Research Article, J Diagn Tech Biomed Anal Vol: 7 Issue: 2
Comparison of 1.5T and 3T Prostate MR Examination using Surface Array Coils in Routine Clinical Practice
Ryznarova Z1,2*, Dezortova M1, Jiru F1, Vik V3, Zachoval R3 and Hajek M1
1Department of Diagnostic and Interventional Radiology, Institute for Clinical and Experimental Medicine, Prague, Czech Republic
2Department of Radiology, Na Homolce Hospital, Prague, Czech Republic
3Department of Urology, Thomayer Hospital, Prague, Czech Republic
*Corresponding Author : Zuzana Ryznarova
Department of Radiology, Na Homolce Hospital, Prague, Czech Republic
Tel: +420 257 272 166
E-mail: zryznarova@seznam.cz
Received: March 12, 2018 Accepted: April 04, 2018 Published: April 11, 2018
Citation: Ryznarova Z, Dezortova M, Jiru F, Vik V, Zachoval R, et al. (2018) Comparison of 1.5T and 3T Prostate MR Examination using Surface Array Coils in Routine Clinical Practice. J Diagn Tech Biomed Anal 7:2. doi: 10.4172/2469-5653.1000131
Abstract
Objective: Our study compares the results of magnetic resonance (MR) examination obtained by the 1.5T and 3T MR scanners using surface coils in patients with prostate carcinoma. Methods: A total of 103 consecutive patients (aged 44-72 years) with biopsy confirmed prostate carcinoma underwent MR examination at 3T or 1.5T with similar protocols including T2-weighted (T2W), diffusion weighted imaging (DWI) including apparent coefficient (ADC) maps, dynamic contrast enhancement (DCE) and magnetic resonance spectroscopy (MRS). MR results were compared with the histopathologic findings of specimen after the radical prostatectomy. The first part of the study assessed local tumor staging. The sensitivity and specificity for extracapsular tumor extension and prediction of seminal vesicles infiltration were calculated and compared using a Fisher exact test. The second part of the study evaluated the accuracy in tumor localization assessment. Results from different MR sequences were compared using a Kruskal-Wallis test. Results: The highest sensitivity and specificity (70% and 100%) as a predictor of extracapsular tumor extension were found in group B, examined at 3T when DCE was included into the standard protocol. The overall accuracy in tumor stage prediction were 66% (1.5T), 90% (3T with (DCE) and 72% (3T without DCE). No significant difference among the three patient groups was found in the accuracy of tumor localization assessment. The best results were obtained when T2 - weighted imaging was combined with DWI or DCE. Conclusion: The highest accuracy of local prostate cancer staging was obtained in patients examined with 3T MR scanner when MR protocol included DCE. No significant difference in tumor localization assessment between 3T and 1.5T MR scanners was found.
Keywords: Prostate; Carcinoma; Magnetic resonance; Multi-parametric MR; 1.5T; 3T; MR spectroscopy
Abbreviations
MR: Magnetic Resonance; MRI: Magnetic Resonance Imaging; T2W: T2-Weighted; T1W: T1-Weighted; DWI: Diffusion Weighted Imaging; ADC: Apparent Diffusion Coefficient; MRS: Magnetic Resonance Spectroscopy; DCE: Dynamic Contrast Enhancement; TSE: Turbo Spin Echo; GE: Gradient Echo; TR: Time To Repeat; TE: Time To Echo; PI-RADS: Prostate Imaging Reporting And Data System; ECE: Extracapsular Extension; SVI: Seminal Vesicle Infiltration; SNR: Signal-to-Noise Ratio
Introduction
Prostate cancer is the third most common cause of deaths in men in Europe [1]. Magnetic resonance (MR) plays an important role in the staging of local prostate carcinoma [2]. Accurate assessment of local staging and tumor extension are important factors in determining the treatment of prostate carcinoma and prognosis of tumor recurrence after treatment [3]. MR imaging (MRI) examination is based on T2-weighted (T2W) sequences generating high resolution anatomical images of zonal anatomy of the prostate and it is usually applied in combination with functional MR techniques such as diffusion weighted imaging (DWI), MR spectroscopy (MRS) and dynamic contrast enhancement (DCE) [4-7]. Multiparametric MR is the combination of T2W high-resolution images with at least two functional techniques and was recommended by the European Society of Urogenital Radiology [8] as a standard MR examination protocol for prostate cancer. However, the optimum field strength and the use of an endorectal or phased-array coils are still being discussed. The endorectal coil and a combination of the endorectal and body phased-array coils at 1.5T was found superior in local tumor staging to just the body phased-array coil, however the differences were not significant [9]. Image quality of prostate cancer from 1.5T with the use of an endorectal coil combined with a body phased-array coil is comparable to that from 3T scanner with a surface coil alone [10,11 ]. However, MR examination of the prostate is practiced in many centers using 1.5T without the use of endorectal coil [12 ].
To the best of our knowledge, there has been no study comparing the diagnostic yield of detection and staging of prostate carcinoma using 1.5T or 3T scanners with only surface coils. Therefore, we focused on the comparison of both the 1.5T and 3T scanners equipped with phased-array coils in preoperative staging of prostate tumors during routine clinical practice with additional analysis. The purpose of our study was to evaluate the accuracy of local prostate carcinoma staging and tumor localization for both scanners (1.5 T and 3T), by the correlation of MRI findings with histology obtained after total prostatectomy.
Material and Methods
Patients
A total of 103 consecutive patients with localized or locally advanced prostate cancer, diagnosed by transrectal prostate biopsy, underwent MR examination followed by radical prostatectomy in the period of 4 weeks after MR examination.
Patients were divided into three groups according to examination protocol. Two different multiparametric protocols were applied. The first protocol was used at 1.5T and 3T and included T1W and T2W imaging, DWI, MRS and DCE examination. The second protocol used only a 3T scanner with all the sequences as the first protocol, but without DCE examination.
In the first group (A) 41 patients (mean age 66.7 ± 6.1 years) were examined in a 1.5T MR scanner using the first protocol. In the second group (B) 30 patients (mean age 66.9 ± 4.4 years) underwent MR examination in a 3T MR scanner also using the first protocol. The third group (C) included 32 patients (mean age 66.0 ± 7.1 years) who underwent MR examination with 3T using the second protocol.
The characteristics of the patients are summarized in Table 1. All MR examinations were performed at least 6-8 weeks after the biopsy to reduce artifacts from post-biopsy hemorrhage. None of the patients had been previously treated with hormone deprivation therapy. Informed consent was taken from all patients for the purpose of the study and approval was given by the local ethical committee.
| Group | A | B | C |
|---|---|---|---|
| 1.5T with DCE** (A) | 3T with DCE (B) |
3T without DCE (C) | |
| Number of patients in the group | 41 | 30 | 32 |
| Average initial PSA* (ng/ml) | 7.7 (range 3.2-14.4) |
6.4 (range 2.3-15.6) |
9.7 (range 4.2-25.5) |
| Prostate risk stratification system [33] | |||
| Low risk | 22% (9) | 17% (5) | 25% (8) |
| Intermediate risk | 41% (17) | 36% (11) | 37.5 % (12) |
| High risk | 37% (15) | 47% (14) | 37.5% (12) |
| Histological staging acording to TNM Classification of Malignant Tumors [13] | |||
| T2 | 61% (25) | 56.6 % (17) | 56 % (18) |
| T3a | 29% (12) | 33.3 % (10) | 34% (11) |
| T3b | 10% (4) | 10 % (3) | 9% (3) |
| T4 | 0 | 0 | 0 |
Table 1: Clinical characteristics of patients groups.
MR examination
Patients were examined in a supine position in 1.5T and 3T scanners (Avanto and Trio, Siemens, Erlangen, Germany) using 8 channels phased-array surface coils. The T1W turbo spin echo (TSE) sequence in axial plane was performed to exclude post-bioptic hemorrhage and pelvic lymphadenopathy; T2W TSE sequences were performed in axial (orthogonal to the urethra), coronal and sagittal planes. Echo-planar DWI was obtained in transverse plane parallel to the transverse T2W to construct ADC maps using the standard Siemens software.
Dynamic contrast enhancement 3D T1-spoiled gradient echo (GE) images were acquired during an intravenous bolus injection of paramagnetic contrast medium (gadobenate dimeglumine) at a dose of 0.2 mmol/kg of body weight for examination at 1.5T and 0.1 mmol/kg of body weight for examination at 3T. A common flow rate of 2.0 ml/s was used, followed by a 20 ml saline flush. The images were acquired every 13 seconds at 1.5T and every 8 seconds at 3T, with each sequence lasting for 4:30 minutes in both scanners. The 3D volume of the entire prostate was covered. Parameters of the sequences are summarized in Table 2.
| Sequence | T1 TSE | T2 TSE | DWI | 3D T1 GRE |
|---|---|---|---|---|
| Slice thikness (mm) | 5 | 3 | 3 | 3 |
| Field-of-view (mm) | 250 | 180 | 250 | 250 |
| Flip angle | 150 | 150 | 10 | |
| Time to repetition (ms) | 700*/700 | 4000*/3000 | 2400*/4500 | 3.62*/7.64 |
| Time to echo (ms) | 11*/13 | 96*/99 | 102*/102 | 1.33*/2.77 |
| Matrix | 320x256* 256x256 |
320x256* 256x256 |
192x192 | 256x192 |
| B-value | - | - | b - 0,50,1000* b - 0,500,1000 |
- |
Table 2: MRI sequence protocol applied at 1.5T and 3T tomographs.
1H MRS was performed by using a point-resolved 3D spectroscopic imaging sequence with the following parameters: 10 acquisitions; repetition time/echo time (TR/TE) = 720/145 ms or 690/120 ms; the total measurement time was 16:17 min or 17:00 min in both 3T and 1.5T scanners, respectively. Spectral suppression of lipid and water signals was applied. The nominal voxel size before apodization was 7.5 x 7.5 × 7.5 mm. Ten patients had to be excluded from MRS evaluation due to poor spectra quality.
MR data evaluation
The first part of the study focused on local carcinoma staging. MR data sets (T2W imaging, DWI, MRS and DCE) were evaluated in the daily work routine by way of consensus of two radiologists. The radiologists were informed that the tumor had been confirmed by biopsy, no information about the clinical stage and prostate specific antigen level was available. The local tumor staging was assessed in each patient using the standard TNM Classification of Malignant Tumors [13].
Positive signs for the extracapsular tumor extension (T3a) were considered as follows: capsular irregularity, bulging of prostatic contour, tissue signal hypointensity spreading in the periprostatic fat, obliteration of rectoprostatic angle and asymmetry of the neurovascular bundle [14,15 ].
The criteria for seminal vesicles invasion (T3b) were as follows: abnormal asymmetric low signal of seminal vesicles on T2W sequences, filling in of the prostate-vesicle angle, seminal vesicles enhancement, seminal vesicles asymmetry [16] and diffusion restriction visible on ADC maps. MR results were compared with the final pathological report.
The second part of the study was focused on tumor localization. T2W images and DWI including apparent diffusion coefficient (ADC) maps, DCE and MRS results were interpreted qualitatively based on PI-RADS scoring system [8] for the tumor detection and localization. A qualitative approach based on a visual characterization of kinetic curves in DCE evaluation was used [8]. A time-intensity curve was analyzed (Figure 1); early wash-in and early wash-out were used as a criterion for prostate cancer presence [17]. MRS data were evaluated qualitatively on a system console (Siemens Medical) comparing peak heights of the metabolites as described Jung et al [18].
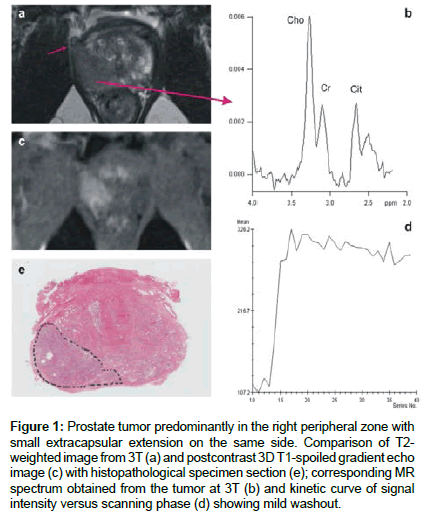
Figure 1: Prostate tumor predominantly in the right peripheral zone with small extracapsular extension on the same side. Comparison of T2- weighted image from 3T (a) and postcontrast 3D T1-spoiled gradient echo image (c) with histopathological specimen section (e); corresponding MR spectrum obtained from the tumor at 3T (b) and kinetic curve of signal intensity versus scanning phase (d) showing mild washout.
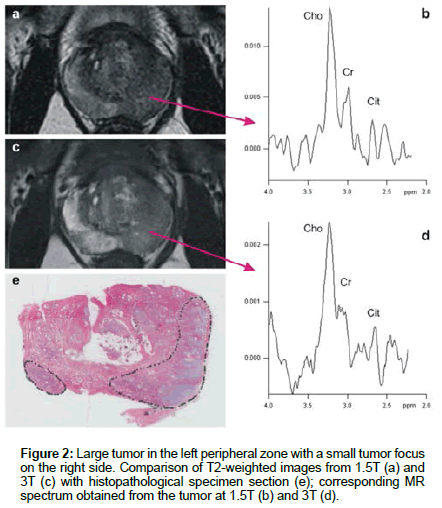
Data obtained from each method were compared step by step with histopathological findings (Figure 2). Based on anatomic landmarks on axial T2W images the prostate was divided into three levels in superior-inferior order: base, middle gland and apex [19]. Each slice obtained from the base, middle gland and apex of the prostate were divided into three partitions (central gland, left and right peripheral zones), i.e. a total of 9 segments.
The MR output in each segment was compared with the histopathologic findings in corresponding segment of the whole mount section.
Histopathology
Prostatectomy specimens were fixed in neutral formaldehyde and coated with Indian ink. Sections parallel to transverse MRI scans with 5 μm thickness were prepared from paraffin whole mount sections and stained with hematoxylin-eosin. On pathologic analysis a Gleason score was assigned to the whole cancer in the specimen. The prostatectomy specimens were staged according to the TNM classification system [13].
Statistics
Sensitivity and specificity of predictions of extracapsular tumor extension and seminal vesicles infiltration were evaluated and compared using a Fisher exact test. Results obtained from different MR methods were compared using a Kruskal-Wallis test. P-values less than 0.05 were considered as statistically significant. Analyses were conducted using R statistical package, version 3.2.3.
Results
In the first part of the study the comparison of final histopathologic and MR results was done in 103 patients. The sensitivity and specificity as a predictor of prostate carcinoma stage were calculated for each group.
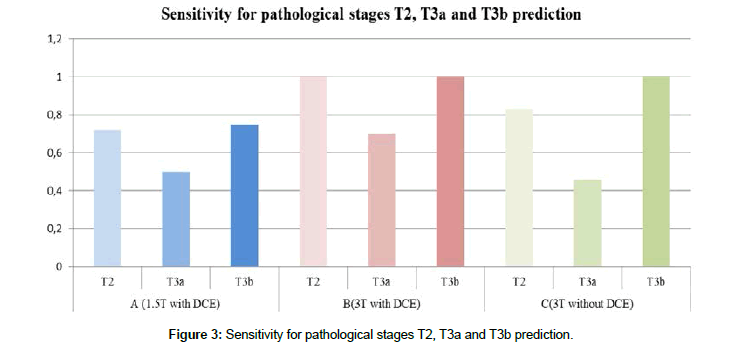
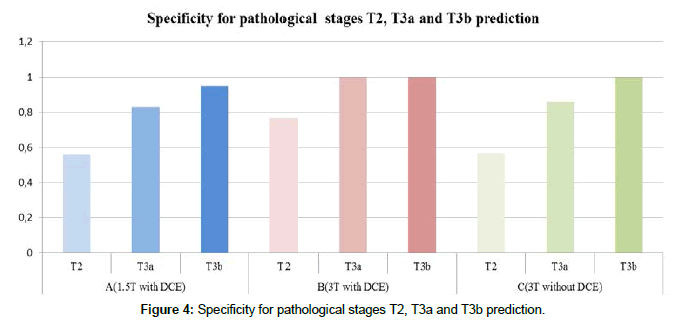
In the first group (A), examined in the 1.5T MR scanner using the first protocol, 27 out of 41 patients were correctly staged. The resulting sensitivity and specificity as a predictor for the tumor localized inside prostate (T2 stage) were 72% and 56%, for extracapsular tumor extension (T3a) 50% and 83%, and for seminal vesicle infiltration (T3b) 75% and 95%. In the second group (B), examined in the 3T MR scanner using standard protocol, 27 out of 30 patients were correctly staged. The sensitivity and specificity were 100% and 77% for T2 stage prediction, 70% and 100% for T3a stage prediction, and 100% and 100% for T3b stage prediction. In the third group (C), examined at 3T without dynamic contrast examination, 23 out of 32 patients were correctly staged. The resulting sensitivity and specificity were 83% and 57% for T2 tumor stage prediction, 46% and 86% for T3a stage prediction, and 100% and 100% for T3b stage prediction (Figures 3 and 4).
The overall accuracy in tumor stage prediction were 66%, 90% and 72% for patients in the groups A, B and C, respectively.
The second part of the study focused on accuracy in tumor localization assessment using T2W images in combinations with DWI with ADC maps, MR spectroscopy and DCE. The results were compared with histopathologic findings and evaluated using a Kruskal-Wallis test. Better accuracy was obtained when T2W images were combined with DWI and ADC or with DCE in all groups. No significant differences of accuracy in tumor localization among the three groups were found. Results are summarized in Table 3.
| A (1.5T with DCE) | B (3T with DCE) | C (3T without DCE) | |
|---|---|---|---|
| T2W+ADC (%) | 82.37 | 82.73 | 75.18 |
| T2W+MRS (%) | 70.97 | 65.11 | 59.17 |
| T2W+DCE (%) | 75.97 | 83.10 | - |
Table 3: The accuracy of tumor localization.
Discussion
The optimum field strength for detection and staging of local prostate cancer is a topic which is continually being discussed. Prostate MR examinations are performed with both 1.5T and 3T scanners in many centers with only surface coils.
This study was focused on the comparison of two scanners (1.5T and 3T) using the only surface phased-array coil in routine prostate MR examination. The correlation between preoperative MR staging of prostate cancer and pathological outcome in patients with confirmed prostate cancer was evaluated for both scanners.
Our results did not show statistically significant difference in sensitivity and specificity of local carcinoma staging between 1.5T and 3T scanners when the phased-array coil was used, in spite of the different image quality obtained at 1.5T and 3T. Using the 3T scanner and adding DCE into the standard protocol led to the improvement of overall accuracy in local tumor staging. The accuracy for extraprostatic extension obtained in our study was similar to other published studies; the similar accuracy was published by Fütterer et al for 1.5T scanner [14] and Somford et al presented the similar accuracy for extraprostatic extension at 3T [20]. Higher field strength led to better spatial resolution in the prostate region, better extracapsular extension detection and seminal vesicles infiltration detection on T2- weighted sequences. Higher signal-to-noise ratio with 3T improved especially T2W images providing better anatomical detail and good T2 contrast [17]. Our results showed higher specificity and lower sensitivity for ECE and SVI detection in all groups, similar to other studies [21].
We found the best accuracy in tumor localization assessment in all the three groups when T2W images were combined with ADC maps in 1.5T and 3T scanners. An improvement in the detection of prostate cancer when T2W images were combined with DWI with ADC maps was described in several studies [22-24]. However, we found no significant difference in tumor localization assessment between 1.5T and 3T scanners in this study when T2W imaging was combined with DWI. The reason could be in higher image artifacts in DWI due to higher magnetic field. It is important to note that image quality is better at 3T and higher SNR in prostate DWI is obtained, nevertheless geometric distortion and ghosting artifacts are more pronounced, which could be limiting for image evaluation [25].
When DCE was evaluated separately, better accuracy in tumor localization was obtained at a higher magnetic field. Increased SNR at 3T leads to an improved image quality with better temporal and space resolution at a higher magnetic field strength [26]. In spite of the fact that worse temporal resolution was used in the study (13s at 1.5T and 8s at 3T), DCE improved the overall accuracy in tumor localization at 3T. However, no statistically significant difference was found in tumor detection among our three patients groups examined in different scanners 1.5T and 3T (p=0.09).
Higher magnetic field strength allowed reduction of a total volume of injected contrast medium. In the 3T scanner a half amount of contrast medium was used in comparison with 1.5T scanner to obtain the same signal intensity of MR images.
Concerning MRS, we did not confirm our primary assumption that 3T improves in the overall accuracy in prostate carcinoma detection [27,28]. We found no statistically significant difference between 1.5 and 3T scanners in tumor detection accuracy in MR spectroscopy (p=0.08).
The main reason for our result is the qualitative approach for MRS data evaluation. Although the qualitative evaluation is recommended by some authors for routine clinical interpretations of prostate tumors [29] as an easier alternative to quantitative analysis by specialized software, our results show that qualitative visual evaluation is not a suitable method for evaluating of spectroscopic data at 1.5T and even less at 3T (as the MR spectra shape is more complicated at 3T, e.g. the citrate is a quadruplet). In addition, qualitative visual evaluation is not able to include signal intensity changes during ageing [30]. Our finding is in agreement with the recommendation of PI-RADS prostate imaging reporting and data system version 2 [31], not to use 1H MR spectroscopy in routine prostate clinical examination.
We are aware of some limitations of our study. The first limitation is that the comparison between 1.5T and 3T and among three patient groups was not done on the same patients. To minimize the effect of the different populations, consecutive patients were examined with the similar clinical indications. The study is also limited by low number of patients with advanced stage of prostate cancer in our group, due to low incidence of these stages in our population. Another possible limitation can be simplification by using of nine-segments framework for the comparison between MRI and histopathology, instead of 27 regions as recommended by Dickinson for prostate cancer detection [32]. The nine-segment framework was chosen for both histopathological specimen and MRI scans for easier anatomical orientation and correlation between these two methods. Several studies recommended sextant framework as a reproducible way of reporting MRI findings [33,34]. We used the similar evaluating system which was extended to the central gland that was evaluated separately at each level.
Conclusions
In conclusion, the best accuracy of local prostate cancer staging was obtained in patients examined in 3T MR scanner in the MR protocol including DCE. No significant difference in tumor localization assessment between 3T and 1.5T MR scanners was found.
Acknowledgements
Authors extend their thanks to the Department of Pathology of Thomayer Hospital and Assoc Prof Hana Malikova, PhD, Prague. Supported by Ministry of Health of the Czech Republic, grants IGA NT13017 and DRO (“Institute for Clinical and Experimental Medicine - IKEM, IN 00023001”).
References
- Reddy MP, Rao DMR, Verma RS, Srinath B, Katiyar RS (1998) Response of S13 mulberry variety to VAM inoculation under semi-arid condition. Ind J Plant Physiol 3: 194-196.
- Bloemberg GV, Wijfjes AHM, Lamers GEM, Stuurmam N, Lugtenberg BJJ (2000) Simultaneous imaging of pseudomonas fluoresens WCS 3655 populations expressing three different autofluorescent proteins in the rhizosphere: new perspective for studying microbial communities. Mol Plant Mic Int 13:1170-1176.
- Goel AK, Laura RD, Pathak DV, Anuradha G, Goel A (1999) Use of biofertilizers: potential, constraints and future strategies review. Int J Trop Agric 17: 1-18.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi