Research Article, J Virol Antivir Res Vol: 10 Issue: 2
An in vitro and in silico Antiviral Evaluation on Phytochemicals from the Leaves of Punica granatum L., and Punicalagin Against Human Herpes Virus-3
Divyadarshini Angamuthua1, Rajarajan Swaminathanb2*, Indu Purushothamana1
1Department of Microbiology & Biotechnology, Presidency College, Chennai, India
2Centre for Designing, Discovery & Development of Drugs, Sri Ramaswamy Memorial Institute of Science and Technology, Sonepet, India
- Corresponding Author:
- Rajarajan Swaminathan
Centre for Designing
Discovery & Development of Drugs
SRM University, Sonepet, Haryana, India
E-mail: drsrajarajan@gmail.com
Received: December 26, 2020 Accepted: March 24, 2021 Published: March 31, 2021
Citation: Angamuthua D, Swaminathanb R, Purushothamana I (2021) An in vitro and in silico Antiviral Evaluation on Phytochemicals from the Leaves of Punica granatum L., and Punicalagin Against Human Herpes Virus-3. J Virol Antivir Res 10:2.
Abstract
The plant Punica granatum L., holds tremendous medicinal properties. The purpose of this research work is to isolate the phytochemicals from the leaves of Punica granatum L., and evaluate its antiviral efficacy under in vitro conditions against the clinical isolates of Human Herpes Virus-3. The phytochemicals were isolated chromatographically and their structures were defined through spectrophotometry. The antiviral activity was performed through post incubation assay and the selectivity index was calculated. Punicalagin, a major phytochemical present in the fruit rind of Punica granatum L., was commercially available and the antiviral potential against the HHV-3 was assessed in parallel for comparison. The antiviral results found that among the isolated leaf phytochemicals, Quercetin-3-O-glucoside and 4-isopropyl -2- methoxy-1-methyl benzene inhibited the HHV-3 induced CPE in post incubation assay. The active leaf phytochemicals and punicalagin were further proceeded to track down the actual inhibition mechanism through time of study antiviral procedures. The observed in vitro antiviral results were found to be consistent with the in silico docking analysis of active leaf phytochemicals and punicalagin with the HHV-3 protease in autodock software. Therefore, the present antiviral study inferred that the phytochemicals of Punica granatum L., inhibited the late events in the replication of HHV-3.
Keywords: HHV-3; antiviral activity; Punica granatum; Punicalagin; in silico docking; Autodock
Keywords
HHV-3; antiviral activity; Punica granatum; Punicalagin; in silico docking; Autodock
Introduction
Human Herpes Viruses belongs to the family of Herpesviridae consisting of eight DNA viruses in three different sub families namely the alpha, beta and gamma herpes viruses. Human Herpes Virus-3 (HHV-3) is an alpha DNA virus that causes Chickenpox in children during which the HHV-3 undergoes latency and upon latent reactivation after several decades to cause Herpes Zoster/shingles in adults. Acyclovir and its analogues namely the valayclovir and famciclovir widely prescribed for the infections caused by HHV-3 [1,2]. These antivirals are prodrugs and exhibit a common mode of action by inhibiting the viral DNA replication with the help of viral thymidine kinase, an enzyme which phosphorylates the prodrug for its action. Mutation in viral thymidine kinase makes the drug un phosphorylated thereby the drug acyclovir remains in its prodrug form and does not exhibit its antiviral action [3]. This renders HHV-3 to gain drug resistance and cross resistance of HHV-3 to all the analogues of acyclovir commonly in use for treating HHV-3 infections are reported in the literature. As a result, inadequate antiviral drug response fuels the recurrent episodes of HHV-3 prolonging the intake of repeated high dosage of acyclovir eventually causes serious adverse events in patients such as renal failure, nephrotoxicity and neurotoxicity [4-9]. Foscarnet, a pyrophosphate inhibitor has been apparently recommended for drug resistant HHV-3 infections however severe nephrotoxicity and renal associated side effects are reported in patients [10-12]. This scenario necessitates the development of novel and effective therapeutic substances from alternate resources.
In recent years, shortage of specific antivirals for various viral infections clearly indicate the space and scope for future research in novel antiviral drug discovery as the scientific society is struggling and unable to battle against the viruses. Natural products are in medicinal practice since from prehistoric period. Phytochemicals can act as templates for the discovery and development of newer efficacious antivirals and thus, research and development unit from various pharmaceutical companies have shifted their interest upon antiviral research from plant origin for various viral infections. The ability of the viruses to gain resistance to the existing antiviral drugs and the ancient knowledge of managing microbial infections with the medicinal plants has led to the success of exploring novel and potential antivirals from plants. Several research publications pertaining to the anti HHV-3 activity speaks of practical utility of finding alternate therapeutic substances from plant origin for treatment of Chickenpox and Herpes Zoster [13-15].
In our previous study, we have investigated the in vitro antiviral activity of various plants against the clinical isolates of Human Herpes Virus-3 in which predominant antiviral activity was seen in the aqueous leaf extract from the leaves of Punica granatum L., [16]. In this present antiviral study, we discussed the isolation of phytochemicals from the leaves of Punica granatum L., followed by its structure elucidation and in vitro antiviral evaluation against the clinical isolates of Human Herpes Virus-3 (HHV-3) along with the time of addition study for anticipating the antiviral mechanism combined with in silico analysis against the HHV-3 specific protease, an assembly protein involved in the capsid assembly of HHV-3. Also, we have evaluated the in vitro anti HHV-3 activity of punicalagin, an active ingredient present in the fruit rind of Punica granatum L., to compare and contrast the anti HHV-3 properties present in the leaf compound (s).
Materials and Methods
Cells & viruses
HEp-2 cells were procured from National Centre for Cell Sciences (NCCS) Pune and successfully maintained at 10% MEM at 37ºC under 5% CO2 atmosphere. The virus stock of the clinical isolates of HHV-3 (HHV-3 isolated from Chickenpox and Herpes Zoster) used in our previous antiviral study were successfully retrieved in HEp- 2 cells at 330C under 5% CO2 atmosphere. The viral isolates were confirmed by amplifying the ORF62 gene. The TCID50 value of the cultivated HHV-3 isolates was estimated by using end point titration technique. Briefly the viral stocks were diluted tenfold and 100 μl from each dilution was added into each well with monolayered cells. The plates were incubated at 330C under 5% CO2 atmosphere for 120 hours to calculate the TCID50 value by using the Reed and Muench formula [17].
Fractionation of the extract
The leaves of Punica granatum L., were collected. air dried and made into a fine powder. A voucher specimen was deposited in the Department of Plant Biology and Biotechnology, Presidency College, Chennai. 100 gm of leaf powder was soaked in 500 ml of aqueous phase and incubated overnight at 40C. The soaked mixture was filtered and the clarified supernatant obtained after centrifugation was filtered using 0.22μm Millipore filter. The filtered aqueous extract was then loaded into a flask and lyophilized to obtain 27 g of the crude aqueous extract. The crude aqueous extract was mixed with 60 g of silica gel (60-120 mesh) and hence the admixture. A 2.4 dia column was packed with the admixture along with hexane (height of 20 cm) and was eluted with increasing solvent polarity from hexane to ethyl acetate. The step wise elution is done in the following ratios of hexane and ethyl acetate: 100%, 95:5, 90:10, 85:15, 80:20, 75:25, 70:30 and 0:100 to yield around 233 fractions. The fractions were concentrated by using the rotary evaporator. An aliquot of all the concentrated fractions were loaded on the activated silica gel TLC plates (20 × 20 cm) and developed using ratio of methanol 8: chloroform 2 dipped in H2SO4. Fractions having the same number of spots with similar Rf values were pooled to yield five compounds. The compounds, their elution details and yield are given in the following Table 1.
Table 1: Separation of phytocompounds from the leaves of Punica granatum L. and their elution details
| S.no | Compounds | Elution details | Compound yield |
|---|---|---|---|
| 1 | C1 | 95:05 | 85mg |
| 2 | C2 | 85:15 | 127mg |
| 3 | C3 | 75:15 | 183mg |
| 4 | C4 | 60:40 | 129mg |
| 5 | C5 | 50:50 | 744mg |
Structural identification of the phytocompounds
The isolated phytocompounds were subjected to identification by using Gas Chromatography-Mass Spectrometry analysis and characterized through Nuclear Magnetic Resonance (NMR). These studies were carried out with the help of facilities available at Sophisticated Analytical Instrument Facility Centre (SAIF), Indian Institute of Technology (IIT) Madras.
The GC-MS analysis was done in a high resolution, double focusing instrument JEOL GCMATE II GC-MS with Data system. The maximum resolution of the instrument is 6000 with 1500 daltons as the maximum calibrated mass. The column was fused silica 50m x 0.25 mm I.D and the phytocompounds were allowed to dissolve in HPLC grade methanol. The column temperature was set at 2350C for 3 minutes and the injector temperature was 2400C. The compound analysis was done for 20 minutes at 1000C with Helium as the carrier gas and the split ratio was 5:4. 1 μl of the sample was evaporated in a split less injector at 300°C. The components were identified by gas chromatography coupled with mass spectrometry. The data obtained was compared with the National Institute of Standards and Technology Library (NISTL).
25mg of the isolated phytochemical compounds was weighed and dissolved in dueterited dimethlysulphoxide (DMSO D6) for NMR spectroscopy. The dissolved solution was transferred into an NMR tube and was placed in the sample collector. The samples were programmed using the Bruker software and the reference standard was Trimethylsilane. The samples were injected and tuned to match the 500 MHz radiofrequency. It was then locked after one complete rotation to monitor the resonance frequency of the deuterium signal and to keep them as constant. After 1 to 35 scans, the samples were ejected and the signals were fourier transformed. The noise to signal ratio was adjusted to produce the end peak and the chemical shift was measured in delta scale.
The above said procedure was adopted for H NMR spectrometry also.
Estimation of CC50 by MTT assay
The phytochemical compounds isolated from the leaves of Punica granatum L., were weighed and diluted in log 2 dilution to reach the required drug concentrations ranging from 500 μg – 3.9 μg/ml. The respective drug dilutions were added to the monolayered HEp-2 cells in a 96 well tissue culture plate. The wells were replenished with 100 μl of 2% MEM and incubated at 370C in a humidified CO2 atmosphere. The experiment was done in triplicates. The plates were then observed under an inverted phase contrast microscope (Nikon Ti Eclipse) for the next 120 hours to observe the maximum non-toxic concentration of the testing compounds. On the final day of observation, 20 μl of MTT was added into each well in all the three plates and incubated at 370C for 4 hours. The wells were emptied by gentle aspiration and DMSO was added into all the wells to dissolve the formed formazon crystals. The plates were then read at 570 nm to record the OD values. The percentage of the cell viability was calculated by using the formula (treated/untreated)* 100. The CC50 values were calculated by plotting dose response curve in excel.
Cytopathic inhibition assay
Confluent HEP-2 cells grown in 96 well microtitre plates were initially infected with 1 MOI and allowed for an hour adsorption at 330C under 5% CO2 atmosphere. Serial dilutions of the phytochemicals, punicalagin and acyclovir (MNTC was the higher drug concentration and serially diluted thereafter) were added into the infected cells after careful removal of the viral inoculum. Virus control and cell control wells were included to which 2% MEM was added. The antiviral assay was done in replicates and incubated for the next 120 hours to observe for the presence or absence of viral induced CPE. MTT was added into the wells as described earlier for the CC50 estimation and the plates were read at 570 nm. The IC50 value was arrived by plotting the log concentration units along the X axis and the percentage of virus infectivity along the Y axis using excel spread sheet. The selectivity index was determined.
Direct virucidal effect
This experiment was done by adopting the procedure described by [18]. In order to check for the direct virucidal effect of the active leaf phytochemicals and punicalagin on the HHV-3 isolates, the TCID50 dose of the HHV-3 isolates were pre incubated with the required drug concentrations at 330C for one hour. The pretreated viral particles are then allowed to infect the fresh monolayered wells and incubated till the appearance of CPE in comparison with controls. The virus titre was estimated by using end point titration technique as described earlier in the section 2.1
Time of removal assay
A 96 well titre plate was seeded with HEp-2 cells and allowed for confluence. The confluent cells were infected with the viral inoculum and allowed for adsorption at 33°C for one hour. After adsorption, the unbound viral particles or excess inoculum was removed by gentle washing and the medium containing different concentrations of the drug was added into the infected cells. The antiviral set up was incubated at 33°C under 5% CO2 atmosphere followed by the removal of drug dilutions from the infected wells at different time points at 24, 48, 72 and 96 hours from the time of addition of the phytochemicals and replaced with 2% MEM (maintenance medium). The set up was incubated at 33°C for 120 hours and the virus titre was calculated as said before.
Time of drug addiction studies
The antiviral effect on the intracellular HHV-3 production was studied by infecting HEp-2 cells at 0 hour and treated with the active drug concentrations at 12, 24, 36, 48, 60, 72, 84, 96 hours (Individual plates were used for each time points). Following the treatment, the infected cells were incubated and the antiviral activity was measured at the 120 hours from the time of inoculation. After 120 hours of incubation, the treated cells were trypsinized, centrifuged and the cell pellets were washed and freeze thawed for three times. The resulting suspension was centrifuged and the virus titre was estimated from the supernatant as described in the earlier sections.
In silico docking studies
The in silico docking study was performed with the scope to predict the possible antiviral target likely the active phytocompounds that exerts antiviral action. The three dimensional structure of the protease of VZV (1VZV) was retrieved from protein data bank (PDB) and optimized by removing the water molecules, 3D protonation and energy minimization. The energy minimized structure of protein was further used for docking analysis. The elucidated structure of the phytocompounds of leaves of Punica granatum L., and standard drug Valacyclovir, Foscarnet and Acyclovir were sketeched using ACD Chemsketch and converted into specific structural file format and used as ligands for the docking study. Preliminary screening was performed using iGEMDOCK and further confirmatry docking was performed for the same. Molecular dokcing was performed using Autodock 4.2. Both ligands and the receptor were converted into PDBQT format before performing docking. For each ligands molecules we obtain 10 docked pose. The best docked pose was visualized using pymol.
Drug likeliness property analysis
The phytochemical compounds were subjected to drug likeliness screening using a web based chemoinformatics tool namely the molinspiration (www.molinspiration.com).
Admet analysis
The ADMET analysis was carried out in order to check for the nature and possible toxicity of the phytochemicals while administering in vivo. The Human Intestinal Absorption (HIA) and skin permeability model was predicted so that oral delivery and transdermal delivery was identified by using drug based models for in vitro CacO2-cell and MDCK cell assay. The blood brain barrier (BBB) penetration of the therapeutic drug in to the central nervous system (CNS) and plasma protein binding model in its disposition and efficacy was identified using PreADMET. Mutagenecity was detected by AMES toxicity and carcinogenecity of the isolated compounds were also perceived in rats and mouse models.
Results
Isolation and spectral identification of the extracts
Five phytochemical compounds were isolated from the Rf value based pooled up fractions of Punica granatum L., leaves through column chromatography and were designated as Compound C1-C5.
The spectral result analysis have found the phytochemicals C1, C3 and C5 were not to be in pure form (data not given) however their antiviral efficacy was evaluated to check for superior activity if any. The phytochemical C2 was structurally identified as Quercetin-3-ortho glucoside. The C NMR spectrum have shown the arrangements of C atoms in the order of 61.32-97.30(glucosidic carbon), 92.65(lactone linkage), 102.4-161.96(all aromatic carbon), 178.0(lactone carbonyl). The H NMR spectrum analysis of C2 revealed the presence of 2.3 (all glycosidic OH protons), 3.0- 4.5 (all CH protons of lactone ring), 5.0 (OH protons of phenolic ring), 6.1(Proton in lactone linkage carbon), 6.3-7.5 (all aromatic protons).
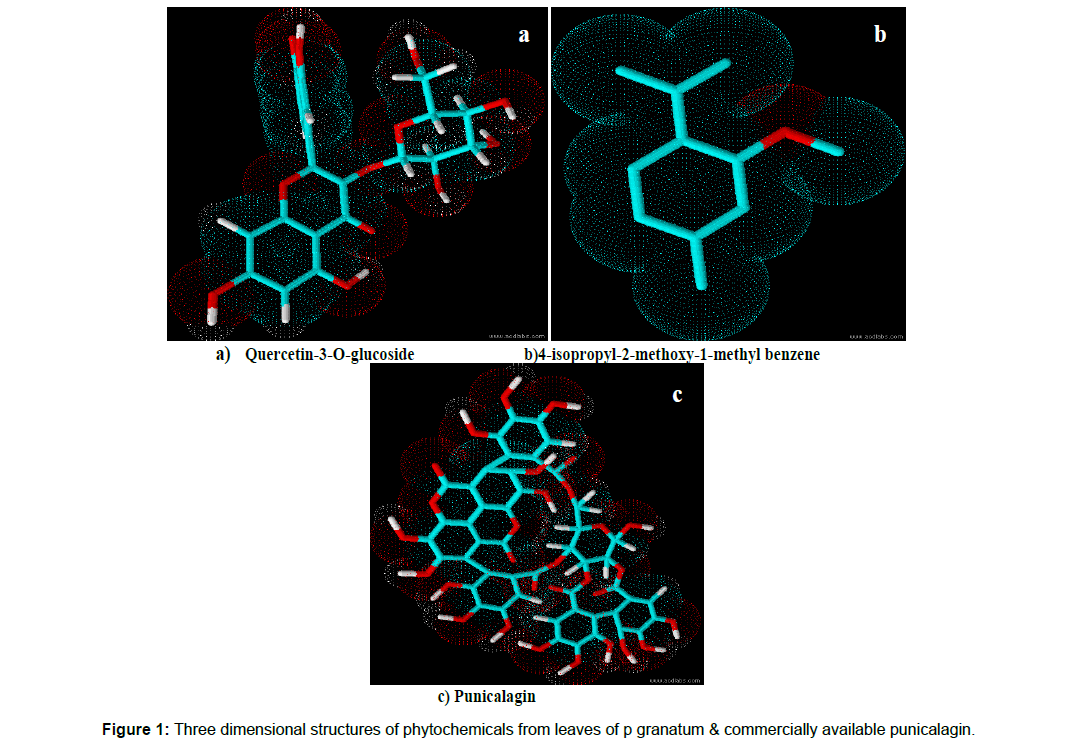
The chemical structure of phytochemical C4 was identified as 4-isopropyl -2- methoxy-1-methyl benzene. The C NMR spectral analysis of phytochemical C4 is as follows: 19.11-24.87(free methyl), 37.01(quarternary carbon), 77.44(methoxy carbon), 125.79-145.72(all aromatic carbons), 167.47(Carbon bearing methoxy). The H NMR spectral analysis shown the presence of H atoms in position: 0.9 – 2.3(free methyl), 3.7(methoxy proton), 7.8(aromatic protons). The three dimensional structure of the isolated active leaf phytochemicals and punicalagin was displayed in Figure 1.
In vitro cytotoxicity of the isolated phytochemical compounds from the leaves of Punica granatum Linn.
The in vitro cytotoxicity of the isolated phytochemical compounds from the leaves of Punica granatum L. was evaluated by MTT assay to calculate the CC50 of the phytochemicals. The morphological indications of cytotoxicity were not observed in the cells treated with the phytochemicals of Punica however milder toxicity was noticed in higher drug concentration. The cytotoxicity results have shown that the phytochemicals of Punica does not inhibited the cell growth conditions in the concentrations used.
In vitro antiviral assay of the phytochemical components from the leaves of Punica granatum L., & punicalagin
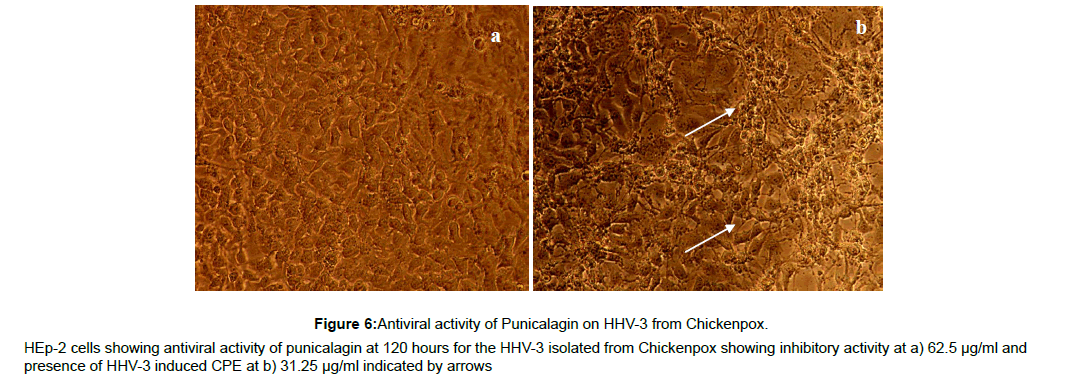
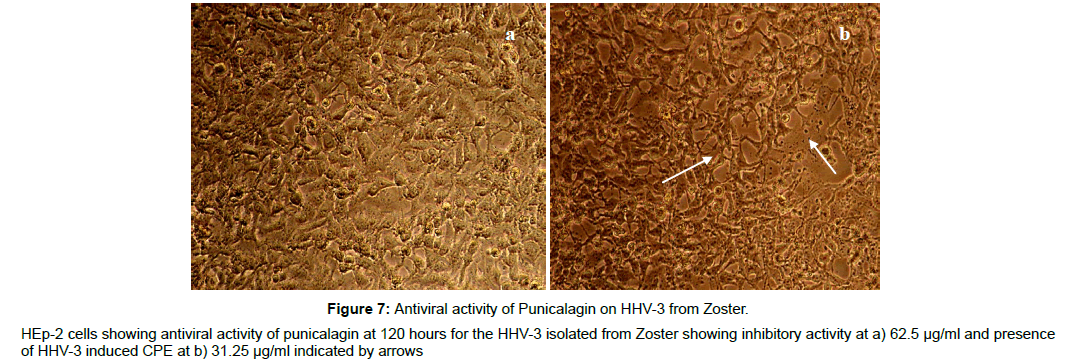
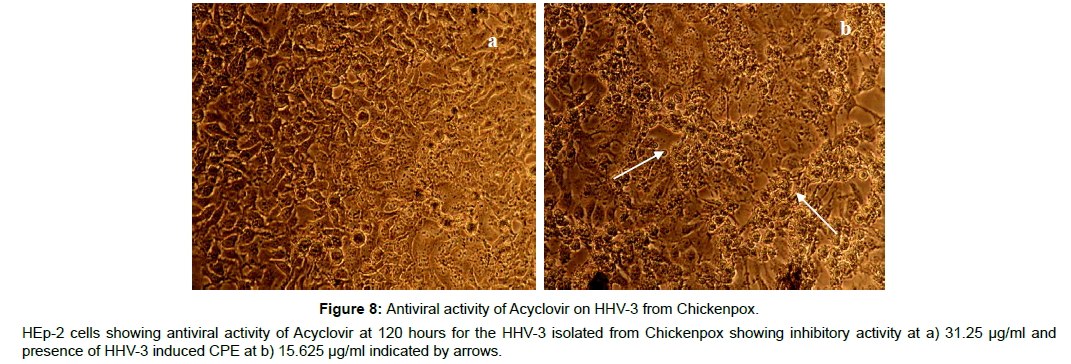
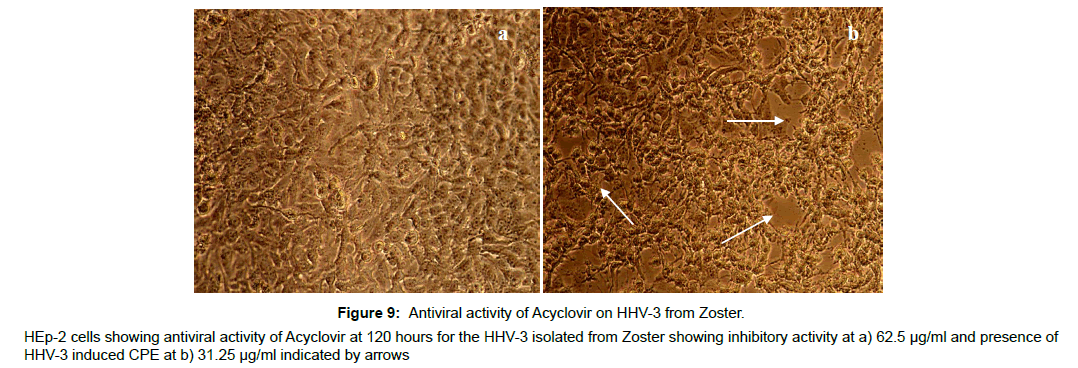
The antiviral assay of the phytochemicals was evaluated individually against the HHV-3 isolated from Chickenpox and Herpes Zoster and the IC50 was calculated. The phytochemicals isolated from the leaves of Punica exhibited substantial activity against the HHV-3 isolates. The phytochemical C2, Quercetin-3- ortho glucoside was effective at 3.21μM and 3.70 μM respectively with the selectivity index value of 106 and 92 against the HHV-3 isolated from chickenpox and Herpes Zoster. The phytochemical C4, 4-isopropyl -2- methoxy-1-methyl benzene exhibited anti HHV-3 activity at 10.97 μM (Chickenpox) and 12.10 μM (Herpes Zoster) with selectivity index of 28.70 and 26.02. Punicalagin was effective at 1.65 and 1.87 μM with selectivity index of 141 and 124. Acyclovir was effective at 1.37 and 1.58 μM with selectivity index of 214 and 186 respectively. The antiviral profile of the leaf phytochemicals, punicalagin and the drug acyclovir were given in Table 2 along with their CC50 values. The corresponding pictures were shown in Figure 2-9.
Figure 2: Antiviral activity of Quercetin-3-O-glucoside on HHV-3 from Chickenpox.
HEp-2 cells showing antiviral activity of Quercetin-3-O-glucoside at 120 hours for the HHV-3 isolated from Chickenpox showing inhibitory activity at 125 μg/ml
and 62.5 μg/ml (a and b)and presence of HHV-3 induced CPE in subsequent drug concentration 31.25μg/ml (c) indicated by arrows.
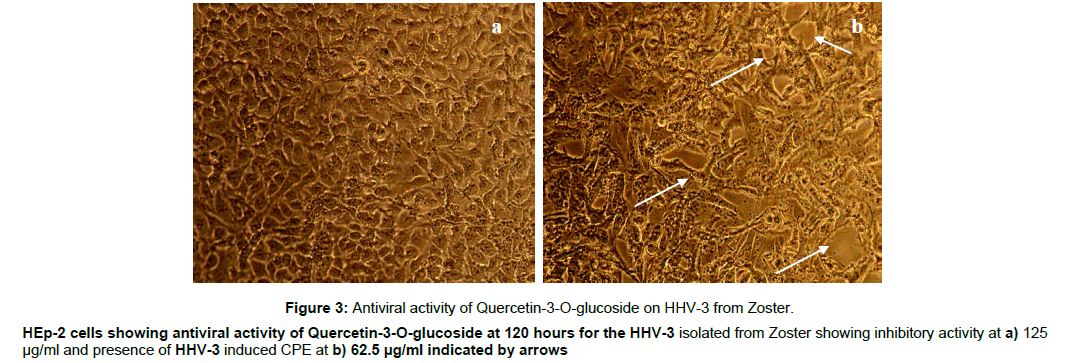
Figure 3: Antiviral activity of Quercetin-3-O-glucoside on HHV-3 from Zoster.
HEp-2 cells showing antiviral activity of Quercetin-3-O-glucoside at 120 hours for the HHV-3 isolated from Zoster showing inhibitory activity at a) 125
μg/ml and presence of HHV-3 induced CPE at b) 62.5 μg/ml indicated by arrows
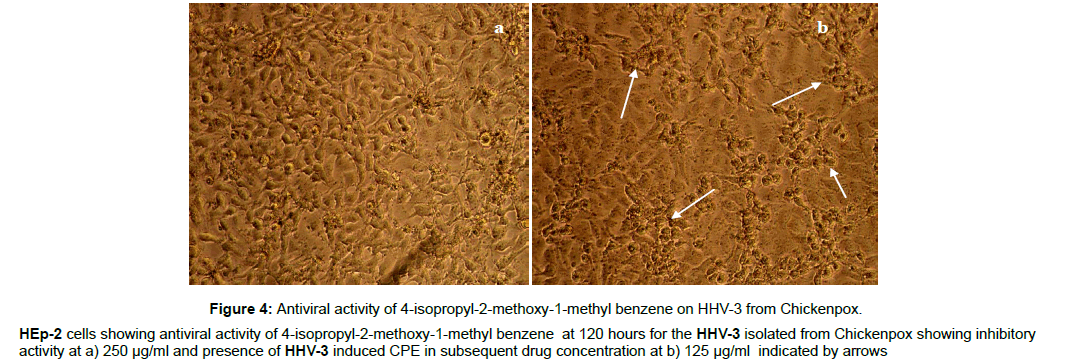
Figure 4: Antiviral activity of 4-isopropyl-2-methoxy-1-methyl benzene on HHV-3 from Chickenpox.
HEp-2 cells showing antiviral activity of 4-isopropyl-2-methoxy-1-methyl benzene at 120 hours for the HHV-3 isolated from Chickenpox showing inhibitory
activity at a) 250 μg/ml and presence of HHV-3 induced CPE in subsequent drug concentration at b) 125 μg/ml indicated by arrows
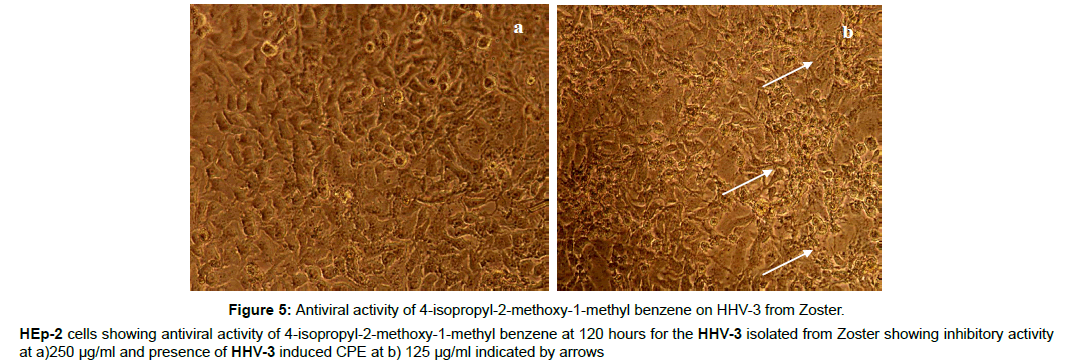
Figure 5: Antiviral activity of 4-isopropyl-2-methoxy-1-methyl benzene on HHV-3 from Zoster.
HEp-2 cells showing antiviral activity of 4-isopropyl-2-methoxy-1-methyl benzene at 120 hours for the HHV-3 isolated from Zoster showing inhibitory activity
at a)250 μg/ml and presence of HHV-3 induced CPE at b) 125 μg/ml indicated by arrows
Table 2: In vitroAntiviral activity of Phytochemicals against HHV-3 isolated from Chickenpox
| S.NO | NAME OF PHYTOCHEMICAL & STANDARD |
CC50 | IC50 | Selectivity index | ||
|---|---|---|---|---|---|---|
| CP | HZ | CP | HZ | |||
| 1. | C1 | 434.31 | 17.83 | - | 24.35 | - |
| 2. | C2 | 342.16 | 3.21 | 3.70 | 106 | 92 |
| 3. | C3 | 281.25 | - | - | - | - |
| 4. | C4 | 314.92 | 10.97 | 12.10 | 28.70 | 26.02 |
| 5. | C5 | 241.73 | 23.26 | - | 10.39 | - |
| 6. | Punicalagin | 233.37 | 1.65±0.014 | 1.87±0.016 | 141 | 124 |
| Acyclovir | 294.54 | 1.37 | 1.58 | 214 | 186 | |
Direct virucidal effect of the compounds
In order to check for the phytochemicals to have any direct antiviral action in the early stages of infection such as blocking the host cell entry by interacting with the viral envelope proteins, direct virucidal activity was performed. No reduction in virus titre was observed in the phytochemical treated virus suspension as when compared with the control. The phytochemicals have not displayed a significant reduction in virus titre upon further incubation for the next 4 hours.
Time of removal assay
This assay was mainly carried out to evaluate the potency of an antiviral drug to achieve an effective antiviral outcome in a limited exposure of time. The drug acyclovir was effective at 250 μg/ml alone in 48 hours of exposure and the activity was declined in 24 hours of time exposure. Punicalagin and the leaf phytochemicals exhibited partial activity when the infected cells were exposed only for 48 hours and did not exhibited significant activity when the exposure was less than 24 hours.
Time of addition studies on phytochemicals
HHV-3 inoculum was allowed to infect the HEp-2 cells followed by adding different concentrations of C2, C4, Punicalagin and Acyclovir in different set points from the time of infection. The results found that the phytochemicals from the leaves of Punica granatum L., and Punicalagin retained the antiviral activity when there was delay in adding the drugs at least for 48 hours however the activity was slowly diminished further in delay of adding the drugs. The drug Acyclovir also has got diminished antiviral activity when there was further delay in adding after 60 hours.
In silico docking studies
The drug fitness properties were analyzed and calculated by using ADMETSAR and MOLINSPIRATION tool. The phytochemicals of Punica were found to be non AMES toxic and non-mutagenic in rats. The phytochemical 4-isopropyl -2- methoxy-1-methyl benzene was found to possess human intestinal absorption, blood brain barrier absorption and skin permeability properties. The phytochemical Quercetin-3-ortho glucoside was found to have human intestinal absorption and the results are interpreted in Table 3, 4.
Table 3: Admetsar properties of phytochemicals.
| MODEL | Quercetin-3-O-glucoside | 4-isopropyl-2-methoxy-1-methyl benzene | Punicalagin |
|---|---|---|---|
| Blood-Brain-Barrier | BBB- | BBB+ | BBB- |
| Human Intestinal absorption | HIA+ | HIA+ | HIA- |
| CaCO2 permeability | CaCO2 - | CaCO2 + | CaCO2 - |
| TOXICITY | |||
| AMES Toxicity | Non AMES toxic | Non AMES toxic | Non AMES toxic |
| Carcinogenecity | Non-Carcinogens | Non-Carcinogens | Non-Carcinogens |
Table 4: Drug likeness properties of the isolated active phytochemicals & punicalagin
| Properties | Quercetin-3-O-glucoside | 4-isopropyl-2-methoxy-1-methyl benzene | Punicalagin |
|---|---|---|---|
| miLogP | -0.36 | 3.88 | 2.04 |
| TPSA | 210.50 | 9.23 | 518.75 |
| n atoms | 33 | 12 | 78 |
| MW | 464.38 | 164.25 | 1084.72 |
| nON | 12 | 1 | 30 |
| nOHNH | 8 | 0 | 17 |
| n violations | 2 | 0 | 3 |
| nrotb | 4 | 2 | 0 |
| Volume | 372.21 | 1776.10 | 800.91 |
The interaction energy of punicalagin and Quercetin-3-Oglucoside, 4-isopropyl -2- methoxy-1-methyl benzene along with standard drug Valacyclovir, Foscarnet and Acyclovir was incorporated in the docking analysis. The preliminary docking was performed using iGEMDOCK and it was found that the compound Punicalagin and Quercetin-3-O-glucoside showed the highest binding affinity and details are given in Table 5.
Table 5 : In silico Preliminary antiviral screening by IGEMDOCK
| S.No | Name of phytochemical/standard | Total energy | Hydrogen bond | Interacting aminoacids |
|---|---|---|---|---|
| 1. | Quercetin-3-O-glucoside | -113.1 | -28.64 | ALA 90, HIS 91, SER 92, PHE 94, ARG 107, GLU 195 |
| 2. | 4-isopropyl-2-methoxy-1-methyl benzene | -56.2 | -3.5 | ILE 64 |
| 3. | Punicalagin | -147.8 | -36.91 | ASP 214,ARG 215, TRP 216, ASP 217 |
| 4. | Acyclovir | -98.01 | -36.4 | PHE 87, ALA 90, SER 92, PHE 95, GLY 96, SER 100, ARG 107 |
| 5. | Valacyclovir | -90.02 | -22.1 | ARG37,LEU40, GLU 66, GLU 172, SER 173, ARG 176 |
| 6. | Foscarnet | -65 | -33.4 | SER 120, LEU 121, ARG 147, ARG 148 |
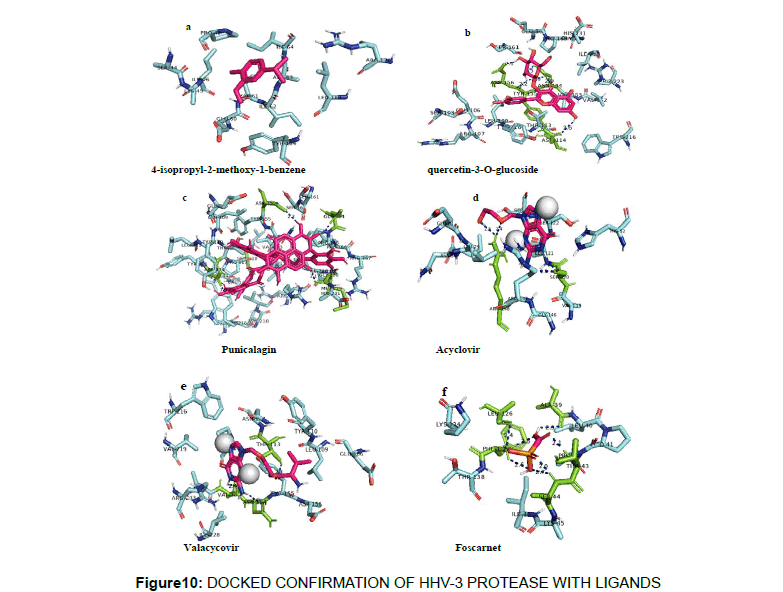
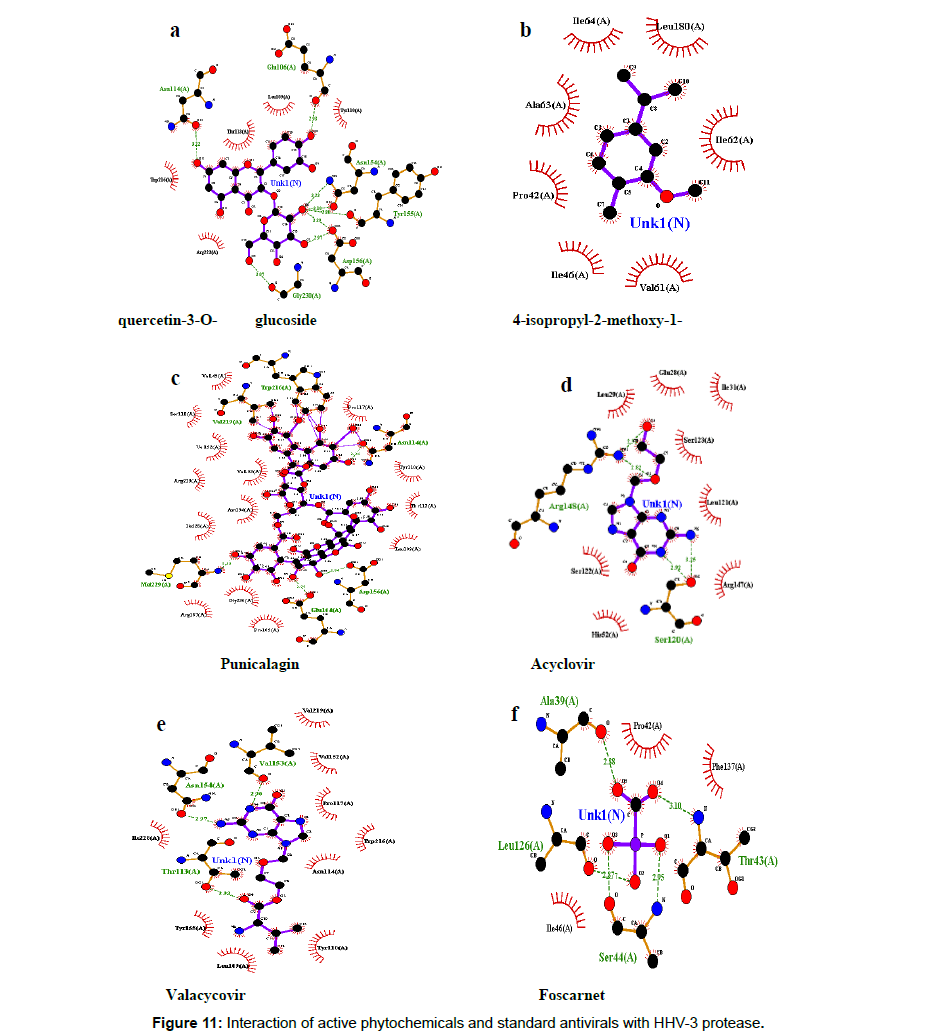
Further confirmatory docking was performed using autodock and it was interesting to note that the phytocompound Punicalagin showed the highest binding energy of -8 kcal/mol with key hydrogen bond interactions with Trp216, Asn114, val219, Asp156,Met229, Glu164,Asp156 followed by Quercetin-3-O-glucoside with a binding energy of -7 kcal/mol and the key hydrogen bonding was observed with Asn114,Glu106,Asn154,Tyr155,Asp156,Gly230 and the interaction profiles of other ligands are furnished in Table 6. The docked confirmation of the phytochemicals with the HHV-3 protease and its interaction was given in Figure 10, 11.
Table 6: Confirmation of preliminary in silico analysis by Autodock
| Ligands | Pubchem ID | Binding energy kcal/mol | HB interactions involved |
|---|---|---|---|
| 4-isopropyl-2-methoxy-1-methylbenzene | 80790 | -3 | - |
| Quercetin-3-O-glucoside | 5280804 | -7 | Asn114,Glu106,Asn154,Tyr155,Asp156,Gly230 |
| Punicalagin | 44584733 | -8 | Trp216,Asn114, val219,Asp156,Met229,Glu164,Asp156 |
| Foscarnet | 3415 | -4 | Leu126, Ala39, Thr43, Ser44 |
| Valacyclovir | 135398742 | -6.1 | Thr113,Asn154,Val153 |
| Acyclovir | 135398513 | -5.7 | Arg148, Ser120 |
Discussion
In our previous publication, we found a superior antiviral efficacy in the leaves of Punica granatum L., against the HHV-3 isolated from Chickenpox and Herpes Zoster [16]. This research finding holds the piece of information that the leaves of the plant Punica has promising anti HHV-3 activity and in order to substantiate this, we attempted to isolate the phytochemicals in the leaves of Punica granatum L., through bioassay guided fractionation, spectral characterization and also to screen the active antiviral principle (s) that could have contributed the worth mentioning anti HHV-3 activity. Earlier antiviral research on the plant Punica was explored against HHV- 1 and HHV-2 and the activity was confined towards blocking the initial cell adsorption and viral entry into the host cells [19,20]. Punicalagin, the major phytochemical present on the fruit rind of Punica granatum L., was reported to have effective antiviral activity against the HHV-2 [21]. The phytochemical punicalagin was yet to address in the context of antiviral activity against HHV-3 and no article in literature deciphering the anti HHV-3 activity on any of the phytochemicals from the pericarp of Punica granatum L. Thus, punicalagin was taken in to account for antiviral evaluation against the HHV-3 and its potency was compared with the anti HHV-3 properties in the phytochemicals isolated from the leaves of Punica granatum L., Therefore, this research work forms the very first effort on exploring the antiviral potency of the phytochemicals from the plant Punica against the HHV-3.
The leaves of plant Punica were fractionated to yield five phytochemicals. The isolated phytochemicals and Punicalagin was evaluated for in vitro antiviral activity against the HHV-3 isolated from Chickenpox and Herpes Zoster by using post incubation assay and the activity was compared with the standard Acyclovir. The isolated phytochemicals from the leaves of Punica granatum L., namely the Quercetin-3-ortho glucoside and 4-isopropyl-2- methoy-1-methyl benzene exhibited potent antiviral activity against the clinical isolates of HHV-3 however the antiviral activity was moderate in the rest of the isolated leaf phytochemicals. Though the leaf phytochemicals have shown effective antiviral activity, the phytochemical punicalagin exhibited superior activity than the leaf phytochemicals and the activity was equivalent and comparable with the drug acyclovir. Hence, the antiviral evaluation was further taken up to interpret or to translate the actual mechanistic action displayed by the phytochemicals.
The isolated effective phytochemicals and Punicalagin which showed superior antiviral efficacy were subjected further to various in vitro antiviral techniques to track down at which particular replication event, the phytochemical is able to exert its antiviral action against the HHV-3. Earlier aspects of antiviral research from phytochemicals anticipated that the interaction of phytochemicals was more efficient with the viral envelope proteins thereby the viral entry into the host cell is blocked [22-23]. However, our results illustrated that no such kind or any direct interaction between the phytochemicals of Punica with HHV-3 envelope proteins as it was evidenced in the direct virucidal activity. The antiviral action was not noteworthy in direct virucidal activity as there was no reduction in the virus titre with the results we observed. Hence the time of exposure was further increased by incubating the virus suspension with the required drug concentration of phytochemicals to an extent of 4hours at 330C conversely the same effect was observed even after extending the incubation time period. Thus, we figure out that none of the active leaf phytochemicals of Punica or its rind phytochemical punicalagin played a significant role in blocking the virus entry into the host cells.
The antiviral efficacy was further evaluated in an aspect of limited exposure of drugs with HHV-3. Acyclovir, the most potent antiviral drug for HHV-3 is effective in vitro only when the time of exposure is 120 hours. However the antiviral potency of Acyclovir was much reduced when the drug is exposed for 48hours and this reduced time exposure resulted in an increase in the effective antiviral concentration to 250μg/ml and dramatic loss of antiviral potency was observed when the time of exposure falls off less than 24 hours.
Though the anti HHV-3 potency of punicalagin was comparable with acyclovir at 120 hours of antiviral evaluation, limited exposure of punicalagin below 24 hours has not achieved potential efficacy. However the antiviral activity was only moderate when punicalagin was exposed for 48 hours. This implies that at least a time exposure of ≥ 48hours is required for punicalagin to exhibit a reasonable antiviral potency despite of higher drug concentration at 125 μ/ml in comparison with parallel 120 hours of time exposure. Among the leaf phytochemicals tested, Quercetin-3-ortho glucoside and 4-isopropyl -2- methoxy-1-methyl benzene did not attain any marked effective antiviral potency when the time of exposure is mere 24 hours however the above said leaf phytochemicals exhibited partial antiviral potency when the cells were exposed for 48-72 hours.
In order to hypothesize the effect of the phytochemicals on the internal steps of viral replication and also to ascertain the effect on the delayed administration of phytochemicals to HHV-3 infected cells, the HEp-2 cells were exposed to HHV-3 inoculum followed by adding the required drug concentrations at different set points from the time of inoculation. The time of addition experiment illustrated that the activity of punicalagin and the phytochemical Quercetin-3- ortho glucoside has not lost its antiviral efficacy when there was delay in adding them till 48 hours from the time of inoculation and the activity was much weakened when there was further delay in adding subsequent hours. We also speculate that these two phytochemicals of Punica granatum L., might have interrupted the late events in the viral replication as there was a significant inhibitory effect in 24- 48 hours. Also, we believe, the replication event of HHV-3 which is possibly going on during 24 hours or between 24-48 hours from the time of infection is the late event replication that eventually includes the viral capsid assembly and its DNA packaging into the viral capsids. Therefore, it is possible that the interference of these two phytochemicals with HHV-3 capsid assembly.
Though the activity of punicalagin and Quercetin-3-ortho glucoside was not much profound beyond 48 hours due to delay in adding them into infected wells, the activity was not much inferior than the drug acyclovir as the acyclovir also did not showed a successful activity when the delay in adding was beyond 60 hours. The antiviral potency of phytochemical 4-isopropyl -2- methoxy- 1-methyl benzene was also not striking as the delay in adding the phytochemical beyond 24 hours to the HHV-3 infected cells did not retained the same efficacious activity as observed in the initial antiviral screening and we anticipate that this phytochemical might have involved in inhibiting the initial stage of replication other than cell entry of HHV-3.
Thus, the promising antiviral activity of punicalagin was proved as it has got an equivalent activity with that of the acyclovir and there is a possibility of phytochemicals interfering with the viral capsid assembly of HHV-3.
In order to establish this in detail, docking study through in silico approach was carried out between the isolated phytochemicals and punicalagin with HHV-3 protease as it is the viral protein principally involved in viral capsid assembly [24-25]. Matsumoto et al., 2001[26] have proposed the protease of HSV-1 and HCMV as an important drug target against the selective non-peptidic inhibitors. Our earlier in silico analysis of phytochemicals with HHV-3 protease found Erysenegalensein E as an effective antiviral drug candidate against the HHV-3 protease [27]. In our previous in vitro antiviral screening of four medicinal plants, we have also interpreted the interaction of phytochemicals reported in the Punica with the aminoacids present in the active site of HHV-3 protease through in silico.
In the present antiviral study, in silico docking with HHV- 3 protease was carried out for the active leaf phytochemicals and punicalagin using autodock 4.2. Interestingly we noticed that the phytocompound Punicalagin showed highest binding energy followed by Quercetin-3-O-glucoside when compared to other phytocompounds. Divyadarshini et al. 2016 [27] had previously reported on the docking study of VZV Protease and stated that phytocompound Erysenegalensein E had showed the highest binding energy with HB interactions with Ile 63, Ile 64. Similar docking studies have been performed against the Thymidine Kinase of VZV and reported that deoxythymidine exhibited effective binding against the three alpha herpes viruses [28]. HHV-3 protease is responsible for cleaving the assemblin protein at its end for subsequent DNA packaging. Therefore we presumed that the antiviral activity of punicalagin and Quercetin-3-O-glucoside could be due to interruption of the capsid assembly of HHV-3. The compounds not only exhibited good binding energy we found it interesting to note that the compounds were non-carcinogens however a mutagenic property that was revealed by in silico ADMET analysis.
The interaction of 4-isopropyl -2- methoxy-1-methyl benzene with HHV-3 protease was much weaker and therefore we predict the possibility for this phytochemical to interrupt any other event in the viral replication of HHV-3. The possible interaction of this phytochemical with other proteins of HHV-3 was not undertaken as there were no three dimensional structures of HHV-3 proteins in the protein data bank which is one of the limitation we find.
Thus we illustrated an in vitro evidence for the plant Punica granatum L. to hold guaranteed antiviral efficacy against the Human Herpes Virus-3. The active phytochemicals reported in this research work can be taken up further for its antiviral evaluation with special reference to the drug resistant VZV isolates. Further safety studies can be attempted on the active phytochemicals of this plant followed by its in vivo antiviral studies.
Conclusion
Through this in vitro antiviral research work, the antiviral effectiveness of the plant Punica granatum L., is well established against the clinical isolates of HHV-3. The isolated active phytochemicals Quercetin-3-ortho glucoside and punicalagin interacts in the aminoacids region contributing for the catalytic site of the protease. Hence, these drugs can be taken into account for the consideration of development of an alternate antiviral for the HHV-3 infections provided when these drugs goes on further for its in vivo safety and antiviral issues followed by clinical phase trials into the pathway of a potential antiviral drug for Human Herpes Virus-3.
Acknowledgements
The authors feel thankful for the Department of Science &Technology, Govt of India for establishing an outstanding laboratory infrastructure facility for carrying out in vitro antiviral research work and Bio Informatics Infrastructure Facility Centre of DBT, Presidency College where the in silico research parameters were analyzed. The authors also express their heartfelt thanks to late Mr.N.Angamuthu.M.A., for his rendered help in the collection of Punica granatum L., leaves from the rural areas of Chidambaram. The authors also acknowledge the help from RKK Medical Centre, Chennai for collecting the clinical samples from HHV-3 infected patients.
Funding
The authors declare that this research work is not supported by any funding agency from Government or non-government organization.
Conflict of Interest
The authors declare that there is no conflict of interest in this research work.
References
- Arvin AM, (2002) Antiviral therapy for varicella and herpes zoster. Semin. Pediatr. Infect. Dis, 13(2002): 12–21.
- Snoeck R, Andrei G, De Clercq E, ( 1999) Current pharmacological approaches to the therapy of varicella zoster virus infections: a guide to treatment. Drugs,57:187-206.
- Gilbert C, Bestman-Smith J, Boivin G, (2002) Resistance of herpesviruses to antiviral drugs: clinical impacts and molecular mechanisms. Drug Resist Updat, 5: 88-114.
- Niibori D, Fujisawa M. Matsuzaki M, (1993) Severe central nervous system symptoms following oral administration of acyclovir in a patient with chronic renal failure: a case report. Hinyokika kiyo. Acta urologica Japonica, 39(5): 459-461
- Helldén A, Odar‐Cederlöf I, Diener P., Barkholt L, Medin C, et al.( 2003)High serum concentrations of the acyclovir main metabolite 9‐carboxymethoxymethylguanine in renal failure patients with acyclovir‐related neuropsychiatric side effects: an observational study. Nephrology Dialysis Transplantation, 18(6): 1135-1141.
- Yavuz B B, Cankurtaran M, Halil M, Dagli N, Kirkpantur A, (2005) Renal dysfunction after oral acyclovir treatment in a geriatric woman: a case report. Scandinavian journal of infectious diseases, 37(8): 611-613.
- Fleischer R, Johnson M, (2010) Acyclovir nephrotoxicity: a case report highlighting the importance of prevention, detection, and treatment of acyclovir-induced nephropathy. Case reports in medicine, 1-3
- Boykin K, Kernan W, Tarchini G, Lurix E, (2011) Case Report-Neurotoxicity Associated With Standard Doses of Valacyclovir in Renal Insufficiency. Hospital Pharmacy, 46(10): 774-778
- MengJ. B, Zheng X, Zhang G, Fang Q, (2011) Oral acyclovir induced acute renal failure. World journal of emergency medicine, 2(4): 310-13
- Safrin S, Berger T.G., wolfe P.R., Wofsy C.B., Mills J et al, (1991) Foscarnet therapy in five patients with AIDS and acyclovir-resistant varicella-zoster virus infection. Ann. Intern.Med, 115(1): 19-21
- Visse B, Dumont B, Huraux J.M, Fillet A.M, ( 1998) Single amino acid change in DNA polymerase is associated with foscarnet resistance in a varicella-zoster virus strain recovered from a patient with AIDS. J. Infect. Dis, 178 (1): 55–57.
- Visse B, Huraux J. M, Fillet A.M, (1999) Point mutations in the varicella-zoster virus DNA polymerase gene confers resistance to foscarnet and slow growth phenotype. J. Med. Virol, 59(1): 84-90
- Yarmolinsky L, Zaccai M, Ben-Shabat S, Huleihel M , (2010) Anti-herpetic activity of Callissia fragrans and Simmondsia chinensis leaf extracts in vitro. The open virology journal,4: 57-63
- Yarmolinsky L, Zaccai M, Ben-Shabat S, Mills D, Huleihel M, et al. (2009) Antiviral activity of ethanol extracts of Ficus binjamina and Lilium candidum in vitro. New biotechnology, 26(6): 307-313
- Shebl R, Amin I, Emad-Eldin M. A, Dajem A, Mostafa S. M. B, et al. (2012) Antiviral activity of liquorice powder extract against varicella zoster virus isolated from Egyptian patients. Chang Gung Medical Journal, 35(3): 231-39.
- Angamuthu D, Purushothaman I, Kothandan S , SwaminathanR,( 2019) Antiviral study on Punica granatum L., Momordica charantia L., Andrographis paniculata Nees. and Melia azedarach L., to Human Herpes Virus-3. European Journal of Integrative medicine, 28: 98-108
- Reed LJ, Muench H. A,( 1938) simple method of estimating fifty per cent endpoints. American journal of epidemiology, 27 (3): 493-97.
- Azhar Jabareen1, Mahmoud Huleihil2, Mahmoud Huleihel, (2013) Effect of Extracts of Passiflora edulis Leaves on Herpes Viruses Infection. J Virol Antivir Res, 2:2
- Zhang J, Zhan B, Yao X, Gao Y, Song J, et al. (1995) Antiviral activity of tannin from the pericarp of Punica granatum L. against genital Herpes virus in vitro. China journal of Chinesemateria medica, 20(9): 556-558.
- Moradi MT, Karimi A, Alidadi S, Gholami-Arjenaki M, (2015) In vitro anti-herpes simplex type-1 activity, antioxidant potential and total phenolic compounds of pomegranate (Punica granatum L.) peel extract. Journal of Chemical and Pharmaceutical Research, 7(8): 82- 88.
- Jagadeesan A, Swaminathan R, (2018) Study on antiviral activities, drug likeness and molecular docking of bioactive compounds of Punica granatum L. to Herpes Simplex Virus-2(HSV-2). Microbial Pathogenesis, 118: 301-309
- Gescher K, Kühn J, Lorentzen E, Hafezi W, Derksen A, et al. (2011) Proanthocyanidin-enriched extract from Myrothamnus flabellifolia Welw. exerts antiviral activity against herpes simplex virus type 1 by inhibition of viral adsorption and penetration. J Ethnopharmacol, 134: 468-474.
- Gescher K, Kühn J, Hafezi W, Louis A, Derksen A, et al. (2011) Inhibition of viral adsorption and penetration by an aqueous extract from Rhododendron ferrugineum L. as antiviral principle against herpes simplex virus type-1. Fitoterapia, (82): 408-413
- Qiu X, JansonC. A, Culp J. S, Richardson S. B, Debouck C, Smith W. W, et al. (1997) Crystal structure of varicella-zoster virus protease. Proceedings of the National Academy of Sciences, 94(7): 2874-2879
- McMillan D. J, Kay J, Mills J. S, (1997) Characterization of the proteinase specified by varicella zoster virus gene 33. Journal of general virology, 78(9): 2153-2157
- Matsumoto M, Misawa S, Chiba N, Takaku H, HayashH,et al. (2001) Selective non peptidic inhibitors of herpes simplex virus type 1 and human cytomegalovirus proteases. Biological and Pharmaceutical Bulletin, 24(3): 236-41
- Angamuthu D, Purushothaman I, Swaminathan R, Ramachandran S, (2016) Screening of potential phytochemicals against protease of Varicella-Zoster Virus- an in silico approach, World J. Pharm. Pharm. Sci,5 (5): 679-693.
- Spadola L, Novellino E, Folkers G, Scapozza L, ( 2003) Homology modelling and docking studies on Varicella Zoster Virus Thymidine kinase. European journal of medicinal chemistry, 38(4): 413-419
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi