Case Report, J Clin Image Case Rep Vol: 2 Issue: 1
Carcinoid: Bringing Closure to PFO
Olisaemeka Achike*, Assad Movahed and Constantin B Marcu
Department of Cardiovascular Sciences, East Carolina University, Brody School of Medicine, East Carolina Heart Institute, USA
*Corresponding Author : Olisaemeka Achike
Department of Cardiovascular Sciences, East Carolina University, Brody School of Medicine, East Carolina Heart Institute 115 Heart Drive, Greenville, NC 27834, USA
Tel: 252-744-5964
E-mail: achikeo16@ecu.edu
Received: January 01, 2018 Accepted: January 10, 2018 Published: January 17, 2018
Citation: Achike O, Movahed A, Marcu CB (2018) Carcinoid: Bringing Closure to PFO. J Clin Image Case Rep 2:1.
Abstract
Background: There is an increased prevalence of patent foramen ovale (PFO) in patients with carcinoid heart disease (CHD).
Case: A 67-year-old woman presented with dyspnea on exertion for 1 month associated with epigastric pain, weight loss and diarrhea. She was tachypneic, with oxygen saturation of 90 % on 30 liters per minute. There was grade 2/6 systolic murmur at left sternal border and raised jugular venous pulse. Alpha-fetoprotein, cancer antigen 19-9 and carcinoembryonic antigen were normal. Urine 5-hydroxyindoleacetic acid, serum serotonin and chromogranin A were high. Computed Tomography (CT) scan of chest ruled out pulmonary embolus. CT scan of abdomen showed hepatic metastatic masses, proven to be carcinoid tumor on biopsy. Transthoracic echocardiogram showed right to left shunt via PFO with early thickening of the mitral valve. There was severe regurgitation of tricuspid and pulmonic valves.
Decision-making: Progression of right valvular CHD and reduced right atrial compliance from endocardial damage causes elevated right heart pressures, hence PFO resumption. Octreotide calmed her symptoms. Right heart catheterization and intra-cardiac echo guided percutaneous PFO closure with a 16 mm Cribriform ASD device was done. Hypoxia resolved.
Conclusion: Relief of hypoxia and mortality benefit from improved function are derived with PFO closure. Screening for PFO is vital because of a high risk of worsening right CHD and developing left CHD as serotonin bypasses lung deactivation.
Keywords: Carcinoid heart disease; PFO; Serotonin
Introduction
A 67-year old African American woman presented with symptoms of shortness of breath for 1 month. Shortness of breath was worse with minimal exertion and associated with orthopnea. She had been on home oxygen for past 1 month.
Her past medical history included hypertension, hyperlipidemia and hypothyroidism. In the past two months, her complaints included persistent upper abdominal pain, episodes of diarrhea, vomiting, hot flashes and weight loss of 20 pounds.
On admission blood pressure was 130/80 mm Hg, pulse rate was 73 beats per minute and respiratory rate was 26 breaths per minute. Oxygen saturation was 90 % on room air and did not improve with high flow nasal cannula rate of 30 litres per minute. Cardiac examination revealed grade 2/6 systolic murmur heard at left sternal border and elevated jugular venous pulse was 5 centimeter above right clavicle. Abdominal and skin examinations were normal. Tumor markers alpha-fetoprotein (AFP), cancer antigen 19-9 (CA 19-9) and carcinoembryonic antigen (CEA) were not elevated. Urine 5-hydroxyindoleacetic acid (5-HIAA) was elevated at 190.5 mg/g (normal range <10 mg/g). Serum chromogranin A level was also elevated at 18.8 ng/ml (normal range 1.9 to 15 ng/ml). Serum serotonin was 1340 ng/ml (normal range 56 to 244 ng/ml).
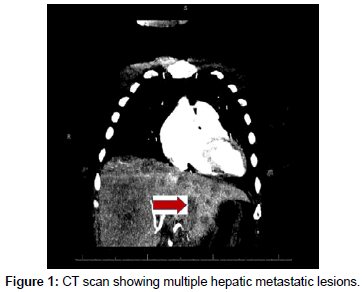
Computed Tomography (CT) scan of chest was negative for pulmonary embolism or masses. CT scan of abdomen showed extensive hepatic metastatic lesions (Figure 1), with subsequent biopsy proving these to be carcinoid tumor. Colonoscopy and Esophagogastroduodenoscopy were normal. Octreotide scan showed extensive hepatic activity.
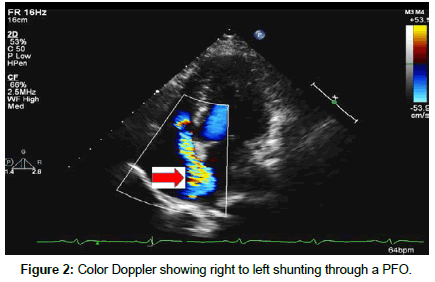
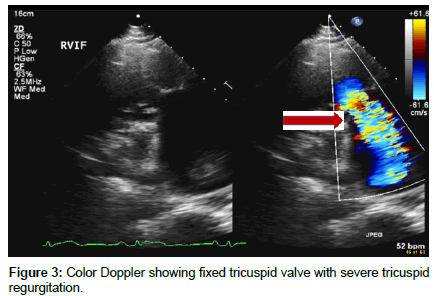
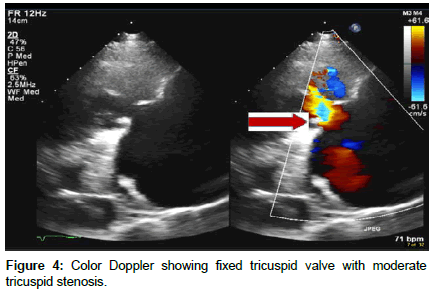
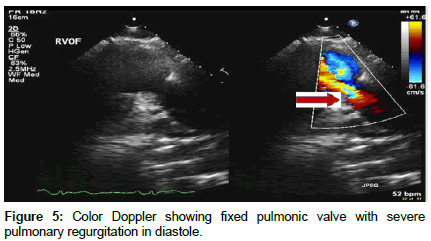
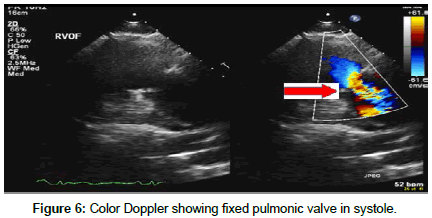
Transthoracic Echocardiography showed severe tricuspid regurgitation with tricuspid regurgitation velocity of 2.48 m/s with associated moderate tricuspid stenosis, severe pulmonic regurgitation, dilated right atrium, and dilated right ventricle with TAPSE of 1.72 cm, and a normal size left ventricle with ejection fraction of 60%. The inter-atrial septum was bowing toward the left atrium consistent with high right atrial pressure. A patent foramen ovale was detected with significant right to left shunt on both color Doppler and agitated saline study (Figures 2-6).
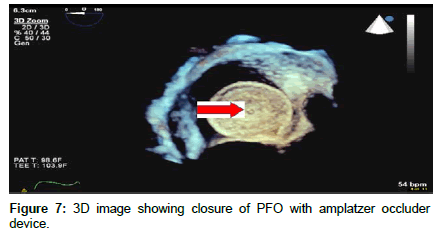
Intravenous octreotide (100 mcg every 8 hours) was administered with dramatic relief of symptoms. The significant hypoxemia, which did not improve after supplemental oxygen was believed to be the result of intracardiac shunting. The patient underwent right heart catheterization followed by intracardiac echo-guided percutaneous PFO closure with a 16 mm Cribriform ASD occluder device which resulted in immediate resolution of hypoxemia (Figure 7).
Discussion
Nowadays, carcinoid tumors are termed neuroendocrine tumors (NETs) [1]. NETs are rare in occurrence and predominantly arise in the gastrointestinal tract. Other sites in which NETs occur include the lungs, pancreas, appendix and ovaries [2]. Carcinoid syndrome (CS) consists of a constellation of symptoms including skin flushing, diarrhea, bronchospasm and cardiac valvular disease.
Carcinoid heart disease (CHD) complicates the clinic course of 50% of patients diagnosed with NETs and carcinoid syndrome (CS) [3]. As seen in our patient, in approximately 18% of cases, the primary NETs site remains unknown [2].
NETs secrete vasoactive substances which promote fibroblast proliferation and plaque deposition. These substances include serotonin (5-HT), prostaglandins, histamine, bradykinin, tachykinins and transforming growth factor beta [1]. In CHD, due partly to serotonin exposure, there is fibrous endocardial thickening resulting in retracted fixed valves and resultant regurgitation [4]. Our case shows the tendency for CHD to occur when there is metastatic hepatic involvement [1,3] due to resulting inability of liver to metabolize these substances.
CHD typically consists of tricuspid and pulmonic valves involvement. This is due to deactivation of serotonin in the lungs, hence mitral and aortic valves are usually not affected. Other rarer manifestations of CHD include left side valvular involvement, intracardiac tumor, metastatic involvement of myocardium and pericardium as well as pericardial effusion [5]. Prognostic markers of survival in CHD include serum chromogranin A and serum NTproBNP [6].
There is an increased prevalence of PFO in patients with CHD and CS as compared to CS alone [4]. With progression of right valvular CHD and reduced compliance of right atrium because of endocardial involvement, there is increased systolic pulmonary arterial pressure, right ventricular and right atrial pressure, hence revitalization of PFO [4,7]. Therefore with right to leave shunting of serotonin via PFO, there is an increased risk of development of left side valvular CHD [7]. Furthermore there is increased risk of debilitating hypoxia as exhibited in our patient.
There is no clear guideline directed approach to management of PFO in the context of CHD. However our patient clearly benefited from closure of PFO, as there was resolution of hypoxia. Predictors of mortality in CHD include moderate to severe right ventricular dilation and New York Heart Association class III or IV (NYHA) [2]. With closure of PFO, there is improvement in New York Heart Association (NYHA) functional class, 6 minute walking distance and arterial blood gas results [4]. It remains unclear whether percutaneous PFO closure offers full protection against progression to left sided CHD, the reason for this being residual shunting due to delay in endothelization of PFO occluder device and persistent elevation of right atrial elevation secondary to valvular involvement.
Conclusion
The benefits of PFO closure includes the symptomatic relief obtained and mortality benefit it offers from improvement in NYHA functional class. The presence of a PFO is associated with increased risk of deterioration of right side CHD and development of left side CHD. Therefore screening for PFO via contrast transthoracic echocardiogram is of essential importance in management of CHD patients.
References
- Grozinsky-Glasberg S, Grossman A, Gross D (2015) Carcinoid Heart Disease: From Pathophysiology to Treatment-‘Something in the Way It Moves’ Neuroendocrinology 101: 263-273
- Moller JE, Pellikka PA, Bernheim AM, Schaff HV, Rubin J, et al (2005) Prognosis of carcinoid heart disease: analysis of 200 cases over two decades. Circulation 112: 3320-3327.
- Fox DJ, Khattar RS (2004) Carcinoid heart disease: presentation, diagnosis, and management. Heart 90: 1224-1228.
- Mansecal N, Mitry E, Pillière R, Lepère C, Gérardin B, et al. (2008) Prevalence of Patent Foramen Ovale and Usefulness of Percutaneous Closure Device in Carcinoid Heart Disease 101: 1035-1038.
- Pellikka PA, Tajik AJ, Khandheria BK, Seward JB, Callahan JA, et al. (1993) Carcinoid heart disease. Clinical and echocardiographic spectrum in 74 patients. Circulation 87: 1188-1196.
- Korse CM, Taal BG, de Groot CA, Bakker RH, Bonfrer JM (2009) Chromogranin-A and N-terminal pro-brain natriuretic peptide: an excellent pair of biomarkers for diagnostics in patients with neuroendocrine tumor. J Clin Oncol 27: 4293-4299.
- Mansecal N, Mitry E, Forissier J, Martin F, Redheuil A, et al. ( 2006) Assessment of patent foramen ovale in carcinoid heart disease. Am Heart J 151: 1129.e1-6.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi