Research Article, J Spine Neurosurg Vol: 8 Issue: 3
Anterior Cervical Discectomy and Fusion Using Porous PEEK Implants at Levels Adjacent to a Previous Fusion
J Kenneth Burkus1* and Christopher Rehak2
1Staff Physician, Spine Service, The Hughston Clinic, Columbus, Georgia
2Spine Service, The Hughston Clinic, Columbus, Georgia
- *Corresponding Authors:
- JK Burkus
The Hughston Clinic, 6262 Veterans Parkwa,
Columbus, Georgia
Tel: (706) 494-3239
Fax: (706) 494-3102
E-mail: jkb66@knology.net
Received:April 15, 2019Accepted: May 14, 2019 Published: May 14, 2019
Citation: Burkus JK, Rehak C (2019)Anterior Cervical Discectomy and Fusion Using Porous PEEK Implants at Levels Adjacent to a Previous Fusion. J Spine Neurosurg 8:3.
Abstract
Objective: We report the results of a prospective, single center clinical study in which anterior cervical discectomy and fusion (ACDF) was performed using a porous PEEK implant and plate at intervertebral disc spaces in patients who had symptomatic, degenerative cervical disc disease at levels adjacent to a previous ACDF. Methods: Twenty-eight consecutive patients (15 females, 13 males; average age, 54 years) with degenerative cervical disc disease at levels adjacent to a previous fusion underwent anterior cervical discectomy and fusion (ACDF) using a porous PEEK interbody fusion cage and plate. There were 22 single-level and 6 two-level adjacent level fusions between C3 and C7. Patients were assessed at 1.5, 3, 6, 12 and 24 months. Standardized clinical outcome measures were used to evaluate the patient’s condition before and after surgery. Plain radiographs were used to assess fusion, bony ingrowth, sagittal plane angulation, subsidence, and migration of the implant. Results: No patients were lost to follow up; all patients were followed for a minimum of 12 months. Six patients were followed for more than 24 months. At the last follow up, NDI and neck and pain scores showed improvement in all patients. All had previous anterior cervical fusions and 22 had at least 1 additional co-morbidly for an adverse event (diabetes, osteoporosis, smoking, obesity). Following revision surgery, all patients showed radiographic fusion with no motion across the instrumented interspaces on dynamic flexion-extension lateral radiographs. Segmental lordosis at the surgical site improved to an average of 5° (2° to 9°) with no evidence of implant migration or subsidence. Average disc space height increased 4 mm. No patient showed radiographic evidence of a pseudarthrosis. No patient developed any radiolucency at the implant interface with the host bone. Conclusion: This prospective, nonrandomized study shows that porous-surface PEEK is a clinically viable alternative for improving the osseointegration and fusion rates of interbody implants in patients undergoing revision fusion surgery at 1 to 2 levels adjacent to previous surgical fusions.
Keywords: Porous PEEK; Anterior cervical interbody fusion; Degenerative cervical disc disease; Multilevel; Spinal fusion; Revision anterior cervical discectomy and fusion
Introduction
Cervical interbody fusion devices have been developed to enhance fusion rates and improve clinical outcomes while avoiding the complications associated with harvesting structural autografts. Contemporary interbody fusion cages and devices have been made from machined allografts [1,2], titanium [3] and polyether-etherketone (PEEK) [4-6]. Allograft and PEEK implants have favorable mechanical properties and imaging characteristics when compared with metal implants. Allograft has been shown to be comparable to autograft with regard to efficacy when used for ACDF in one-level instrumented procedures [2,7]. However, fusion rates for allograft often fall below the fusion rate for autograft [8], and are also reduced in smokers [9-11] and diabetic patients [12], and in patients undergoing multilevel [13-15] and revision surgeries [16,17].
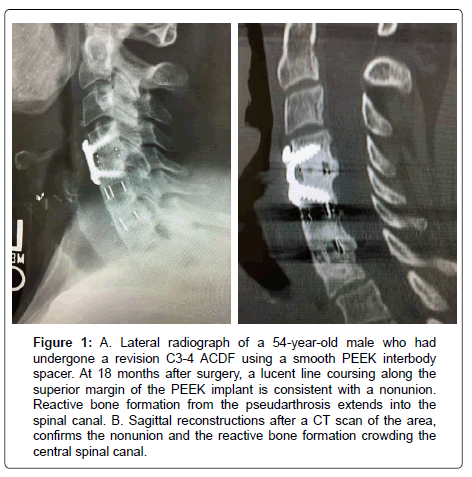
Polyetheretherketone (PEEK) has been utilized in cervical interbody constructs because of its high strength, radiolucency, and similar modulus to bone [5,6,8]. However, smooth-surfaced PEEK implants (Figure 1) have been associated with fibrous tissue formation, migration, and subsidence [5].
Figure 1:A. Lateral radiograph of a 54-year-old male who had undergone a revision C3-4 ACDF using a smooth PEEK interbody spacer. At 18 months after surgery, a lucent line coursing along the superior margin of the PEEK implant is consistent with a nonunion. Reactive bone formation from the pseudarthrosis extends into the spinal canal. B. Sagittal reconstructions after a CT scan of the area, confirms the nonunion and the reactive bone formation crowding the central spinal canal.
Reactive bone formation from the pseudarthrosis extends into the spinal canal. B. Sagittal reconstructions after a CT scan of the area, confirms the nonunion and the reactive bone formation crowding the central spinal canal.
The innovative application of new porous surface technology has been shown to enhance osseointegration and fixation of PEEK implants. In vitro and in vivo data have demonstrated enhanced osteoconductivity and osseointegration of the porous PEEK interbody fusion devices [18-20]. Mechanical testing has shown the porous PEEK surface architecture to withstand significant loading stresses [21].
Adverse clinical outcomes and complications after surgery have been associated with several clinical conditions [22]. Clinical risk factors associated with pseudarthrosis [23] and other adverse events include current or former smokers [9-11], diabetes [12], and obesity [24]. Additional cervical surgery is associated with significantly increased risk of multiple adverse outcomes, including thromboembolic events, surgical site infections, return to the operating room, blood transfusion, and readmission within 30 days [16,17]. Patients who undergo revision surgery or multilevel fusions have a greater incidence of and risk for perioperative wound infections, hematomas, dysphagia, and neurologic complications relative to the primary procedures [13-15,25]. Our goal in this study was to evaluate the clinical and radiologic outcomes in patients undergoing a second anterior cervical surgery in which a porous PEEK implant and plate was used at levels adjacent to a previous instrumented ACDF.
Materials and Methods
Twenty-eight consecutive patients with degenerative disc disease, cervical spondylosis and associated radiculopathy at levels adjacent to a prior anterior interbody fusion were treated by an anterior cervical discectomy and interbody fusion using a porous PEEK implant, cervical plate and autogenous iliac crest bone graft. They underwent 22 singlelevel and 6 two-level revision fusion surgeries between C3 and T1. Patients’ clinical and radiographic outcomes were followed prospectively.
Patient demographics
Thirteen males (36%) and 15 females (54%) with an average age of 54 years (range, 37 to 67 years) were treated between May 2016 and June 2017 (Table 1). All patients had clinical complaints of neck pain and radiating arm pain that was unresponsive to a minimum of 8 weeks of nonoperative treatment that included immobilization, traction, modalities, medications, and physical therapy. In addition to recurrent or persistent complaints of pain, all patients had an objective neurologic deficit which included 1 or more of the following: an asymmetric deep tendon reflex, a sensory deficit in a dermatomal pattern, or motor weakness. All patients had a correlative neuroradiographic study. We recorded patients’ smoking status and presence of diabetes, osteoporosis, and obesity.
Table 1: Demographic and surgical characteristics in 28 patients undergoing ACDF in which a porous PEEK implant and plate was used at levels adjacent to a previous ACDF. ACDF=anterior cervical discectomy and fusion.
| Patient | Sex/Age | Second Surgery Levels | Previous Surgery Levels | Smoke | Osteo | Diabetic | Obese |
|---|---|---|---|---|---|---|---|
| 1 | F/64 | C3-4 | C4-5, C5-6 | N | N | N | N |
| 2 | F/42 | C5-6 | C4-5, C6-7 | N | N | N | Y |
| 3 | M/62 | C6-7 | C5-6 | N | N | N | N |
| 4 | F/63 | C4-5 | C5-6, C6-7 | N | Y | N | N |
| 5 | F/53 | C3-4 | C4-5, C5-6, C6-7 | Y | Y | N | N |
| 6 | F/53 | C4-5, C6-7 | C5-6 | N | Y | N | N |
| 7 | M/54 | C4-5 | C5-6, C6-7 | N | N | N | Y |
| 8 | F/67 | C3-4, C5-6 | C4-5 | Y | Y | Y (Type 2) |
N |
| 9 | F/47 | C6-7 | C4-5, C5-6 | N | N | N | Y |
| 10 | M/49 | C6-7 | C5-6 | N | N | N | N |
| 11 | F/55 | C4-5 | C5-6, C6-7 | N | N | N | Y |
| 12 | F/63 | C4-5 | C5-6, C6-7 | N | N | N | Y |
| 13 | F/52 | C4-5 | C5-6, C6-7 | Y | N | N | N |
| 14 | M/56 | C3-4 | C4-5 | N | Y | Y (Type 2) |
Y |
| 15 | M/59 | C4-5 | C5-6, C6-7 | N | N | N | Y |
| 16 | M/58 | C3-4 | C4-5 | Y | N | Y (Type 2) |
N |
| 17 | F/53 | C5-6 | C6-7 | N | N | N | N |
| 18 | M/50 | C5-6, C6-7 | C7-T1 | Y | Y | N | N |
| 19 | M/51 | C7-T1 | C5-6, C6-7 | N | Y | N | N |
| 20 | M/61 | C3-4, C4-5 | C5-6 | N | N | Y (Type 2) |
N |
| 21 | M/65 | C5-6, C6-7 | C3-4, C4-5 | Y | N | N | N |
| 22 | F/42 | C4-5 | C5-6 | N | N | N | Y |
| 23 | M/51 | C4-5 | C5-6, C6-7 | N | N | Y (Type 2) |
Y |
| 24 | F/56 | C4-5 | C5-6, C6-7 | N | Y | Y (Type 2) |
N |
| 25 | F/37 | C4-5, C6-7 | C5-6 | N | N | N | N |
| 26 | M/54 | C5-6 | C4-5 | N | N | N | Y |
| 27 | F/46 | C4-5, C5-6 | C6-7 | N | N | N | N |
| 28 | M/56 | C4-5 | C5-6 | N | N | N | Y |
Clinical and radiographic follow up
Patients were examined at 1.5, 3, 6, 12, and 24 months. Functional outcomes were assessed by the Neck Disability Index questionnaire (NDI), which was used to measure the effects of neck pain associated with activities of daily living [26,27]. Numeric rating scales were used to assess neck and arm pain intensity. Patients rated their neck pain duration and intensity on a scale from 0-10, with a score of 0 representing “no pain,” and a score of 10 representing “pain as bad as it could be.” Neurological success was defined as maintenance or improvement in 3 objective clinical findings: sensory, motor, and reflex testing.
Neutral anteroposterior and lateral radiographs were obtained at each visit. Dynamic flexion-extension lateral radiographs were taken at 6, 12, and 24 months. Sagittal plane angulation was measured on neutral lateral radiographs and determined by Cobb’s criteria. Intradiscal distraction and subsidence was measured by assessing the vertical distance between the midpoints of the adjacent vertebral endplates. Intradiscal motion ad implant migration was assessed on the dynamic radiographs.
Surgical technique
A standard left-sided approach to the cervical spine was used, and a transverse skin incision was made over the appropriate interspaces. The platysma was divided in the direction of the skin incision and blunt dissection was carried out separating the carotid sheath and the trachea, esophagus and strap muscles. Dissection was limited within the neck to this avascular plane. The anterior plate from the earlier surgery was exposed with bunt dissection and removed, and the interbody fusion was explored. The adjacent degenerated disc levels were exposed.
Anterior cervical discectomy was performed; anatomic disc space height and segmental sagittal contours were restored. The posterior annulus, the posterior longitudinal ligament, and the uncovertebral joint spurs were removed. The bony endplates were preserved. A porous PEEK implant was packed with autogenous graft and impacted into the interspace. An anterior cervical plate was applied over the implants.
Results
All patients in the study had a minimum follow up of 12 months; 7 were followed for 24 months. No patients were lost to follow up.
Clinical outcomes
After surgery, patients’ mean neck and arm pain scores showed improvement from preoperative scores. At last follow up, average neck pain improved to 3.9 from a preoperative average score of 7.6. Similarly, arm pain showed improvement at last follow up from a preoperative average of 6.9 to a postoperative average of 3.7 points. Neurological success was seen in all study patients with no patient showing a loss in neurological functioning for the duration of the study. Preoperative NDI scores averaged 56.8 points. At last follow up, NDI scores averaged 30.1 points for a mean overall NDI score improvement of 26.1 points when compared with preoperative scores.
Radiographic outcomes
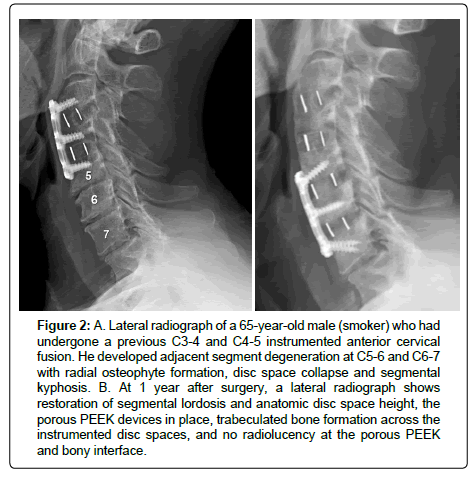
All patients showed radiographic evidence of fusion at 6 months after surgery. No patient showed motion across the fused interspace on dynamic flexion-extension lateral radiographs (Figure 2). There was no evidence of implant migration or hardware failure on postoperative radiographs. No patient showed radiographic evidence of a pseudarthrosis, and no patient had lucency around the implant.
Figure 2:A. Lateral radiograph of a 65-year-old male (smoker) who had undergone a previous C3-4 and C4-5 instrumented anterior cervical fusion. He developed adjacent segment degeneration at C5-6 and C6-7 with radial osteophyte formation, disc space collapse and segmental kyphosis. B. At 1 year after surgery, a lateral radiograph shows restoration of segmental lordosis and anatomic disc space height, the porous PEEK devices in place, trabeculated bone formation across the instrumented disc spaces, and no radiolucency at the porous PEEK and bony interface.
The average disc space height was increased by 4 mm after surgery. The sagittal plane alignment was improved in all patients at surgery and was maintained at last follow up. The preoperative sagittal plane angulation was -1° (lordosis) and ranged from +4° (kyphosis) to -3° (lordosis). Average sagittal plane angulation improved to an average of -5° (-2° to -9° lordosis).
Adverse events
No adverse events occurred at surgery or were identified during postoperative follow-up examinations. No patient underwent additional surgical procedures, and no patient underwent a posterior decompression or supplemental posterior fixation.
Discussion
Cervical disc herniations with associated radiculopathy can be effectively treated with anterior discectomy and interbody fusion. This procedure has been found to be safe and reliable in achieving improved patient outcomes and satisfaction [28-30]. Fusion in the spine is a complex process that does not always heal successfully. This process is, in part, a race among resorption of the graft material, cellular apoptosis, and the formation of new bone growing through the graft connecting the 2 adjacent, mobile vertebral bodies. Several factors influence the rates of healing after interbody fusion. The type of graft and interbody spacer used influences biomechanical stress across the interspace [18]. Autogenous grafts have a higher rate of healing when compared to allografts [1,7,8]. The surgical technique involving endplate preparation and graft insertion also influences the rate of fusion [31,32].
In this study, the high rate of fusion, preservation of disc space height and sagittal contours was associated with the use of the unique porous surface of the interbody fusion cages. With this surgical technique, the bony endplates were preserved. The porous PEEK implant surface technology promotes a superior osteogenic response and improves the bone-implant interface for spinal interbody. The improved integration of the implant surface to the host bone facilitates fusion by stabilizing the vertebral motion segment and reducing the contact stresses at the graft-vertebral interface. The deleterious effects of smoking, diabetes, osteoporosis and obesity on rates of fusion may be overcome by utilizing this technique of endplate preparation, interbody grafting, and stabilization.
Conclusion
Our purpose was to assess the bone healing response to a porous PEEK interbody implant used in challenging healing environments in patients with degenerative cervical disc disease. Porous-surface PEEK is a clinically viable alternative for improving the osseointegration and fusion rates of interbody implants in patients undergoing revision surgery at 1 to 2 levels adjacent to previous surgical fusions.
References
- Miller LE, Block JE (2011) Safety and effectiveness of bone allografts in anterior cervical discectomy and fusion surgery. Spine (Phila Pa 1976) 36: 2045-2050.
- Zdeblick TA, Ducker TB (1991) The use of freeze-dried allograft bone for anterior cervical fusions. Spine (Phila Pa 1976) 16: 726-729.
- Seaman S. Kerezoudis P, Bydon M, Torner JC, Hitchon PW (2017) Titanium vs. polyetheretherketone (PEEK) interbody fusion: Meta-analysis and review of the literature. J Clin Neuro Sci 44: 23-29.
- Burkus JK (2018) Early outcomes of anterior cervical discectomy and fusion using a porous PEEK interbody fusion device. J Spine Neuro Surg 7: 2-7.
- Kersten RF, Gaalen SM, Gast A, Oner FC (2015) Polyetheretherketone (PEEK) cages in cervical applications: a systematic review. Spine J 15: 1446-1460.
- Liu JM, Xiong X, Peng AF, Xu M, Chen XY, et al. (2017) A comparison of local bone graft with PEEK cage versus iliac bone graft used in anterior cervical discectomy and fusion. Clin Neurol Neurosurg 155: 30-35.
- Shapiro S (1996) Banked fibula and the locking anterior cervical plate in anterior cervical fusions following cervical discectomy. J Neurosurg 84: 161-165.
- Burkus JK, Dryer RF, Arnold PM, Foley KT (2017) Clinical and radiographic outcomes in patients undergoing single-level anterior cervical arthrodesis: a prospective trial comparing allograft to a reduced dose of rhbmp-2. Clin Spine Surg 30: E1321-E1332.
- Beerman D, Oren JH, Bendo J, Spivak J (2017) The effect of smoking on spinal fusion. Int J Spine Surg 28: 29.
- Brown CW, Orme TJ, Richardson HD (1986) The rate of pseudoarthosis in patients who are smokers and patients who are not smokers: A comparison study. Spine (Phila Pa 1976) 11: 942-943.
- Hilibrand AS, Fye MA, Emery SE, Palumbo MA, Bohlman HH (2001) Impact of smoking on the outcome of anterior cervical arthrodesis with interbody or strut-grafting. J Bone Joint Surg Am 83: 668-673.
- Phan K, Kim JS, Lee NJ, Kothari P, Cho SK (2017) Relationship between ASA scores and 30-day readmissions in patients undergoing anterior cervical discectomy and fusion. Spine (Phila Pa 1976) 42: 85-91.
- Laratta JL, Reddy HP, Bratcher KR, McGraw KE, Carreon LY, et al. (2018) Outcomes and revision rates following multilevel anterior cervical discectomy and fusion. J Spine Surg 4: 496-500.
- Nandyala SV, Marquez-Lara A, Fineberg SJ, Singh K (2014) Comparison of revision surgeries for one-to two-level cervical TDR and ACDF from 2002 to 2011. Spine J 14: 2841-2846.
- Tumialán LM, Pan J, Rodts GE, Mummaneni PV (2008) The safety and efficacy of anterior cervical discectomy and fusion with polyetheretherketone spacer and recombinant human bone morphogenetic protein-2: a review of 200 patients. J Neurosurg Spine 8: 529-535.
- Schroeder GD, Boody BS, Kepler CK, Kurd MF, Silva S, et al. (2018) Comparing health-related quality of life outcomes in patients undergoing either primary or revision anterior cervical discectomy and fusion. Spine (Phila Pa 1976) 43: 752-757.
- Carpenter RD, Klosterhoff BS, Torstrick FB, Foley KT, Burkus JK, et al. (2018) Effect of porous orthopaedic implant material and structure on load sharing with simulated bone ingrowth: a finite element analysis comparing titanium and PEEK. J Mech Behav Biomed Mater 80: 68-76.
- Torstrick FB, Evans NT, Stevens HY, Gall K, Guldberg RE, et al. (2016) Do surface porosity and pore size influence mechanical properties and cellular response to PEEK? Clin Orthop Relat Res 474: 2373-2383.
- Torstrick FB, Safranski DL, Burkus JK, Chappuis JL, Lee CSD, et al. (2017) Getting PEEK to stick to bone: the development of porous PEEK for interbody fusion devices. Tech Orthop 32: 158-166.
- Evans NT, Torstrick FB, Lee CS, Dupont KM, Safranski DL, et al. (2015) High-strength, surface-porous polyether-ether-ketone for load-bearing orthopedic implants. Acta Biomater. 13: 159-67.
- Gerling MC, Radcliff K, Isaacs R, Bianco K, Jalai CM, et al. (2017) Two-year results of the prospective spine treatment outcomes study: an analysis of complications rates, predictors of their development, and effect on patient derived outcomes at 2 years for surgical management of cervical spondylotic myelopathy. World Neurosurg 106: 247-253.
- Hofler RC, Swong K, Martin B, Wemhoff, Jones GA (2018) Risk of pseudoarthosis after spinal fusion: analysis from the healthcare cost and utilization project. World Neurosurg 120: 194-202.
- Narain AS, Hijji FY, Haws BE, Kudaravalli KT, Yom KH, et al. (2018) Impact of body mass index on surgical outcomes, narcotics consumption, and hospital cost following anterior cervical discectomy and fusion. J Neurosurg Spine 28:160-166.
- Veeravagu A, Cole T, Jiang B, Ratliff JK (2014) Revision rates and complication incidence in single and multilevel anterior cervical discectomy and fusion procedures: an administrative database study. Spine J 14: 1125-1131
- Vernon H, Mior S (1991) The Neck Disability Index: a study of reliability and validity. J Manipulative Physiol Ther 14: 409-15.
- Westaway M, Stratford P, Binkley J (1998) The patient-specific functional scale: validation of its use in persons with neck dysfunction. J Orthop Sports Phys Ther 27: 331-338.
- Buttermann GR (2018) Anterior cervical discectomy and fusion outcomes over 10 years: a prospective study. Spine (Phila Pa 1976) 43: 207-214.
- Gore DR, Sepic SB (1984) Anterior cervical fusion for degenerated or protruded discs. A review of on-hundred forty-six patients. Spine (Phila Pa 1976) 9: 667-671.
- Katsuura A, Hukuda S, Imanaka T, Miyamoto K, Kanemoto M (1996) Anterior cervical plate used in degenerative disease can maintain cervical lordosis. J Spinal Disorders 9: 470-476.
- Brodke DS, Zdeblick TA (1992) Modified Smith-Robinson procedure for anterior cervical discectomy and fusion Spine (Phila Pa 1976) 17: 427-430.
- Emery SE, Bolesta MJ, Banks MA, Jones PK (1994) Robinson anterior cervical fusion: Comparison of the standard and modified techniques. Spine (Phila Pa 1976) 19: 660-663.
- Krag MH, Robertson RA, Johnson CC, Stein AC (1977) Anterior cervical fusion using a modified tricortical bone graft: A radiographic analysis of outcome. J Spinal Disord 10: 420-430.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi