Research Article, J Vet Sci Med Diagn Vol: 8 Issue: 1
Capillary Electrophoresis of Serum Proteins across the Lifespan and Different Metabolic Stages in Sheep
- *Corresponding Authors:
- Isabel R Dias
Department of Veterinary Sciences, School of Agricultural and Veterinary Sciences (ECAV)
University of Trás-os- Montes e Alto Douro (UTAD), Quinta de Prados, P.O. Box 1013, 5000-801 Vila Real, Portugal
Tel: +00351 259 350632
Fax: +00351 259 350480
E-mail: idias@utad.pt
Received: September 11, 2018 Accepted: January 22, 2019 Published: January 28, 2019
Abstract
In recent years, sheep have been used extensively in animal and veterinary sciences research and also as a large animal model for pre-clinical and translational research due to similarities with humans. The objectives of this study were the characterization and evaluation of the serum concentration of total protein and their fractions, as determined by free-solution capillary electrophoresis in ewes of different ages and physiological states. For that purpose, 80 ewes were divided according to age or physiological state in eight groups. Blood samples were drawn during the same season and in the same flock, and serum protein electrophoresis was performed by an automated system. There were significant differences between the reference range of different ages, for total protein, albumin, alpha 1 and alpha 2, and gamma globulins. Physiological states showed significant differences between dry and lactation groups for total protein and beta globulin values. Total protein and protein fractions gradually increased across the lifespan, and decreased after 8 years of age. The knowledge obtained via this study help validate the influence of age and physiological states on serum protein variability, which could be used for diagnostic and research purposes.
Keywords: Ageing; Capillary electrophoresis; Physiological stages; Serum proteins; Sheep
Introduction
Sheep have been used as an animal model in human and veterinary research mainly due to the widespread acceptance of their use as a large animal experimental model for pre-clinical and translational studies, and their low cost, easy housing and handling, and availability [1]. The Churra-da-Terra-Quente is a typical sheep breed from the northeast of Portugal. This autochthonous breed is well adapted to wide seasonal variation and it provides high quality milk for Terrincho cheese and also lamb meat [2]. The ovine species has a great diversity of breeds that are consequently allocated in varying climates and with different nutritional support. It is therefore difficult to establish blood reference values, such as total protein and electrophoretic serum protein fraction profile, for this species [3].
Protein electrophoresis is a standard method for fractionation and quantification of the serum proteins, and it has become a routine analysis in human clinical diagnosis. It has not been commonly used, however, in veterinary routine profiles. Serum protein electrophoresis can be an important and useful tool in flock veterinary medicine due to the correlations that can be made between protein profile changes and several pathologies. The protein fractions are basically grouped into five fractions; albumin and globulins (alpha-1, alpha-2, beta, and gamma). Protein fractions are responsible for several crucial functions. Albumin, for instance, is osmotically active, and is essential for maintaining intravascular colloidal pressure, as well as for nutrition of the body (since it contains essential amino acids), and is also a carrier for many substances (e.g. fatty-acids, calcium ions, drugs). Alpha, beta, and gamma globulins are responsible for immune responses (e.g. immunoglobulins or antibodies), for inflammatory response, for the transport of molecules (e.g. cholesterol, iron, vitamins, hormones); and fibrinogen is involved in hemostatic and fibrinolytic processes in plasma protein electrophoresis [4].
Concerning the standardization of serum protein profiles, several factors may influence the creation of a reference range. Considering the possible intra-species variability, whether due to gender, age, physiological state, or breed, the interval could be wider than that seen in the literature [5-7]. Therefore, a detailed knowledge of this interval could help farmers minimize financial losses by knowing the nutritional requirements of the animals and also provide researchers with more reliable numbers [8].
Age [9] and physiological state [6] are important factors when analyzing an animal’s protein profile since these variables may affect the concentration of serum protein fractions. The reported variation in ruminants, such as buffaloes [7] and calves [5], show that protein patterns can be influenced by age, as well as pregnancy and lactation, where metabolic functions vary considerably due to demands of the fetus or milk synthesis, which could affect blood serum chemistry values [6,10].
Capillary electrophoresis is an advanced method compared to other conventional electrophoretic methods such as cellulose acetate or agarose gel, due to multiple factors, including ease, high speed, and high resolution separations, fully automated process and detection methods (based on analytic nature) that facilitates obtaining an analysis in a short period of time [11]. In this technique, lateral diffusion is minimized (as glass capillaries used usually have internal diameters of 200–500 μm), and there are temperature differences between the center of the capillary and the walls [11].
Only a few studies have described reference values for the ovine species, and some of these studies did not consider age and physiological state as factors leading to variation. Therefore, the main purpose of the present study was to characterize the physiological serum protein electrophoretic pattern for Churra-da-Terra-Quente sheep, and to describe the variation among different ages and physiological states such as lactation, gestation, and dry periods.
Materials and Methods
Animals
Eighty ewes (Churra-da-Terra-Quente sheep) from the same flock, located in Carrazeda de Ansiães, a municipality in the district of Bragança, in northern Portugal, were used. These animals were chosen from the flock based on their age or physiologic state and divided into 8 groups of 10 animals each. The groups were as follows: 6 months old, 1 year old, 2 years old, 3 to 5 years old (no pregnant or lactating ewes), 3 to 5 years old ewes with 2 or 3 months of pregnancy, 3 to 5 years old ewes with 2 or 3 months of lactation, 6 to 8 years old, and the last group with animals over the age of 8 years. The ewes were kept in a natural pasture during the day and housed overnight. The diet was composed of grass hay, supplemented with 0.250 kg of concentrate feed per animal per day, and water provided ad libitum. Dry matter and chemical composition of grass hay is made up of dry matter per kg feed (88.5 g), ash per kg dry matter (5.9 g), neutral detergent fiber per kg (73.3 g) dry matter and crude protein per kg dry matter (6.1 g). Dry matter and chemical composition of feed concentrate is made up of dry matter per kg feed (90.4 g), ash per kg dry matter (8.5 g), neutral detergent fiber per kg dry matter (31.6 g) and crude protein per kg dry matter (20.7 g).
All animal handling practices followed Directive 2010/63/EU of the European Parliament and of the Council on the protection of animals used for scientific purposes.
Blood sampling
Blood was drawn in December, during the Northern hemisphere winter. Blood samples were drawn from the jugular vein and placed into serological tubes (S-Monovette®, SARSTEDT, Nümbrecht, Germany). Blood collection took place between 9:00 a.m. and 10:00 a.m., and the blood was immediately carried in a thermal box to the laboratory facilities. Blood was centrifuged (3000 rpm for 10 min) and the serum was stored in Eppendorf tubes at −20°C until analysis.
Determination of total protein
The biuret method was used for quantitative determination of total protein concentration in serum using an automated analyzer (Mindray BS-380; Shenzhen Mindray Bio-Medical Electronics, Nanshan, China).
Separation of serum proteins by capillary electrophoresis
Electrophoresis was performed using an automated system (Capillarys 2; SEBIA, Paris, France) according to the procedures described by the manufacturer. The electrophoresis system uses the principle of capillarity in free solution to allow multiple simultaneous analyses. Electrophoretic mobility occurs due to charged molecules that are separated with the aid of an alkaline buffer. The pH of the electrolytes and the electro-osmotic flows are responsible for this separation.
Statistical analysis
Serum protein fractions and total protein values are presented as mean ± standard deviation (SD) and median ± interquartile range (IQR). Statistical normality was checked using the Shapiro-Wilk test of all groups. ANOVA and the post-hoc Tukey test were used for data with normal distribution. The Kruskal-Wallis test was used for testing the non-parametric statistical hypothesis and the Kruskal- Wallis pairwise method for multiple comparisons. Statistical analyses were performed using SPSS software (version 23.0, SPSS, Inc., IBM Company, NY, USA). The level of significance was set at p ≤ 0.05.
Results
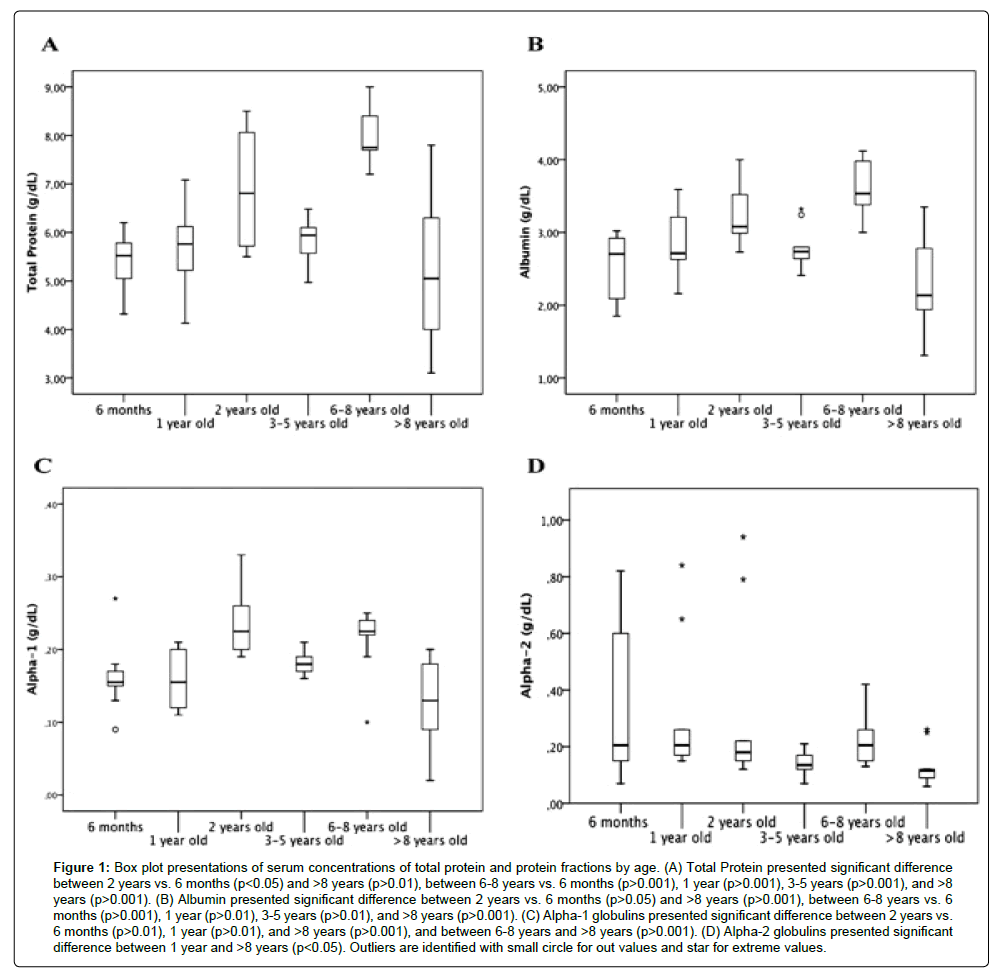
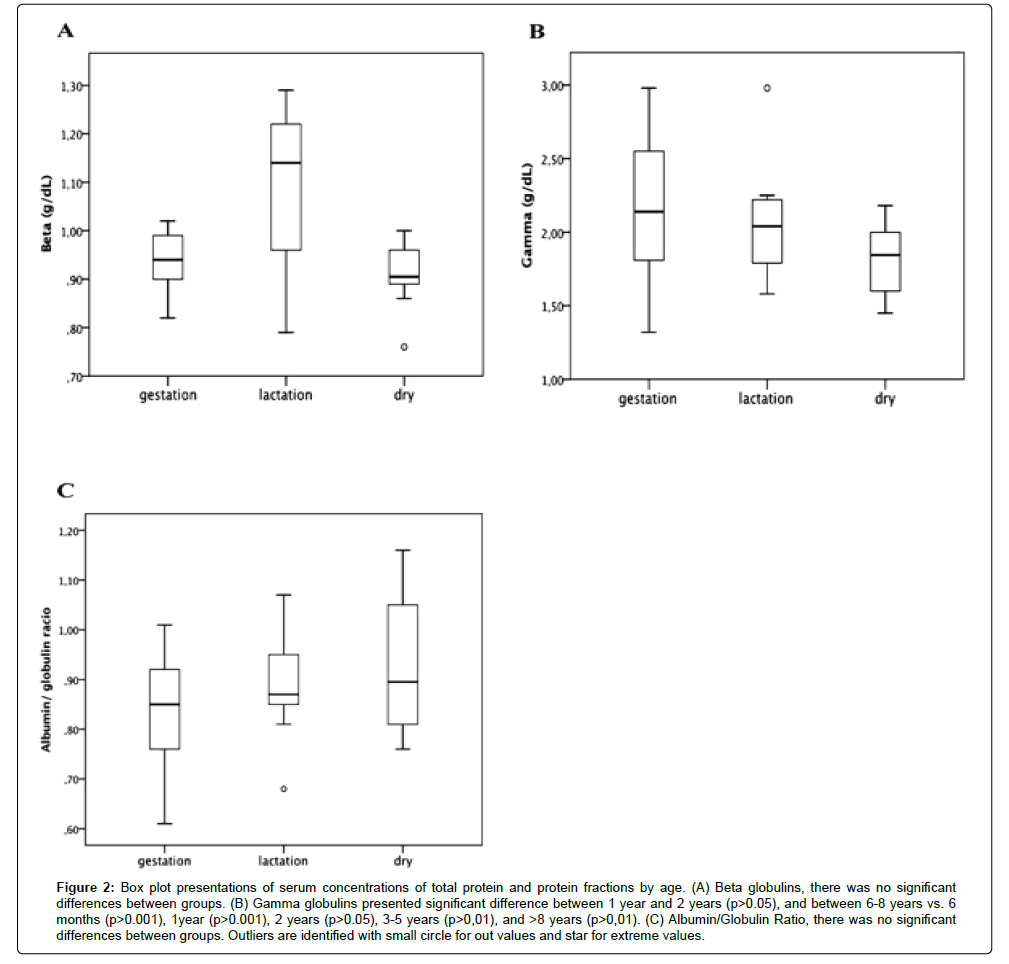
Table 1 shows mean values ± SD and median values ± IQR for studied parameters across different ages. ANOVA and the post-hoc Tukey test showed a significant difference (p<0.001) between age groups “6-8 years” and all others, except “3-5 years”, for total protein values (Figure 1A, Table 1), with the “6-8 years” group having the highest values, and a significant difference (p<0.01) between the “6-8 years” group and the other age groups for gamma globulin (Figure 2B). Regarding albumin (Figure 1B), we observed a similar significance as that observed for total protein, with the highest values seen in the “6-8 years” group (Table 1), but there was no significant difference (p>0.05) between the different age groups for albumin/globulin ratio (Figure 2C). The Kruskal-Wallis test and pairwise method did not show a significant difference (p>0.05) between the groups with different ages for beta globulin (Figure 2A). Alpha-1 (Figure 1C) and alpha-2 globulins (Figure 1D) presented a significant difference along the lifespan (p<0.01 and p<0.05, respectively).
Figure 1:Box plot presentations of serum concentrations of total protein and protein fractions by age. (A) Total Protein presented significant difference between 2 years vs. 6 months (p<0.05) and >8 years (p>0.01), between 6-8 years vs. 6 months (p>0.001), 1 year (p>0.001), 3-5 years (p>0.001), and >8 years (p>0.001). (B) Albumin presented significant difference between 2 years vs. 6 months (p>0.05) and >8 years (p>0.001), between 6-8 years vs. 6 months (p>0.001), 1 year (p>0.01), 3-5 years (p>0.01), and >8 years (p>0.001). (C) Alpha-1 globulins presented significant difference between 2 years vs. 6 months (p>0.01), 1 year (p>0.01), and >8 years (p>0.001), and between 6-8 years and >8 years (p>0.001). (D) Alpha-2 globulins presented significant difference between 1 year and >8 years (p<0.05). Outliers are identified with small circle for out values and star for extreme values.
Figure 2:Box plot presentations of serum concentrations of total protein and protein fractions by age. (A) Beta globulins, there was no significant differences between groups. (B) Gamma globulins presented significant difference between 1 year and 2 years (p>0.05), and between 6-8 years vs. 6 months (p>0.001), 1year (p>0.001), 2 years (p>0.05), 3-5 years (p>0,01), and >8 years (p>0,01). (C) Albumin/Globulin Ratio, there was no significant differences between groups. Outliers are identified with small circle for out values and star for extreme values.
Table 1: Values for the studied parameters across the life-span in sheep.
| Parameters (g/dL) | 6 Months | 1 Year | 2 Years | 3-5 Years | 6-8 Years | >8 Years |
|---|---|---|---|---|---|---|
| Total Protein | 5.42 ± 0.56c | 5.64 ± 0.85 | 6.85 ± 1.14 | 5.84 ± 0.43 | 7.99 ± 0.55a | 5.20 ± 1.53c |
| Albumin | 2.54 ± 0.45c | 2.84 ± 0.47 | 3.20 ± 0.38 | 2.78 ± 0.28 | 3.60 ± 0.38a | 2.20 ± 0.64c |
| Alpha-1 globulins | 0.16 ± 0.04c | 0.15 ± 0.03c | 0.23 ± 0.04 | 0.17 ± 0.01 | 0.21 ± 0.04d | 0.12 ± 0.06c |
| Alpha-2 globulins | 0.15 ± 0.02 | 0.15 ± 0.06 | 0.22 ± 0.05 | 0.18 ± 0.01 | 0.22 ± 0.01 | 0.13 ± 0.08b |
| Beta globulins | 0.79 ± 0.30 | 0.80 ± 0.26c | 1.07 ± 0.45 | 0.90 ± 0.06 | 1.12 ± 0.08 | 0.87 ± 0.32 |
| Gamma globulins | 1.54 ± 0.38 | 1.54 ± 0.28c | 1.88 ± 1.06 | 1.84 ± 0.33 | 2.84 ± 0.51a,c | 1.67 ± 0.82 |
| Albumin/ Globulin Ratio | 0.91 ± 0.26 | 1.02 ± 0.12 | 0.92 ± 0.20 | 0.92 ± 0.13 | 0.83 ± 0.14 | 0.82 ± 0.18 |
a Vs 6 months, 1 year, 3-5 years and >8 years
b Vs 1 year
c Vs 2 years
d Vs >8 years
Total Protein, albumin, alpha-1, beta and albumin/globulin Ratio have Mean ± SD; alpha-2 and Gamma globulins have Median ± IQR displayed
Letters represent significant difference between groups for a same parameter (p<0.05)
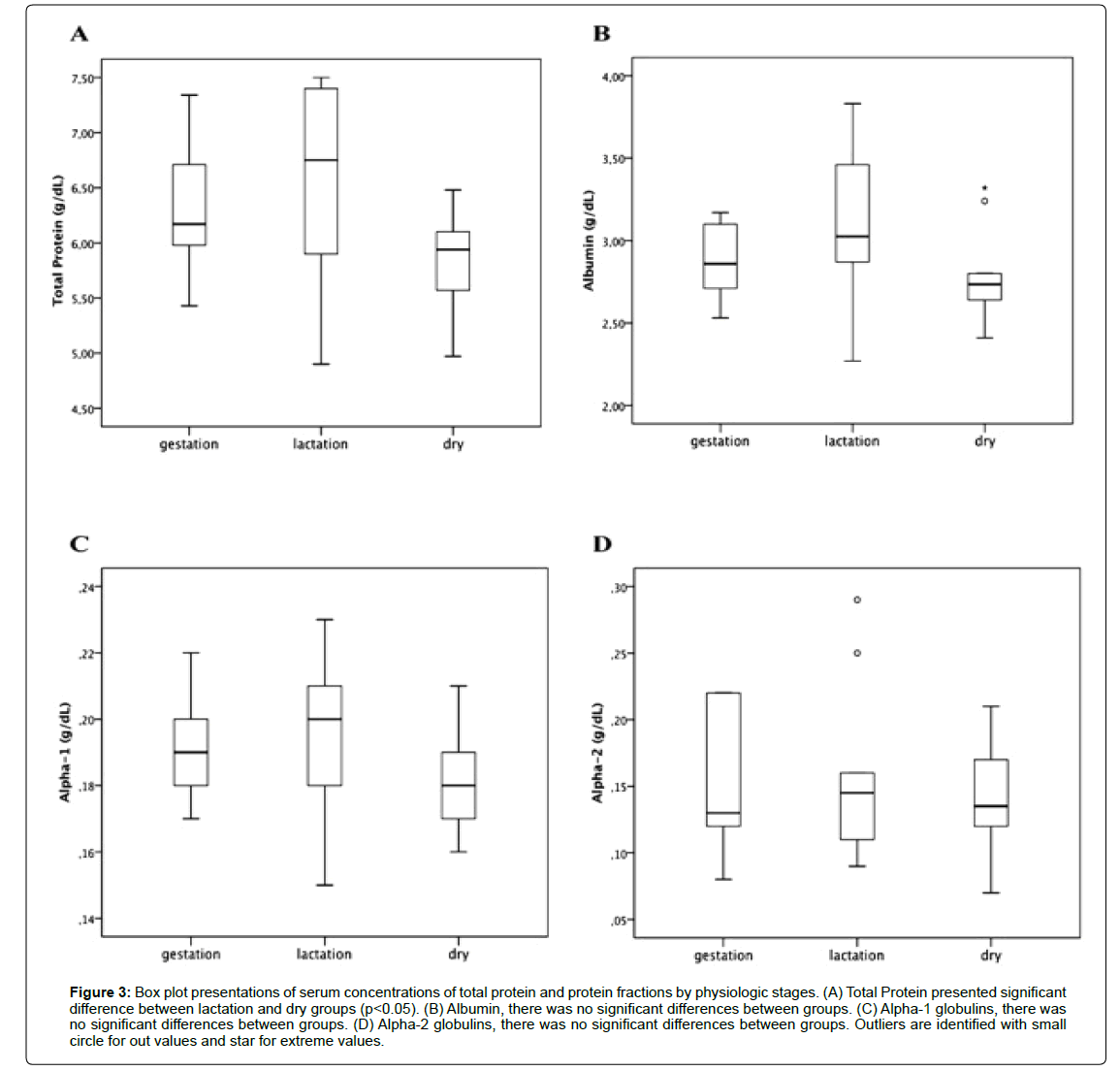
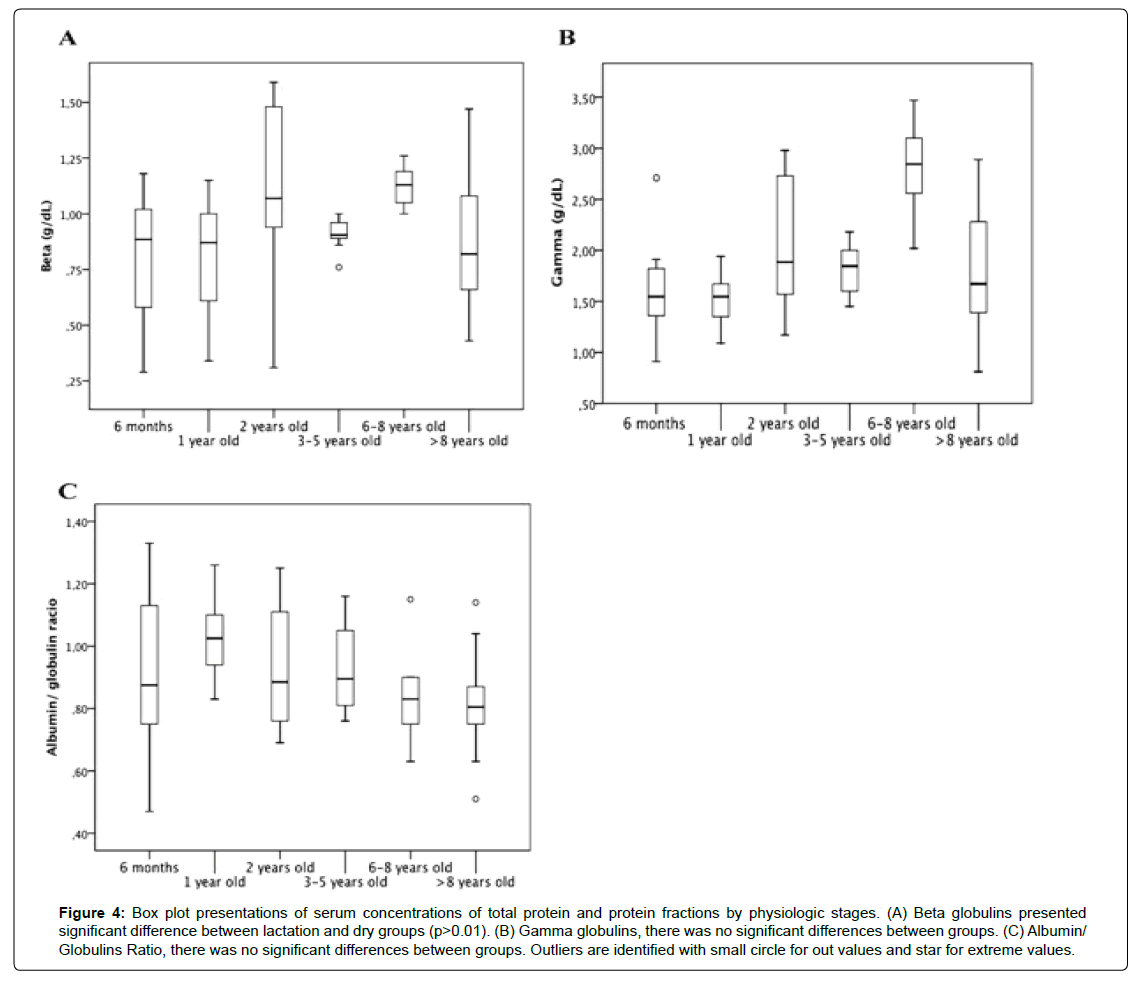
Regarding the analyses for the groups with animals between 3 and 5 years of age in different physiological phases, Table 2 shows mean values ± SD and median ± IQR for the parameters in this study. ANOVA and post-hoc Tukey test showed a significant difference (p<0.05) between the different physiological states for total protein (Figure 3A). Albumin (Figure 3B), alpha-1 (Figure 3C), alpha-2 globulins (Figure 3D), and albumin/globulin ratio (Figure 4C) did not present a significant difference (p>0.05). Kruskal-Wallis test and
Figure 3:Box plot presentations of serum concentrations of total protein and protein fractions by physiologic stages. (A) Total Protein presented significant difference between lactation and dry groups (p<0.05). (B) Albumin, there was no significant differences between groups. (C) Alpha-1 globulins, there was no significant differences between groups. (D) Alpha-2 globulins, there was no significant differences between groups. Outliers are identified with small circle for out values and star for extreme values.
Figure 4:Box plot presentations of serum concentrations of total protein and protein fractions by physiologic stages. (A) Beta globulins presented significant difference between lactation and dry groups (p>0.01). (B) Gamma globulins, there was no significant differences between groups. (C) Albumin/ Globulins Ratio, there was no significant differences between groups. Outliers are identified with small circle for out values and star for extreme values.
Table 2: Values for studied parameters per physiological stage in sheep.
| Parameters (g/L) | Gestation | Lactation | Dry |
|---|---|---|---|
| Total Protein | 6.32 ± 0.59 | 6.62 ± 0.86a | 5.84 ± 0.43 |
| Albumin | 2.86 ± 0.23 | 3.09 ± 0.47 | 2.78 ± 0.28 |
| Alpha-1 globulins | 0.19 ± 0.01 | 0.19 ± 0.02 | 0.17 ± 0.01 |
| Alpha-2 globulins | 0.15 ± 0.05 | 0.15 ± 0.06 | 0.14 ± 0.04 |
| Beta globulins | 0.94 ± 0.09 | 1.14 ± 0.23a | 0.90 ± 0.06 |
| Gamma globulins | 2.14 ± 0.74 | 2.04 ± 0.39 | 1.84 ± 0.33 |
| Albumin/ Globulin Ratio | 0.84 ± 0.12 | 0.88 ± 0.10 | 0.92 ± 0.12 |
aVs dry
Total Protein, Albumin, Alpha-1 and 2, Albumin/ Globulin Ratio have Mean ± SD;
Beta globulins, Gamma globulins have Median ± IQR
Letters represent significant difference
Discussion
The aim of our study was to determine the reference range for serum total protein and the corresponding protein fractions through electrophoresis analysis over the lifespan and different physiological stages in sheep. There are few scientific reports analyzing these protein fractions in sheep and there are no reports concerning the influence of age and physiological phases on protein fraction variations in sheep using the capillary electrophoresis method.
Evaluation of serum proteins and their electrophoretic pattern may be an important tool for diagnosing many diseases and metabolic states in veterinary medicine since this is an established method for clinical diagnosis in humans. The importance of a larger reference range, which considers not only the species, but other important factors, such as age and physiological state, enables a reduction of false positives in the diagnosis of pathologies.
Characterization of the electrophoretic pattern for sheep presents some obstacles, such as a wide variety of breeds, varying ages, physiological states, electrophoresis methods, and possibly also gender differences, which were not the focus of this study. These obstacles indicate that further similar studies are needed for establishing a more reliable range for serum protein values for the species. In this study, the variation in protein bands between different ages and physiological states represents the difficulty in obtaining a range of values for ovines. Serum protein electrophoretic pattern for Merino sheep at the age of 2-4 years old determined by [12], however, the data presented by those authors do not correspond to the results obtained in the present study. This might be due to multiple factors, such as type of electrophoresis method used [13], the different breed, and management conditions, such as feeding.
In the present study, serum total protein was found to be within the reference range (60 – 79 g/L) published by [14], except for the 6 months old age group, which presented a slightly lower value.
The reference range of albumin presented by [14] was 24 – 30 g/L, and in the present study, only the “6-8 years” old age group presented values outside that range. However, the range of values published by [12] for serum total protein, albumin, and beta and gamma globulin in Merino sheep (ages between 2-4 years old), were similar to those we encountered. Nevertheless, [12] reported higher values for alpha-1 and alpha 2 globulins in comparison with our study.
Regarding the analysis of the serum protein bands and total protein with respect to the different age groups, only beta and albumin/globulin ratio did not show statistical difference, with clear interference of age in the reference values for sheep, mainly evidenced by the highest values found in the “6-8 years” old age group.
In relation to the oscillation of values between different ages, with increasing values along the sheep lifespan until the “6-8 years” old age group and a decrease in the 8 years old age group for serum total protein, albumin, and beta and gamma globulins. This fact may be directly related to a continuous increase in serum protein parameters with age. Another author Ahmadi et al. [15] has also described this finding in other species, that is, higher values for adults in comparison with young individuals. However, the “3-5 years” old age group showed lower values than the 2 years old age group, with no explanation for this reduction.
During gestation and lactation, the mother organism undergoes physiologic changes [6,10] the role of serum proteins during these phases is directly linked to productive performance and consequently to economic profit or loss. Analyzing the different protein bands in the different physiological stage groups, only the total serum proteins and beta globulins presented significantly different values (p<0.05 and p<0.01, respectively) between dry and lactation groups. The high serum protein value in the lactation group is probably due to the need for a large protein supply for milk production. Beta globulins presented the same variation as the total protein, with values increasing from dry to pregnant states, and also increasing from pregnant to lactating ewes, and decreasing from the lactating to the dry ewes. However, the results reported by Piccione et al. [6] in Comisana ewes, with 3 ± 0.6 years old, reported slightly higher values for total protein during pregnancy, but similar for dry and lactation. We were unable to compare our findings to [6] for beta- 1 and beta-2 globulins since we did not obtain these electrophoretic bands, possibly due to variation in electrophoresis technique.
Analysis of different physiological states showed beta globulins and serum total protein with significant difference between dry and lactation states, nevertheless, a detailed month-by-month analysis would likely result in more accurate values. Nevertheless, our study confirmed the influence of the different physiological phases on protein metabolism in ewes.
The values collected from the different physiological stage groups remain within the range of values of the different age groups along lifespan. Therefore, during the formation of a general reference interval for the sheep, the different physiological stages should not influence this range.
Nevertheless, the presented data on the different physiological stages become an important financial tool to curb the excess or lack of protein in the feeding of sheep during different phases of their lives, leading to an increase in profit.
During the comparison of conventional electrophoresis methods for serum protein band assessment, agarose gel has been considered more accurate than cellulose acetate due to the relatively neutral characteristic of agarose, the minimal electroosmotic flow, the porosity, and the optical clarity [16]. However, the capillary electrophoresis method compared with agarose gel presents advantages such as higher sensitivity, full automation, ease of use, and rapidity; on the other hand, it has lower specificity [17]. Advantages seen with the capillary method may increase its use, thus requiring new reference profiles to take into account the variation that could occur in the results obtained using agarose gel or cellulose acetate for capillary electrophoresis [13].
Another important factor that was not evaluated in this study is the influence of diet. Although concentrate has a fixed nutritional value for protein, the use of grass hay needs a deeper analysis because it does not present a fixed value during the entire year, with higher values near the winter [18]. Therefore, future studies could investigate the influence of feeding compared to other factors, such as age and different physiological stages.
Conclusion
The results presented herein provide a variety of values detailing the serum protein profile of sheep of the Churra-da-Terra-Quente breed across its lifespan and different physiologic stages. This information may be a useful tool not just in animal and veterinary studies, but also in pre-clinical and translational research studies. Further studies are necessary to establish if there are significant variations between sheep breeds and thereby form a more reliable reference range for serum protein values and pattern in the ovine species.
Acknowledgements
José A. Camassa acknowledges the National Council for Scientific and Technological Development (CNPq-Brazil) for his PhD scholarship 202248/2015-1.
References
- Dias I, Camassa JA, Bordelo JA, Babo PS, Viegas CA, et al. (2018) Preclinical and translational studies in small ruminants (sheep and goat) as models for osteoporosis research. Curr Osteoporos Rep 16:182-197.
- Dias IR, Viegas CA, Silva AM, Pereira HF, Sousa CP, et al. (2010) Haematological and biochemical parameters in Churra-da-Terra-Quente ewes from the northeast of Portugal. Arq Bras Med Vet Zootec 62: 265-272.
- Metson AJ, Saunders WMH (1978) Seasonal variations in chemical composition of pasture. New Zeal J Agr Res 21: 341-353.
- Tothova C, Nagy O, Kovac G (2016) Serum proteins and their diagnostic utility in veterinary medicine: A review. Vet Med 61: 475-496.
- Piccione G, Casella S, Giannetto C, Vazzana I, Niutta P, et al. (2009) Influence of age on profile of serum proteins in the calf. Acta Veterinaria (Beograd) 59: 413-422.
- Piccione G, Alberghina D, Marafioti S, Giannetto C, Casella S, et al. (2009) Electrophoretic serum protein fraction profile during the different physiological phases in comisana ewes. Anim Sci Pap Rep. 27: 321-330.
- França RT, Costa MM, Martins DB, Pagnoncelli M, Leal ML, et al. (2011) Protein profile of buffaloes of different ages. Acta Sci Vet 39: 995-999.
- Chiaradia E, Avellini L, Tartaglia M, Gaiti A, Just I, et al. (2012) Proteomic evaluation of sheep serum proteins. BMC Vet Res 8:1-13.
- Fayos M, Couto CG, Iaybik MC, Wellman ML (2005) Serum protein electrophoresis in retired racing greyhounds. Vet Clin Pathol 34: 397-400.
- Yokus B, Cakir DU, Kanay Z, Gulten T, Uysal E (2006) Effects of seasonal and physiological variations on the serum chemistry, vitamins and thyroid hormone concentrations in sheep. J Vet Med A Physiol Pathol Clin Med 53: 271-276.
- Whatley H (2001) Basic principles and modes of capillary electrophoresis: Clinical and forensic applications of capillary electrophoresis. (1st Edtn), Humana Press Inc, Totowa, USA.
- Nagy O, Tóthová C, Nagyová V, Kováč G (2015) Comparison of serum protein electrophoretic pattern in cows and small ruminants. Acta Vet Brno 84:187-195.
- Giordano A, Paltrinieri S (2010) Interpretation of capillary zone electrophoresis compared with cellulose acetate and agarose gel electrophoresis: Reference intervals and diagnostic efficiency in dogs and cats. Vet Clin Pathol 39: 464-473.
- Kaneko JJ (1997) Serum proteins and the dysproteinemias: Clinical Bio-chemistry of Domestic Animals. (5th edtn), Academic Press, San Diego, USA.
- Ahmadi-hamedani M, Ghazvinian K, Kokhaei P, Barati M, Mahdavi A (2014) Comparison of effects of age and sex on serum protein electrophoretic pattern in one-humped camels (Camelus Dromedarius) in semnan, Iran. Open Vet J 4: 4-8.
- Rosenfeld L (1974) Serum Protein Electrophoresis. A comparison of the use of thin-layer agarose gel and cellulose acetate. Am J Clin Pathol 62: 702- 706.
- Lissoir B, Wallemacq P, Maisin D (2003) Serum protein electrophoresis: Comparison of capillary zone capillary technical® (Sebia) and agarose gel electrophoresis Hydrasys® (Sebia). Ann Biol Clin 61: 557-562.
- Demanet R, Mora ML, Herrera MÁ, Miranda H, Barea JM (2015) Seasonal variation of the productivity and quality of permanent pastures in adisols of temperate regions. J Soil Sci Plant Nutr 15: 111-128.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi