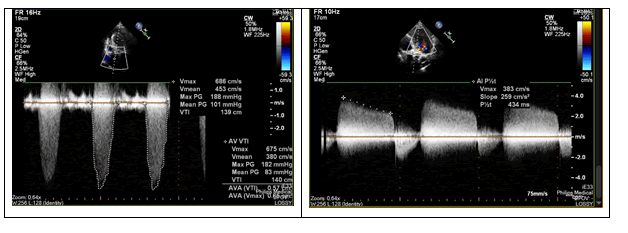
Inclusion criteria for transcatheter aortic valve implantation were (1) severe aortic stenosis, (2) valve’s calcification or fibrosis and (3) age >75 years. Patients were identified as having severe AS if any of the following echocardiographic criteria were achieved: aortic valve (AV) mean gradient ≥ 40 mmHg; AV peak velocity ≥ 4.0 m per second (m/s); AV area (AVA) ≤ 1.0 cm2; or dimensionless performance index (DPI) ≤ 0.25, as per the criteria outlined in the joint statement from the European Association of Cardiovascular Imaging and the American Society of Echocardiography.
Group characteristics
A number of patients with isolated aortic stenosis in TAVI group was 4 (40, 0%). Just over half the sample (59, 4%) was diagnosed with coronary heart disease, which is considered to be an aggravating factor and adversely affects the life expectancy prognosis. In SAVR group, 72 patients (86, 7%) were diagnosed with coronary heart disease and 11 patients (15, 27%) with type II diabetes mellitus. The chronic obstructive pulmonary disease was diagnosed in 4 patients (5%). The TAVI procedures were performed in a hybrid operating room (HOR) under general anaesthesia. Two access routes for TAVI implantation were performed: transapical and transfemoral (7 procedures and 3 procedures, respectively). In all transapical aortic valve implantation cases we used the self-expandable novel XPand prosthesis, created in collaboration between German, French and Ukrainian scientists. Device consists of two parts: (1) tubular slotted stainless-steel stent frame with unidirectional trileaflet bovine pericardial tissue and a (2) fabric skirt. Trileaflet bovine pericardial tissue sutured into a wire frame of nitinol, a nickel (55%)-titanium (45%) alloy that has temperature-associated shape memory features.
Currently, XPand device could be used only for the transapical access. It has been second standard approach after transfemoral, with stable rates although the advancement of alternative options. Transapical access, aside from the ability to deliver a large device in a patient with inadequate iliofemoral vessels, may offer additional advantages in complex patients. Undoubtable advantage of this approach is the shortest and direct access to the aortic valve, which makes the procedure of prosthesis delivery and positioning more controllable. The risk of surgery using EuroSCORE II and STS was calculated for each patient and showed higher values in TAVI group. The statistical significance was not estimated. However, each risk stratification model suffers from limitations and still not perfect [3].
| Overall (N = 93) | TAVI (N = 10) | p-value |
|---|---|---|
| Age, median (IQR) | 86 ± 10,3 | <0.001 |
| Female Gender, n (%) | 9(90%) | 0.42 |
| BMI, median (IQR) | 27.4 (24.6, 30.6) | 0.15 |
| NT-proBNP (ng/mL), median (IQR) | 1421 | 0.099 |
| EGFR (mL/min/1.73 m2), median (IQR) | 72 | <0.001 |
| Prior HF, n (%) | 2 (20 %) | |
| Prior IHD, n (%) | 6 (60%) | 11 (48%) |
| Prior COPD, n (%) | 0 (0%) | 2 (9%) |
| Prior PVD, n (%) | 3 (30%) | 2 (9%) |
| Mitral Valve Disease â?? Mod/Sev, n (%) | 3 (30%) | 0 (0%) |
| Prior Diabetes, n (%) | 2 (20%) | 6 (26%) |
| Prior AF/Flutter, n (%) | 0 (0%) | 6 (26%) |
| Prior CABG, n (%) | 0 (0%) | 4 (17%) |
| HFA Score, median (IQR) | 1 (1, 3) | 1 (1, 3) |
| STS Score (%), median (IQR) | 7,85 | |
| EF (%), median (IQR) | 55 | |
| AV Mean Gradient (mmHg), median (IQR) | 60,25 | 47.2 (40.7, 57.2) |
| AV Area (cm2), median (IQR) | 0.5 | |
| AV Peak Velocity (m/s), median (IQR) | 4.4 | 4.50 (4.10, 4.90) |
| E/e’, median (IQR) | 15.2 | 14 (13.0, 18.5) |
| Left Atrial Area (cm2), median (IQR) | 24.3 | 25.2 (21.0, 28.3) |
| Systolic BP (mmHg), median (IQR) | 152 (136, 166) | 150 (143, 160) |
| Diastolic BP (mmHg), median (IQR) | 56 (50, 87) | 59 (50, 86) |
| MAP (mmHg), median (IQR) | 104 (95, 112) | 104 (100, 112) |
| Pulse Pressure (mmHg), median (IQR) | 70 (60, 83) | 66 (56, 75) |
| Heart Rate (bpm), median (IQR) | 68 (60, 80) | 66 (59, 78) |
| Central Arterial Pressure (mmHg), median (IQR) | 142 (127, 157) | 139 (133, 152) |
| Central Pulse Pressure (mmHg), median (IQR) | 59 (48, 72) | 55 (46, 61) |
| Zva, median (IQR) | 4.3 (3.8, 5.4) | 3.9 (3.6, 4.3) |
| NYHA Class, median (IQR) | 3 (2, 3) | 2 (2, 3) |
Table 1: Group baseline characteristics.
Statistical analysis
Continuous variables were described as mean and standard deviation, and categorical variables as absolute number and percentage. Statistical significance was analysed using analysis of variance and t-tests as appropriate. Student's t-test (or the Mann-Whitney test) was used to compare continuous variables for independent samples, and the chi-squared test or Fisher's exact test was used for categorical variables, as appropriate. To assessif the means of two groups are significantly different the ANOVA was used. All analyses were performed using Stata ICand Excel software version. Statistical significance level was set at 5%.
Outcomes
It should be noted that all patients had a good postprocedural outcomes, confirmed by the obtained data. No critical complications requiring additional treatment (eg, ECMO and dialysis) have been identified. If we now turn to the average length of stay (ALOS from admission to discharge, transfer) ranged as 21.35 ± 8.1 days. The most striking result to emerge from the data is that average ICU length of stay (LOS), time of the artificial ventilation (ventilation) and duration of inotropic support were significantly lower in TAVI group. Patients undergoing TAVI were older, with worse renal function, lower aortic valve areas and higher STS scores.
After TAVI, there was a significant reduction in systolic and diastolic blood pressure (178 ± 25 versus 134 ± 17 mm Hg, P<0.001 and 45 ± 16 versus 63 ± 15 mm Hg, P=0.001, respectively), albeit that at follow-up blood pressures approached baseline values (135 ± 15 and 60 ± 20 mm Hg, respectively). The pulse pressure, however, was lower immediately after TAVI and remained so at 1-year. Overall, there was a significant decrease in systemic arterial compliance.
We found that pre-procedural LV EF (r = 0.284, p < 0.001) and ZVA (r = 0.523, p < 0.001) were independent factors associated with the LV mass regression after TAVR (1 – year follow up), no significant data regarding the SAVR group was obtained. The difference in echocardiographic characteristics after TAVR and SAVR showed a positive correlation with ZVA (r=0.491, p <0.001). According to the obtained data, an elevated Zva was present in all patients with severe AoS undergoing both TAVR and SAVR. Despite significant improvements in Zva following TAVR, 54% of the patients continued to have an elevated Zva after TAVR in an early post-operative period, although Zva in SAVR group was elevated both in early post-operative period and in 1-year follow-up. Effective AVA increased from 0.5 ± 0.2 to 1.9 ± 0.4 cm2 after TAVR (p < 0.001) and 0.65 ± 0.5 to 2.0 ± 0.75 in SAVR group.
Discussion
Systemic arterial compliance was calculated as the ratio of stroke volume index to arterial pulse pressure in 93 patients with at severe AS. As a measure of global afterload, we calculated the valvulo-arterial impedance, which theoretically accounts for the effects of both AS and SAC. This research has thrown up many questions in need of further investigation. Numerous clinical challenges including paravalvular insufficiency, vascular complications, coronary artery occlusion, valve ring rupture, ventricular perforation, stroke etc. still remain unsolved in both SAVR and TAVI groups. A reasonable approach to tackle this issue could be to manage this group of patients by the Heart Team [4].
A significant decrease in Zva was obtained in TAVI group immediately after procedure and in follow-up periods, which could testify for an earlier adaptation and decreased global afterload in this group comparing with the SAVR [5]. Several questions still remain to be answered, although the findings of this study have a number of practical implications. We found numerous advantages for TAVI in elderly patients:
- More rapid valvulo arterial impedance
- Minimize blood loss.
- Reduction of surgery duration.
- Artificial blood circulation is not required.
- Possibilities to perform TAVI in patients of high surgical risk with severe comorbidities and patients required re-intervention.
However, with a limited sample size and potential biases, caution must be applied, as the findings might not be extrapolated and generalized to all the study population, future studies on the current topic are therefore recommended.
Conclusion
No significant difference in mortality rate between TAVI and SAVR groups was found. Elevated Zva was present in all patients with severe AoS undergoing both TAVR and SAVR. Pre-procedural LV EF and ZVA were independent factors associated with the LV mass regression after TAVR. The difference in echocardiographic characteristics after TAVR and SAVR showed a positive correlation with ZVA. Despite significant improvements in Zva following TAVR, 54% of the patients continued to have an elevated Zva after TAVR in an early post-operative period.
References
- Mallis P, Michalopoulos E, Dimitriou C, Kostomitsopoulos N, Stavropoulos-Giokas C (2017) Histological and biomechanical characterization of decellularized porcine pericardium as a potential scaffold for tissue engineering applications. Biomed Mater Eng 28: 477-488.
- Falk V, Baumgartner H, Bax JJ, De Bonis M, Hamm C, et al. (2017) 2017 ESC/EACTS guidelines for the management of valvular heart disease. Euro J Cardio Thorac Surg 52: 616-664.
- Khan AA, Murtaza G, Khalid MF, Khattak F (2019) Risk stratification for transcatheter aortic valve replacement. Cardiol Res 10: 323.
- Reardon MJ, Van Mieghem NM, Popma JJ, Kleiman NS, Sondergaard L, et al. (2017) Surgical or transcatheter aortic-valve replacement in intermediate-risk patients. N Engl J Med 376: 1321-1331.
- Ferrari E, Eeckhout E, Keller S, Muller O, Tozzi P, et al. (2017) Transfemoral versus transapical approach for transcatheter aortic valve implantation: Hospital outcome and risk factor analysis. J Cardiothor Surg 12: 1-11.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi