Review Article, J Pulm Med Vol: 5 Issue: 4
A Review of Risk Factors for COVID-19 Disease: Opportunities for Personalized Prevention and Immunization
Hector G Ortega*
Department of Respiratory and Allergic Diseases, Genentech, USA
*Corresponding Author: Hector G Ortega Department of Respiratory and Allergic Diseases, Genentech, USA E-mail: hortega@gossamerbio.com
Received: August 09, 2021; Accepted: August 23, 2021; Published: August 30, 2021
Citation: Hector Ortega (2021) A Review of Risk Factors for COVID-19 Disease: Opportunities for Personalized Prevention and Immunization. J Pulm Med 5:4
Abstract
During the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) pandemic, the clinical, scientific, and public health communities have had to react to new viral genetic variants. These have triggered a range of reactions from the public health community raising concerns about the effectiveness of the current vaccines and questions about the need for booster vaccinations. This is further complicated by vaccine hesitancy and the “perceived” benefit-risk of available vaccines. In the US alone there is approximately 3% of immunocompromised individuals. While the obese and elderly individuals do not fall into the classical spectrum of immunocompromised, they are at a high risk for a more severe clinical course of COVID-19. Obesity is a strong independent risk factor for hospitalization in COVID-19 patients with increased morbidity and mortality. Recently, there has been enormous progress on the understanding of vaccines benefit-risk profile. There is an opportunity for providing vaccine personalized approaches across different groups by age and by gender. This review focuses on the obese and the elderly populations but also presents opportunities for personalized interventions with different vaccines. Strategies are urgently needed to systematically address the underlying public health problems such as obesity to effectively manage this COVID-19 pandemic as well as the rapid introduction of tailored approaches for vaccination.
Keywords: Obesity; Pro-inflammatory; Elderly; Vaccine Immunocompromised
Introduction
The United States (US) currently has one of the highest mortality rates from COVID-19 worldwide and has a higher prevalence of obesity compared to other nations [1,2]. It is well established that obesity is linked to chronic diseases associated with increased morbidity and mortality; it is now clear that obesity also is a strong risk factor for hospitalization related to COVID-19 in patients affected by these diseases [1]. Previous experience with obesity-related mortality during the H1N1 influenza epidemic [3] provides some based on similarities with the current SARS-CoV-2 pandemic, where an increased number of obese individuals were admitted to intensive care units. For example, obesity is associated with a greater decrease in lung function compared to the non-obese. Furthermore, the obese must devote a larger percentage of total oxygen consumption to respiratory work, even during quiet breathing [4]. This relative inefficiency creates a decreased Hector: ventilatory? reserve and a propensity to respiratory failure, even with mild pulmonary insults. In addition to these mechanical limitations, obese individuals have an underlying impaired immune response further affecting the ability to deal with infections. Thus, the already impacted respiratory system of obese individuals can be severely compromised from any respiratory insult, like COVID-19 disease. Additionally, comorbidities that are commonly associated with obesity such as cardiovascular disease, hypertension, diabetes mellitus, and chronic lung diseases [1,5] have also been found to be linked with a severe clinical course of COVID-19 and increased mortality. Hence, obesity contributes substantially to the increase in morbidity and mortality of SARSCoV- 2 infection.
Pathophysiology and Immune Response
Adipose tissue is perhaps the most structurally dynamic tissue in the adult human body. Its capacity to grow and shrink in size by large magnitudes is critical to human health, metabolism, fitness, and adaptation. At the cellular level, hypertrophic lipid engorged adipocytes are more likely to activate endoplasmic reticulum and mitochondrial stress responses along with mechanical stress inducing shear on the extracellular environment [6]. This promotes the activation of a chronic, proinflammatory state within the adipose tissue. Crown- like structures (CLSs) are adipose microenvironments of macrophages engulfing adipocytes [7]. Their histological density in visceral adipose tissue (VAT) predicts metabolic disorder progression in obesity and is believed to initiate obesity-related comorbidities. Hector: do you need both obese and VAT here? contains large CLSs absent from lean tissues and is located near the tissue center, while lean CLSs have higher volumetric cell densities and prolate shapes. These features are consistent with inefficient adipocyte elimination in obesity that contributes to chronic inflammation [7].
Animal and Human Studies
Mice with high-fat, diet-induced obesity who become infected with influenza viruses exhibit a significantly higher mortality and more severe lung pathology than their lean counterparts [8]. These findings may be related to the abundance of proinflammatory mediators in adipose tissue, which leads to dysfunction of innate immunity in obesity [9,10]. In a diet-induced obesity model, increases in proinflammatory mediators were associated with augmented tissue inflammation and increased mortality upon infection [11]. Dietary fat can also affect pulmonary surfactant function in mice [12]. The chronic inflammatory state also blunts antigen activation of macrophages and reduces proinflammatory cytokine production upon macrophage stimulation [13]. This blunted activation of macrophages upon antigen presentation may explain the potential inability for mounting a strong response following vaccination in obese individuals. Studies have demonstrated that obesity may be linked to a lower vaccine response following influenza and Hepatitis B immunization [14-16]. This is experience is critical as health authorities consider booster immunization strategies.
Obese individuals may have defective responses in both innate and adaptive immunity; for example, virus in obese individuals alters endothelial function resulting in the leakage of pathogens facilitating the development of pneumonia in the more severe cases. Obesity should not be ignored in older individuals, particularly since their immune system weakens with age, creating further challenges to fight infections. Human studies have shown that the obese state is associated with a decrease in CD8+ T cells and an increase or decrease in CD4+ T cells [17,18]. B and T cell responses are also disrupted in obese individuals, with a reduced lymphocyte proliferative response. This disruption results in an increased susceptibility to infection and delay in the resolution of viral infections. Obesity is also characterized by impaired memory CD8+ T cell responses to influenza virus infections, elevated viral titers in lung, and worsened lung pathology [19]. Moreover, low vitamin D levels in obese individuals may have a negative impact on its anti-inflammatory and antimicrobial properties against COVID-19 disease [20]. Taken together, these features may contribute to increased susceptibility to COVID-19 related lung infections. Interactions between influenza virus (pH1N1) and lipopolysaccharide (LPS) in mice have resulted in a synergistic increase in IL-6, IL-1β, and TNF-α levels in lung tissue, promoting a hypercytokinemia and proinflammatory immune response [21].
Role of the Renin Angiotensin System
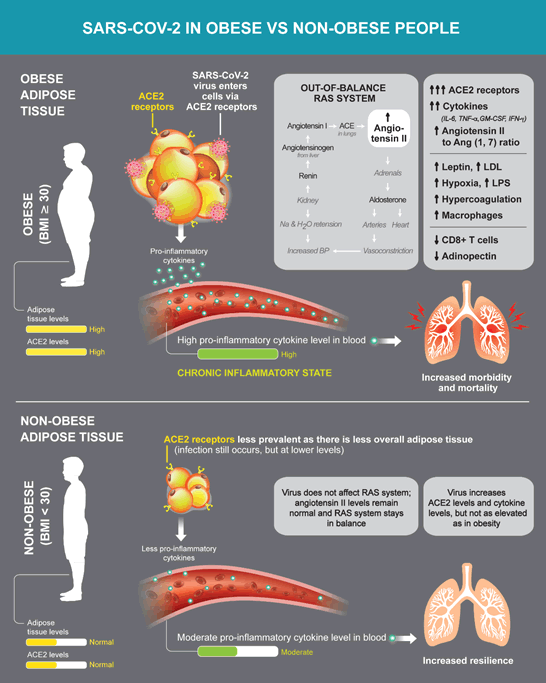
Obesity, both in humans and in experimental animals, is also associated with an imbalance in the renin–angiotensin system (RAS), resulting in an overexpression of the Angiotensin II (ANG II) and Angiotensin II receptor type 1 (AT1R) axis locally in adipose tissue and systemically. Angiotensin converting enzyme II (ACE2) is the key enzyme of the alternative RAS and counterbalances Angiotensin II activity by enzymatically converting Angiotensin II to Angiotensin 1-7 (Figure 1).
The figure represents a contrast between obese and non-obese people. ACE2 serves as the key receptor for SARS-CoV-2 infection. ACE2 is highly expressed the adipose tissue. The renin angiotensin system (RAS) involves Ang II, ACE, ACE2, and Ang [1-7] for regulating blood pressure, electrolyte balance, and vascular resistance. Renin, produced by the kidneys, cleaves Ang I, which is then converted to Ang II by ACE; Ang II promotes inflammation, salt and water reabsorption, oxidative stress, and vasoconstriction. Reduced ACE2 activity as well as initial high levels of ACE2 expression may play a key role in increasing disease severity leading to worsened prognosis. A decreased level of ACE2 secondary to SARS-CoV-2 infection followed by an increase in Ang II can exacerbate cardiovascular symptoms and promote disease complications. Adipose cells produce biologically active agents, known as adipokines (leptin, adiponectin). In adipose tissue, excess production of leptin and other proinflammatory cytokines such as IL- 6, TNF-α, GM-CSF, INF-ɣ contribute to a proinflammatory state.
Anchored in the membrane of type-II alveolar and lung epithelial cells, ACE2 can be cleaved by the metalloproteinase ADAM17 [22]. Thus, in obese individuals, especially those with extensive visceral adipose tissue, upregulation of the ANG II and AT1R-axis might lead to the development of a more severe course of infection. Data have shown that LPS induces lung injury through the suppression of ACE2 and the upregulation of ANG II, ACE, and AT1 receptors, significantly modulating the whole RAS [23].
Protection against Infection
Several factors influence the endotoxicity of LPS, some of them acting as enhancers (LPS binding protein and CD14), others as suppressors (haptoglobin); these factors are strongly dysregulated in the porcine respiratory coronavirus (PRCV) model [24]. Importantly, human plasma levels of LPS binding protein are increased in obesity, demonstrating a positive correlation with BMI [25]. In addition, CD14 expression is enhanced in obese individuals [26]. CD14 acts as a coreceptor (along with the Toll like receptor TLR-4 and MD-2) for the detection of bacterial LPS. CD14 can bind LPS only in the presence of LPS binding protein (LBP). CD14 is required for influenza A virus-induced cytokine and chemokine production [27]. Of note, in COVID-19, the plasma concentration of sCD14 increases markedly with severity of the disease [28]. Entry of the SARS-CoV-2 virus into the cell is mediated by fusion of the viral spike protein and cellular membrane through interaction with cell surface ACE2. The ACE2 receptor is highly expressed in the lungs and heart, among other tissues, including cornea, esophagus, ileum, colon, liver, gallbladder, kidney, and testis [29]. In adipose tissue, the expression of ACE2 is greater than that in lung tissue, the main target tissue affected by SARS-CoV-2 virus [30]. Thus, individuals with increased adipose tissue have an increased number of ACE2 expressing cells and therefore a larger number of ACE2 receptors [31] (Figure 1). This higher number of adipocytes may lead to a greater viral load and prolonged viremia. In fact, adipose tissue can act as a reservoir for different viruses including influenza A virus, HIV, human adenovirus Ad-36, and SARS-CoV-2. As such, SARS-CoV-2 can preferentially accumulate in adipose tissue and attack other organs, worsening the disease [31].
Cytokine Activation and Regulation
Adipose cells produce biologically active agents, known as adipokines (leptin, adiponectin, and other cytokines). Obese individuals have lower concentrations of adiponectin (anti-inflammatory) and higher levels of leptin (proinflammatory) than non-obese individuals. Human eosinophils, a key inflammatory cell, express the leptin receptor Ob-Rb [32]. Leptin is structurally related to IL-6 and is the main regulator of appetite [33]. Leptin stimulates eosinophils to produce inflammatory cytokines [34]. In adipose tissue, excess production of leptin and other proinflammatory cytokines such as IL-6, IL-1β, MCP-1, and TNF-α support proinflammatory stimulation. Interestingly, during the H1N1 pandemic in 2009, leptin was identified as cofactor in disease pathogenesis [35].
Adiponectin is synthesized and secreted by the adipocyte; in addition to its anti-inflammatory properties, it also regulates glucose and fatty acid metabolism. In obese individuals, adipose tissue becomes infiltrated with proinflammatory macrophages. The adipocytes and activated macrophages in turn produce proinflammatory adipokines and cytokines that together with the decreased adiponectin levels generate “metabolic inflammation”. The metabolic dysfunction associated with obesity may lead to systemic leptin resistance [32-35]. In fact, this resistance may be associated with reduced bone marrow hematopoiesis and decreased T cell production [36,37]. Noteworthy is the finding that, obesity may be associated with a pro-coagulant profile that may have a role in the thromboembolic complications seen in COVID-19 disease [38]. Also notable is that a decrease LDL levels is associated with higher severity and mortality of COVID-19 disease [39]. Viral infections induce proinflammatory cytokines that modulate lipid metabolism including oxidation of LDL (elevated in obesity), by reactive oxygen species to facilitate LDL clearance. Thus, serum LDL measurement, during disease progression, may be useful in identifying patients with a poor prognosis at an early stage. Remarkably, in SARS-CoV patients, alterations in lipid metabolism have been detected as late as 12 years after the initial infection [40].
During and after the 2009 influenza A/H1N1 pandemic, higher BMI was recognized as an independent risk factor for increased disease severity and duration of viral shedding; in fact, symptomatic obese individuals shed the virus 42% longer than their non-obese counterparts [41-43]. Moreover, both animal and human studies have shown a delayed capacity to produce interferons in the obese state [44,45]. This delay enables further viral RNA replication to increase the chance of new, more virulent viral strains emerging [45]. This is potentially relevant since the SARS-CoV-2 virus continues to adapt, resulting in a series of viral variants. It is unclear to date, however, the extent that these new variants will escape recognition by vaccine-induced immunity but currently the Delta variant represents a significant proportion of the COVID-19 cases, presenting with more severe disease.
Obesity and Aging
It is known that older individuals have an increased risk for the development of severe forms of COVID-19 [46]. Notably, aging is associated with increased visceral fat; data suggest that visceral fat could influence longevity. Studies in mice and in humans have shown that white adipose tissue (WAT) accumulation could negatively influence longevity, whereas a surgical or genetically engineered reduction in WAT mass in rodents could prolong lifespan [47-50]. Animals with reduced fat mass were protected against age-related obesity and its subsequent metabolic abnormalities, although their food intake was normal. Fat-specific insulin receptor knockout (FIRKO) mice had an 18% increase in mean lifespan, with parallel increases in median and maximum lifespans [51]. Thus, a reduction of fat mass without caloric restriction can be associated with increased longevity in mice, possibly through effects on insulin signaling. It has been postulated that obesity could be a condition of premature metabolic aging.
There is an apparent decline in the relative added risk of obesity with increasing age despite the negative impact of obesity on the risk of death from all-cause mortality [52]. This has led some experts to conclude that obesity should not necessarily be viewed as a disease in individuals older than 55 years. This perception of adiposity during the latter phases of life could have a negative impact by not only discouraging attempts for weight loss, but also demotivating older individuals to establish and maintain healthy nutritional and lifestyle changes. Despite claims that obesity is not harmful in older individuals, several largescale studies would argue otherwise. In the 10-year follow-up of the National Institutes of Health AARP cohort, both overweight and obesity, at all ages and in both sexes, and particularly in those individuals who had never smoked and without history of disease, are linked to increased mortality [52]. Although, the risk associated with a high BMI may decline with advancing age, the absolute rise in mortality rates associated with a high BMI is still much greater in elderly subjects, simply due to increased death rates in this age range [53]. This relationship may no longer exist for the very old, in whom mortality rates may be driven by other conditions such as malignancy or aging of the cardiovascular system that evolved throughout life. In the follow-up study of the First National Health and Nutrition Examination Survey (NHANES I), 1,259 Caucasian obese women aged 65-74 years showed a 50% increased risk of coronary heart disease during a mean follow-up period of 14 years, which was 2.5-fold higher than in non-obese women [54].
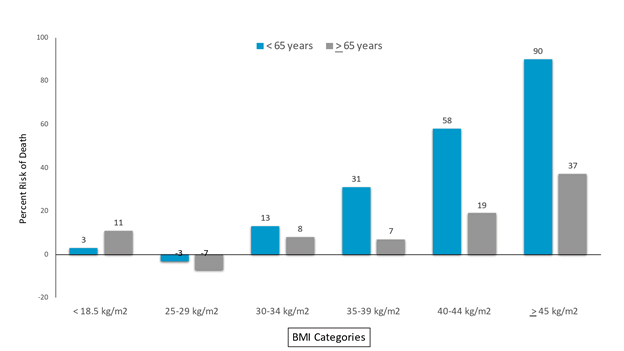
The Centers for Disease Control and Prevention (CDC) reported data [55] from a large, hospital-based, all-payer database during April 1 to December 31, 2020 demonstrating that both adults <65 and ≥ 65 years old have an increased risk of death in parallel to increase in BMI (Figure 2).
In Premier Healthcare Database Special COVID-19 Release (PHD-SR) dataset. Data from the original report were obtained from a large, hospital- based, all-payer database during April 1 December 31, 2020. Data included 71,491 patients hospitalized with COVID-19. The data show that adults <65 and ≥ 65 years old have increased risk of death in parallel to increase in BMI. BMI of 18.5 to 24 kg/m2 was used as reference for comparisons across BMI ranges. Modified from Kampaniyets L, et al. (MMWR March 8, 2021).
Added to the obesity risk, there are age-related changes of the immune system that contributes to increased incidence and severity of infections in the elderly. Most currently used vaccines are less immunogenic and therefore, less effective in the elderly compared to younger adults. Potential strategies to improve their immunogenicity include use of a higher antigen dose, alternative routes of administration, and adjuvants, which were all implemented for influenza vaccines, and may induce moderately higher antibody concentrations [56]. Thus, it will be important to determine soon the durability of the immune response in the elderly with the current vaccines available against SARS-CoV-2.
Public Health Implications
The COVID-19 pandemic has occurred at a time when the prevalence of individuals with obesity is increasing in virtually all countries worldwide. In the US, the prevalence of adult obesity in 2017 to 2018 has risen to 42% [2,57]. Notably, almost all countries worldwide report that at least 20% of the population is classified as either overweight or obese [58,59]. To date, no country has experienced a reduction in the prevalence of individuals with obesity. In fact, no state or territory in the US has reported a prevalence of obesity less than 20% [60]. Notably, 12 states in the US have ≥ 35% prevalence of obesity. Importantly, there is a disproportionate rate of obesity among Blacks and Hispanics. In many areas of the US, Blacks and Hispanics are more than twice as likely as Whites to die from COVID-19 [61]. Recently (Feb 18, 2021) the CDC reported that age-adjusted COVID-19 hospitalization rates for Blacks and Hispanics was 2.9 and 3.2 times higher than those of Whites, respectively. Similarly, the death rate for Blacks and Hispanics was 1.9 and 2.3 times higher than those of Whites, respectively [62].
A study in 5,700 US patients hospitalized with severe forms of COVID- 19 reported that 41% of affected individuals were classified as obese [63]. Another study conducted in New York showed that for those under the age of 60, obesity was twice as likely to result in COVID-19- related hospitalization, and that BMI >40 kg/m2 was the second strongest independent predictor of hospitalization after old age [64]. These observations are consistent across different countries. For example, in a study in China [65], where obesity is less prevalent than in Western countries, the investigators concluded that overweight patients had an 86% higher risk, and those with obesity a 142% higher risk of developing COVID-19-related severe pneumonia compared with normal weight patients. Consistent with these findings, a large UK-based cohort study examined the factors associated with COVID-19-related hospital death using linked electronic health records of 17 million adult National Health Service patients [66]. The study found that the risk of death increased with a higher degree of obesity; the adjusted HR was 1.27 for BMI 30-34.9 kg/m2, increasing to 2.27 for BMI ≥ 40 kg/m2. Lastly, a recent meta-analysis showed that the odds of COVID-19 positivity in obese individuals were 46% higher than in those individuals who were not obese. Moreover, obese individuals were more likely to have unfavorable outcomes with a 48% increase in death [67].
Need for Booster Immunizations
Obese and elderly
Obese individuals exhibit impaired immunological memory. In fact, influenza vaccination in adults with and without obesity results in equivalent influenza-specific antibody titers at 30 days after vaccination, but antibody titers wane significantly more in adults with obesity (as in older individuals) compared with adults who are lean at 1 year after vaccination [68]. Compared with influenza vaccinated lean adults, vaccinated adults with obesity have impaired CD8+ T and CD4+ cell production of key inflammatory cytokines IFN-γ and granzyme B [69]. Adults with obesity also have two times greater odds of influenza or influenza-like illness despite a robust antibody response [70]. While the mRNA vaccines have shown excellent protection of approximately 95% in obese individuals [71], this observation is limited to a few months after the second injection. It will be critical to determine if the protection in obese individuals and the elderly is durable after a longer period and comparable to other demographic subgroups. It is also important to highlight that protection with currently available vaccines is extremely promising. For example, the Janssen (Ad26.COV2.S) single dose vaccine, demonstrated full protection in the more severe COVID-19 cases. Of the reported COVID-19 related deaths in their Phase 3 study, all seven occurred in the placebo group. Notably, all were obese except for one individual [72].
Immunocompromised
Immunocompromised individuals represent approximately 3% of US adults [73]. These include people with solid tumor and hematologic malignancies, recipients of solid organ or hematopoietic stem cell transplant, severe primary immunodeficiencies, individuals with HIV, or treatment with immunosuppressive medications including cancer chemotherapeutic agents, TNF blockers, certain biologic agents (e.g., rituximab), and chronic use of high-dose systemic corticosteroids. These individuals are more likely to get severe disease from COVID-19 due to prolong shedding, and low antibody/neutralization titers to SARS-Cov-2 variants [74]. Studies indicate reduced antibody response in immunocompromised individuals following a primary vaccines series, compared to healthy vaccine recipients. In fact, among individuals vaccinated, 44% of hospitalized breakthrough cases involve immunocompromised individuals [75]. While the vaccines are quite effective, the vaccine effectiveness in terms of COVID-19 hospitalizations ≥ 14 days after the second dose of mRNA vaccines was 59% among immunocompromised individuals vs. 91% without immunocompromised [75]. Data have shown that among those who had no detectable antibody response to an initial mRNA vaccine series, 33% to 50% developed an antibody response to an additional dose [76,77]. These findings highlight the importance of booster immunizations among the immunocompromised. Importantly, no serious adverse events were reported in a small series (n=99) after the administration of the third dose in solid organ transplant recipients [78]. There is need to further assess who may benefit most from an additional dose and to determine the acceptable intervals and mix and match schedules. (e.g., mRNA vaccine recipients who subsequently receive the Janssen vaccine and vice versa) Ultimately, the regulatory authorities need to rapidly make decisions considering the increase in the number of cases due to the delta (B.1.617.2) variant, which currently represents 83% of reported cases in the US (July of 2021) [79].
Personalized Approaches and Benefit Risk of the Current Vaccines
The US is currently facing a challenge because of vaccine hesitancy [80]. The number cases of COVID-19 hospitalizations have doubled during the second quarter of 2021 across many states. These numbers are by far driven by unvaccinated individuals. While vaccine hesitancy is multifactorial, it is important to highlight the overall positive benefit-risk profile of the current vaccines. This provides an opportunity to tailor immunizations to specific groups with intrinsic lower risk. Some of the key adverse events reported after vaccination are the Gillian Barre Syndrome (GBS), Thrombosis with Thrombocytopenia Syndrome (TTS) and myocarditis. Recently the CDC reported a benefit-risk analysis across the Janssen and mRNA (Moderna and Pfizer) vaccines [81]. Table 1 illustrates the number of serous events (i.e., hospitalization, ICU and death) prevented by these vaccines stratified by age and gender. These data can be used to make informed decisions and introduce a personalized approach for vaccination. Specifically, one could consider the mRNA vaccine among females 30 to 49 years old and ≥ 65 years old to minimize the risk for GBS and TTS, respectively and for males 50 to 64 years old to minimize the risk for GBS. Conversely, one could consider the Janssen vaccine in males 18 to 29 years old to minimize the risk for myocarditis. The remaining groups could benefit substantialy from any of the available vaccines. Overall, the potential harm is quite small after COVID-19 vaccination. The risk for TTS from the Janssen vaccine is 3.0 per million doses among adults, and for GBS 7.8 cases per million doses among adults. Regarding mRNA vaccines, the risk for myocarditis is 3.5 cases per million doses among adults [81]. It is important to highlight that the risk for each potential harm varies by age and by gender as presented in Table 1.
| Janssen COVID-19 Cases | mRNA COVID-19 vaccines | |||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Age | Severe COVID-19 outcomes prevented | GBS Cases | TTS Cases | Severe COVID-19 outcomes prevented | Myocarditis Cases | |||||
| Hospitalization | ICU | Death | Hospitalization | ICU | Death | |||||
| Females | ||||||||||
| 18-29 years | 700 | 50 | 5 | 1 | 4-5 | 750 | 50 | 5 | 3-4 | |
| 30-49 years | 900 | 140 | 20 | 6-7 | 8-10 | 950 | 140 | 20 | 1-2 | |
| 50-64 years | 1,600 | 350 | 120 | 7-8 | 3-4 | 1,700 | 375 | 125 | 1 | |
| 65+ years | 5,900 | 1,250 | 840 | 8-10 | 0 | 6,200 | 1,300 | 900 | <1 | |
| Males | ||||||||||
| 18-29 years | 300 | 60 | 3 | 2 | 2-3 | 300 | 60 | 3 | 22-27 | |
| 30-49 years | 650 | 150 | 25 | 7-8 | 1-2 | 700 | 160 | 25 | 5-6 | |
| 50-64 years | 1,800 | 480 | 140 | 14-17 | 1-2 | 1,900 | 500 | 150 | 1 | |
| 65+ years | 11,800 | 3,300 | 2,300 | 7-8 | 0 | 12,500 | 3,500 | 2,400 | <1 | |
| *For every million doses of vaccine given with US exposure risk and hospitalization rates from June 19, 2021; red color denotes increased risk and opportunities for personalized approaches. Modified from reference 81. | ||||||||||
Table 1: Benefits and Risks after COVID-19 Vaccine Stratified by Age and Sex Groups.``
Conclusion
Overall, lessons learned from the devastating effects of the pandemic have solidified the belief that obesity is a significant societal and clinical problem that requires a strategic approach to address it as a major public health problem. Hence, it is critical to promote effective and healthy nutritional changes. Cost-effective community-based prevention, including monitoring and screening of body weight is critical. There is need for tailored behavioral, pharmacological, nutritional, and physical activity interventions. There is evidence that weight loss is associated with lower all-cause mortality, regardless of age. Low levels of spontaneous physical activity is a major predictor of adipose tissue accumulation in humans, and reduced total body movement, most of which is related to ambulation, is negatively correlated with fat mass. The obese and the elderly (including those in long term care facilities) may require a booster immunization to augment protection as well as the immunocompromised. Given the overall favorable safety profile of the current vaccines, a personalized approach offers an opportunity to maximize immunizations in a more targeted manner and perhaps increase vaccine acceptability among hesitant individuals.
References
- Ko YJ, Danielson LM, Town M (2020) Risk factors for coronavirus disease 2019 (covid-19)-associated hospitalization: Covid-19-associated hospitalization surveillance network and behavioral risk factor surveillance system. Clin Inf Dis
- Bilinski A, Emanuel EJ (2020) COVID-19 and excess all-cause mortality in the us and 18 comparison countries. J American Med Asso;324:2100-2102
- Maier HE, Lopez R, Sanchez N (2018) Obesity increases the duration of influenza A virus shedding in adults. J Infect Dis; 218:1378-82.
- Kress JP, Pohlman AS, Alverdy J (1999) The impact of morbid obesity on oxygen cost of breathing (Vo2 RESP) at rest. Am J Resp Crit Care Med; 160:883-6.
- Leung NY, Bulterys MA, Bulterys PL (2020) Predictors of COVID-19 incidence, mortality, and epidemic growth rate at the country level. medRxiv.
- Deng J, Liu S amd Zou L (2012). Lipolysis response to endoplasmic reticulum stress in adipose cells. J Biol Chem; 287:6240-9.
- Geng J, Zhang X, Prabhu S, Shahoei HS, Nelson RE et al. (2021) 3D microscopy and deep learning reveal the heterogeneity of crown-like structure microenvironments in intact adipose tissue. Science Advances. 8;2480.
- Smith AG, Sheridan PA, Harp JB, Beck MA (2007) Diet-induced obese mice have increased mortality and altered immune responses when infected with influenza virus. J Nutrition; 137:1236-1243.
- Bennett BD, Solar GP, Yuan JQ (1996) A role for leptin and its cognate receptor in hematopoiesis. Curr Biol.; 6:1170-80.
- Richard C, Wadowski M, Goruk S (2017) Individuals with obesity and type 2 diabetes have additional immune dysfunction compared with obese individuals who are metabolically healthy. BMJ Open Diabetes Res Care;5:e000379.
- Nieman DC, Nehlsen-Cannarella SI, Henson DA (1996) Immune response to obesity and moderate weight loss. Int J Obes Relat Metab Disord; 20:353–60.
- Schipke J, Jutte D, Brandenberger C (2021) Dietary carbohydrates and fat induce distinct surfactant alterations in mice. Am J Respir Cell Mol Biol;3: 379-390.
- Takahashi K, Mizuarai S, Araki H (2003) Adiposity elevates plasma MCP-1 levels leading to the increased CD11b-positive monocytes in mice. J Biol Chem 21; 47:46654-60.
- Ahn SY, Sohn SH, Lee SY (2015) The effect of lipopolysaccharide-induced obesity and its chronic inflammation on influenza virus-related pathology. Environ Toxicol Pharmacol. 40:924-30.
- Neidich S D, Green WD, Rebeles J (2017) Increased risk of influenza among vaccinated adults who are obese. Inter J Obesity. 9:1324-1330.
- Young MD, Gooch WM 3rd, Zuckerman AJ (2001) Comparison of a triple antigen and a single antigen recombinant vaccine for adult hepatitis B vaccination. J Med Virol; 64:290-298.
- Nieman DC, Henson DA, Nehlsen-Cannarella SL (1999) Influence of obesity on immune function. J Am Diet Assoc; 99:294-9.
- O’Rourke RW, Kay T, Scholz MH (2005) Alterations in T-cell subset frequency in peripheral blood in obesity. Obes Surg; 15:1463-68.
- Karlsson EA, Sheridan PA, Beck MA (2010) Diet-induced obesity in mice reduces the maintenance of influenza-specific CD8+ memory T cells. J Nutr; 140:1691-7.
- Grant WB, Lahore H, McDonnell SL (2020) Evidence that vitamin D supplementation could reduce risk of influenza and COVID-19 infections and deaths. Nutrients. 12:988.
- Koch RM, Diavatopoulos DA, Ferwerda G (2018) The endotoxin-induced pulmonary inflammatory response is enhanced during the acute phase of influenza infection. Intensive Care Med Experimental; 6:15.
- Vaduganathan M, Vardeny O, Michel T (2020) Renin Angiotensin-Aldosterone System Inhibitors in Patients with Covid-19. N Engl J Med; 382: 1653-1659.
- Ye R, Liu Z (2019) ACE2 exhibits protective effects against LPS-induced acute lung injury in mice by inhibiting the LPS-TLR4 pathway. Exper Mol Pathology. 113:104350.
- Van Gucht S, Atanasova K, Barbe´ F, Cox E, Pensaert M et al. (2006) Effect of porcine respiratory coronavirus infection on lipopolysaccharide recognition proteins and haptoglobin levels in the lungs. Microbes and Infection; 8:1492-1501.
- Sato J, Kanazawa A, Ikeda F (2014) Gut dysbiosis and detection of "live gut bacteria" in blood of Japanese patients with type 2 diabetes. Diabetes Care; 37:2343-2350.
- Fernandez-Real JM, Perez del Pulgar S, Luche E (2011) CD14 modulates inflammation-driven insulin resistance. Diabetes. 2011; 60:2179-2186.
- Pauligk C, Nain M, Reiling N (2004) CD14 is required for influenza A virus-induced cytokine and chemokine production. Immunobiology; 209:3-10.
- Messner CB, Demichev V, Wendisch D (2020) Clinical classifiers of COVID-19 infection from novel ultra-high-throughput proteomics. medRxiv preprint. Cell Systems.
- Sungnak W, Huang N, Bécavin C (2020) SARS-CoV-2 entry factors are highly expressed in nasal epithelial cells together with innate immune genes. Nat Medicine. 26: 681-687.
- Al-Benna S (2020) Association of high level gene expression of ACE2 in adipose tissue with mortality of COVID-19 infection in obese patients. Obesity Medicine.19:100283.
- Bourgeois C, Gorwood J, Barrail-Tran A (2019) Specific biological features of adipose tissue and their impact on HIV persistence. Frontiers Microbiol. 10:2837.
- Conus S, Bruno A, Simon HU (2005) Leptin is an eosinophil survival factor. J Allergy Clin Immunol. 116:1228-1234.
- Bennett BD, Solar GP, Yuan JQ (1996) A role for leptin and its cognate receptor in hematopoiesis. Curr Biol; 6:1170-80.
- Wong CK, Cheung PF, Lam CW (2007) Leptin-mediated cytokine release and migration of eosinophils: Implications for immunopathophysiology of allergic inflammation. Eur J Immunol;37:2337-2348.
- Zhang AJ, To KK, Li C (2013) Leptin mediates the pathogenesis of severe 2009 pandemic influenza A (H1N1) infection associated with cytokine dysregulation in mice with diet-induced obesity. J Infect Dis. 207:1270-80.
- Yang R, Barouch L (2007) Leptin signaling and obesity: Cardiovascular consequences. Circ Res. 101:545–59.
- Howard JK, Lord GM, Matarese G (1999) Leptin protects mice from starvation-induced lymphoid atrophy and increases thymic cellularity in ob/ob mice. J Clin Invest. 104:1051-9.
- Ranucci M, Ballotta A, Di Dedda U (2020) The procoagulant pattern of patients with COVID-19 acute respiratory distress syndrome. J Thromb Haemost;18: 1747-51.
- Fan J, Wang H, Ye G (2020) Low-density lipoprotein is a potential predictor of poor prognosis in patients with coronavirus disease 2019. Metab Clin Exp 107;1542-43.
- Wu Q, Zhou L, Sun X (2017) Altered lipid metabolism in recovered SARS patients twelve years after Infection. Sci Rep. 2017; 7: 9110-18.
- Luzi L, Radaelli MG (2020) Influenza and obesity: Its odd relationship and the lessons for COVID-19 pandemic. Acta Diabetol. 57: 759-64.
- Maier H, Lopez R, Sanchez L (2018) Obesity increases the duration of influenza a virus shedding in adults. J Infect Dis. 218:1378-82.
- Meschi S, Selleri M, Lalle E (2011) Duration of viral shedding in hospitalized patients infected with pandemic H1N1. BMC Infect Dis;11: 140.
- Honce R, Karlsson EA, Wohlgemuth N (2020) Obesity-related microenvironment promotes emergence of virulent influenza virus strains. mBio 11:1-16.
- Klinkhammer J, Schnepf D, Ye L (2018) INF-lambda prevents influenza virus spread from the upper airways to the lungs and limits virus transmission. Elife 7:e33354.
- Mehra MR, Desai SS, Kuy S, Henry TD, Patel AN (2020) Cardiovascular disease, drug therapy and mortality in COVID-19. New Eng J Med. 382:e102.
- Muzumdar R, Allison DB, Huffman DM (2008) Visceral adipose tissue modulates mammalian longevity. Aging Cell. 7: 438-440.
- Olshansky SJ, Passaro DJ, Hershow RC (2005) A potential decline in life expectancy in the United States in the 21st century. N Engl J Med. 352: 1138-1145.
- Chiu CH, Lin WD, Huang SY (2004) Effect of a C/EBP gene replacement on mitochondrial biogenesis in fat cells. Genes Dev. 18: 1970-1975.
- Gates AC, Bernal-Mizrachi C, Chinault SL (2007) Respiratory uncoupling in skeletal muscle delays death and diminishes age-related disease. Cell Metab. 6: 497-505.
- Bluher M, Kahn BB, Kahn CR (2003) Extended longevity in mice lacking the insulin receptor in adipose tissue. Science. 299: 572-574.
- Adams KF, Schatzkin A, Harris TB, (2006) Overweight, obesity, and mortality in a large prospective cohort of persons 50 to 71 years old. N Engl J Med. 355:763-778.
- Stevens J, Cai J, Pamuk ER, Williamson DF, Thun MJ, Wood JL (1998) The effect of age on the association between body mass index and mortality. N Engl J Med. 338:1-7.
- Harris TB, Ballard-Barbasch R, Madans J (1993) Overweight, weight loss, and risk of coronary heart disease in older women: The NHANES I Epidemiologic Follow-Up Study. Am J Epidemiol. 137:1318-1327.
- Kompaniyets L, Goodman AB, Belay B (2020) Body mass index and risk for COVID-19-related hospitalization, Intensive Care Unit admission, invasive mechanical ventilation, and death - United States. MMWR Morb Mortal Wkly Rep. ePub: 8 March 2021.
- Weinberger B (2018) Vaccines for the elderly: Current use and future challenges. Immun Ageing. 2018: 22; 15:3.
- Hales K, Carroll MD, Fryar CD (2020) Prevalence of obesity and severe obesity among adults: United States, 2017-2018. NCHS Data Brief, no. 360. Hyattsville, MD: National Center for Health Statistics;
- COVID-19 and Obesity: The 2021 Atlas. The cost of not addressing the global obesity crisis. Obesity Federation 107-111 Fleet Street, London, EC4A 2AB.
- James TP (2004) Obesity: The worldwide epidemic. Clinics Dermatology. 22;276-280.
- Centers for Disease Control and Prevention. Adult Obesity Prevalence Maps; (Internet). Atlanta (GA): CDC; 2021
- Racial data dashboard. The Covid Tracking Project (serial on the Internet). 2020 Jun 12 (cited 2020 Jul 15).
- Centers for Disease Control and Prevention. COVID-19: Risk for COVID-19 Infection, Hospitalization and Death by Race/Ethnicity (Internet). Atlanta (GA): CDC; 2021 February 18, 2021 (cited 2021 March 3).
- Richardson S, Hirsch JS, Narasimhan M (2020) Presenting characteristics, comorbidities, and outcomes among 5700 patients hospitalized with COVID-19 in the New York City area. J American Med Asso. 323: 2052.
- Petrilli CM, Jones SA, Yang J (2020) Factors associated with hospital admission and critical illness among 5279 people with coronavirus disease 2019 in New York city: Prospective cohort study. BMJ. 2:m1966.
- Qingxian C, Fengjuan C, Fang L (2020) Obesity and COVID-19 severity in a designated hospital in Shenzhen, China. SSRN 2020, preprint
- Williamson E, Walker AJ, Bhaskaran KJ (2020) Open SAFELY: Factors associated with COVID-19-related hospital death in the linked electronic health records of 17 million adult NHS patients. medRxiv 2020
- Popkin BM, Du S, Green WD (2020) Individuals with obesity and COVID-19: A global perspective on the epidemiology and biological relationships. Obesity Reviews. 21:e13128.
- Sheridan PA, Paich HA, Handy J (2012) Obesity is associated with impaired immune response to influenza vaccination in humans. Int J Obes. 36:1072-1077.
- Paich HA, Sheridan PA, Handy J (2013) Overweight and obese adult humans have a defective cellular immune response to pandemic H1N1 influenza A virus. Obesity. 21:2377-2386.
- Neidich SD, Green WD, Rebeles J (2107) Increased risk of influenza among vaccinated adults who are obese. Int J Obes. 41: 1324-1330.
- Vaccines, Related Biological Products Advisory Committee December 17, 2020 Meeting Presentation - FDA Review of Efficacy and Safety of Moderna COVID-19 Vaccine Emergency Use Authorization Request.
- Vaccines and Related Biological Products Advisory Committee February 26, 2021 Meeting Presentation - FDA Review of Efficacy and Safety of Janssen COVID-19 Vaccine Emergency Use Authorization Request.
- Harpaz R, Dahl MR (2016) Dooling LK. Prevalence of Immunosuppression Among U.S. Adults, 2013. J American Med Asso;23:2547-2548.
- Strengert M, Becker M, Morilla Ramos G (2021) Cellular and Humoral Immunogenicity of a SARS-CoV-2 mRNA Vaccine Inpatients on Hemodialysis. medRxiv
- Tenforde WM, Patel MM, Ginde AA (2021) Effectiveness of SARS-CoV-2 mRNA Vaccines for Preventing Covid-19 Hospitalizations in the United States medRxiv 2021.
- Werbel AW, Boyarsky JB, Ou TM (2021) Safety and Immunogenicity of a Third Dose of SARS-CoV-2 Vaccine in Solid Organ Transplant Recipients: A Case Series. Annals Int Med 2021,
- Maxime E, Charmetant X, Barba T (2021) Justification, safety, and efficacy of a third dose of mRNA vaccine in maintenance hemodialysis patients: A prospective observational study. medRxiv
- Kamar N, Abravanel F, Marion O Three Doses of an mRNA Covid-19 Vaccine in Solid-Organ Transplant Recipients. New England Journal of Medicine.
- Variant Proportions. Centers for Disease Control and Prevention.
- Ortega H (2021) A vaccine is coming but prevention is still up to us. Open J Nursing. 11:1-6.
- COVID-19 Vaccines in Adults: Benefit-Risk Discussion. ACIP Meeting, July 22, 2021. Centers for Disease Control and Prevention..
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi