Case Report, J Pulm Med Vol: 2 Issue: 1
A Rare Case of Sporadic Lymphangioleiomyomatosis with Manifestation of Recurrent Pneumothorax in a Premenopausal Female
Stacey Ho, Ja cqueline Marie Gowan, Tania Zavalza Jimenez* and Fred Javdan
Dignity Health, California Hospital Medical Center, Ross University School of Medicine, Los Angeles California, USA
*Corresponding Author : Tania Zavalza Jimenez
Dignity Health, California Hospital Medical Center, Ross University School of Medicine, Los Angeles California, USA
Tel: 2096485826
E-mail: tzavalza90@yahoo.com
Received: April 04, 2018 Accepted: May 16, 2018 Published: May 21, 2018
Citation: Jimenez TZ, Ho S, Gowan JM, Javdan F (2018) A Rare Case of Sporadic Lymphangioleiomyomatosis with Manifestation of Recurrent Pneumothorax in a Premenopausal Female. J Pulm Med 2:1.
Abstract
Sporadic lymphangioleiomyomatosis (LAM) is a rare multisystem disorder affecting approximately 3 to 8 per million women. Our patient, a 42-year-old nulligravida Hispanic female with recurrent pneumothoraces presented to the Emergency Department (ED) with a chief complaint of shortness of breath and exertional chest tightness that progressively worsened over four days. Upon arrival to the ED she was tachypneic and hypoxic, with chest x-ray revealing large bilateral pneumothoraces and chest computed tomography (CT) from a previous encounter showed dozens of air-filled cavities in both lungs. Already possessing the diagnosis of LAM upon presentation and having had her most recent pneumothorax 3 months prior, our patient reported to the hospital once she recognized her symptoms as another relapse. Cavitated cystic blebs within the lung parenchyma are believed to be the reason behind recurrent pneumothoraces in those with LAM, such as with our patient. The importance of educating patients with rare conditions is illustrated in this case. Our patient was aware of the importance of going to an ED upon onset of dyspnea due to her condition, and was therefore able to receive appropriate care.
Keywords: Lymphangioleiomyomatosis;Emergency Department ;computed tomography ;pneumothorax.
Introduction
Lymphangioleiomyomatosis (LAM) is a rare multisystem perivascular epithelioid (PEComa) neoplasm, which can manifest itself as cystic lung destruction, chylous pleural effusions and/or renal angiomyolipomas [1]. Characterized by LAM smooth muscle cell invasion and proliferation within the interstitium of healthy lung parenchyma [2] causing multiple thin-walled cysts, chylous pleural effusions, and recurrent pneumothoraces. Typically occurring in a female population exclusively [3] two forms of LAM have been studied: the subtype associated with Tuberous Sclerosis, and the spontaneous subtype not associated with tuberous sclerosis. The development of LAM occurs in 30-40% of females with tuberous sclerosis complex (TSC) mutations, and only in 3 to 8 individuals per 1 million in the sporadic version [4] as experienced by our patient. Normally, TCS works as a tumour suppressor, which deactivates the DNA replication promoter mTOR in dividing cells such as smooth muscle, lymphatic cells, and leukocytes. Without TCS suppression, mTOR disinhibition leads to the overactivation and proliferation of smooth muscle cells in the parenchyma of the lungs, leading to the formation of cystic lesions1. Additionally, LAM cells are known to possess lymphangiogenic growth factors, such as vascular endothelial growth factors C and D, which allow for spread of LAM cells throughout the lymphatics, lymphatic obstruction, as well as expansion and fusion of existing cysts [5].
The invasiveness and metastatic manuscript potential of LAM cells with mutations specific to the TSC2 component of the complex are further promoted by progesterone and estrogen through activation of the MEK1/2 and PI3K/Akt signalling pathways.
Although etiology of what triggers the sporadic development of LAM in patients with normal functioning TSC remains unknown [6,7] the interplay of estrogen, lymphatic proliferative factors, as well as mTOR autophosphorylation in response to hypoxia all play a synergistic role in the development of this disease.
Objective
To report a rare case of sporadic lymphangioleiomyomatosis (LAM) with manifestation of recurrent pneumothorax in a premenopausal female and explore factors that drive the disease process of LAM forward, as well as the role of sirolimus in slowing that process down.
Case Report
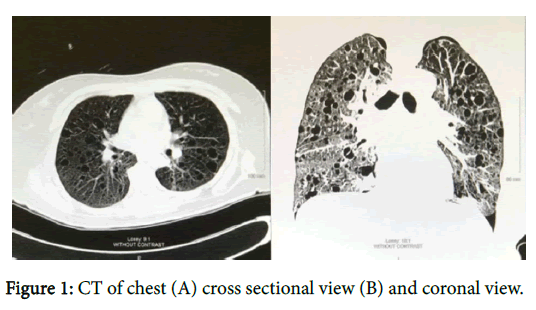
A 42-year-old nulligravida Hispanic female with a past medical history of recurrent bilateral pneumothoraces secondary to lymphangioleiomyomatosis (LAM) presented to the Emergency Department (ED) with chest tightness upon exertion as well as shortness of breath that progressively worsened over 4 days, despite the usage of her as needed albuterol inhaler at home. Having had two previous pneumothoraces, with her most recent 3 months prior, our patient reported to the hospital once she recognized her symptoms as yet another relapse. Chest computed tomography (CT) from her last episode showed dozens of air filled cavities in both lungs (Figures 1A and 1B). Past medical history was negative for asthma, COPD, or heart failure, however surgical history was significant for a left sided nephrectomy several years prior for reasons that the patient cannot recalled.
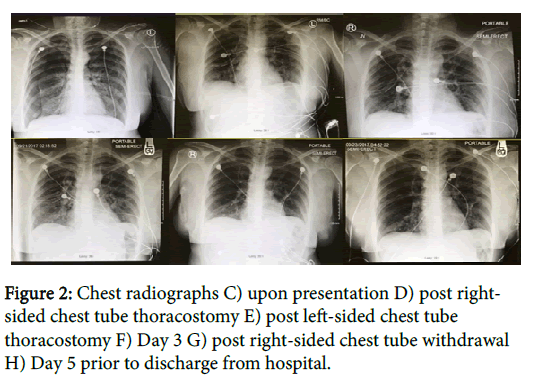
On examination, the patient was tachypneic with respiratory rate of 35 breaths/min and hypoxic with SPO2 of 87%. Chest radiography at presentation revealed large bilateral pneumothoraces, with greater severity on the right side (Figure 2C). After an attempt of Bipap therapy in the ED did not relieve her symptoms, the decision to place right and left sided chest thoracostomy tubes was made. Mild reexpansion of the lungs was appreciated on repeat chest radiograph (Figure 2D), and the patient was transferred to the Intensive Care Unit (ICU) where her lungs continued to improve. On day 3 the left lung showed significant improvement and the tube was removed (Figures 2E and 2F), however the right lung showed a remaining small apical pneumothorax and the tube remained for several more hours until the pneumothorax had resolved (Figure 2G). The patient was transferred to the floor for observation and was discharged on day 5 with instructions to follow-up with outpatient pulmonology for further evaluation and management.
Discussion
Invasion and proliferation of LAM smooth muscle cells within the interstitium of the lung parenchyma leads to the multitude of thin walled cystic lesions seen in patients with LAM. These cysts, ranging from a few millimeters to centimeters in diameter, have the ability to trigger pneumothoraces from either direct cystic rupture when in contact with the pleural space, or indirectly through alveolar wall disruption [8,9]. Furthermore, the ability to induce hyperplasia of the lymphatic vessels via VEGF-C and VEGF-D causes lymphatic obstruction within the lungs of these LAM patients, resulting in lymphedema, and in some cases contribute to expansion and fusion of existing cysts.
The lifetime risk of pneumothorax in patients with LAM ranges from 39-81% with a recurrence rate of 73% after their initial or sentinel pneumothorax, and are likely to experience at least two or more pneumothoraces after their initial presentation. Studies following the lifetime incidence of pneumothorax in LAM include: Urban et al. [10] study that reported 47 of 69 patients (68%) had at least one pneumothorax over the course of their disease McCormack et al. [1] demonstrated 29 of 49 (60%) patients experienced a pneumothorax, Johnson and Tattersfield [11] Kitaichi et al. [12] reported pneumothoraces in 18 of 46 (39%) of LAM patients.
Although what triggers LAM smooth muscle cells to proliferate initially in the spontaneous form of the disease remains unknown, the normal physiological response of the lung tissues exposed to chronic hypoxia caused by air trapping and cyst formation is known to contribute to disease progression. A major driving factor of LAM progression is mTOR, a serine/threonine kinase which promotes DNA synthesis, survival, and proliferation of pulmonary arterial vascular smooth muscle cells (PAVSM) cells in response to chronic hypoxia. Pulmonary artery hypertension (PAH) as a consequence of proliferation over activity of PAVSM cells in response to chronic hypoxia significantly contributes to the morbidity and mortality of several chronic lung diseases including chronic obstructive pulmonary diseases, cystic fibrosis, and LAM. It is therefore logical to assume that targeting and inhibiting mTOR as a potential therapeutic means of alleviating symptoms associated with PAH and chronic lung disease states.
mTOR is composed of two separate subunits, and as demonstrated by Krymskaya et al. [13], must work together in order to influence PAVSM cell proliferation. In this study, PAVSM cell proliferations in hypoxic states were suppressed when either rapamycin sensitive mTORC1 or rapamycin mTORC2 components were inactivated. Twenty-four hours of hypoxic exposure of PAVSM cells in this study treated with rapamycin were shown to inhibit phosphorylation of mTORC1 Ser 2481 and S6 subunits (required for its activation), which resulted in PAVSM proliferation suppression in comparison to cells not treated with rapamycin. Additionally, siRNA used to specifically target and inhibit the Akt (Ser473) phosphorylation site on mTORC2 were also shown to decrease proliferation when exposed to hypoxic conditions in comparison to those not treated.
The Krymskaya et al. study also demonstrated the role of autophosphorylation of mTOR in PAVSM cells in comparison to other cell lines [13]. In several cell lines, anaerobic glycolysis in response to hypoxia and decreased availability of ATP promotes AMPK, an enzyme responsible for phosphorylation and activation of tumour suppressor tuberous sclerosis complex (TSCI/TSC2).
In these lines, TSCI/TSC2 activation is responsible for mTOR suppression, and thus puts a halt cellular proliferation. In PAVSM cell lines exposed to hypoxia however, AMPK targeted inhibition by siRNA did not show significant difference in the amount of proliferation undertaken by the treatment vs. control groups. This evidence suggests that mTOR activation within PAVSM cells during hypoxic states is AMPK independent, and may therefore instead rely on autophosphorylation of itself for activation and cell proliferation. Furthermore, when autophosphorylation of mTORC1 S6 subunit was inhibited by Metformin, cell division was suppressed. In light of this evidence, treatment that directly targets mTOR itself may be more advantageous in slowing the progression of PAH due to PAVSM proliferation in chronic hypoxic states, rather than targeting signaling molecules in its activation pathway.
Currently, the treatment of choice for patients with LAM is via the inhibition of mTOR with sirolimus (rapamycin), thereby restoring homeostasis in cells with the defective TSC gene [10].
Although the decision to initiate sirolimus therapy should be individualized for the patient, and with contribution of pulmonary and immunosuppressive therapy experts, typical indication for its use is in patient was decreased lung function (FEV1<70%), patients who are pre-menopausal (higher estrogen levels and therefore enhanced progression of the disease in comparison to postmenopausal patients), and in those who are symptomatic. It should be noted however that Sirolimus is only useful for reducing the rate LAM progression rather than a cure. The function of sirolimus was demonstrated in the [1] trial in 2011 which conducted a year long study comparing FEV1 response to Sirolimus treatment vs. place.
The study demonstrated the slope of decline of FEV1 significantly improved with the use sirolimus compared to the placebo during the treatment period. Once taken off Sirolimus after the trial had ended, patients in the treatment group again experienced a functional decline in parallel to those in the placebo group.
The disadvantage of sirolimus is that due to the immunosuppressive nature; white blood cell (WBC) counts must be monitored indefinitely. In addition to inhibiting the proliferation of PAVMS cells, Sirolimus has also made a name for itself as one of the staple prophylactic drugs of choice when it comes to transplant rejection because of its ability to suppress B and T cell proliferation, thus leaving patients vulnerable to infection in 2014 reported that of the LAM cohort they followed on Sirolimus therapy, 40% had reported neutropenia/leukopenia [14].
More recently however, Gopalakrishnan et al. [15] conducted an observational study which aimed to determine whether the diurnal phenomenon of pseudoneutropenia/eudoleukopenia (morning WBC counts are generally lower in the morning in healthy persons) was influenced by Sirolimus in LAM patients, and whether this made a significant difference in their susceptibility to infection compared to LAM patients not on sirolimus [14]. While results showed that Sirolimus treatment enhanced the cyclic nature of WBC and neutrophil counts in LAM patient taking the drug, isolated morning neutropenia/leukopenia in these patients did not increase the risk of infection [15]. Therefore stressed the importance of physicians taking into consideration the diurnal nature of pseudoneutropenia/ pseudoleukopenia is enhanced in patients taking Sirolimus, and not to attribute an abnormally low morning WBC to Sirolimus toxicity and to instead retest blood later in the day.
In summary, LAM is a proliferative, invasive, smooth muscle neoplasm that manifests as thin walled cystic lesions that significantly increase the risk of pneumothorax development in patients.
Overactivation of mTOR in LAM cells due to either TSC loss of function, or by spontaneous activation have been described as the underlying pathology of LAM, with the normal physiological response to hypoxia driving the disease process forward. The current treatment requires mTOR to be directly targeted, as focus on upstream targets such as AMPK have been shown to not be effective due to mTOR’s ability to autophosphorylate when exposed to chronic hypoxic states.
The fact that Sirolimus is an immunosuppressive agent should also factor into the treatment plan of patient with LAM. Furthermore, physicians should not mistakenly take Sirolimus toxicity when monitoring morning WBC counts as pseudoleukopenia and pseudoneutropenia are known to be exaggerated when using this agent, and this misrepresentation of true leukocyte and neutrophil concentration have not been shown to significantly contribute to an increased risk of opportunistic infections in patients with LAM.
References
- McCormack FX, Inoue Y, Moss J, Singer LG, Strange C, et al. (2011) Efficacy and safety of sirolimus in lymphangioleiomyomatosis. N Engl J Med 364:1595.
- Zhe X, Yang Y, Jakkaraju S, Schuger L (2003) Tissue inhibitor of metalloproteinase-3 downregulation in lym- phangioleiomyomatosis: potential consequence of abnormal serum response factor expression. Am J Respir Cell Mol Biol 28: 504–511.
- Johnson S (1999) Lymphangioleiomyomatosis: clinical features, management and basic mechanisms. Thorax 54: 254.
- Torre O, Elia D, Caminati A, Harari S (2017) New insights in lymphangioleiomyomatosis and pulmonary Langerhans cell histiocytosis. Eur Respir 26: 170042
- Tobino K, Hirai T, Johkoh T, Fujimoto K, Kawaguchi A, et al. (2017) Difference of the progression of pulmonary cysts assessed by computed tomography among COPD, lymphangioleiomyomatosis, and Birt-Hogg-Dube Ì syndrome. Plose 12.
- Sun Y, Zhang E, Lao T, Pereira AM, Li C, et al. (2014) Progesterone and estradiol synergistically promote the lung metastasis of tuberin-deficient cells in a preclinical model of lymphangioleiomyomatosis.Hormones and cancer 5: 284-298.
- Glassberg MK, Elliot SJ, Fritz J, Catanuto P, Potier M (2008) Activation of the estrogen receptor contributes to the progression of pulmonary lymphangioleiomyomatosis via matrix metalloproteinase-induced cell invasiveness. J Clin Endocrinol Metab 93: 1625-33.
- Almoosa KF, Ryu JH, Mendez J, Huggins JT,Young LR, et al. (2006) Management of pneumothorax in lymphangioleiomyomatosis - Effects on recurrence and lung transplantation complications. Chest 129: 1274-1281.
- Tattersfield AE, Harrison TW (1999) Step 3 of the asthma guide- lines. Thorax 54: 753–754.
- Urban T, Lazor R, Lacronique J, et al (1999) Pulmonary lymphangio- leiomyomatosis: a study of 69 patients; Groupe d’Etudes et de Recherche sur les Maladies Orphelines Pulmonaires (GERM“O”P). Medicine (Baltimore) 78: 321–337.
- Johnson SR, Tattersfield AE (2000) TattersfieldAE.Clinicalexperienceoflymphangio- leiomyomatosis in the UK. Thorax 55: 1052–1057.
- Kitaichi M, Nishimura K, Itoh H, Izumi T (1995) Pulmonary lym-phangioleiomyomatosis: a report of 46 patients including a clinicopathologic study of prognostic factors. Am J Respir Crit Care Med 151: 527–533.
- Krymskaya VP, Snow J, Cesarone G, Khavin I, Goncharov DA, et al. (2011) mTOR is required for pulmonary arterial vascularsmooth muscle cell proliferation under chronic hypoxia. The FASEB J 25: 1922-1933.
- Yao J, Taveira-DaSilva AM , Jones AM, Julien-Williams P, Stylianou M, et al. (2014) Sustained effects of sirolimus on lung function and cystic lung lesions in lymphangioleiomyomatosis. Am J Respir Crit Care Med 190: 1273–1282.
- Gopalakrishnan V, Jones AM, Julien-Williams P, Machado T, Danner RL, et al. (2017) Pseudoneutropenia in lymphangioleiomyomatosis (LAM) patients receiving sirolimus: evaluation in a 100 patient cohort. ERJ Open Res 4: 00121.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi