Case Report, J Vet Sci Med Diagn Vol: 7 Issue: 2
A Case of Poorly Differentiated Cutaneous Canine Mast Cell Tumor with an Atypical Dermatologic Presentation
Vichi G1*, Binanti D2, Ciaccini R3 and Costantini L4
1CIMIE lab, Via C.Morelli 23, 62100, Macerata (MC), Italy
2AbLab, Via privata Massa Neri 13, 19038 Sarzana (SP), Italy
3Clinica Veterinaria S.Silvestro, Località Montecchio, 52043, Castiglion Fiorentino (AR), Italy
4Clinica Veterinaria S.Antonio, Contrada Fiastra 69/F, 62020, Colmurano (MC), Italy
*Corresponding Author : Gaia Vichi
DVM, CIMIE lab, Via C.Morelli 23, 62100, Macerata (MC), Italy
Tel: +39 071 7930720
E-mail: gaiavichi@gmail.com
Received: April 09, 2018 Accepted: May 09, 2018 Published: May 14, 2018
Citation: Vichi G, Binanti D, Ciaccini R, Costantini L (2018) A Case of Poorly Differentiated Cutaneous Canine Mast Cell Tumor with an Atypical Dermatologic Presentation. J Vet Sci Med Diagn 7:2. doi: 10.4172/2325-9590.1000255
Abstract
Canine cutaneous mast cell tumors occur as nodular dermal lesions. A condition of mastocytosis is also described in dogs. The purpose of this study is to report the clinical and histopathological findings in a dog with high grade non-nodular cutaneous mast cell tumor. An 11-year-old intact male German Shepherd dog presented a severe erythematous dermatitis and erythematous plaques on the left thoracic wall, rapidly increasing in thickness, ulcerating, and spreading to the head. The fine needle cytology was indicative of a round cell neoplasm. The histological appearance of the lesions was suggestive of a high grade cutaneous mast cell tumor. Neoplastic cells were immunoistochemically Tryptase positive and c-kit positive (focal cytoplasmic staining), with a high Ki67 index. On the basis of this report high grade canine cutaneous mast cell tumors may show an atypical non-nodular presentation and should be included in the differential diagnosis for different lesions grossly appearing as erythematous dermatitis/plaques.
Keywords: Dog; Erythematous plaques; Haired skin; Mast cell tumor
Abbreviations
MCT: Mast Cell Tumor; FNA: Fine Needle Aspiration; HPF: High Power Field
Introduction
Canine mast cell tumors (MCTs) generally occur as nodular dermal proliferations of mast cells (in some cases also with the infiltration of the subctutis). A different condition of diffuse dermal infiltrate of well-differentiated mast cells (mastocytosis) is also described in dogs [1]. To the authors’ knowledge, high grade canine cutaneous MCTs with an atypical non-nodular presentation have not been reported. The purpose of this study is to report the dermatologic and histopathologic findings in a dog with high grade non-nodular cutaneous MCT (presenting as erythematous dermatitis and multifocal erythematous plaques).
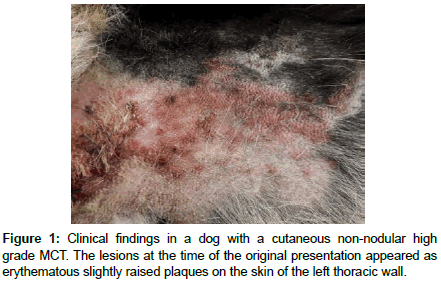
An 11-year-old intact male German Shepherd dog (recently treated with Cyclosporine because of perianal fistulas) was presented to a private clinic for evaluation of intensely pruritic lesions on the skin of the left thoracic wall. Grossly a severe locally extensive erythematous dermatitis with hypotrichosis and then also erythematous cutaneous plaques (developed in a few days) were observed. These lesions were initially only slightly raised on the cutaneous surface (1-2 mm of thickness) (Figure 1). In the subsequent two weeks the lesions were ulcerated and increased in thickness (up to 3 cm of thickness) and spread to the head (skin of the occipital region), with a similar appearance. Differential diagnoses, on the basis of the gross appearance of these lesions and their distribution were toxic epidermal necrolysis, erythema multiforme, or epitheliotropic cutaneous lymphoma (mycosis fungoides/Sézary synfrome). Fine needle aspiration (FNA) was performed and multiple cutaneous punch biopsies were taken and collected in neutral buffered formalin (10%) for histopathologic, and further immunohistochemical evaluation.
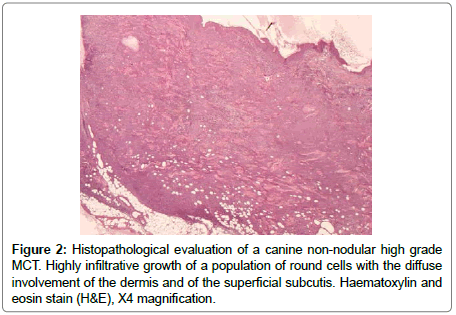
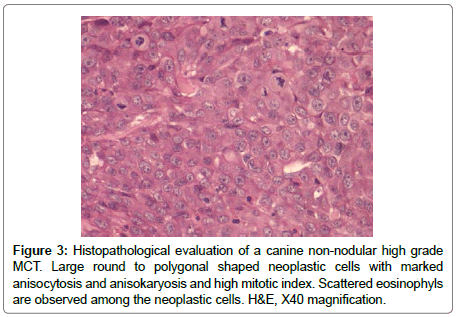
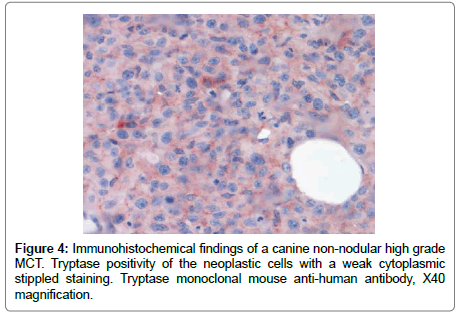
The FNA samples (Diff-Quick stain) showed, on a hematic background, the presence of an abundant cellular population composed of large round cells singularly distributed or organized in large sheets. These neoplastic cells showed an abundant variably bluish cytoplasm, in some cases with a finely granular appearance, and large, central to paracentral, round-shaped nuclei, with coarse chromatin pattern and prominent nucleoli (up to three for each nucleus). There were also some binucleated cells and scattered atypical mitoses. In this context the presence of sparse well differentiated mastocytes and some eosinophils were also noted. The histology (from paraffin embedded tissue routinely stained with haematoxylin and eosin) revealed the presence of a neoplastic highly infiltrative growth of a population of round cells diffusely involving the dermis and the superficial subcutis (Figure 2). The overlying epidermis was multifocally ulcerated. The neoplastic cells were organized in large sheets, separed by thin collagen bundles. The neoplastic cells were large (average diameter: 70 μm), round to polygonal in shape, with abundant eosinophilic, finely granular cytoplasm, with well distinct borders (Figure 3). Toluidine Blue stain revealed lightly metachromatic cytoplasmic granules in almost the 10% of these neoplastic cells. The nuclei appeared large (modelately dysmetric), centrally to paracentrally located, round to polygonal, with reticulated chromatin and prominent nucleoli (up to 3 for each nucleus). As observed with cytologic examination there were also some binucleated cells. Marked anisocytosis and anisokaryosis were noted. The mitotic index was high (45 mitoses/10 HPF) with 0 to 5, prevalently atypical mitoses for each HPF. In this context there was also the presence of sparse well differentiated mastocytes, multifocal lymphocytic aggregates and many eosinophyls, also with focal areas of degranulation and formation of flame figures. Finally also few neoplastic emboli were noted in small dermal lymphatic vessels. Immunohistochemistry was performed with multiple antibodies for a definitive diagnosis (Supplementary Table 1). CD18, CD3, CD79A were negative. The neoplastic cells were Tryptase positive, prevalently with a weak cytoplasmic stippled staining (Tryptasestaining pattern III) (Figure 4) [2]. Finally a positivity for CD117 (c-kit) was observed in scattered neoplastic cells with an intense focal (prevalently perinuclear) or stippled cytoplasmic staining (pattern II) [3]. The Ki67 index was also evaluated and resulted greater than the cut-off 10.6% [4]. These findings permitted a diagnosis of a high grade (and poorly differentiated) cutaneous MCT [3].
The dog was also evaluated with a radiographic and ecographic examination and FNA to exclude invasion of local lymh nodes and systemic involvement. The FNA from the axillary lymph node revealed its invasion by numerous metastatic neoplastic cells, however RX and ecographic images of internal organs were normal and FNA from spleen and liver were free of neoplastic elements. The therapeutic approach comprised a combination of Prednisolone (2 mg/kg once a day), Vinblastine (1.6 mg/m2 once a week) and then Toceranib (2.2 mg/Kg every other day), with concomitant administration of gastroprotectors (Omeprazole and then Sucralfate) and Maropitant. A progressive reduction of ulceration, bleeding and swelling of the cutaneous lesions was observed. Clinicopathological findings during therapy revealed a moderate progressive non-regenerative anemia and neutrophilic leucocytosis with left shift (23.7*109/L WBC, 16.3*109/L neutrophils and 3,49*109/L nonsegmented neutrophils) After 5 weeks the subject was euthanasized for a concumitant gastric dilatation-volvulus.
Canine cutaneous MCTs generally are seen as single or multiple nodular lesions. A different condition of diffuse dermal infiltrate of well-differentiated mast cells (mastocytosis) is also described in dogs (similar to urticaria pigmentosa in humans) [1]. This case presents its clinical importance as an atypical non-nodular presentation of a cutaneous high grade MCT, diagnosed on the basis of cytologic, histologic and immunohistochemical findings. The neoplastic cells were Tryptase positive, with a weak cytoplasmic stippled staining (Tryptase-staining pattern III). This finding agrees with the diagnosis of MCT: the identification of Tryptase using immunohistochemistry has shown excellent specificity and sensitivity to mast cells [5,6]. However the neoplasm was very poorly differentiated, showed an infiltrative growth and it spread also to the axillary lymph node and to a cutaneous distant site (skin of the occipital region). On the basis of these findings, suggesting a highly aggressive behavior, we expected a c-kit cytoplasmic diffuse staining pattern (pattern III) since it is proven that a more aggressive biologic behavior of canine cutaneous MCTs is associated with the increase in cytoplasmic staining for c-kit [2]. Interestingly, in this case, the neoplastic cells showed an intense focal (prevalently perinuclear) or stippled cytoplasmic staining (pattern II). The initial intensely pruritic lesions on the skin of the left thoracic wall appeared grossly as a severe locally extensive erythematous dermatitis with hypotrichosis. Then also erythematous cuteneous plaques were observed. These lesions were initially only slightly raised on the cutaneous surface (1-2 mm of thickness). In the subsequent two weeks the lesions were ulcerated and increased in thickness (up to 3 cm of thickness) and spread to the head. Lesions grossly similar to the initial changes seen in this case are clinically most compatible with other dermatologic conditions such as toxic epidermal necrolysis, erythema multiforme, or with other cutaneous neoplasms, such as epitheliotropic cutaneous lymphoma (mycosis fungoides/Sézary synfrome). To the authors’ knowledge, high grade canine cutaneous MCTs with an atypical non-nodular presentation have not been reported, but on the basis of this report, high grade canine cutaneous MCTs should be included in the differential diagnosis for non-nodular dermatologyc lesions grossly appearing as erythematous dermatitis/plaques.
Acknowledgements
The authors would like to thank Barbara Brunetti (Associate Professor at Department of Veterinary Medical Sciences, University of Bologna) for contributing to completition of the immunohistochemical panel, and Stefania Vichi for the linguistic revision of the manuscript.
References
- Davis BJ, Page R, Sannes PL, Meuten DJ (1992) Cutaneous mastocytosis in a dog. Vet Pathol 29: 363-365.
- Kiupel M, Webster JD, Kaneene JB, Miller R, Yuzbasiyan-Gurkan V (2004) The use of KIT and tryptase expression patterns as prognostic tools for canine cutaneous mast cell tumors. Vet Pathol 41: 371-377.
- Kiupel M, Webster JD, Bailey KL, Best S, DeLay J, et al. (2011) Proposal of a 2-Tier histologic grading system for canine cutaneous mast cell tumors to more accurately predict biological behavior. Vet Pathol 48: 147-155.
- Vascellari M, Giantin M, Capello K, Carminato A, Morello EM, et al. (2013) Expression of Ki67, BCL-2, and COX-2 in canine cutaneous mast cell tumors-association with grading and prognosis. Vet Pathol 50: 110-121.
- Walls AF, Jones DB, Williams JH, Church MK, Holgate ST (1990) Immunohistochemical identification of mast cells in formaldehyde-fixed tissue using monoclonal antibodies specific for tryptase. J Pathol 162: 119-126.
- Yang F, Tran TA, Carlson JA, Hsi ED, Ross CW, et al. (2000) Paraffin section immunophenotype of cutaneous and extracutaneous mast cell disease: comparison to other hematopoietic neoplasms. Am J Surg Pathol 24: 703-709.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi