Research Article, J Electr Eng Electron Technol Vol: 10 Issue: 3
Air-made, Flexible, Schottky Junction Diode Using DPP-DTT Co-polymer System
Pawan Gaire*, Shubhendu Bhardwaj and John VolakisDepartment of Electrical and Computer Engineering, Florida International University, Miami, USA
*Corresponding Author : Pawan Gaire
Department of Electrical and Computer Engineering,
Florida International University, Miami, FL 33174, USA
Tel: (+1) 804-213-2426 E-mail: pgair001@
fiu.edu
Received: September 17, 2020, Accepted: September 24, 2020, Published: September 30, 2020
Citation: Pawan Gaire et al. (2020) Air-made, Flexible, Schottky Junction Diode Using DPP-DTT Co-polymer System. J Electr Eng Electron Technol
Keywords: Schottky diode, co-polymer, DPPDTT, charge conduction mechanism, air-made
Introduction
Rectifier diodes are the building block for RF to DC rectification. For supplying power to wearable devices, such diodes must be grown on a flexible substrate, and this problem represents the bottleneck for wireless powering of wearable devices. Recently, flexible diodes have been grown by using organic or by using a combination of organic and inorganic materials [1]–[3]. These diodes have been investigated for their use for sensors, detectors, and RFID applications. Furthermore, solution-based processing and fabrication of flexible diodes has been noted recently due to advantages of low temperature processing and low cost for mass production [4].
A donor-acceptor (D-A) co-polymer system exhibits increased mobility, where the transport of electrons is favored by between electron rich and electron deficient molecules. In certain D-A co-polymeric systems, a π-π stacking is available, which further provides a strong intermolecular interaction [5] to form an ordered, lamellar polymer thin film. The D-A pair is also known to improves the carrier transport due to injection of carriers at the metal-semiconductor interface [6].
In prior works, DPP polymeric system has reported mobility between 0.1 cm2V−1s−1 to 2 cm2V−1s−1 with a typical value of 1 cm2V−1s−1. However, for the co-polymeric system of DPP and DTT, the mobility is consistently increased and reported between 1 and 3 cm2V−1s−1, where the films were grown by using low-cost spin coating processes [6]–[11]. Therefore, a D-A type co-polymeric system of DPP-DTT offers features of solution processability, chemical stability and increased mobility. To build on these reporting, we investigate the air fabrication of Schottky barrier junctions of aluminum metal and DPP-DTT co-polymeric system, while realizing them on flexible substrates for wearable applications.
In related prior works, DPP-DTT has been used as an active layer material among field effect transistors [6], [12], photovoltaic cells [13], and Schottky diodes [14]. However, these reported devices are fabricated and tested in nitrogen box. From practical standpoint of daily use, here we report the performances of the Schottky diodes that are fabricated and tested in air. Such airmade devices offer lower cost of fabrication but have not been given attention in the prior studies. Similarly, a detailed study of conduction mechanism for such Schottky diodes has not been reported.
Objective of the study is to determine the performance of Schottky diode that uses DPPDTT as the active material layer. Specifically, we study Schottky diode with Au/DPPDTT/ Al stack and an also conduct investigation into the carrier transport mechanisms to verify the measurement results. The aim is to understand the possible currents in the DPPDT based diodes for future RF applications. We also further estimate the diode parameters, such as barrier height, saturation current and ideality factor of the DPPDTT based Schottky diode. In section II, fabrication of the diodes in air is shown. In section III, the measurement results are presented and compared with analytical models. Our analysis shows that a charge conduction transport mechanism is the best suited theory to quantify the I-V characteristics exhibited by the diodes. Conclusions are presented in section IV.
II. Fabrication and experiments
A. Electrodes: Ohmic and Schottky Contacts
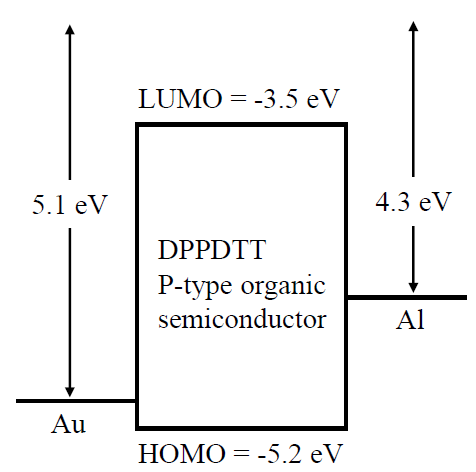
Mobility edge energy level separates the localized and delocalized states of carriers within the disordered (or imperfect) crystals. The position of mobility edge with respect to Fermi level of a metal determines the nature of metal contact, i.e. determines if the contact is ohmic or Schottky. An ohmic contact is formed between the metal and semiconductor if the Fermi level of the metal is close to the mobility edge of semiconductor [15]. For p-type semiconductor, high work function metal like gold, indium tin oxide (ITO), platinum is required to form ohmic contact. These metals allow proximity of the metal Fermi level with the valence band conduction edge (see Fig. 1). Likewise, a low work function metal like calcium, aluminum, magnesium is needed to form Schottky contact. These metals allow barrier due to sufficient gap between the valence band mobility edge and metal Fermi level. For ohmic contact at anode, gold (work function φAu = 5.1 eV [16]) is chosen over platinum and ITO. Platinum is expensive and not readily available. ITO is difficult to deposit due to transparent characteristics, which does not allow visual feedback after deposition. For creating a Schottky barrier at cathode, aluminum (φAl = 4.3 eV [16]) is chosen over calcium and magnesium. Although calcium has lower work function, polymer-calcium junction degrades with diffusion of calcium ion into the polymer layer. Magnesium is difficult to deposit because of its reactivity [17].
DPP-DTT has band gap of 1.7 eV and electron affinity of 3.5 eV [12]. Energy diagram of Au/DPP-DTT/Al structure in equilibrium is shown in Fig. 1, where the polymer layer is sandwiched between our choice of metal contact. As shown, the structure allows for a Au/DPPDTT ohmic contact and Al/DPP-DTT Schottky barrier junction for intended diode like
B.Fabrication of Schottky Diode
The device fabrication and characterization were done in atmosphere and in room temperature as opposed to inert environment in previous studies. DPP-DTT was obtained from Ossila in powdered form with number average molecular weight (Mn) of 183,332. Chloroform and O-Dichlorobenzene (DCB) solvents were obtained from Lab Alley and used in original form. DPP-DTT solution in chloroform and DCB was prepared. 250 mg of the polymer powder was added to 30 ml of chloroform and the solution was placed in a magnetic stirrer for 6 hours. The resulting solution with DPPDTT in chloroform liquid was 8.3 mg/ml. Similarly, 8.3 mg/ml of DPP-DTT in DCB liquid was prepared. For the solution processing, the polymer concentration in the solvent is important. High polymer concentration results in a viscous solution, which prevents us from getting a uniform and thin film. On the other hand, low concentration may result in a film with pinholes. Therefore, the above solution concentrations were chosen after careful optimization and several attempts of creating the polymer films.
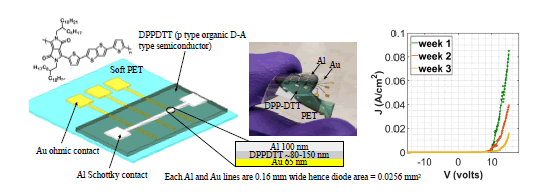
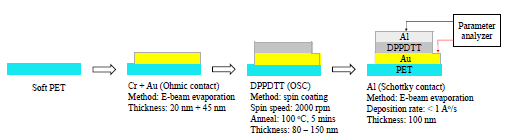
Fig. 2 shows the fabrication process of Schottky diode presented in this paper. Devices were deposited on flexible PET substrate. First, a fabrication recipe was optimized by using hard substrate of SiO2. On this substrate, chromium and gold, 20 nm and 45 nm respectively were deposited by e-beam evaporation at a rate of 2 Ao/s in vacuum. Chromium was used as an adhesion layer, as it forms alloy with gold and has an affinity to silicon substrate. Following this, organic polymer was spin coated on top of gold electrodes, at 2000 rpm for 40 seconds, which gives about 100 nm thick and uniform polymer film. Each sample was annealed at 100 oC for 5 minutes to remove residual solvent. The samples were then kept in vacuum at pressure less than 10−6 torr for about 12 hours to remove unintentional water and oxygen doping from the surface of the polymer. These dopants could make the intended Schottky junction to behave as ohmic if the junction is highly doped [18], [19]. The shadow mask evaporation of aluminum in vacuum was done to deposit 100 nm of aluminum at a rate of 0.7 Ao/s. Aluminum deposition was done as a last step of fabrication, to eliminate the possibility of alumina formation at the junction. The shadow masks for both gold and aluminum were created using laser cutting printer. Fig. 3 shows the schematic of fabricated devices, epitaxial stack of the device and a photo of realized diode on soft PET substrate.
The deposition rate for the last metal layer also played an important role. If the deposition rates are too high, the metal penetrates the thin film organic layer and causes metal-polymer interface to behave as metal-metal contact [15]. This will degrade the diode I-V characteristic from exponential to linear. To retain the exponential diode behavior, the deposition rate less than 1 Ao/s was optimized in the fabrication.
C. Instrumentation
The polymer solution was prepared using Corning PC-320 magnetic stirrer. The masks for gold and aluminum were printed using laser cutting printer. Gold and aluminum were deposited using CHA Evaporator by electron beam evaporation technique. The thickness of deposited materials was measured using a profilometer and width of the metal contact was measured using an optical microscope. The output I-V measurements were carried out with Keithley 4200 SCS by applying bias voltage in the desired range and by measuring the resulting current.
III. Results and Discussion
Device characterization was done within 2 hours of fabrication at room temperature and in atmosphere as opposed to inert environment. First, the diodes were fabricated with DPP-DTT in DCB solution which resulted in a non-uniform polymer film because of the high viscosity of the solvent. Then the diodes were fabricated with DPP-DTT in chloroform solvent; lower viscosity of chloroform as compared to DCB resulted in relatively uniform film thickness. Fig. 3 shows the exponential nature of current with voltage for the fabricated devices. The knee voltage is approximately 10 volts which is quite high for the required low power operation for power rectification for wearables. Ideally the desired voltage value is under 1 volt. The knee voltage reported for other organic polymers like MEH-PPV, rr-P3HT, etc. (these polymers has lower mobility compared to DPP-DTT) is less than 1 volt; it is reasonable to assume that knee voltage was high for fabricated devices due to presence of non-idealities in our device. Interface states or metal oxide layer in polymer-metal interface as well as water induced impurities in bulk of organic semiconductor are possible non-idealities in the fabrication process [20].
The device performance is observed for 3 weeks. The devices were kept in the air after fabrication. Device characterization was done in 1 week interval and the observed I-V performance is shown in Fig. 3 (right). The current density decreased as the device aged. This is due to addition of aforementioned non-idealities while the device aged in air.
A. Theoretical Model
Theoretical models exist to quantitatively describe the charge conduction mechanism in semiconducting material used for Schottky diodes. For organic semiconductors, the nonlinear I-V behavior is explained using Poole-Frankel emission, or thermionic emission, or space charge limited conduction (SCLC) [21]. To determine the model applicable to explain electronic transport in our Schottky barrier junctions, we consider these analytical models to understand which fits the measured I-V curve the best. This further allows us to deduce material parameters of semiconductor used for Schottky diode.
Poole-Frenkel emission explains how electrical insulators can conduct electricity. Electrons trapped in localized states move to conduction band because of random thermal fluctuations, more frequently in presence of electric field and require less thermal energy, move to different location for brief time, and fall back to other localized state. The resulting current density as per Poole-Frenkel emission is expressed by [22]
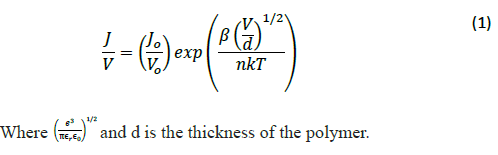
SCLC conduction mechanism is dependent on trap states between metal and polymer interfacial layer [19]. The resulting current density because of SCLC is expressed by
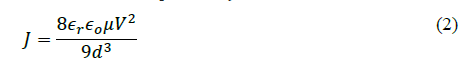
where μ denotes carrier mobility.
For thermionic conduction model, electrons cross the work function barrier because of thermal energy or applied electric field and move to the conduction band. The charge carriers then follow band transport mechanism. The resulting current density is expressed by [19], [23], [24]

The ideality factor, n, the ratio of width of band tail and temperature, approaches 1 for highly ordered semiconductor. Other processes, e.g. tunneling or recombination in the depletion region may also be involved and accounted for in the model by choosing a higher value of ideality factor. For organic semiconductors, n varies between 1.2 to 8 whereas 1 to 1.6 for inorganic semiconductor. In the above equation, Jo is the saturation current in reverse bias of the diode; once known, Richardson equation can be used to calculate the barrier height (φb) [19], [23]
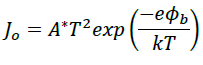
where A* is Richardson constant. The value of A∗ is 120 A/cm2K2 for free electron. Similarly, the value for ideality factor can be calculated as

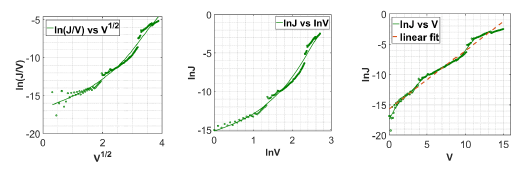
To determine the dominant charge transport mechanisms, we analyze the I-V curve. For Poole-Frenkel emission, ln(J/V ) vs V1/2 is linear. For thermionic emission, ln(J) vs V is linear. Finally, if ln(J) vs ln(V ) is linear, the conduction is due SCLC mechanism and expected slope is 2. These plots are shown in Fig. 4. The circle marker represents the values obtained from measurement, while the solid lines are the curve fit using cubic spline interpolation on the measured data.
B. Measured Results
For the device characterized within 2 hour after fabrication, log(J/V ) vs V 1/2 is non-linear, as shown in Fig. 4a. Hence it removes the possibility of Poole-Frenkel conduction mechanism. Moreover, ln(J) vs ln(V ) is nonlinear as shown in Fig. 4b and the slope when fitted is 4 for the whole plot and 8 for the high voltage region, while the required slope is 2 for SCLC. Hence this observation eliminates the SCLC transport mechanism. ln(J) vs V is linear as in Fig. 4c, which suggests that thermionic emission mechanism is the dominant charge conduction mechanism in the fabricated DPP-DTT/Al Schottky diode.
ln(J) vs V plot when fitted to a straight line using linear regression (see dotted line in Fig. 4c), the value of y intercept and slope of the straight line gives saturation current and ideality factor of diode, respectively. Using saturation current, we find the barrier height using equation 4. The calculated saturation current, barrier height and ideality factor are Jo = 1.55 x 10−7 A/cm2, φb = 0.8 eV, and n = 30 respectively.
One of the most widely used organic polymer, MEHPPV is reported to have maximum current density of 0.01 A/cm2 [25], and an ideality factor of 10.7. Although our proposed devices show higher current, the ideality factor is also high. This high value of ideality factor arises from the interface states in the active layer, formation of oxide layer at the polymer-metal interface as well a from the water induced impurity states in the bulk of organic semiconductor. These nonidealities deviate the device performance from an ideal exponential type I-V characteristic. High ideality-factor is also the reason for the diode to show a high knee-voltage when compared to other polymers.
Prior made DPPDTT Schottky diode in nitrogen chamber [14] has shown current densities less than 0.01 A/cm2. Comparison of our air made DPPDTT Schottky shows that the active layer is rather robust to fabrication steps. Nevertheless, it is known that the DPPDTT layer is susceptible to moisture [14]. We also notice a higher knee voltage in the device due to non-uniformity in the barrier height. Because of aforementioned non-idealities, the structure of polymermetal interface is disordered; the effective environment for all metal atoms are different, resulting in Gaussian barrier height distribution [26]. This implies an inhomogeneity at the interface of organic semiconductor. In future work, this inhomogeneity should be reduced to obtain device with a lower knee voltage. A potential approach is use of additives in organic semiconductors to blend with solution processable organic semiconductors. The resulting hybrid material has demonstrated crystal growth that can be controlled [27]. The additive could be small molecule additives, polymer additives, or nanoparticles. This is known to improve material crystallization, enhance uniformity, and improve charge transport. Additives will be blended in DPPDTT polymer in future work achieve improve device performance in terms of ideality factor and maximum current density.
IV. Conclusion
Air made DPP-DTT Schottky diode was fabricated using gold as ohmic contact and aluminum as Schottky contact. The diode is explored to reduce production costs and make the fabrication process simpler as compared to fabrication inside nitrogen chamber. Current-voltage characteristics and charge transport methods were studied, and conclusion was made that the thermionic emission is the dominant mechanism in DPP-DTT/Al barrier junction diodes. Electronic parameters including barrier height, ideality factor and reverse saturation current are calculated to be φb = 0.8 eV, n = 30, and Jo = 1.55 x 10−7 A/cm2 respectively. The DPPDTT Schottky diode presented here is fabricated on PET substrate for proof of concept, but it can also be fabricated in other substrates such as PEN tape and transferred to clothing materials, which makes it appealing for possible wearable applications.
Acknowledgment
This project was supported by the NASA MIRO Center for Research and Education in 2D Optoelectronics, Grant no.
80NSSC19M0201
References
- Lin CY (2011) High-frequency polymer diode rectifiers for flexible wireless power-transmission sheets, Org. Electron 12: 1777–1782.
- Li M, Heljo PS, Lupo D (2014) Organic rectifying diode and circuit for wireless power harvesting at 13.56 MHz, IEEE Trans. Electron Devices 61: 2164–2169.
- Sani N (2014) All-printed diode operating at 1.6 GHz, Proc Natl Acad Sci 111: 11943–11948.
- Semple J, Georgiadou DG, Wyatt-Moon G, Gelinck G, and T. D. Anthopoulos (2017) Flexible diodes for radio frequency (RF) electronics: a materials perspective, Semicond. Sci. Technol., 32:012-123002.
- H. N. Tsao (2009)The influence of morphology on high-performance polymer field-effect transistors. Adv. MateR 21: 209–212.
- Y. Li, S. P. Singh, and P. Sonar (2012) A high mobility P-type DPP-thieno[3,2-b]thiophene copolymer for organic thin-film transistors. Adv. Mater 22: 4862–4866.
- H. Bronstein (2011) Thieno [3, 2-b] thiophene- diketopyrrolopyrrole-containing polymers for high-performance organic field-effect transistors and organic photovoltaic devices. J. Am. Chem. Soc 133: 3272–3275.
- X. Zhang et (2011)Molecular packing of high-mobility diketo pyrrolo-pyrrole polymer semiconductors with branched alkyl side chains. J. Am. Chem. Soc 133: 15073–15084.
- Y. Li, P. Sonar, S. P. Singh, M. S. Soh, M. van Meurs, and J. Tan (2011) Annealing-Free High-Mobility Diketopyrrolopyrrole- Quaterthiophene Copolymer for Solution-Processed Organic Thin Film Transistors. J. Am. Chem. Soc 133:2198–2204.
- Y. Li, P. Sonar, S. P. Singh, W. Zeng, and M. S. SoH (2011)3, 6-Di (furan-2-yl) pyrrolo [3, 4-c] pyrrole-1, 4 (2H, 5H)-dione and bithiophene copolymer with rather disordered chain orientation showing high mobility in organic thin film transistors. J. Mater. Chem 21: 10829–10835.
- T. L. Nelson (2010) Transistor paint: high mobilities in small bandgap polymer semiconductor based on the strong acceptor, diketopyrrolopyrrole and strong donor, dithienopyrrole. Adv. Mater 22: 4617–4621.
- J. Li (2012)A stable solution-processed polymer semiconductor with record high-mobility for printed transistors, Sci. Rep 2:1-9.
- G. Zhang, Y. Fu, Z. Xie, and Q. Zhang (2001) Synthesis of low bandgap polymer based on 3, 6-dithien-2-yl-2, 5-dialkylpyrrolo [3, 4-c] pyrrole-1, 4-dione for photovoltaic applications, Sol. energy Mater. Sol. Cells 95:1168–1173.
- M. Nikolka (2009) High-mobility, trap-free charge transport in conjugated polymer diodes. Nat. Commun 10:1-9.
- H. Campbell and D. L. Smith (2000) Semiconducting Polymers: Chemistry, Physics and Engineering, ed. G. Hadziioannou, and PF van Hutten. Weinheim: Wiley-VCH.
- K. Barbalace, Periodic Table of Elements. [Online]. Available: https://environmentalchemistry.com/yogi/periodic
- R. H. Friend (1999) Electroluminescence in Conjugated Polymers. Nature 121–128.
- E. J. Meijer, C. Detcheverry, P. J. Baesjou, E. Van Veenendaal, D. M. De Leeuw, and T. M. Klapwijk (2003) Dopant density determination in disordered organic field-effect transistors. J. Appl. Phys 93: 4831–4835.
- S. M. Sze(2008)Semiconductor devices: physics and technology. John wiley & sons.
- K. Hess( 2000)Advanced theory of semiconductor devices.
- D. R. Lamb (1967) Electrical conduction mechanisms in thin insulating films. Methuen.
- J. Frenkel (1938) On pre-breakdown phenomena in insulators and electronic semi-conductors. Phys. Rev 54:8- 647.
- R. K. Gupta and R. A. Singh (2004) Schottky diode based on composite organic semiconductors. Mater. Sci. Semicond. Process 7:83–87.
- H. Tomozawa, D. Braun, S. Phillips, A. J. Heeger, and H. Kroemer (1987) Metal-polymer Schottky barriers on cast films of soluble poly (3-alkylthiophenes). Synth. Met 22: 63–69.
- M. Zhu, T. Cui, and K. Varahramyan (2004) Experimental and theoretical investigation of MEH-ppv based Schottky diodes. Microelectron. Eng 75: 269–274.
- S. Bandyopadhyay, A. Bhattacharyya, and S. K. Sen (1999) Measurements and modelling of the barrier heights and ideality factors in the metal/conducting polymer composite Schottky device. J. Appl. Phys 85: 3671–3676.
- Z. He, Z. Zhang, and S. Bi (2020) Nanoparticles for organic electronics applications, Mater. Res. Express, vol. 7:1- 12004.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi