Review Article, J Virol Antivir Res Vol: 7 Issue: 2
An Overview of Risk Factors of Vertical Transmission of Hepatitis C Virus and Treatment Options for Chronic HCV
1Department of Medicine, St. George's University of London, London, UK
2Reader in Department of Cardiovascular Immunology at St. George’s University of London, London, UK
*Corresponding Author : Elisavet Arsenaki
Department of Medicine, St. George's University of London, UK
Tel: +020 8672 9944
E-mail: elisavet.arsenaki@hotmail.com
Received: September 03, 2018 Accepted: October 15, 2018 Published: October 22, 2018
Citation: Arsenaki E, Christina B (2018) An Overview of Risk Factors of Vertical Transmission of Hepatitis C Virus and Treatment Options for Chronic HCV. J Virol Antivir Res 7:2. doi: 10.4172/2324-8955.1000185
Abstract
This essay focuses on hepatitis C transmission, the methods used for viral screening and the risk factors involved in mother-to-child transmission. Bearing in mind that HCV infected children rarely clear the infection after the age of four, different treatment options for chronic HCV will be discussed. Additionally, to identify the most effective treatment for children and adults, monotherapy will be compared to combined therapy.
Keywords: Hepatitis; Chronic HCV; Vertical transmission; Toxins
Introduction
Acute defining Hepatitis
Hepatitis is defined as inflammation of the liver and is mostly spread through viruses. Less commonly, hepatitis can emerge as a result of excessive alcohol consumption, certain drugs and/or toxins. When hepatitis is caused by a virus, it is termed viral hepatitis and can emerge from five different viruses: A, B, C, D or E.
Hepatitis C
Genotypes: There are six major HCV genotypes and they all cause liver disease, yet their infectivity varies [1-3]. The six genotypes vary in their nucleotide sequence by 30%-35% and “within a HCV genotype subtype”, the nucleotide sequence differs by 20%-25% [3]. Besides, there are more than fifty subtypes for each of the six major genotypes [4].
Chronic and acute infection: Acute hepatitis C is a short-term infection that emerges within the first 6 months after exposure to the virus. Once in the host, the virus can be spontaneously cleared, but this rarely occurs. In fact, acute infection generates chronic infection in approximately 70%-85% cases. In chronic hepatitis C, the virus can last a lifetime, resulting in serious complications such as liver cirrhosis and/or hepatic cancer. [5,6].
Epidemiology of HCV: The World Health Organization estimates that on a global scale, there were 71 million people living with chronic HCV in 2015. HCV distribution is variable “across and within countries”, with no regular pattern, but the areas classified as most affected include European and Eastern Mediterranean regions [7]. Concerning the epidemiology of HCV in the UK, according to NHS choices, approximately 215,000 people are infected, while the “Hepatitis C in the UK 2017 report” considers the number to be closer to 214,000. Injecting drug use is the leading cause of infection in the UK, with prevalence varying around the country [8-10].
HCV transmission
Hepatitis C is a blood borne infection and can be transmitted in two ways; horizontal transmission, which involves the spread of a particular disease among people who do not have a parent-child relation and vertical transmission, where spread occurs from mother to child [11]. Horizontal transmission of HCV can occur though the sharing of drug-injecting equipment or accidents in healthcare procedures, such as needle stick injuries, yet the leading mode of transmission is “percutaneous exposure to blood”, mainly via illegal drug injections [12]. HCV transmission can also potentially occur through sexual intercourse with an infected person, but the risk is currently perceived as low [6]. Still, the risk of sexual transmission does increase if an individual is co-infected with HIV and/or has multiple sexual partners [13]. Vertical transmission of HCV is the dominant cause of infection in infants. One third to one half of mother-to-child transmission takes place in utero, prior to the last month of pregnancy and the rest occurs either during the ninth month of pregnancy or delivery [12]. The prevalence of paediatric HCV infection varies from 0.05% to 0.36% in developed countries, while in the developing world, these rates range between 1.80% and 5.00% [14]. In general, the risk of vertical transmission is considered low, since only 6 out of 100 infants born to HCV infected mothers are found to be infected [14].
HCV transmission is unlikely to occur as a result of exposure to other body fluids, such as saliva and semen. The virus does not spread through coughing and sneezing, sharing eating utensils or through physical contact. Furthermore, there is no evidence that viral transmission occurs via human breast milk, so it is safe for HCV infected mothers to breastfeed their babies [12]. However, women with cracks and/or bleeding in their breasts, or acute hepatitis C during pregnancy, are advised to avoid breastfeeding [12,15].
Factors that influence vertical transmission of HCV
A risk factor for vertical transmission of hepatitis C is circulating HCV RNA, which is present in 70% of anti-HCV positive pregnant women [15]. Pregnant women that present with no HCV RNA, or have irregular HCV RNA levels, rarely transmit the viral infection to their babies, with the rate of transmission being 0.3% [15]. It is supported that mother-to-child transmission has a high chance of occurring only if a woman has detectable HCV RNA levels while she is pregnant [12]. In addition, HIV co-infection is considered to be a risk factor for vertical transmission. In an article assessing the risk of vertical transmission, women having HCV were associated with a 5.8% risk, while for those co-infected with HIV, the risk rose to 10.8% [16]. Furthermore, mode of delivery has not been found to influence the rate of transmission [12].
A study that followed 214 HCV positive women identified circulating HCV RNA in 69% of them, with all mothers of infected children having HCV viraemia [17]. 26% of the mothers were coinfected with HIV and in those, the risk of mother-to-child transmission was found to be three-fold higher. Children were tested for being HCV infected at birth, 3, 6 and 12 months of age. At 3 months of age, a total of 12 children were found to have circulating HCV RNA and 50% of them were identified as being born to HCV/ HIV-co-infected mothers. As for HCV RNA levels, it was concluded that the risk of transmission is increased in mothers having HCV RNA that is higher than 6.0 log-copies/ml (Which is equivalent to >1,000,000 copies per ml) [17]. In HCV/HIV-co-infected mothers, the risk of vertical transmission was independent of HIV status of the mother if HCV RNA levels were at least 6.0log-IU/ml, but if they were lower, the risk of mother-to-child transmission elevated. This indicates that additional mechanisms may be involved in HCV vertical transmission. What is more, vertical transmission was not found to be influenced by the HCV genotype and the number of pregnancies did not affect the transmission of the virus either. Therefore, the chances of a mother transmitting HCV to her second child did not increase or decrease according to whether her first child was HCV-infected. There was no significant difference identified when comparing vaginal and caesarian delivery, while in the case of emergency caesarean sections, children were not found to be infected [17]. Thus, the study concluded that different modes of delivery, including vaginal delivery, elective and emergency caesarian sections, do not influence vertical transmission in children of HCV/HIV-co-infected women.
Similar results were obtained in a clinical review that found no correlation between the HCV genotype and the rate of vertical transmission [18]. The occurrence of vertical transmission in one pregnancy was also not found to influence subsequent pregnancies. Furthermore, the same clinical review cites numerous studies that found the risk of infection to be increased in children whose mothers had a high hepatitis C viral load, in contrast to two Italian studies that are mentioned, which found no significant difference in the viral load of women who infected their children and others who did not. As for HCV/HIV-co-infection, it is stated that “the frequency of HCV transmission was consistently higher”, in multiple different studies, when mothers were HIV-co-infected. A study carried out in the UK and Ireland is particularly discussed, where a 3.8 times higher risk of vertical transmission was reported in mothers possessing HIV antibodies [18].
Screening for HCV in pregnant women and their babies
When are pregnant women screened?: The UK National Screening Committee does not recommend routine screening, due to the fact that vertical transmission rates are low. Accordingly, the HCV guidance provided by the American Association for the Study of Liver Diseases, regards routine HCV screening as unnecessary. However, both agree that pregnant women with “known or suspected risk factors for HCV infection” must undergo screening.
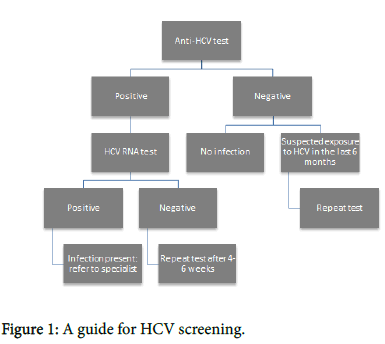
What are the different screening methods used to check for HCV?: A hepatitis C antibody blood test is initially carried out and if the result is negative, there is no infection. Still, if there is suspected exposure of the patient to HCV in the last six months, the test should be repeated. A positive anti-HCV test means that the patient has been infected, without this necessarily meaning that he/she currently has HCV, since the virus can be spontaneously cleared or remain in the blood. In order to differentiate between these scenarios, a follow up RNA test is required; most commonly a PCR test. HCV RNA is only identified in individuals with current infection [11]. Therefore, a negative result verifies the absence of HCV infection. Nevertheless, this must be confirmed with a second HCV RNA test, four to six weeks later. If the HCV RNA test is positive, referral to a specialist is required [6,8] (Figure 1).
When should babies be screened?: Babies born to HCV infected mothers are screened for HCV antibodies once they reach 18 months of age [12]. However, the American Academy of Pediatrics, supports that antibody testing can be carried out even earlier, after 12 months of age. This is because although the time period during which maternally derived HCV antibodies persist in babies is unknown, it is “unlikely to be more than 12 months” [19].
Treatment of chronic HCV
Children who are younger than three years are not offered treatment, due to the chance of spontaneous viral clearance taking place [20]. However, after the age of four, this is unlikely to occur and 80% of the children that have been infected via vertical transmission develop chronic HCV [21]. NICE recommends the use of a peginterferon and ribavirin for treating chronic hepatitis C. This combined treatment is deemed appropriate for adult patients with hepatitis of “mild or moderate to severe” character, while the length of treatment is usually six to twelve months. Treatment is contraindicated in patients with a history of past medical conditions, such as liver, kidney and heart problems and also in pregnant women, due to the teratogenic effects of drugs like ribavirin on the fetus [12]. As a result, treatment needs to be transferred postpartum when it comes to pregnant women. In addition, ribavirin is contraindicated in ribavirin intolerant patients (NICE) and individuals with haemolytic anaemia or renal failure, where interferon monotherapy is used as the alternative treatment plan [22]. NICE guidelines state that peginterferon and ribavirin must be used for the treatment of chronic HCV, in HCV genotypes 2 and 3. For genotypes 1, 4, 5, 6, or multiple HCV genotype infections, the same treatment should be followed for twelve weeks, at which point viral load must be checked. If the viral load presents a decrease of “less than 1% of its level at the start of treatment”, the combined therapy needs to be adhered to for another 36 weeks. However, if there is no reduction in viral load, treatment is to be withdrawn.
The combined therapy of peginterferon alfa-2a and ribavirin is approved by NICE for use in children. It is appropriate for children who are at least five years old, have serum HCV RNA and have not received previous treatment for CHC. The administered dose depends on the child’s body surface area, with total treatment duration ranging from 24 to 48 weeks, depending on the presence or absence of a virological response after the first 24 weeks. Children with “a body surface area of less than 0.71 m2” cannot receive this therapy. Peginterferon alfa-2b and ribavirin constitute another approved therapy for children with CHC, who have not received previous treatment. To be offered this treatment, children must be at least three years old, have no hepatic decompensation and also be HCV RNA positive (NICE).
A published paper quoting a randomized controlled trial concludes that combination therapy of ribavirin and peginterferon is more successful than monotherapy in children [23]. The results obtained also suggest that HCV genotype 1 is less responsive to treatment than other genotypes. In the study, a total of 114 children were treated for 48 weeks. One group was treated using peginterferon alfa-2a and ribavirin, while the second group only received peginterferon alfa-2a. A sustained virological response was reached in 53% of the children following combined treatment, while 21% was observed in the monotherapy group (55 children were treated using combined therapy and 59 were treated with monotherapy). Comparing the effectiveness of the treatments in different HCV genotypes, a 47% sustained virological response was reported in genotype 1 infected children following the combination therapy, with this dropping to 18% in genotype 1 infected patients following monotherapy. In children with all other HCV genotypes, an 80% sustained virological response was noted when receiving combined therapy and 36% when following monotherapy [24].
Another study (A total of twelve children had acquired HCV through vertical transmission, twenty-three were infected with HCV genotype 1, six with genotype 3 and one with genotype 2) , compared peginterferon alfa-2b to alfa-2a. A total of twenty children were treated with peginterferon alfa-2b and ten with alfa-2a (All being given a ribavirin dose of 15 mg/kg), while the treatment duration differed according to the HCV genotype. Children infected with genotypes 2 and 3 were treated for 24 weeks, whereas those infected with genotype 1, followed the treatment for 48 weeks. Genotype 1 infection was found to be less responsive to treatment, in comparison to all other HCV genotypes, since “treatment success” was 56.5% for genotype 1 infected children and 100% for all other HCV genotype infections. Still, the difference was not statistically significant (P=0.1) . In terms of comparing the two peginterferons, the group being treated with alfa-2a presented a 90% success rate, in contrast to a rate of 55% in the group receiving alfa-2b. This result was also statistically insignificant (P=0.064) [20].
Monotherapy vs. combined therapy
Two separate stages of viral decline have been identified when IFNα is used to treat HCV. The first one represents the antiviral efficacy of the interferon, while the second phase consists of hepatocyte destruction. Ribavirin appears to have “little or no effect on the first phase”, but it reinforces the decrease in HCV levels in phase two. However, ribavirin monotherapy is deemed insufficient, because it fails to decrease HCV RNA levels significantly (Henry et al., 1997). Clinical trials have identified that ribavirin only leads to a small decrease in HCV RNA levels; therefore it lacks “direct antiviral activity against HCV” [25]. Likewise, the decrease in HCV RNA levels is minimal when using IFN or peginterferon monotherapy (Both on a daily basis).
The sustained virological responses noted following combined treatment with ribavirin plus a peginterferon, were 43%, 47% and 40% in three different controlled trials (Carried out in Taiwan, Italy and Sweden respectively) [26]. All three were higher in comparison to the rates yielded following IFN-a monotherapy, with sustained virological responses there being 6%, 13% and 20% (Ming-Yang, 2000). Correspondingly, in a randomized clinical trial, sustained virological response was elevated in patients given IFN-a (3 million units, three times per week) and ribavirin (1000-1200 mg per day, given orally), in comparison to IFN monotherapy [27]. The sustained virological response observed also varied depending on total treatment duration. The sustained virological response obtained after following combined treatment was 31% for a treatment length of 24 weeks and 38% for 48 weeks. Accordingly, sustained virological response after IFN monotherapy was 6% if the duration of treatment was 24 weeks and 13% if it was 48 weeks [28].
A study that compared peginterferon alpha-2a and ribavirin to peginterferon alpha-2a administered with a placebo, but also nonpegylated interferon alpha-2b and ribavirin, concluded that more patients receiving pegylated alfa-2a and ribavirin presented a sustained virological response, in comparison to the other two groups [22]. A higher sustained virologic response of 56% was reported in patients treated with peginterferon alfa-2a and ribavirin, in comparison to 44% in those receiving interferon alfa-2b and ribavirin. The ones receiving peginterferon alfa-2a and the placebo achieved a sustained virological response of 29%. In HCV genotype 1 infected patient, a sustained virological response of 46% was observed following peginterferon alpha-2a and ribavirin, in comparison to 21% in the peginterferon alpha-2a and placebo group. Lastly, the response recorded in patients receiving interferon alfa2-b and ribavirin was 36%. [22]. Thus, taking all the above into consideration, ribavirin plus peginterferon alpha-2a is the most effective treatment.
Treating different HCV genotypes
HCV genotypes 2 and 3 are susceptible to treatment combining pegylated IFNα and ribavirin, while genotype 1 is significantly less susceptible, with HCV genotype 1 patients presenting a virological response lower than 50% [3]. A research paper identified that pegylated IFNα and ribavirin treatment used for a total of 11 months produced virological response rates of 50 to 60% [25]. These rates were elevated in genotype 2 and 3 HCV infections, reaching a 75 to 80% response, whereas the response rates observed in patients with HCV genotype 1 ranged from 46 to 52% (The higher sustained virological response rates detected in genotypes 2 and 3, after the 24 weeks of combined therapy, were reached using a lower daily ribavirin dosage of 800 mg instead of 1000-1200 mg) [25]. As a result, the standard treatment plan for HCV genotypes 2 and 3 is pegylated IFNα and ribavirin.
A more recent triple therapy for HCV genotype 1 infected patients, adds a NS3 protease inhibitor (Telapravir and boceprevir include protease inhibitors that have been tested in adults) to the pegylated IFNα and ribavirin [3]. In comparison to dual treatment (Increasing the SVR from 38% to 41% with dual therapy up to 67% to 75% with triple therapy), this triple therapy resulted in an increase of up to 37% in sustained virological response [23]. Nevertheless, the only approved treatment for children is ribavirin and peginterferon combined therapy since other treatment options lack a license [29-32].
Conclusion
In conclusion, there is no evidence to support that mode of delivery affects the rate of vertical transmission of hepatitis C, while HIV coinfection is considered to increase the risk of mother-to-child transmission. In addition, the majority of published papers agree that the higher the HCV RNA serum levels in mothers, the higher the risk of vertical transmission occuring. When it comes to treating HCV, combined therapy of ribavirin and pegylated IFNα is the first line treatment in adults and the only currently approved therapy for children; with the peginteferon used being either alfa-2a or alfa-2b. This combined treatment is offered to patients that are infected with HCV genotypes 2 and/or 3, while a triple therapy of ribavirin, pegylated IFNα and a NS3 protease inhibitor is advised for HCV genotype 1 infection. Alternatively, NICE guidelines state that peginterferon and ribavirin are suitable for use regardless of the HCV genotype, but, in genotypes 1, 4, 5 and 6, or in the case of multiple HCV genotype infections, if there is no significant decrease in viral load after the first twelve weeks of therapy, the treatment must be discontinued.
References
- Ashfaq U, Javed T, Rehman S, Nawaz Z, Riazuddin S (2011) An overview of HCV molecular biology, replication and immune responses. Virology Journal 8: 161.
- Interferon alfa (pegylated and non-pegylated) and ribavirin for the treatment of chronic hepatitis C. (2004) National Institute for Health and Care Excellence, United Kingdom.
- Heim M (2012) Interferons and hepatitis C virus. Swiss Med Wkly 142: 1-2.
- CDC (2013) HEPATITIS C What to Expect When Getting Tested. US Department of Health & Human Services, USA.
- CDC (2016) Hepatitis C FAQs for the Public. US Department of Health & Human Services, USA.
- CDC (2013) Testing for HCV Infection: An Update of Guidance for Clinicians and Laboratorians. US Department of Health & Human Services, USA.
- Hutin Y, Hess S, Bulterys M, Hirnschall G (2017) Global Hepatitis Report 2017. World Health Organization.
- Hepatitis C Quick reference guide for primary care (2011) NHS, UK.
- Hepatitis C Conditions (2015) NHS, UK.
- Hepatitis (2016) NHS, UK.
- CDC (2016) What is Viral Hepatitis? US Department of Health & Human Services, USA.
- Hughes B, Page C, Kuller J (2017) Hepatitis C in pregnancy: screening, treatment, and management. Am J Obstet Gynecol 217: 3-9.
- Picco M (2014) Hepatitis C: How common is sexual transmission? Mayo Clinic, USA.
- Yeung C, Lee H, Chan W, Jiang C, Chang S, et al. (2014) Vertical transmission of hepatitis C virus: Current knowledge and perspectives. World J Hepatol 6: 643-651.
- Resti M, Bortolotti F, Vajro P, Maggiore G (2003) Guidelines for the screening and follow-up of infants born to anti-HCV positive mothers. Dig Liver Dis 35: 453-457.
- Benova L, Mohamoud Y, Calvert C, Abu-Raddad L (2014) Vertical transmission of Hepatitis C virus: Systematic review and meta-analysis. Clin Infect Dis 59: 765-773.
- Mariné-Barjoan E, Berrébi A, Giordanengo V, Favre S, Haas H, et al. (2007) HCV/HIV co-infection, HCV viral load and mode of delivery: risk factors for mother-to-child transmission of hepatitis C virus?AIDS Society Journal 21: 1811-1815.
- Conte D, Colucci A, Minola E, Fraquelli M, Prati D (2001) Clinical course of pregnant women with chronic hepatitis C virus infection and risk of mother-to-child hepatitis C virus transmission. Dig Liver Dis 33: 3-4.
- Joan C, Margaret CF, Michael A, Donald S, Steve K (1998) Hepatitis C virus infection. Official Journal of the American Academy of Pediatrics 101: 3-4.
- Rosen I, Kori M, Adiv O, Yerushalmi B, Zion N, et al. (2013) Pegylated interferon alfa and ribavirin for children with chronic hepatitis C. World J Gastroenterol 19: 1-4.
- Jara P, Hierro L (2010) Treatment of Hepatitis C in children. Expert Rev Gastroenterol Hepatol 4: 1-3.
- Fried M, Shiffman M, Reddy K, Smith C, Marinos G, et al. (2002) Peginterferon alfa-2a plus ribavirin for chronic Hepatitis C Virus infection. N Engl J Med 347: 975-982.
- Hu J, Doucette K, Hartling L, Tjosvold L, Robinson J (2010) Treatment of Hepatitis C in children: A systematic review. Plos One 7: 4-6.
- Peginterferon alfa and ribavirin for treating chronic hepatitis C in children and young people (2013) National Institute for Health and Care Excellence. UK.
- Chung R, Polyak S, Lemon S, Liang T, Hoofnagle J, et al. (2008) Mechanisms of action of interferon and ribavirin in chronic Hepatitis C: Summary of a workshop. Hepatology 47: 306-320.
- Ming-Yang L (2000) First line treatment for hepatitis C: combination interferon/ribavirin vs. interferon monotherapy. J Gastroenterol Hepatol 15: E130–E133.
- McHutchison JG, Gordon SC, SchiffInterferon ER (1998) Alfa-2b alone or in combination with ribavirin as initial treatment for chronic Hepatitis C. The New England Journal of Medicine 339: 3-5.
- McHutchison J, Gordon S, Schiff E, Shiffman M, Lee W, et al. (1998) Interferon Alfa-2b Alone or in Combination with Ribavirin as Initial Treatment for Chronic Hepatitis C. N Engl J Med 339: 1485-1492.
- Squires J, Balistreri W (2017) Hepatitis C Virus Infection in children and adolescents. Hepatol Communic 1: 87-98.
- Costella A, Craine N, Goldberg D, Harris H, Hutchinson S, et al. (2017) Hepatitis C in the UK 2017 Report. Public Health England, London.
- Cutler N (2006) Understanding Hepatitis C Interferon Therapy. Hepatitis Central, University of Rochester, USA.
- HCV in Pregnancy (2017) American Association for the Study of Liver Diseases and the Infectious Diseases Society of America, Virginia, USA.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi