Research Article, J Soil Sci Plant Health Vol: 2 Issue: 1
Are Artificial Media Valid For Root Analysis? A Case Study Comparing Root Traits of Five Lentil Genotypes in Artificial Media versus Soil
Linda Yuya Gorim1*, Eliza Mohsenzadeh Rabani1, Brent Barlow2, Devini De Silva2 and Albert Vandenberg1,2
1Department of Plant Sciences, College of Agriculture and Bio resources, University of Saskatchewan, Saskatoon, SK, Canada
2Crop Development Centre, Department of Plant Sciences, College of Agriculture and Bioresources, University of Saskatchewan, Saskatoon, SK, Canada
*Corresponding Author : Linda Yuya Gorim
Department of Plant Sciences, College of Agriculture and Bio-resources, University of Saskatchewan, Saskatoon, SK, Canada
Tel: +1 306 966 1956
E-mail: linda.gorim@usask.ca
Received: December 15, 2017 Accepted: January 01, 2018 Published: January 10, 2018
Citation: Gorim LY, Rabani EM, Barlow B, De Silva D, Vandenberg A (2018) Are Artificial Media Valid For Root Analysis? A Case Study Comparing Root Traits of Five Lentil Genotypes in Artificial Media versus Soil. J Soil Sci Plant Health 2:1.
Abstract
Background: Interest in plant root systems by plant physiologists and breeders has grown in recent years as challenges compounded by global warming further increases the complexity of crop production systems. Given the difficulties of assessing roots under field conditions, work or roots are increasingly being carried out using artificial media in containers indoors. This study addresses the question of which media to use by comparing above and below ground plant growth characteristics of lentil genotypes grown under fully watered and drought conditions in both inorganic and organic media compared to soil.
Methods: Five lentil genotypes were grown indoors until flowering in these media. Above/below ground biomass was assessed in each media. Root traits were evaluated with the aid of the root program, WinRhizoTM.
Results: This study demonstrated that under fully watered and drought conditions, lentil genotypes partitioned resources to above and below ground structures differently in soil as opposed to artificial media indoors. The organic compared to the inorganic medium, had similar root traits as soil for most lentil genotypes under both moisture
levels.
Conclusions: The inorganic media cannot be recommended for lentils cultivation because plants were stunted with large root systems. Secondly, preliminary projects on below ground parameters should be carried out in artificial media to see how they compared to soil before plants are grown in artificial media.
Keywords: Below and above ground traits; Soil; Artificial media; Drought; Lenti genotypes
Introduction
Above ground plant structures have been the subject of considerable research resulting in huge datasets, but roots have not been the focus of as much scientific scrutiny largely due to the technical difficulty of studying them [1]. In the 1990s, roots became the focus of research, mostly in the context of water and nutrient uptake for increased crop productivity in different ecological zones [2]. In more recent times, growing awareness of the increasing variability of climatic conditions has shifted the focus of plant physiologists and plant breeders to pay attention to roots. In particular, how roots interact with the environment and affect yield potential have become critical in terms of feeding the increasing world population [3]. The plasticity of root growth and development in response to moisture deficit and nutrient availability can be exploited to enhance crop productivity [4]. The attention given to plant root systems has gained momentum with the development of rhizotrons, magnetic resonance and computed tomography techniques, and 3-D root phenotyping platforms to facilitate nondestructive spatial and temporal investigations into root systems [5-10].
Elucidating the genetic and developmental basis of root architecture presents many challenges that must be addressed through a combination of field, greenhouse, and laboratory approaches [1]. Field studies provide the “ground truth” about plant growth in a particular environment, but this type of root study is hindered by intensive labor and excavation processes that destructively sample root systems at a single point during development [1-11]. Furthermore, heterogeneity within and along the soil profile [2], combined with physical and chemical interactions between various components of the rhizosphere, can drastically impact root architecture, even under presumably controlled conditions [12]. Soil conditions such as hardiness, soil drying, and excessively large pores or loose soil structure influence both above and below ground plant traits [13]; therefore, most root analyses are carried out in artificial media, not soil [14-16]. However, artificial media also affect some root parameters. For example, the number of root hairs in the root tip of white clover were affected by the concentration of nutrient solution added [17]. Different types of artificial media abound and most are marketed by creating the impression that they enable more aeration, increase nutrient access by plants, and allow easy separation of roots [18]. Recently, other soil-less environments such as hydroponics and aeroponics have been extensively used. Growing plants in pots in artificial media is the predominant way to carry out plant assessment indoors [9,14-16]. The reproducibility of the results obtained using these different phenotyping platforms is crucial in terms of producing quality results from scientific experiments, and an important factor is the type of media used. In this respect, a pressing question is whether the data obtained from experiments using artificial media are comparable to those from soil.
This study compares two artificial media to soil in terms of their effect on above and below ground biomass of both wild and cultivated lentils at flowering. We further compared the effects of drought and its interaction with media on above and below ground plant traits. Although root biomass has been commonly evaluated, some studies in rice have shown that genotypes with similar root biomass may differ in other root traits [19]. Therefore, we evaluated both root biomass and root traits such as total root length, total root surface area, root average diameter, root volume etc. The rationale was to determine whether media and drought have a significant effect on plant parameters in different media and which medium has results that are comparable to those from soil.
Materials and Methods
Media, growth conditions, and experimental design
The experiment was carried out from August 2015 to May 2016 at the controlled environment facility at the College of Agriculture and Bioresources at the University of Saskatchewan, Canada (lat. 52.133; long. -106.631) using three media for comparative analysis. The first was the inorganic medium (PROFILE® Greens Grade™), which has been successfully used in root analysis under both fully watered and drought conditions for wheat and rye grass [18]. PROFILE® Greens Grade™ (GG) is an inorganic growth medium with a pH of 5.5 (± 1). It is marketed as a medium that enables water retention and rapid separation of roots with minimal damage to the root system. GG contains silica, aluminum oxide, iron (III) oxide, and crystalline quartz. It has limited amounts of total nitrogen, only 5.9 mg.Kg-1. Other components of GG include very small amounts (total<5%) of calcium, magnesium, sodium, and titanium oxides. Its cation exchange capacity is 33.6 mEq/100 g. The second medium used for this study was Sunshine Mix #4 (SSM4). It is commonly used for growing lentil plants [14-16]. SSM4 is an organic growth medium with a pH and electrical conductivity ranging from 5.5-6.5 and 0.8-2.0 mS cm-1, respectively. SSM4 consists of Canadian sphagnum peat moss, coarse perlite, and dolomitic limestone, a mixture that promotes moisture and nutrient retention. This medium also had 13.7 mg kg-1 total nitrogen. The third medium was soil was classified as an orthic dark brown Chernozem with the A and B horizons both described as loam and the C horizon as clay loam [20].
The experiments were conducted in a randomized complete block design with four replicates carried out in three runs; each medium was used in a different growth room. In each room, the temperature was set to 21°C day/15°C night with a day length of 16 h. Light intensity ranged from 308 to 392 μmol·m-2·s-1 depending on tube position and plant height. The light bulbs in the room were of two types: Phillips T-5 Fluorescence bulb # 835 (ON, Canada) and LED light bars, 730 mm Far Red (Fluence Bioengineering, Austin, TX, USA). Plant positions within each block were re-randomized at each weighing throughout the experiment to minimize light position effects. Temperature and humidity loggers (ibutton DS1923, Embedded Data Systems, Lawrenceburg, KY, USA) were placed randomly within each block to monitor environmental conditions.
Poly tubes 60 cm in length by 10 cm diameter were filled with soil or GG. SSM4 medium was used in poly pots 17.5 cm in height by 15.5 cm in diameter. Ten random tubes or pots were filled with water to saturation, their tops covered with aluminum foil, and settling allowed for 48 h until no water was observed to leave the base. The amount of water held by each tube or pot (100% field capacity, FC) was then calculated by subtracting the weight of the empty tube/pot and dry soil. Fully watered tubes/pots were maintained at 80% FC to avoid flooding while drought treatment tubes/pots were maintained at 40% FC. The moisture level in each tube (pot) was maintained by weighing, recording the value, and topping up with water as needed every other day throughout the experiment. Before sowing, each tube received an application of 200 mL of modified Hoagland solution that included calcium chloride (60.5 mM) - replacing calcium nitrate, micronutrients (12.1 mM), FeEDTA (12.1 mM), potassium hydrogen phosphate (12.1 mM), and magnesium sulphate (4.1 mM).
Plant materials
The plant species considered in this study included one genotype from each of five lentil species: the cultivated genotype Lens culinaris ‘Eston’ and the four wild lentil species genotypes (abbreviation in parentheses) were: Lens tomentosus (L. tom.) IG 72805, L. odemensis (L. ode.) IG 72623, L. lamottei (L. lam.) IG 110813 and L. ervoides (L. erv.) L-01-827A. Based on genotyping- by-sequencing classification, L. cul. Eston and L. tom. IG 72805 belong to the primary gene pool, L. ode. IG 72623 and L. lam. IG 110813 to the secondary gene pool, and L. erv. L-01-827A to the tertiary gene pool (21). Seeds of wild and cultivated species were scarified, washed in bleach, and then pre-germinated in a dark chamber at 22 °C. After three days, seedlings with a radicle length longer than 2 cm were selected and transplanted into each tube or pot. Rhizobium inoculum (Rhizobium leguminosarum biovar viceae strain 1435; Nodulator XL SCG, Becker Underwood, Canada) was placed approximately one cm from the transplanted seedlings at a rate of 6 g per tubeor pot. At flowering, plant height was measured from the soil level in tubes to the tip of the uppermost leaf, then above ground biomass (AGB) was harvested and roots washed. The procedures for root washing and measuring root traits [20]. Root traits evaluated in addition to below ground biomass (BGB), included total root length (TRL), total root surface area (TRSA), root length density (RLD), total root volume (TRV), total number of root tips (TNT), and total number of root forks (TNF).
Data analysis
Different root traits and plant height were compared between media and genotypes. Least significant differences between means were calculated using the PROC GLM procedure in Statistical Analysis System software (SAS 9.4, SAS Institute, Cary, NC, USA). Testing the effects of media, moisture, and their interaction for root traits was conducted using the PROC MIXED procedure in SAS. Significant differences are presented at alpha equals 5%. All figures were drawn with the aid of Sigma Plot (Sigma Plot 10, Systat Software Inc., and Germany). Results are arranged phylogenetically.
Results
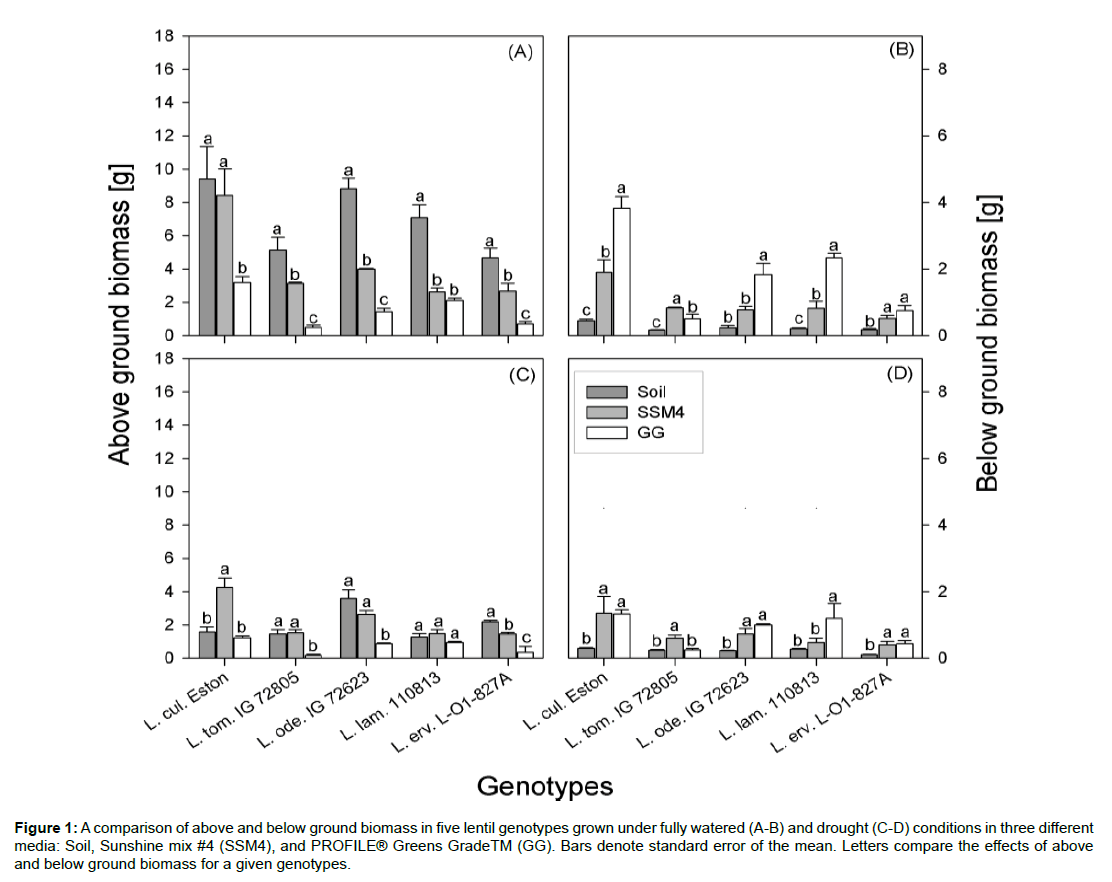
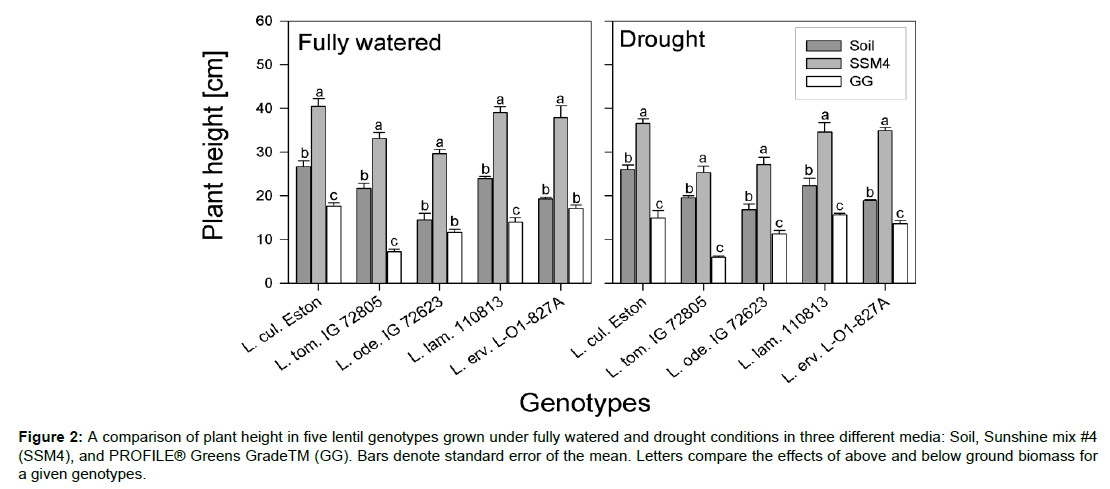
There were significant effects of media, moisture and their interaction on both above and below ground biomass. Under fully watered conditions, AGB was significantly higher in genotypes grown in soil compared to when they were grown in GG; Lens culinaris Eston, unlike its wild relatives, had similar AGB under both soil and SSM4 (Figure 1A). Under drought conditions, the cultivated and wild lentils responded differently (Figure 1B). Significantly higher AGB was observed in L. cul. Eston grown in SSM4, with similar AGB in soil and GG. Similar amounts of AGB were observed in L. tom. IG 72805 when grown in soil and SSM4, and both were significantly higher than the AGB produced GG. In both L. ode. IG 72623 and L. erv. L-01- 827A, the AGB in each media was significantly different while in L. lam. IG 110813, there was no significant difference in AGB among media. Although AGB between genotypes varied and was dependent on moisture levels, plant height was significantly higher in SSM4 compared to both soil and GG irrespective of moisture level. However, plant height in soil was significantly higher than that in GG for all lentil genotypes tested at both moisture levels (Figure 2).
Figure 1: A comparison of above and below ground biomass in five lentil genotypes grown under fully watered (A-B) and drought (C-D) conditions in three different media: Soil, Sunshine mix #4 (SSM4), and PROFILE® Greens GradeTM (GG). Bars denote standard error of the mean. Letters compare the effects of above and below ground biomass for a given genotypes.
Figure 2: A comparison of plant height in five lentil genotypes grown under fully watered and drought conditions in three different media: Soil, Sunshine mix #4 (SSM4), and PROFILE® Greens GradeTM (GG). Bars denote standard error of the mean. Letters compare the effects of above and below ground biomass for a given genotypes.
Under fully watered conditions, BGB was significantly higher in both artificial media compared to soil for both cultivated and wild lentils (Figure 1c). Under drought conditions, L. cul. Eston and L. erv. L-01-827A had similar BGB when grown under both artificial media and this was significantly higher than BGB in soil. Similar BGB was observed in L. tom. IG 72805 grown in soil and GG, a significantly lower BGB compared to that in SSM4. Lens odemensis IG 72623 and L. lam. IG 110813 had significant different BGB for each medium (Figure 1D). BGB has been traditionally assessed in plants grown under well-watered conditions, but in drought studies there is the need to further identify traits that will be advantageous under moisture deficit conditions. Table 1 summarizes the effects of different parameters on root traits. Neither media, moisture nor their interaction had an effect on root length density. The interaction between genotype and moisture did not affect TRSA, TRV or RAD. RAD was significantly affected by genotypic differences and media only. The interaction between genotype and moisture had a significant effect only on BGB and TRL, while the interaction of media and moisture had no significant effect on both TRV and RAD. Given that TRL and TRSA are traits frequently assessed because of their role in water and nutrient uptake [22], we evaluated the how these traits are affected by growth media under both moisture levels. Under fully watered conditions, both TRL and TRSA were variable for each genotype grown in the different media (Tables 2 and 3). In L. cul. Eston, TRL and TRSA were significantly lower in soil compared to both artificial media. Lens tomentosus IG 72805 had similar TRL in all media but its TRSA was significantly higher when grown in SSM4 and GG compared to soil. Lens odemensis IG 72623 had significantly higher TRL in SSM4 compared to both soil and GG but its TRSA was similar in SSM4 and GG; this was significantly higher than that in soil. Lens lamottei IG 110813 and L. erv. L-01-827A both had significantly higher TRL and TRSA when grown in GG; similar TRL and TRSA were observed in soil and SSM4.
| Root traits | ||||||
|---|---|---|---|---|---|---|
| BGB | TRL | RLD | TRSA | TRV | RAD | |
| Effects | P values (a = 5%) | |||||
| Genotype | <.0001 | <.0001 | 0.1775 | <.0001 | 0.0075 | 0.0421 |
| Media | <.0001 | 0.0006 | 0.1702 | <.0001 | <.0001 | <.0 0 0 1 |
| Moisture | <.0001 | 0.0012 | 0.0693 | 0.0019 | 0.0493 | 0.7227 |
| Media×Moist | <.0001 | 0.0356 | 0.1239 | 0.0404 | 0.4157 | 0 2 7 6 2 |
| Geno×Media | <.0001 | 0.0235 | 0.0641 | 0.0055 | 0.0209 | 0.0566 |
| Geno × Moist | <.0001 | 0.0056 | 0.1120 | 0.1123 | 0.3296 | 0. 6 3 9 1 |
| Geno × Media × Moist | 0.0011 | 0.6289 | 0.5766 | 0.7583 | 0.1418 | 0.7362 |
Table 1: Effects of media, moisture, and their interaction with genotypes on root traits in cultivated and wild lentil genotypes.
| Lens species/ genotypes | Fully watered | Drought | ||||
|---|---|---|---|---|---|---|
| Soil | SSM4 | GG | Soil | SSM4 | GG | |
| culinaris Eston | 1936 (± 435)b | 15232 (± 5394)b | 15365 (± 3641)a | 1946 (± 192)b | 3339 (± 126)ab | 4984 (± 813)a |
| tomentosus G72805 | 1431 (± 191)a | 1852 (± 148)a | 1266 (± 197)a | 1678 (± 163)a | 1495 (± 319)a | 1386 (± 117)a |
| odemensis IG72623 | 1666 (± 120)c | 9580 (± 1386)a | 4899 (± 719)b | 1395 (± 218)b | 3039 (± 424)a | 3684 (± 744)a |
| lamottei IG110813 | 1731 (± 174)b | 2616 (± 139)b | 5624 (± 750)a | 2504 (± 293)ab | 1960 (± 130)b | 2992 (± 374)a |
| ervoides L-01-827A | 2787 (± 287)b | 1821 (± 205)b | 6656 (± 1313)a | 1962 (± 212)b | 876 (± 122)b | 3640 (± 171)a |
Table 2: Total root length (cm) in soil, Sunshine mix #4 (SSM4) and Greens grade (GG) grown under fully watered and drought conditions in cultivated and wild lentils.
| Lens species/ genotypes | Fully watered | Drought | ||||
|---|---|---|---|---|---|---|
| Soil | SSM4 | GG | Soil | SSM4 | GG | |
| culinaris Eston | 491 (± 24)b | 2519 (± 752)a | 4104 (± 716)a | 557 (± 41)b | 1319 (± 216)b | 3049 (± 101)a |
| tomentosus IG72805 | 396 (± 22)b | 713 (± 68)a | 899 (± 95)a | 422 (± 33)a | 564 (± 120)a | 764 (± 92)a |
| odemensis IG72623 | 542 (± 83)b | 2173 (± 212)a | 3417 (± 371)a | 262 (± 22)b | 1115 (± 129)ab | 2165 (± 482)a |
| lamottei IG110813 | 410 (± 38)b | 966 (± 111)b | 3995 (± 431)a | 475 (± 38)c | 1078 (± 199)b | 1910 (± 123)a |
| ervoides L-01-827A | 700 (± 92)b | 626 (± 35)b | 2087 (± 211)a | 510 (± 59)b | 364 (± 101)b | 1884 (± 199)a |
Table 3: Total root surface area (cm2) in soil, Sunshine mix #4 (SSM4) and Greens grade (GG) grown under fully watered and drought conditions in cultivated and wild lentils.
p>Under drought conditions, significantly higher TRL and TRSA were observed in L. cul. Eston grown in GG compared to soil. For L. tom. IG 72805 TRL and TRSA were similar in all media, a behavior that was observed under fully watered conditions. Lens odemensis IG 72623 had similar TRL when grown in both artificial media, and this was significantly higher than that in soil.
However, TRSA in SSM4 was similar to that of soil and GG. Lens lamottei IG110813 had similar TRL when grown in soil and GG but there was a significant difference in TRSA in all three media (Table 3). Lens ervoides L-01-827A had similar TRL and TRSA in soil and SSM4 which was significantly lower than that in GG. It is noteworthy that under drought conditions, L. cul. Eston and other wild lentil genotypes such as L. tom. IG 72805 and L. lam. IG 110813 tended to invest more resources into their TRL and TRSA when grown in soil; this was not observed in artificial media. We observed no similarities between root traits AGB or BGB in the different media that can be attributed to the categorization of either cultivated or wild genotypes into a given gene pool.
Discussion
Media has a significant influence on both above ground and below ground biomass with variable genotypic responses. The choice of media in past research was often based on previous experience and recommendations. Different media are now available for use in assessment of plant growth, and hydroponics or aeroponics is also used in high input indoor systems. Given the increasing emphasis placed on the contribution of the rhizosphere, especially roots, to plant survival, this study compared some commonly assessed plant traits in plants grown in soil vs. two artificial media at two moisture levels.
Media played a significant role in the partition of resources to either above or below ground structures (Figure 1). In soil under fully watered conditions, much of the dry matter fixed was allocated to above ground structures such as leaves and shoots, and less to the roots. In artificial media such as GG, for most genotypes, the reverse was true (Figure 1A, C). The differences in dry matter allocation might be due to the inability of soilless media to buffer pH changes, and therefore, to supply sufficient nutrients during plant growth given that pH plays a crucial role in nutrient uptake in soilless media [23]. Therefore, in GG where pH varied from 4.5 to 6.5, some nutrients may have been immobilized, causing plants to allocate more of their resources below ground compared to above ground. Differences in pore sizes resulting in different water holding capacities between the media may have also played a role in resource allocation.
Drought significantly reduced AGB in all genotypes. The proportion of this decrease was higher in L. cul. Eston compared with some wild lentil genotypes such as L. ode. IG 72623 (Figure 1A, B). The variable response observed in the AGB of lentil genotypes grown in different media subjected to drought conditions is worth taking into consideration when planning or designing drought experiments given that this experiment was terminated at flowering. We have shown that above ground and root parameters are significantly higher post-flowering in most of the assessed genotypes [22]. Media significantly affected the heights of both cultivated and wild lentil genotypes. The tallest plants were found in SSM4 and the shortest in GG, with those grown in soil were intermediate (Figure 2). This implies that using artificial media for experiments may result it values that either over or underestimate plant height compared to growth in soil. This, coupled with the fact that wild lentils showed varied responses to drought implies that the use of artificial media in experiments needs to take these genotypic responses into consideration. For example, under fully watered conditions, L. cul. Eston had similar AGB when grown in soil or SSM4, implying that either of these media would produce similar results, but when subjected to drought conditions, AGB was significantly higher in SSM4. SSM4 is promoted for its water holding capacity and may have had higher available water reserves under drought that resulted in higher AGB compared to soil.
BGB was also influenced by both media and moisture. For all lentil genotypes except L. tom. IG 72805 grown under fully watered conditions, BGB was significantly higher when plants were grown in GG (Figure 2C). The enormous root biomass produced might have resulted from the interaction of GG and lentil roots whereby nutrients are immobilized in the medium and low supply results in plants dedicating resources towards root production rather than shoot production even under fully watered conditions. This behavior is similar to that reported for plants grown in soil containing additives such as Geohumus®, which is known to immobilize some nutrients and make them unavailable to plants during growth [24]. However, allocation of resources towards root production is not only associated with nutrient deficiencies like phosphorus [25,26]. Large root biomass with less shoot biomass s has also been associated with lack of contact with media in hydroponic systems [13]. Root traits such as TRL and TRSA need to be characterized in drought studies. Different rice genotypes with similar root biomass do not necessarily have similar root traits [19]. This study demonstrates that under fully watered conditions, the TRL and TRSA of L. cul. Eston will be overestimated if assessed in artificial media. Under drought conditions, either soil or SSM4 would be appropriate for root trait assessments. For wild lentil genotypes, a case by case analysis has to be done. For example: wild genotypes such as L. erv. L-01-827A can be accurately assessed in either soil or SSM4 under both moisture levels, while L. ode. IG 72623 could only be assessed in soil under fully watered conditions (Tables 2 and 3).
Root traits such as TRL, TRSA, and TRV significantly differed between media for L. cul. Eston, implying that their assessments of this genotype in media other than soil are not reliable at the growth stage investigated here. Assessments at other growth stages need to be investigated for concrete conclusions to be drawn. The root traits of wild lentil genotypes such as L. erv. L-01-827A and L. lam. IG110813 were similar when grown in either soil or SSM4, implying they can only be assessed in these media. Other root traits such as RAD are significantly affected by the media in which plants are grown while others such as RLD are not affected by media, moisture, or their interaction (Table 1). Therefore, breeders and other scientists focusing their assessments on above ground traits, such as days to flowering and yield in combination with root traits need to ascertain the reproducibility of these traits obtained from other media to accurately depict plant growth, especially under drought conditions.
Finally, container size has been shown to affect root traits [9]. It is expected that similar root trends will be observed in genotypes grown in GG and soil given they were cultivated in tubes of similar size. However, our study demonstrates that media (Tables 2 and 3), rather than container size, had a significant effect on root growth. Although GG has been used successfully to grow and easily analyze wheat roots [18], this study suggests that it is not an appropriate medium for growing lentil plants given the disproportionate mobilization of resources towards the roots rather than shoots, a condition that results in dwarf plants.
Conclusions
The type of media chosen for assessment of lentil growth is dependent on a specific set of plant traits and genotypes. This study demonstrates that, when artificial media are used, the above ground growth relative to that below ground does not reflect what is observed with field soil, with considerable variability displayed by both cultivated and wild lentils. Furthermore, growth media that enable root trait assessments under drought in cereals (wheat) do not necessarily work for legumes, as shown in the case of GG. SSM4 was a better medium for assessing root traits given the similar results or trends to soil observed for different root traits. Gene pool categorization did not influence any trait evaluated in all media. We recommend that further analysis be carried out for crops at different growth stages in the three media so that definitive conclusions can be made.
References
- Paez-Garcia A, Motes CM, Scheible WR, Chen R, Blancaflor EB, et al. (2015) Root traits and phenotyping strategies for plant improvement. Plants 4: 334-355.
- Lynch J (1995) Root architecture and plant productivity. Plant Physiol 109: 7-13.
- Hamdy A, Ragab R, Scarascia-Mugnozza E (2003) Coping with water scarcity: water saving and increasing water productivity. Irrigation and Drainage 52: 3-20.
- Kano M, Inukai Y, Kitano H, Yamauchi A (2011) Root plasticity as the key root trait for adaptation to various intensities of drought stress in rice. Plant and Soil 342: 117-128.
- Taylor HM, Upchurch DR, McMichael BL (1990) Applications and limitations of rhizotrons and minirhizotrons for root studies. Plant and Soil 129: 29-35.
- Gregory PJ, Hutchison DJ, Read DB, Jenneson PM, Gilboy WB, et al. (2003) Non- invasive imaging of roots with high resolution X-ray micro-tomography. Plant and Soil 255: 351-359.
- Tracy SR, Roberts JA, Black CR, McNeill A, Davidson R, et al. (2010) X- factor: visualizing undisturbed root architecture in soils using X-ray computed tomography. J Exp Bot 61: 311-313.
- Clark RT, MacCurdy RB, Jung JK, Shaff JE, McCouch SR, et al. (2011) Three-dimensional root phenotyping with a novel imaging and software platform. Plant Physio 156: 455-465.
- Judd LA, Jackson BE, Fonteno, WC (2015) Advancements in root growth measurement technologies and observation capabilities for container-grown plants. Plants 4: 369-392.
- Jeudy C, Adrian M, Baussard C, Bernard C, Bernaud E, Bourion V, et al. (2016) Rhizotubes as a new tool for high throughput imaging of plant root development and architecture: test, comparison with pot grown plants and validation. Plant Methods 12:31
- Smit AL (2000) Root Methods: A Handbook. Springer, Berlin, Germany.
- Ward JT, Lahner B, Yakubova E, Salt DE, Raghothama KG (2008). The effect of iron on the primary root elongation of Arabidopsis during phosphate deficiency. Plant Physiol 147: 1181 1191.
- Passioura JB (2002) Soil conditions and plant growth. Plant, Cell and Environment 25: 311-318.
- Zakeri H, Bueckert RA, Schoenau J (2013) Effects of nitrogen on reproductive duration and yield in lentil cultivars. J Plant Nutri 36: 877-894.
- Bazghaleh N, Hamel C, Gan Y, Tar'an B, Knight JD (2015) Genotype-specific variation inthe structure of root fungal communities is related to chickpea plant productivity. Appl Environ Microbiol 81: 2368-2377.
- Yuan HY, Saha S, Vandenberg A, Bett, KE (2017) Flowering and growth responses of cultivated lentil and wild Lensgermplasm toward the differences in red to far-red ratio and photosynthetically active radiation. Front Plant Sci 386: 1-10.
- Crush JR, Care DA, Gourdin A, Woodfield DR (2005) Root growth media effects on root morphology and architecture in white clover. New Zeal J Agr Res 48: 255-263.
- Becker SR, Byrne PF, Reid SD, Bauerle WL, Mckay JK, et al. (2016) Root traits contributing to drought tolerance in synthetic hexaploid wheat in a greenhouse study. Euphytica 207: 213-224.
- Asch F, Dingkuhn M, Sow A, Audebert A (2005)Drought-induced changes in rooting patterns and assimilate partitioning between root and shoot in upland rice. Field Crops Res 93: 223-236.
- Gorim LY, Vandenberg A (2017a) Evaluation of wild lentil species as genetic resources to improve drought tolerance in cultivated lentil. Front Plant Sci 8: 1129.
- Wong MML, Gujaria-Verma N, Ramsay L, Yuan HY, Caron C, et al. (2015) Classification and characterization of species within the genus Lens using genotyping- by-sequencing (GBS). PLoS ONE 10: e0122025.
- Gorim LY, Vandenberg A (2017b) Root traits, nodulation and root distribution in soil for five wild lentil species and Lens culinaris (Medik.) grown under well-watered conditions. Front Plant Sci 8: 1632.
- Peterson JC (1982) Effects of pH upon nutrient availability in a commercial soilless root medium utilized for floral crop production. Research circular 268: 16-19.
- Lynch JP (2011) Root phenes for enhanced soil exploration and phosphorus acquisition: Tools for future crops. Plant Physiol 156: 1041-1049.
- Ho MD, Rosas JC, Brown KM, Lynch JP (2005) Root architectural tradeoffs for water and phosphorus acquisition. Funct Plant Biol 32: 737-738.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi