Research Article, Int J Ophthalmic Vol: 9 Issue: 2
A fatal case of acute bilateral orbital apex syndrome with mucormycosis
Hiromitsu Kunimi1, Yusaku Katada1*, Masaki Fukui1,2, Shinji Ideta3, Ohde Hisao1,4, Kazuo Tsubota1
1Department of Ophthalmology, Keio University School of Medicine, Japan
2Department of Ophthalmology, Tokyo Dental College Ichikawa General Hospital, Japan
3Department of Ophthalmology, Ideta Eye Hospital, Japan
4Department of Ophthalmology, Makuhari Ode Eye Clinic, Japan
*Corresponding Author: Yusaku Katada
Department of Ophthalmology, Keio University School of Medicine, Tokyo, Japan
E-mail: yusakukatada@z2.keio
Received: May 25, 2020 Accepted: June 20, 2020 Published: June 26, 2020
Citation: Kunimi H, Katada Y, Fukui M, Ideta S, Hisao O, et al. (2020) A fatal case of acute bilateral orbital apex syndrome with mucormycosis. Int J Ophthalmic Pathol 9:2. doi: 10.37532/iopj.2020.9(2).237
Abstract
We report a fatal case of mucormycosis that started with an acute bilateral orbital apex syndrome. A 44-year-old man with severe alcoholic hepatitis visited our hospital with a chief complaint of sudden visual loss in his left eye. The cranial computed tomography scan and magnetic resonance imaging revealed a lesion in his left orbital apex. After being diagnosed with orbital apex syndrome due to infection, he was prescribed antibiotic and antifungal therapy. Despite the treatment, he showed no improvement, and he lost vision in the contralateral eye. He also developed cranial nerve symptoms. His general condition worsened rapidly; within a week, he died of multiple organ failure. Pathologic examination confirmed the diagnosis of invasive mucormycosis.
Keywords: orbital apex syndrome, mucormycosis
Introduction
Orbital Apex Syndrome (OAS) is a syndrome with a complex of symptoms, including visual loss, blepharoptosis, ophthalmoplegia, hypoesthesia of the face and cornea, and Horner’s syndrome. These symptoms occur because the lesion of the orbital apex and cavernous sinus includes the superior orbital fissure and optic canal [1]. That means the optic nerve (II), oculomotor nerve (III), trochlear nerve (IV), abducens nerve (VI), and ophthalmic branches of the trigeminal nerve (V1, V2) are damaged [2]. Primary causes of OAS are inflammation, tumour, infection, trauma or vascular disease1.
One of the causes of OAS is mucormycosis. It is not a common disease and should be diagnosed and treated as early as possible, due to its fatal outcome if any delay. Here, we report a fatal case of mucormycosis which started with an acute bilateral orbital apex syndrome.
Case Report
A 44-year-old Caucasian man who had severe alcoholic hepatopathy developed a sudden visual loss of the left eye and he totally lost his vision in nine hours. He was conscious and afebrile with normal vital signs. His visual acuity was 18/20 in the right eye and no light perception in the left eye. The intraocular pressure of his eyes was 10 and 18 mmHg respectively. His pupils were isochoric, but the direct light reflex was lost in the left eye and the consensual light reflex was lost in both eyes. The movement of the left eye was limited in every direction with blepharoptosis. Ophthalmic examination also revealed yellow conjunctiva and superficial punctate keratitis in the left eye. There were no significant funduscopic lesions in both eyes. The facial perception on the left side was lost. His blood test showed a mild increase in C-reactive protein (CRP) and white blood cells (Table 1). However, the CRP stayed almost equivalent to the previous value, and the increase in white blood cells might be the side effect of oral prednisolone (20mg/day) for hepatopathy. The computed tomography and the Magnetic Resonance Imaging (MRI) revealed sinusitis, but no sign of orbital invasion like bone destruction (Figure 1A,1B). The initial clinical diagnosis was OAS due to some inflammation, because there are less findings suggestive of infection; normothermia, negative beta-D-glucan and aspergillus antibody test, and mild sinusitis without bone destruction. Therefore, we first started steroid pulse therapy (methylprednisolone 1000 mg/day) as diagnostic treatment.
| Test | Value | Reference |
|---|---|---|
| Leukocyte (/µL) | 16100 | 3300-8600 |
| Hemoglobin (g/dL) | 8.2 | 13.7-16.8 |
| Platelet count (/µL) | 14.3×104 | 15.8-34.8×104 |
| C-reactive protein (mg/dL) | 2.25 | <0.14 |
| Blood urea nitrogen (mg/dL) | 15.5 | 20-8 |
| Creatinine (mg/dL) | 0.73 | 0.65-1.07 |
| Sodium (mEq/L) | 122 | 139-145 |
| Potassium (mEq/L) | 4.2 | 3.6-4.8 |
| Chlorine (mEq/L) | 92 | 101-108 |
| Calcium (mg/dL) | 6.9 | 8.8-10.1 |
| Aspartate aminotransferase (lU/L) | 77 | 13-30 |
| Alanine aminotransferase (lU/L) | 63 | 10-42 |
| y3Glutamyltransferase (lU/L) | 97 | 13-64 |
| Activated Partial thromboplastin time (sec) | 36.3 | 23-40 |
| International normalized ratio of prothrombin time | 1.95 | 0.80-1.20 |
| Fibrinogen (mg/dL) | 232 | 150-400 |
| Angiotensin-converting enzyme1 (lU/L) | 27.4 | 8.3-21.4 |
| Proteinase 3 anti-neutrophil cytoplasmic antibody (lU/mL) | <1.0 | <2.0 |
| Myeloperoxidase anti-neutrophil cytoplasmic antibody (lU/L) | <1.0 | <3.5 |
| ß-D-glucan (pg/mL) | <2.7 | <20.0 |
| Aspergillus antigen | Negative | |
| Hepatitis B surface(HBs)antigen | Negative | |
| Hepatitis B surface(HBs)antibody | Negative | |
| Hepatitis C virus(HCV)antibody | Negative | |
| Human immunodeficiency virus (HIV)-1,2 antibody | Negative |
Table 1: Laboratory data on the first day.
In the next morning, his symptoms did not become better and his general condition grew worse. With an increased possibility of infectious OAS, the steroid therapy was stopped, and his treatment was changed to administrations of ceftriaxone sodium hydrate 2g/day and voriconazole 150mg/day for the possibility of fungous infection.
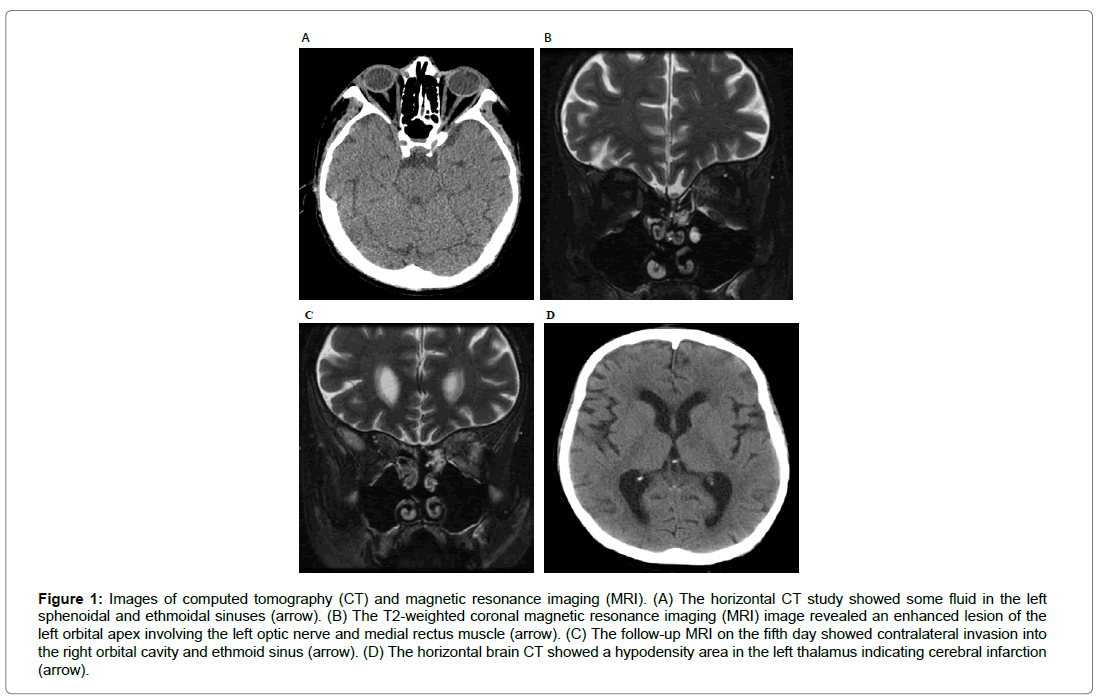
On the fifth day, his condition went worse and the symptoms became bilateral; he lost the light perception and had blepharoptosis, no extraocular movement and proptosis in both eyes. The follow-up MRI showed contralateral orbital invasion (Figure 1C). Funduscopic examination showed retinal ischemia and a pale optic disc in the left eye. With high suspicion of mucormycosis, the antifungal therapy was changed from voriconazole to amphotericin B (AMB), 1.0 mg/kg/day on the same day. On the sixth day, impaired consciousness and right partial hemiplegia occurred. The brain CT revealed the infarction of the left thalamus (Figure 1D). His general condition got worse, and he died with the diagnosis of multiple organ failure on the seventh day.
Figure 1: Images of computed tomography (CT) and magnetic resonance imaging (MRI). (A) The horizontal CT study showed some fluid in the left sphenoidal and ethmoidal sinuses (arrow). (B) The T2-weighted coronal magnetic resonance imaging (MRI) image revealed an enhanced lesion of the left orbital apex involving the left optic nerve and medial rectus muscle (arrow). (C) The follow-up MRI on the fifth day showed contralateral invasion into the right orbital cavity and ethmoid sinus (arrow). (D) The horizontal brain CT showed a hypodensity area in the left thalamus indicating cerebral infarction (arrow).
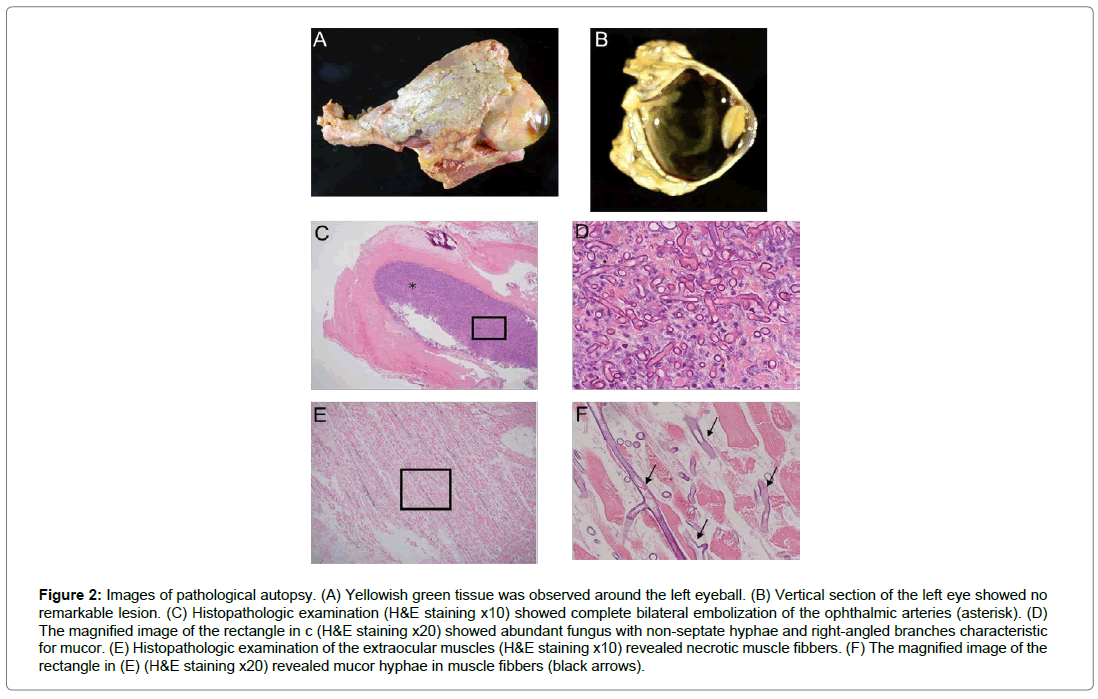
In the result of pathological autopsy, there was a yellowish green creamy tissue suggestive of mucor infection around the eyeball (Figure 2A, 2B). Complete embolization of mucor hyphae was shown in the bilateral ophthalmic arteries and internal carotid arteries (Figure 2C, 2D). Mucor hyphae were also detected around the extraocular muscles and optic nerves (Figure 2E, 2F).
Figure 2: Images of pathological autopsy. (A) Yellowish green tissue was observed around the left eyeball. (B) Vertical section of the left eye showed no remarkable lesion. (C) Histopathologic examination (H&E staining x10) showed complete bilateral embolization of the ophthalmic arteries (asterisk). (D) The magnified image of the rectangle in c (H&E staining x20) showed abundant fungus with non-septate hyphae and right-angled branches characteristic for mucor. (E) Histopathologic examination of the extraocular muscles (H&E staining x10) revealed necrotic muscle fibbers. (F) The magnified image of the rectangle in (E) (H&E staining x20) revealed mucor hyphae in muscle fibbers (black arrows).
Discussion
Mucormycosis is an especially fatal cause of, and a risk factor for poor prognosis in infectious OAS 2. In general, the risk factors of mucormycosis are immunocompromised states such as diabetes (especially diabetic ketoacidosis), hematological malignancy or steroid administration [2,3]. OAS by mucormycosis begins with nasal, paranasal sinuses or palate infection by inhalation of the pathogen, and then it spreads into the orbit [4]. Mucor infiltrates the body angiogenically because they have strong affinity for vessels [5]. Then, mucor proliferates and forms multiple thromboembolisms in the internal carotid arteries and ophthalmic arteries [2]. Primary symptoms of mucormycosis are headache, fever and ophthalmalgia, and then they sometimes progress to blepharoptosis, extraocular palsy, low vision and trigeminal nerve palsy in a unilateral eye [6,7]. Analysis of biological specimens from clinically involved sites is mandatory for diagnosis [8]. Standard treatment is excision of the original lesion as wide as possible and administration of AMB [9]. If it progresses intracranially into the brain, patients will fall into a critical condition [10]. In the case of sudden OAS in immunocompromised patients, it is necessary to consider the possibility of mucormycosis. However, mucormycosis invades hematogenously and no circulation antigen detection test such as Beta-D-glucan is available [11]. Furthermore, the fact that the surgical removal or biopsy of the lesion is essential for the definitive diagnosis of mucormycosis makes prompt treatment difficult [12].
Due to these difficulties, and a lack of extraocular symptoms, we could not get to the diagnosis of mucormycosis primarily in this case. From the rapid clinical progression, we clinically diagnosed mucormycosis and started AMB immediately. However, even the short delay of AMB treatment might have led to the fatal outcome [13].
The pathologic examination confirmed that the systemic embolism such as the ischemic retina, the infarction of the left thalamus, and the multiple organ failure was caused by embolization of mucor hyphae.
Misdiagnosing an orbital apex lesion as inflammation and treating patients primarily with corticosteroids can be life-threatening. If mucormycosis is suspected in patients having basal disease, AMB has to be administered as fast as possible [13]. However, this therapy has critical side effects such as renal failure, and mucormycosis has poor visual and vital prognosis [2]. It is not easy to decide whether to start the AMB treatment.
Nevertheless OAS, especially by the invasion from mucormycosis, is an emergent and fatal syndrome, not widely recognized. We conclude that due to the fatal poor prognosis, making a prompt diagnosis and administering the appropriate treatment must be done without delay if OAS from mucormycosis is suspected.
Acknowledgement
This work was supported by JSPS KAKENHI Grant Number JP19K24053
Conflict of Interest
The authors report no conflict of interest.
References
- Yeh S, Foroozan R (2004) Orbital apex syndrome. Curr Opin Ophtalmol 15: 490-498
- Gamaletsou MN, Sipsas NV, Roilides E, Walsh TJ (2012) Rhino-Orbital-Cerebral mucormycosis. Curr Infect Dis Rep 14: 423–434.
- Hisao O, Kamoshita I, (1992) A case of rhinocerebral mucormycosis with ocular symptoms. Folia Ophthalmol Jpn43: 507–511.
- Abedi E, Sismanis A, Choi K, Pastore P (1984) Twenty-five years’ experience treating cerebro-rhino-orbital mucormycosis. Laryngoscope 94:1060–1062.
- Spellberg B, JohnEdwards Jr, Ibrahi A (2005) Novel perspectives on mucormycosis: pathophysiology, presentation, and management. Clin Microbiol Rev 18 : 556–569.
- Yasuko K, Hiroyuki M, Mamiko A, Nobuko I, (2002) Bilateral central retinal artery occlusion in a case of rhinocerebral mucormycosis. Japanese J Clin Ophthalmol 96: 981–983.
- Masakazu H, Akira K, Takashi M, Hiroshi T, Hideyuki K et al. (1997) A case of rhinocerebral mucormycosis in a normal host. Pract Otorhinolaryngol (Basel) 90: 1343–1350.
- Toshiko H, Katsuhiko N (1999) A case of sudden death preceded by unilateral exophthalmos. Japanese Rev Clin Ophthalmol 93: 408–410.
- Cornely OA, Arikan Akdagli S, Dannaoui E, Groll AH, Lagrou K, Chakrabarti A et al. (2014) ESCMID and ECMM joint clinical guidelines for the diagnosis and management of mucormycosis 2013. Clin Microbiol Infect 20: 5–26.
- Greenberg RN, Scott LJ, Vaughn HH, Ribes JA (2004) Zygomycosis (mucormycosis): emerging clinical importance and new treatments. Curr Opin Infect Dis 17: 517–525.
- Lanternier F, E Dannaoui, G Morizot, C Elie, D Garcia-Hermoso, et al. (2012) A Global Analysis of Mucormycosis in France: The RetroZygo Study (2005–2007). Clin Infect Dis 54: 35–43.
- Masahiko S, Yasuyuki I, Yasuhiko I, (2002) An immunocompromised patient of rhino cerebral mucor mycosis presenting orbital apex syndrome. Neurol Med 56: 67–71.
- Chamilos G, Lewis RE, Kontoyiannis DP (2008) Delaying Amphotericin B – Based Frontline Therapy Significantly Increases Mortality among Patients with Hematologic Malignancy Who Have Zygomycosis. Clin Infect Dis 47: 503–509.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi