Case Report, J Clin Image Case Rep Vol: 2 Issue: 1
Branchio-Oto-Renal Syndrome-A Rare Case
Ishwar Singh1, Poonam Sagar1*, Bhandari PS2 and Anil Kalra2
1Department of ENT, Lok Nayak Hospital, New Delhi, India
2Department of Plastic Surgery, Lok Nayak Hospital, New Delhi, India
*Corresponding Author : Poonam Sagar
Department of ENT, Lok Nayak Hospital, C-11A, Ram Dutt Enclave, Uttam Nagar, Pincode-110059, New Delhi, India
Tel: 91-9818979820
E-mail: poonamsgr.mamc@gmail.com
Received: February 24, 2018 Accepted: March 14, 2018 Published: March 21, 2018
Citation: Singh I, Sagar P, Bhandari PS, Kalra A (2018) Branchio-Oto-Renal Syndrome-A Rare Case. J Clin Image Case Rep 2:1.
Abstract
Branchio-otorenal syndrome (BOR) is an autosomal dominant syndrome with high penetrance and variable expressivity. Congenital hearing loss, auricular malformations, branchial sinus and renal anomalies are major components. Minor manifestations may be variable. The present case report describes BOR in a 19 year boy whose father is also having same syndrome. Pedigree chart of five generations revealed strong family history. The present case had an uncommon presentation of facial palsy.
Keywords: Branchio-oto-renal syndrome; Facial paralysis; Deafness
Introduction
Branchio-oto-renal (BOR) syndrome is an autosomal dominant syndrome characterized by branchial cleft anomalies, hearing loss with auricular malformations and renal anomalies. Branchial cleft anomalies were first described by Von Ascherson (1832) but it was not until Melnick and Fraser who described associated renal anomalies and gave term Branchio-oto-renal syndrome also known as Melnick Fraser syndrome [1-3]. The diagnostic criteria were described by Chang et al. [4]. The major criteria were branchial anomalies, deafness, pre-auricular pits and renal anomalies. Minor criteria included external ear anomalies, middle ear anomalies, inner ear anomalies, pre-auricular tags, facial asymmetry and palatal abnormalities. An affected individual must have at least three major criteria; two major criteria and at least two minor criteria; or one major criteria and an affected first degree relative meeting criteria of BOR. We describe a case of BOR syndrome with pedigree showing dominant inheritance and both son and father showing features of BOR syndrome with a bilateral facial nerve palsy in son.
Case Report
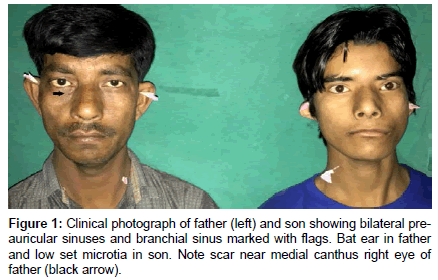
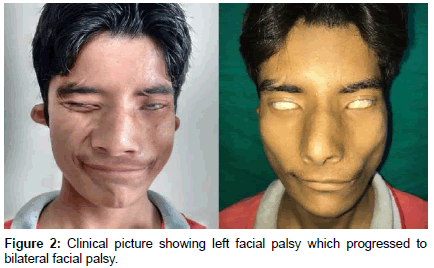
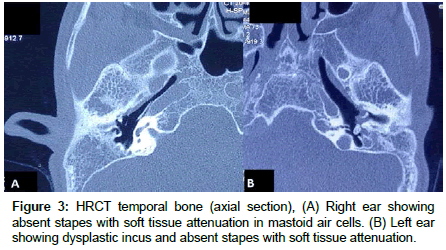
A nineteen year male presented to OPD with chief complaints of congenital deformity of bilateral ear, bilateral profound hearing loss by birth (Figure 1). Hearing loss was progressive during the last two years. Patient also complained of left facial weakness since birth. There was history of excessive bilateral eye watering during mastication. The patient had strong family history of congenital deafness running in ancestry associated with various sinus defects. Patient’s father has bilateral bat ear, bilateral pre-auricular sinuses, left branchial sinus and past history of congenital lacrimal fistula repair surgery in childhood. Patient’s uncle and their children also had similar history of ear deformities, sinuses and hearing loss (chart). On examination, patient had bilateral low set ears, pre-auricular sinuses and external auditory canal (EAC) atresia. Patient also had slanting eyes, mongoloid facies and high arched palate. Pure tune audiometry revealed mixed hearing loss, sensorineural component 35-40db with 50-55db conductive component. The profound hearing loss led defective speech development. Patient had congenital unilateral facial palsy of left side but for the past one year, patient developed facial palsy of the right side leading to bilateral facial nerve palsy (Figure 2). Ultrasonography of abdomen revealed normal kidneys. Skeletal survey revealed normal spine. High resolution computed tomography (HRCT) of temporal bone was done. Right ear showed absent stapes with soft tissue attenuation in mastoid air cells. Scans through left ear showed dysplastic incus with absent lenticular process and absent stapes with soft tissue attenuation of mastoid and middle ear. Left vestibule was dilated with dysplastic lateral semicircular canal (Figure 3). Bilateral EAC atresia was also seen. Sinogram done for right neck sinus revealed internal opening in right tonsillar fossa suggestive of 2nd arch branchial sinus. Conchal suturing was done for making antihelix in both the ears, pre-auricular and branchial sinuses were excised. Late onset facial palsy right side was treated with conventional oral steroid regimen to which it did not respond and persisted.
Discussion
BOR syndrome is an autosomal dominant syndrome with high penetrance and variable expression. The estimated prevalence is 1:40,000 and accounts for 2% of congenital hearing loss [5]. The chr 8.13.3 and mutations in EYA 1 gene is responsible for this syndrome [6]. Ruf et al. identified another locus on chr 14q23.1 as a cause for BOR. SIX1 gene mutations were found responsible for the same [7].
Hearing loss is most prevalent feature of BOR found in 93% of patients, followed by ear pits (82%). Branchial fistulae are found in 36% of patients. For this association it was known as “ear pits deafness syndrome” [8]. Hearing loss can be mixed (52%), conductive (33%) and sensorineural (29%) type [8,9]. Hearing loss is not always static; it can be progressive or fluctuant [10]. In the present case report, hearing loss was mixed and had progressive pattern. The external ear deformity is characteristic underdeveloped antihelix leading to lop ear. The present case had bilateral microtia with lop ear and bilateral external auditory canal atresia.
Branchial fistula is typically found at the anterior border of sternocleidomastoid. BOR should be differentiated from the branchio-oculo-facial (BOF) syndrome which is also autosomal dominant syndrome but characterized by post aural cervical fistulae, cleft lip/palate, low set ears and external nasal deformities [11]. In the present study, both the patient and his father had bilateral preauricular sinus and unilateral 2nd branchial cleft sinus. Present patient lacked characteristic facial features of BOF syndrome.
Renal anomalies in BOR ranges from mild dysplasia to complete agenesis and affects 13% of individuals [5,9]. Renal anomalies are better detected on intravenous pyelography than ultrasonography. The most common renal anomaly is reduced renal volume with loss of cortico-medullary differentiation. Renal involvement may lead to end stage renal failure during 2nd-4th decade of life.
Minor phenotypic abnormalities are found in 20% of affected individuals. These includes pre-auricular tags, lacrimal duct aplasia, gustatory lacrimation, short palate, facial paralysis, meatal atresia, retrognathia, benign intracranial tumour, non-rotation of gastrointestinal tract, pancreatic duplication cyst and congenital hip dysplasia [4,9]. The present patient had gustatory lacrimation, slanting eyes and notched eyebrows. CT cervical spine was done to rule out vertebral anomalies related to oculo-facial-vertebral syndrome.
Propst et al. studied radiology of temporal bones in BOR. The most common findings were hypoplastic apical turn of cochlea, facial nerve deviated to medial side of the cochlea, and funnel shaped internal auditory canal and patulous eustachian tube [12]. Enlarged endolymphatic sac has been associated with higher hearing loss [10]. The present case study had dysplastic incus, lateral semicircular canal, funnel shaped internal auditory canal and dilated vestibule. The present case also had congenital bilateral absence of stapes which is a rare [13].
The present case study is novel in few aspects. The patient’s father who also has BOR, had history of congenital lacrimal fistula of right eye for which he was operated in childhood. Lacrimal aplasia is associated anomaly not congenital lacrimal fistula. The second novel feature present in the patient was late onset facial nerve palsy coexistent with congenital facial palsy that has not been reported in association with BOR with best of our knowledge. The facial nerve lesions may arise due to interruption of their trajectory caused by structural anomalies in the inner ear [14]. Congenital lesions/ syndromes causing facial nerve palsy may manifest in later period of life as happened in our case [15].
The present case study highlights the typical inheritance pattern of BOR in five generations. The present case report has some unique and uncommon findings such as bilateral absence of stapes and congenital unilateral palsy which progressed later in life to bilateral facial palsy.
Conclusion
Ear manifestations of BOR may vary from simple pre-auricular sinuses to aural atresia, middle and inner ear anomalies as happened in present case. Hearing loss and facial palsy may have different manifestation such fluctuant or progressive hearing loss or late onset facial palsy which is very rare.
References
- Ascherson FM (1832) De fistulis colli congenitis, adiecta fissurarum branchialium in mammalibus avibusque historia succincta: commentatio pro venia docendi. The Bavarian State Library, Berlin, Germany
- Melnick M, Bixler D, Silk K, Yune H, Nance WE (1975) Autosomal dominant branchiootorenal dysplasia. Birth Defects Orig Artic Ser 11: 121-128.
- Fraser FC, Ling D, Clogg D, Nogrady B (1978) Genetic aspects of the BOR syndrome-branchial fistulas, ear pits, hearing loss, and renal anomalies. Am J Med Genet 2: 241-252
- Chang EH, Menezes M, Meyer NC (2004) Branchio-oto-renal syndrome: the mutation spectrum in EYA1 and its phenotypic consequences. Hum Mutat 23: 582-589
- Fraser FC, Sproule JR, Halal F (1980) Frequency of the branchio-oto-renal (BOR) syndrome in children with profound hearing loss. Am J Med Genet 7: 341-49
- Abdelhak S, Kalatzis V, Heilig R, Compain S, Samson D, et al. (1997) A human homologue of the Drosophila eyes absent gene underlies branchio-oto-renal (BOR)syndrome and identifies a novel gene family. Nat Genet 15: 157-164
- Ruf RG, Xu PX, Silvius D, Otto EA, Beekmann F, et al. (2004) SIX1 mutations cause branchio-otorenal syndrome by disruption of EYA1-SIX1-DNA complexes. Proc Natl Acad Sci USA 101: 8090-8095.
- Cremers CW, Fikkers-Van Noord M (1980) The earpits-deafness syndrome. Clinical and genetic aspects. Int J Pediatr Otorhinolaryngol 2: 309-322.
- Chen A, Francis M, Ni L, Cremers CW, Kimberling WJ, et al. (1995) Phenotypic manifestations of branchio-oto-renal syndrome. Am J Med Genet 58: 365-370.
- Lindau TA, Cardoso ACV, Rossi NF, Giacheti CM (2014) Anatomical changes and audiological profile in branchio-oto-renal syndrome: A literature review. Int Arch Otorhinolaryngol 18: 68-76.
- Trummer T, Muller D, Schulze A, Vogel W, Just W (2002) Brancio-oculo-facial syndrome and branchio-otic/ branchio-oto-renal syndromes are distinct entities. J Med Genet 39: 71-73
- Propst EJ, Blaser S, Gordon KA, Harrison RV, Papsin BC (2005) Temporal bone findings on computed tomography imaging in branchio-otorenal syndrome. Laryngoscope 115: 1855-1862.
- Undabeitia JI, Undabeitia J, Cianci L, Padilla L, Petreñas E, et al. (2014) Bilateral congenital absence of the stapes superstructure in two siblings. Case Reports in Otolaryngology 2014: 3.
- Jankauskienė A, Azukaitis K (2013) Congenital unilateral facial nerve palsy as an unusual presentation of BOR syndrome. Eur J Pediatr 172: 273-275.
- Jemec B, Grobbelaar AO, Harrison DH (2000) The abnormal nucleus as a cause of congenital facial palsy. Arch of Dis in Childhood 83: 256-258.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi