Research Article, J Regen Med Vol: 11 Issue: 5
An Acellular Biologic Extracellular Matrix Envelope for Cardiovascular Implantable Electronic Devices: Preclinical Evaluation
Daniel Deegan1, Shawn K Piasecki2, Jerome B Riebman3*
1Principal Scientist at Aziyo Biologics Inc, Silver spring, United States
2Senior Director, Medical Affairs at Aziyo Biologics Inc, Silver spring, United States
3Chief Medical Officer, Aziyo Biologics Inc, MD, Silver spring, United States
*Corresponding Author: Jerome B Riebman
Chief Medical Officer, Aziyo Biologics Inc, MD, Silver spring, United States
Tel: 240-247-1170
E-mail: jriebman@aziyo.com
Received: 20-Aug-2022, Manuscript No. JRGM-22-72434;
Editor assigned: 22-Aug-2022, PreQC No. JRGM-22-72434(PQ);
Reviewed: 05-Sep-2022, QC No. JRGM-22-72434;
Revised: 07-Sep-2022, Manuscript No. JRGM-22-72434(R);
Published: 14-Sep-2022, DOI:10.4172/2325-9620.1000226
Citation: Deegan D, Piasecki SK, Riebman JB (2022) An Acellular Biologic Extracellular Matrix Envelope for Cardiovascular Implantable Electronic Devices: Preclinical Evaluation. J Regen Med 11:5.
Abstract
Background: Cardiovascular ?mplantable Electronic Device (CIED) implant sites are prone to fibrotic capsule formation which can lead to device movement, infection risk, and reoperative challenges. Using a biologic CIED envelope at implant may promote constructive tissue remodeling of the pocket and improve outcomes.
Methods: Eleven rabbits received Pacemakers (PMs) in porcine small intestinal submucosa extracellular matrix (SIS ECM) envelopes (PM+ECM) and ten without (PM ONLY). Implant sites were analyzed by observation and histopathology at either week 2, 13, or 26.
Results: PM+ECM animals had a 5-fold reduction in PM subcutaneous movement (device flipping) compared to the PM ONLY group (p=.027). ECM envelopes showed progressive resorption and increasing vascularization at 13 and 26 weeks compared to the PM ONLY group. PM+ECM animals had a lower average capsule thickness compared to the PM ONLY group at 13 and 26 weeks, however the differences were not statistically significant at either time point (p>0.05). Neovascularization scored significantly higher in the PM+ECM group vs. PM ONLY at both 13 and 26 weeks (p=0.046 and p<0.001, respectively).
Conclusion: Use of decellularized ECM envelopes promoted PM implant site healing remarkable for stabilization and increased vascularity, compared to PMs without envelopes in a preclinical model. ECM envelopes may provide meaningful clinical benefits and warrant further investigation.
Keywords: Extracellular Matrix; Cardiovascular Implantable Electronic Device; Biologic Envelope; CIED; CIED Envelope; TYRX; CanGaroo; Pacemaker; Implantable Cardioverter Defibrillator.
Introduction
Treatment of cardiac arrhythmias and conduction issues may include pharmacotherapy, surgery, or implantation of a Cardiac İmplantable Electronic Device (CIED), such as a pacemaker, İmplantable Cardioverter-Defibrillator (ICD), or a device for Cardiac Resynchronization Therapy (CRT). The expansion of indications for these life saving devices has led to more patients, and younger patients, benefitting from them. However, the increasing rate of implantation has led to an even greater increase in complications, potentially due to more comorbid patients receiving these devices [1,2]. Additionally, younger patients and geriatric patients surviving longer pose a unique challenge as they potentially survive long enough to require multiple generator changes and/or lead revisions throughout their lifetime [1,2].
Upon initial (de novo) implantation, postoperative complications such as infection, hematoma, lead dislodgement, seroma, generator migration and/or erosion, and implant site discomfort occur and mostly affect patients in high-risk groups (comorbid, corticosteroid use, end-stage renal disease, etc.) [3,4]. Following standardized guidelines and proper surgical technique can mitigate many of these complications [5,6]. However, some complications arise due to issues related to the surgical implant site of the CIED (migration, erosion, infection, discomfort, seroma, hematoma) or issues relating to the leads attached to the CIED generator (dislodgement, perforation, infection), which can result in additional procedures and risk for patients [6,7]. Device envelopes have been commercially developed to promote stabilization of implanted devices, which can help mitigate complications related to movement of the device within the subcutaneous pocket, such as generator migration, erosion, lead movement/dislodgement, seroma, discomfort, or even changes in electrical vectors for ICDs [8,9].
Complication rates increase significantly after generator or lead exchange and upgrade or revision surgery, which has been linked to fibrotic adhesions on the generator and/or leads within the device pocket [2,10-12]. Medical device implants (such as CIEDs) evoke a chronic macrophage activation as part of the foreign body reaction in the subcutaneous tissues that may result in significant fibrotic tissue surrounding the CIED and/or leads, which can make future removal or repositioning of both components difficult [11-14]. Once the CIED and/or leads are encased in fibrotic tissue, the capsule can also act as a safe harbor for bacterial growth. Cultures from fibrotic capsules have tested positive for Staphylococci species even in clinically asymptomatic patients, which may be released during re-intervention procedures, causing an increased infection risk for patients undergoing multiple re-operations [10]. In addition to an increased risk of infection, other complications include increased procedure time, damage to the leads during attempts to free them from the fibrotic tissue, and/or bleeding and hematoma as a result of capsulectomy, pocket debridement, and/or lead dissection [15,16]. Therefore, interventions that potentially simplify future dissection of the device pocket and reduce potential fibrotic tissue burden may have a positive effect on complication rates and future revision surgeries [17].
Porcine Small İntestinal Submucosa Extracellular Matrix (SIS ECM) is a biologic material that has been well-characterized in multiple regenerative medicine applications and is rich in growth factors, such as basic fibroblast growth factor (bFGF) and Vascular Endothelial Growth Factor (VEGF), Glycosaminoglycans (GAGs), and structural proteins, such as collagens and fibronectin [18- 20]. Non-biologic biomaterials (such as CIEDs or synthetic nonabsorbable or absorbable materials) do not naturally possess these biocompatible or bioactive characteristics. The native SIS ECM structures and bioactive factors have the ability to shift the foreign body response to stimulate healthy tissue growth and angiogenesis [19,21]. Upon implantation, SIS ECM naturally degrades over time, which releases its intrinsic biologic signals that have been shown to mitigate hypovascular fibrotic encapsulation normally caused by an inflammatory response to non-biologic implanted devices [22]. Techniques to increase vascularity of capsules surrounding nonbiologic implants may be important since it would allow circulating immune cells direct access to the area, which may decrease the risk of bacterial colonization on or around the device and therefore potential subsequent infection.
To help mitigate device implant site complications due to migration of the device and the foreign body reaction, an envelope manufactured from SIS ECM has been created. The potential clinical role biologic SIS ECM envelopes play in CIED device implantation was evaluated in a preclinical model.
Materials and Methods
Animal model
A subcutaneous implantation was performed in New Zealand White (NZW) rabbits using dorsal bilateral surgeries to implant pacemaker devices with or without supportive decellularized SIS ECM envelopes. The NZW rabbit was chosen because it provided enough surface area on the dorsal surface for implantation of full-size clinical test and control articles (pacemakers approximately 5.5 x 5.5 x 0.5 cm). The rabbit is an appropriate subcutaneous model for evaluating biocompatibility and local effects of implanted materials according to the current International Organization for Standardization testing standards (ISO 10993-6). Tissue erosion, cardiac device migration, and local tissue responses including neovascularization and capsule formation were evaluated.
Study design
The study protocol was approved by the Institutional Animal Care and Use Committee (IACUC) and all animals received humane care in compliance with “The Guide for the Care and Use of Laboratory Animals,” published by the US National Institutes of Health. The study rabbits were non-pregnant females, aged 15–16 weeks, weighing 2.6 – 3.3 kg, individually housed in cages, fed with certified commercial feed, and provided water ad lib for the duration of the study.
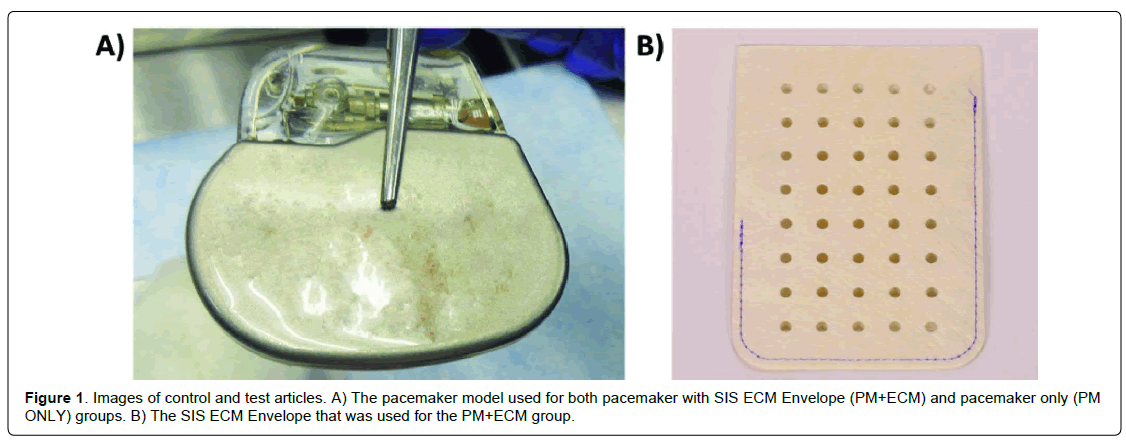
Twenty-one NZW rabbits received Pacemaker (PM) implants divided into two groups –eleven with PMs inside SIS ECM envelopes (PM+ECM) and ten with no envelope (PM ONLY) (Figure 1). After anesthesia and aseptic preparation, bilateral subcutaneous pockets were created through midline incisions on the dorsal lumbar aspect of each animal. Minimal necessary pocket dissection was performed to accommodate one implant per pocket, and all pockets were the same size.
For the eleven PM+ECM animals, single-chamber pacemakers (St. Jude Medical – now Abbott Laboratories, Chicago, IL) without the lead were each placed within a decellularized SIS ECM envelope (CanGaroo® Envelope, size Medium, Aziyo Biologics, Silver Spring, MD) prepared per manufacturer instructions. The pacemaker lead header was placed nearest the opening of the envelope and the lead header was oriented in the implant pocket closest to the incision. Each implant was laid flat with no overlapping sections/folds of the envelope within the pocket. The PM+ECM animals had their devices secured with a single, non-absorbable suture (e.g. 3-0 Prolene) passed through the pacemaker header suture hole, across the envelope opening, and secured to the underlying chest wall. As a control, ten PM ONLY animals received identical pacemakers implanted following the same surgical technique as described above, but without envelopes. Similarly, the devices were laid flat within the subcutaneous pocket, with the lead header oriented closest to the incision. PM ONLY devices were secured with a single, non-absorbable suture passed through the pacemaker header suture hole and secured to the underlying chest wall. The subcutaneous tissues for both groups were closed using a continuous stitch and non-absorbable suture. Standard surgical techniques were used to finish closing the implant sites, and the animals were allowed to recover after being returned to standard individual housing.
One PM+ECM animal was evaluated at 2 weeks post implant (timepoint selected to allow for skin incision healing) to provide a histology comparison point for baseline SIS ECM implantation. For the remaining ten animals in each group, four animals were evaluated at week 13 and six were evaluated at week 26. Implant sites were then assessed using a standard protocol including gross observation, surgical dissection and exposure with macroscopic observation, and tissue specimen harvesting for histopathology.
All animals were humanely euthanized at the end of their scheduled duration. For gross necropsy, the area over the implant sites was shaved, and the lateral (outer) capsule surrounding the pacemaker at each implant site was surgically exposed and photographed by removing the layers of epidermis and dermis. Macroscopic observations were documented, including skin erosion or wound healing issues (prior to surgical exposure), and any evidence of envelope resorption and pacemaker migration from the envelope or implant area. Presence of fluid, capsule thickening, and/or discoloration were also noted. The pacemaker was then removed, and the full capsule was excised and marked with tissue dye for orientation. The tissue was fixed in formalin for histopathological analysis.
Histopathology and capsule measurements
Two tissue pieces 1 cm in length were excised for histology from the middle and the dorsal corner of each capsule. Samples were processed by standard histopathology techniques including paraffin embedding, sectioning (two serial sections at each sample midpoint), and staining (one section per stain with Hematoxylin and Eosin [H&E] and Masson’s Trichrome). Slides were evaluated by a blinded veterinary pathologist for local tissue responses, envelope resorption, and capsule formation. This analysis scored the appearance of mineralization, necrosis (including cellular debris from degenerating inflammatory cells), neovascularization, and capsule formation on an established scale of 0 to 4 (0 meaning absent and 4 meaning marked presence). The average of these scores was used to compare results between groups, and student’s t-test was used to calculate p values. Values were considered statistically significant if p<0.05.
For assessment of SIS ECM envelope resorption, a baseline estimation of envelope area was made in the slides from an initial 2-week time point in a PM+ECM animal. Subsequent measurements of remnant SIS ECM in the capsular tissue were made at 13 and 26 weeks and standardized to the 2-week time point for the remaining PM+ECM animals. In addition to resorption, thickness measurements of the tissue capsule were made at three (3) representative places from each individual H&E and trichrome slide of each tissue sample, excluding remnant envelope material in the measurements and was measured with Nikon Elements (Nikon Instruments Inc., Melville, NY). The average of these measurements was used to estimate fibrotic capsule thickness from every individual implant site and subsequently averaged for each experimental group.
Results
All envelopes and pacemakers were prepared and implanted without incident, and all animals remained healthy during the study duration and survived until their scheduled termination at week 2, 13, or 26.
Gross necropsy observations
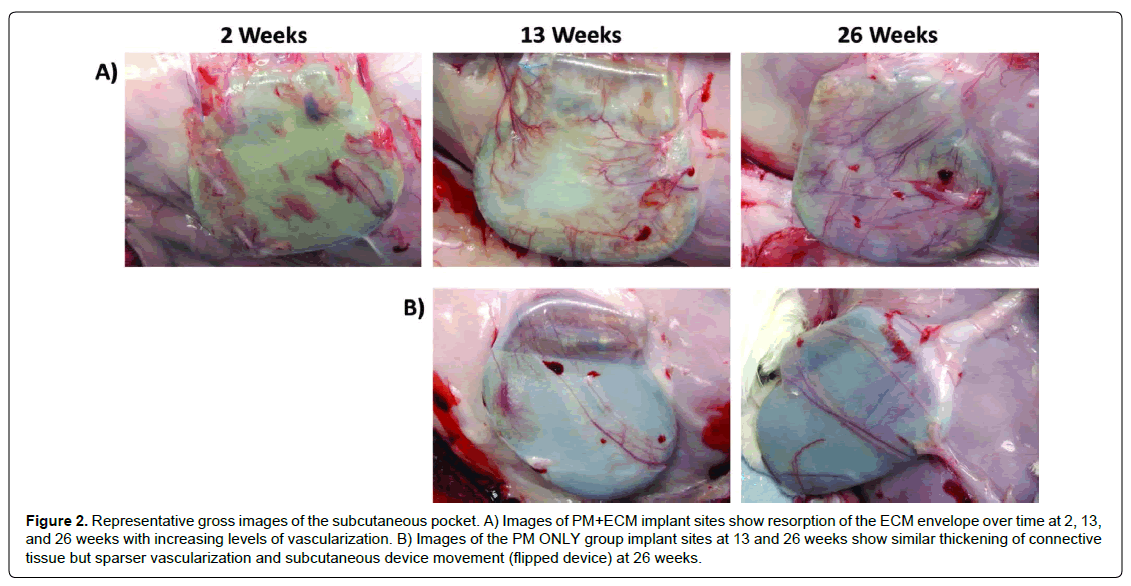
No erosions of the tissue overlying the pacemaker implants were observed in either the PM+ECM or PM ONLY groups through 26 weeks. Subcutaneous device movement (manifested as device flipping 180° cranially within the pocket) was documented during necropsy in 50% (5 of 10 total, 2 at 13 weeks and 3 at 26 weeks) of the PM ONLY group sites (Table 1, Figure 2), compared to only 10% (1 of 10 total, at 26 weeks) in the PM+ECM group (p=0.027).
| Pacemaker + SIS ECM Envelope (PM+ECM) |
Pacemaker only (PM ONLY) |
p-values (PM+ECM vs. PM ONLY) |
||||
|---|---|---|---|---|---|---|
| Implant Duration | 13 wks | 26 wks | 13 wks | 26 wks | 13 wks | 26 wks |
| Neovascularization Score (0 – 4)* | 2.0 ± 0.0 | 2.1 ± 0.3 | 1.6 ± 0.7 | 1.6 ± 0.5 | .046 | <.001 |
| Capsule Thickness (μm) | 349.3 ± 165.2 | 427.5 ± 116.0 | 417.3 ± 152.5 | 437.9 ± 141.4 | .235 | .820 |
| Subcutaneous Device Movement (total %) | 1/10 (10%) | 5/10 (50%) | .027 | |||
Table 1. Comparison between independent histopathologic evaluation of the pacemaker + SIS ECM Envelope (PM+ECM) group and the pacemaker only (PM ONLY) group at 13 and 26 weeks.
Figure 2: Representative gross images of the subcutaneous pocket. A) Images of PM+ECM implant sites show resorption of the ECM envelope over time at 2, 13, and 26 weeks with increasing levels of vascularization. B) Images of the PM ONLY group implant sites at 13 and 26 weeks show similar thickening of connective tissue but sparser vascularization and subcutaneous device movement (flipped device) at 26 weeks.
Upon examination of the subcutaneous pocket, thickening of the capsule increased with implant duration time, and mild thickening of all capsules was observed in both groups by 26 weeks. Capsule thickness was quantified at the 13- and 26-week time points (see Histopathology section). Capsule color was highly variable within and between groups, and vascularization was clearly visible in all sites (PM+ECM and PM ONLY) at 26 weeks. Upon gross inspection, the observed amount of vascularization was visually greater in the PM+ECM group than in the PM ONLY group at 13 and 26 weeks (Figure 2), which was quantified during pathologic evaluation.
Histopathology
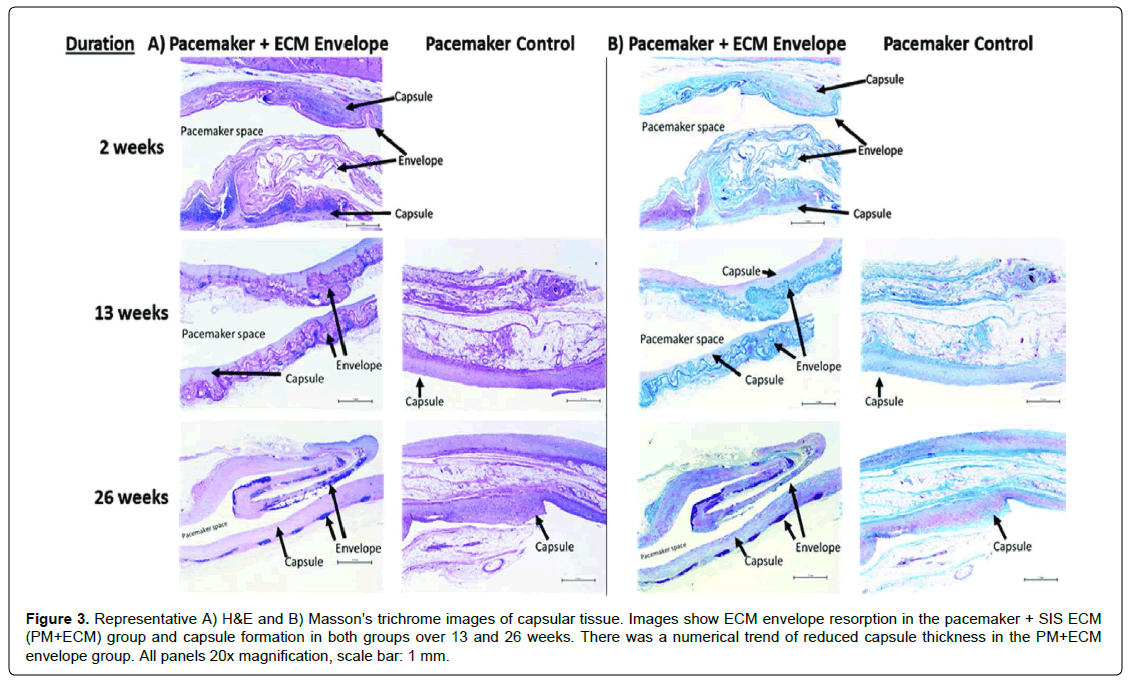
Histology was performed to quantify and score the capsular tissue response. Trichrome and H&E stains demonstrated similar normal collagen formation and fibrous connective tissue organization in both groups (Figure 3). The PM+ECM implant sites had a lower average capsule thickness compared to PM ONLY sites at 13 weeks (349.3 ± 165.2 μm vs. 417.3 ± 152.5 μm) (mean ± SD) and at 26 weeks (427.5 ± 116.0 μm vs. 437.9 ± 141.4 μm) (Table 1). However, these differences did not reach statistical significance.
Figure 3: Representative A) H&E and B) Masson’s trichrome images of capsular tissue. Images show ECM envelope resorption in the pacemaker + SIS ECM (PM+ECM) group and capsule formation in both groups over 13 and 26 weeks. There was a numerical trend of reduced capsule thickness in the PM+ECM envelope group. All panels 20x magnification, scale bar: 1 mm.
Histology specimens were also scored for neovascularization by an independent blinded pathologist using a 5-point scale (0-absent to 4-marked) (Table 1). Mean neovascularization scores were similar at both 13 and 26 weeks within each individual group, however, the PM+ECM group had a significantly higher average score at both timepoints compared to the PM ONLY group: 2.0 ± 0.0 vs. 1.6 ± 0.7 (13 weeks, p=.046) and 2.1 ± 0.3 vs. 1.6 ± 0.5 (26 weeks, p<0.001). Minimal to no mineralization or necrotic tissue was observed in both groups, and only low levels of apoptotic cells normally seen in surgical sites were found.
Envelope resorption in the PM+ECM group was quantified in histology slides as the average area of remnant material at each timepoint standardized to material present at the 2-week baseline timepoint. A decreasing trend of progressive envelope resorption was seen at both 13 and 26 weeks (average of 60.6% and 73.2% resorbed, respectively (Table 2).
| Implant Duration | 2 weeks (baseline) |
13 weeks | 26 weeks |
|---|---|---|---|
| Average area of implanted envelope material within implant sites (mm2) | 14.52 | 5.72 | 3.89 |
| % change from baseline | -- | 60.6% | 73.2% |
Table 2. Comparison of the average area of implanted material and percent envelope resorption within the capsular tissue slides taken from the pacemaker + SIS ECM Envelope (PM+ECM) implant sites at 2-, 13-, and 26-week duration.
Discussion
SIS ECM is bioactive and promotes tissue remodeling
Scaffolds derived from non-crosslinked SIS ECM contain a natural three-dimensional architecture and native bioactive components that attract and allow cells to migrate throughout the matrix. The backbone of the SIS ECM matrix consists of collagen, fibronectin, laminin, and proteoglycans, which supply attachment sites to enable cells to remodel and organize new tissue [23]. The release of bioactive components (such as growth factors, antimicrobial peptides, and other cytokines) from the SIS ECM during degradation by native host tissue proteinases is paramount for neovascularization and tissue remodeling [19,21,24-26]. Basic fibroblast growth factor (bFGF) which accelerates wound closure and regulates fibroblast activity, and vascular endothelial growth factor (VEGF) which stimulates angiogenesis, are only two of the many native growth factors that are released from SIS ECM during tissue remodeling [18,27]. Importantly, angiogenesis provides a path for host immune cells to directly access the wound bed during the remodeling process [19,21]. Host immune cell attraction and release of bioavailable factors from SIS ECM signals for a variety of other cell types which are important for tissue remodeling to come to the area including endothelial cells, epithelial cells, and mesenchymal stem cells [21,26]. Further, degradation products of SIS ECM have been shown to possess inherent antimicrobial properties, thereby assisting in the prevention of bacterial infections as the matrix remodels [23,24,27]. The natural bioactivity of SIS ECM supports an immunomodulatory pathway to promote constructive tissue remodeling, which is a native hallmark of biologic tissues [21,23]. Non-biologic materials (such as a CIED generator or the leads) do not naturally posess this bioactivity.
In this study, gross necropsy and histopathology confirmed implanted SIS ECM envelopes remodeled within surgical pockets, which aligns with previous pre-clinical observations on this material [28,29]. The biologic SIS ECM envelopes showed steady resorption over time with about 26.8% of the starting material remaining after 26 weeks. This timeline may differ in clinical application and between individual patients, however, the ability of the host tissue to utilize and remodel the biologic SIS ECM envelope into native tissue is expected to be conserved.
SIS ECM stabilizes implanted devices within remodeled, vascularized tissue
Device envelopes are intended to securely hold implantable electronic devices (CIEDs or neurostimulators) to create a stable environment once implanted in the body. This report demonstrates the ability of SIS ECM envelopes to secure pacemaker devices through cellular and vascular tissue capsules in a clinically relevant rabbit model. This may explain why in a recent clinical publication, there were no reports of device migration, erosion, or need for pocket revision after CIED implantation with the same ECM envelope [17]. Similarly, no migration from the implant site or skin erosions were found in either group at 26 weeks in this model. However, the PM+ECM group had significantly less device movement within the subcutaneous pocket. The subcutaneous device movement (flipping) seen in the PM ONLY group creates a relative clinical concern, as device movement at the implant site is a cause of lead dislodgement in clinical patients, potentially leading to complications and/or reoperation [30]. Additionally, device movement within the subcutaneous pocket can create friction and abrasion, which in longer term models or clinical cases could contribute to erosion and subsequent infection [31].
Growing indications for CIED implantation, increasing life expectancy, and younger patients receiving these devices means that reoperation, and sometimes multiple reoperations, are a reality for patients living with these devices [1,2]. Implanted non-biologic devices (such as CIEDs) trigger a foreign body reaction, which potentially leads to significant fibrosis encapsulating the device generator and/ or leads [11–14]. Fibrotic entrapment of the generator and/or leads increases the difficulty of revision procedures and contributes to the increased complication rates observed for revision procedures compared to those reported for de novo implantations [11,12,15,16]. Securing devices in vascular tissue within the pocket may ease device changeouts by reducing generator and/or lead dissection from a thick fibrous capsule, which could potentially reduce complications during revision procedures [17].
SIS ECM stimulates more vascularized capsules (vs. no envelope)
Histologically, SIS ECM envelope use was significantly correlated with more angiogenesis and neovascularization versus no envelope use. The neovascularization scoring (where a greater score represented more capillary proliferation) showed that capsular tissue in the PM+ECM group scored significantly higher than the capsules of the PM ONLY group at both timepoints. This result is at least partially due to SIS ECM’s native growth factor milieu, including transforming growth factor beta (TGF-β), bFGF, and VEGF. These bioactive proteins are not only anti-inflammatory but promote angiogenesis by upregulating endothelial cell migration and proliferation [18,23,27]. Natural degradation of the SIS ECM matrix also releases matricryptic peptides, which have similar angiogenic effects on both endothelial cells and perivascular stem cells [25,32]. The natural porosity of SIS ECM scaffolds aids cell infiltration and ultimately stimulates fast and efficient vessel and capillary growth [23]. The angiogenic benefits observed in this study have also been described in a variety of other clinical cardiovascular applications of SIS ECM, such as intracardiac repair and pericardial reconstruction [21,33,34].
SIS ECM is associated with a reduction in fibrous capsule formation (vs. no envelope)
In addition to stabilizing CIEDs within vascular, remodeled tissue, SIS ECM in the PM+ECM group correlated with a general reduction in the thickness of the device capsule. This numerical trend was consistent at each timepoint but did not reach statistical significance. Since thickening of the capsule increased with implant duration in both groups, future studies are needed to uncover potential long-term differences that cannot be addressed from this study.
As mentioned previously, SIS ECM promotes a favorable environment for constructive tissue remodeling which can mitigate excessive fibrosis. The mechanism of action for this characteristic is that non-crosslinked SIS ECM elicits M2 macrophage polarization and production of interleukin 10 (IL-10) and other anti-inflammatory cytokines upon implantation [35-38]. This unique ability is not observed for the implantation of devices such as CIEDs, which stimulate chronic activity of the pro-inflammatory M1 macrophage phenotype, often resulting in a thick, fibrous capsule [14,36,38]. Mitigation of a thick, fibrous capsule may ease device changeouts during future revision procedures, which could lead to a reduction in clinical complications [17].
Conclusion
Several clinically relevant biologic advantages of using SIS ECM envelopes during the implantation of CIEDs were observed in this study. The small number of animals included in this study and used as a control is a limitation, however, the purpose was to observe trends in the data to direct future studies. Pacemakers implanted with SIS ECM envelopes (PM+ECM) in general had thinner fibrous capsules that were significantly more vascularized than the pacemaker only (PM ONLY) group. This study aligns with the hypothesis that SIS ECM envelopes steadily resorb and are replaced by native, organized collagen over time that is highly vascularized and free of mineralization. These tissue characteristics were associated with a significant reduction in pacemaker movement observed within the subcutaneous pocket in the PM+ECM group compared to the PM ONLY group. This study demonstrates that SIS ECM envelopes foster a pro-remodeling microenvironment to secure CIEDs after implantation.
References
- Beck H, Boden WE, Patibandla S, Kireyev D, Gutpa V, et al. (2010) 50th anniversary of the first successful permanent pacemaker implantation in the United States: Historical review and future directions. Am J Cardiol, 106(6): 810-818.
- Tarakji KG, Ellis CR, Defaye P, Kennergren C (2016) Cardiac implantable electronic device infection in patients at risk. Arrhythm Electrophysiol Rev, 5(1): 65-71.
- Tompkins C, McLean R, Cheng A, Brinker JA, Marine JE, et al. (2011) End-stage renal disease predicts complications in pacemaker and ICD implants. J Cardiovasc Electrophysiol, 22(10): 1099-104.
- Lin YS, Hung SP, Chen PR, Yang CH, Wo HT, et al. (2014) Risk factors influencing complications of cardiac implantable electronic device implantation: infection, pneumothorax and heart perforation: a nationwide population-based cohort study. Medicine (Baltimore), 93(27): e213.
- Blomstrom-Lundqvist C, Traykov V, Erba PA, Burri H, Nielsen JC, et al. (2020) European Heart Rhythm Association (EHRA) international consensus document on how to prevent, diagnose, and treat cardiac implantable electronic device infections-endorsed by the Heart Rhythm Society (HRS), the Asia Pacific Heart Rhythm Society (APHRS), the Latin American Heart Rhythm Society (LAHRS), International Society for Cardiovascular Infectious Diseases (ISCVID) and the European Society of Clinical Microbiology and Infectious Diseases (ESCMID) in collaboration with the European Association for Cardio-Thoracic Surgery (EACTS). Europace, 22(4): 515-549.
- Kusumoto FM, Schoenfeld MH, Wilkoff BL, Berul CI, Birgersdotter-Green UM, et al. (2017) 2017 HRS expert consensus statement on cardiovascular implantable electronic device lead management and extraction. Heart Rhythm, 14(12): e503-e551.
- Borleffs CJ, Thijssen J, de Bie MK, van Rees JB, van Welsenes GH, et al. (2010) Recurrent implantable cardioverter-defibrillator replacement is associated with an increasing risk of pocket-related complications. Pacing Clin Electrophysiol, 33(8): 1013-1019.
- Xiang K, Levine JD, Elayi SC, Su J, Hsu S, et al. (2019) A novel bio envelope and suture orientation for stabilization of the subcutaneous implantable cardioverter-defibrillator generator to the chest wall. HeartRhythm Case Rep, 5(8): 430-432.
- Xiang K, Catanzaro JN, Elayi C, Esquer Garrigos Z, Sohail MR (2021) Antibiotic-Eluting Envelopes to Prevent Cardiac-Implantable Electronic Device Infection: Past, Present, and Future. Cureus, 13(2): e13088.
- Wilkoff BL (2011) Infections associated with cardiac implantable electronic devices are misunderstood. Tex Heart Inst J, 38(4): 353-354.
- Biefer HR, Hurlimann D, Grunenfelder J, Salzberg SP, Steffel J, et al. (2013) Generator pocket adhesions of cardiac leads: Classification and correlation with transvenous lead extraction results. Pacing Clin Electrophysiol, 36(9): 1111-1116.
- Maytin M, John RM, Epstein LM (2015) Device Pocket Scar Predicts Transvenous Lead Extraction Difficulty. J Innov Card Rhythm Manag, 6(11): 2173-2177.
- Perry L, Karp F, Hauch K, Ratner BD (2007) Explanted Pacemakers : Observations of the Long-term Foreign Body Response. J Undergrad Res in Bioeng, 7: 13-21.
- Steckiewicz R, Swieton EB, Kolodzinska A, Bogdanska M, Fus L, et al. (2017) Morphometric parameters of cardiac implantable electronic device (CIED) pocket walls observed on device replacement. Folia Morphol (Warsz), 76(4): 675-681.
- Sood N, Martin DT, Lampert R, Curtis JP, Parzynski C, et al (2018) Incidence and Predictors of Perioperative Complications With Transvenous Lead Extractions. Circ Arrhythm Electrophysiol, 11(2): e004768.
- Poole JE, Gleva MJ, Mela T, Chung MK, Uslan DZ, et al. (2010) Complication rates associated with pacemaker or implantable cardioverter-defibrillator generator replacements and upgrade procedures: Results from the REPLACE registry. Circulation, 122(16): 1553-1561.
- Woodard DA, Kim G, Nilsson KR (2022) Risk Profiles and Outcomes of Patients Receiving Cardiovascular Implantable Electronic Devices With and Without Antibacterial Envelopes. Cureus, 14(5): e24739.
- Hodde JP, Record RD, Liang HA, Badylak SF (2001) Vascular endothelial growth factor in porcine-derived extracellular matrix. Endothelium, 8(1): 11-24.
- Brown BN, Badylak SF (2014) Extracellular matrix as an inductive scaffold for functional tissue reconstruction. Transl Res, 163(4): 268-285.
- Badylak SF (2014) Decellularized allogeneic and xenogeneic tissue as a bioscaffold for regenerative medicine: Factors that influence the host response. Ann Biomed Eng, 42(7): 1517-1527.
- Piterina AV, Cloonan AJ, Meaney CL, Davis LM, Callanan A, et al. (2009) ECM-Based Materials in Cardiovascular Applications: Inherent Healing Potential and Augmentation of Native Regenerative Processes. Int J Mol Sci, 10(10): 4375-4417.
- Badylak SF, Gilbert TW (2008) Immune response to biologic scaffold materials. Semin Immunol, 20(2): 109-116.
- Brown-Etris M, Cutshall WD, Hiles MC (2002) A new biomaterial derived from small intestine submucosa and developed into a wound matrix device. Wounds, 14(4): 150-166.
- Velnar T, Bailey T, Smrkolj V (2009) The wound healing process: An overview of the cellular and molecular mechanisms. J Int Med Res, 37(5): 1528-1542.
- Brennan EP, Reing J, Chew D, Myers-Irvin JM, Young EJ, et al. (2006) Antibacterial activity within degradation products of biological scaffolds composed of extracellular matrix. Tissue Eng, 12(10): 2949-2955.
- Reing JE, Zhang L, Myers-Irvin J, Cordero KE, Freytes DO, et al. (2009) Degradation products of extracellular matrix affect cell migration and proliferation. Tissue Eng Part A, 15(3): 605-614.
- Voytik-Harbin SL, Brightman AO, Kraine MR, Waisner B, Badylak SF (1997) Identification of extractable growth factors from small intestinal submucosa. J Cell Biochem, 67(4): 478-491.
- Shadrina DV, Venediktov AA, Evdokimov SV, Vaskovskii VA, Artyukhina EA, et al. (2020) Assessment of Biocompatibility and Local Action of Biomaterial for Production of an Envelope for Implanted Heart Electronic Devices. Bull Exp Biol Med, 168(3): 375-377.
- Ip JE, Xu L, Lerman BB (2021) Differences between cardiac implantable electronic device envelopes evaluated in an animal model. J Cardiovasc Electrophysiol, 32(5): 1346-1354.
- Fuertes B, Toquero J, Arroyo-Espliguero R, Lozano IF (2003) Pacemaker lead displacement: Mechanisms and management. Indian Pacing Electrophysiol J, 3(4): 231-238.
- Baddour LM, Epstein AE, Erickson CC, Knight BP, Levison ME, et al. (2010) Update on cardiovascular implantable electronic device infections and their management: A scientific statement from the american heart association. Circulation, 121(3): 458-477.
- Londono R, Badylak SF (2015) Biologic Scaffolds for Regenerative Medicine: Mechanisms of In vivo Remodeling. Ann Biomed Eng, 43(3): 577-592.
- Boyd WD, Johnson WE 3rd, Sultan PK, Deering TF, Matheny RG (2010) Pericardial reconstruction using an extracellular matrix implant correlates with reduced risk of postoperative atrial fibrillation in coronary artery bypass surgery patients. Heart Surg Forum, 13(5): 311-316.
- Stelly M, Stelly TC (2013) Histology of CorMatrix bioscaffold 5 years after pericardial closure. Ann Thorac Surg, 96(5): e127-e129.
- Brown BN, Londono R, Tottey S, Zhang L, Kukla KA, et al (2012) Macrophage phenotype as a predictor of constructive remodeling following the implantation of biologically derived surgical mesh materials. Acta Biomater, 8(3): 978-987.
- Brown BN, Sicari BM, Badylak SF (2014) Rethinking regenerative medicine: A macrophage-centered approach. Front Immunol, 5: 510.
- Sicari BM, Dziki JL, Siu BF, Medberry CJ, Dearth CL, et al. (2014) The promotion of a constructive macrophage phenotype by solubilized extracellular matrix. Biomaterials, 35(30): 8605-8612.
- Dziki JL, Wang DS, Pineda C, Sicari BM, Rausch T, et al. (2017) Solubilized extracellular matrix bioscaffolds derived from diverse source tissues differentially influence macrophage phenotype. J Biomed Mater Res A,105(1):138-147.
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
Indexed at, Google scholar, Cross ref
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi