Research Article, J Trauma Stress Disor Treat Vol: 6 Issue: 3
Complex Real Life-Related Prospective Memory in Soldiers with and Without Post-Traumatic Stress Disorder
Katharina Glienke1,2*, Gerd Dieter Willmund3, Peter Zimmermann3 and Martina Piefke1,2
1Neurobiology and Genetics of Behavior, Department of Psychology and Psychotherapy, Germany
2Centre for Biomedical Education and Research, Witten/Herdecke University, Germany
3Centre for Military Mental Health, Hospital of the German Armed Forces, Germany
*Corresponding Author : Katharina Glienke, M.Sc.
Witten/Herdecke University, Faculty of Health, Department of Psychology and Psychotherapy, Neurobiology and Genetics of Behavior, Alfred-Herrhausen-Str.50, D-58448 Witten, Germany
Tel: 00492302/926-785
Fax: 00492302/926-407
E-mail: katharina.glienke@uni-wh.de
Received: July 24, 2017 Accepted: August 10, 2017 Published: August 17, 2017
Citation: Glienke K, Willmund GD, Zimmermann P, Piefke M (2017) Complex Real Life-Related Prospective Memory in Soldiers with and Without Post- Traumatic Stress Disorder. J Trauma Stress Disor Treat 6:3. doi: 10.4172/2324-8947.1000176
Abstract
Objective: Posttraumatic stress disorder (PTSD) is known to influence memory functions in a complex fashion. However, little is known about the impact of PTSD on future directed mnemonic functions like prospective memory (PM).
Methods: The present study aimed at examining performance of 13 soldiers with PTSD, 12 without PTSD and 21 non-military controls without PTSD across different phases of a real life-related PM paradigm. In addition, neuropsychological tests of memory and the influence of salivary cortisol levels on memory performance were
assessed.
Results: Soldiers with PTSD performed significantly worse during the planning and retrieval phase of time- and event-dependent PM compared to the non-military controls, but not to soldiers without PTSD. Moreover, PTSD symptom severity correlated negatively with PM retrieval. In soldiers without PTSD time- and eventdependent PM was significantly declined during retrieval phase compared to the non-military controls. Significantly decreased salivary cortisol levels were detected only for soldiers with PTSD. Standard neuropsychological assessment showed significantly impaired working- and retrospective long-term memory in both
soldiers with and without PTSD.
Conclusion: Our results indicate for the first time that exposure to combat-related stress may have selective deteriorating effects on real-life-related PM and other memory functions in the absence of a PTSD diagnosis. We conclude that stress-related alterations of memory functions in soldiers may not only be related to
PTSD-related changes in cortisol metabolism. Rather, additional neuropeptides and/or neurohormones may be involved in the emergence of memory dysfunctions resulting from combat-related stress.
Keywords: Posttraumatic stress disorder; Prospective memory; Cortisol; Combat-related stress; Implementation of intention; Future memory; Deployment-related PTSD; HPA-axis
Introduction
Posttraumatic stress disorder (PTSD) is understood as mental disorder that originates from a traumatic event (Diagnostic and Statistical Manual of Mental Disorders-5) [1]. PTSD symptoms have frequently been reported after sexual and/or physical abuse [2,3], environmental disasters [4], and in combat veterans [5]. Epidemiological studies of German soldiers who have served in combat show service related PTSD incidence of about 0.9% and a twelve months prevalence of about 2.9% [6-9]. It is well known that PTSD is accompanied by cognitive disturbances. Long-term and working memory, executive functions, and attention are particularly affected. So far, neurocognitive alterations associated with PTSD have been best documented for retrospective declarative memory and learning. For instance, patients with PTSD have repeatedly been reported to show deteriorated performance in retrospective declarative memory and learning tasks compared to controls [10-14]. In addition, prior research demonstrated impairments in working memory, attention, and executive functions (except for planning performance) in patients with PTSD [15,16]. These deficits result at least in part from morphological and/or functional abnormalities of brain regions involved in memory and other higher cognitive functions. These are mainly the hippocampus, the amygdala, and regions of the prefrontal cortex [17,19]. It is known that alterations of the physiological stress reaction, in particular the metabolism of the stress hormone cortisol, play a critical role in the emergence of cognitive disturbances associated with PTSD [12,20,21].
To date, little is known about the influence of PTSD on prospective memory (PM). PM contains future-directed mnemonic functions, which proceed through different phases of processing [22,23]. PM allows us to plan intentions (planning-phase), retain them in memory (retention-phase), retrieve them at a certain point of time (timedependent PM) or in response to a particular event (event-dependent PM) in the future, and translate them into action (performancephase) [22-24]. Neuroanatomically, PM relies on functions of medial temporal (in particular the hippocampus) and prefrontal brain structures (in particular the medial prefrontal and frontopolar cortices) [25-28]. Although, PM is understood as a distinct memory concept it also shares functional and neuroanatomical overlaps with retrospective memory (RM) [26,29]. Given the overlap of brain structures which are relevant in both PM and RM it is likely that PTSD may deteriorate both memory of the past and memory of the future to a comparable extent [18,25-28]. Studies of episodic future thinking in PTSD patients showed that the disorder has deteriorating effects on imagination of the future [30-32]. Although it is a cognitive ability, which is directed to the future it is distinct from PM in that it does not require the planning, retention, and execution of intentions in the future [33-35]. Nonetheless, authors found a significant correlation between PM and episodic future memory [35]. They suggested that episodic future thinking may contribute to PM by the imagination of the context that will be encountered at the moment the intentions will be brought into action.
Yet, there are only two studies published which addressed PM in combat veterans [36,37]. In one of the two studies, authors tested event-dependent PM in combat veterans with PTSD [36]. They applied an ongoing PM distraction task and tested event-dependent PM. The task contained of multiple-choice trivia questions which had to be answered and participants were asked to press a computer button every time they saw a certain cue word. They found a negative correlation between the severity of PTSD symptoms and event-based PM. In the other study authors tested PM in combat veterans with and without PTSD and consulted the Memory for Intention Test (MIST) to investigate the influence of PTSD on both event- and time-based PM [37-39]. The MIST consists of eight PM trials over 30 min. that are counterbalanced by PM type (i.e., time-based vs. eventbased), response (i.e., physical vs. verbal), and delay (i.e., 2 min vs. 15 min) [37-39]. They found a significant decline in the sum score of PM and a significantly increased error rate in time-dependent PM in combat veterans with PTSD compared to combat veterans without PTSD.
Taken together, the few studies addressing the impact of PTSD on PM draw an inconsistent picture of results. Additionally, studies of PTSD patients aiming at a differentiation of the influence of the disorder on different phases of PM have not been published, yet. Thus, previous paradigms may have not tapped the main high cognitive demands of everyday-related PM. The current study extends prior research by investigating the impact of PTSD on the different phases of PM in a complex real life-related PM and including an additional control group that is free of combat or another trauma, besides the combat veteran control group. To our knowledge, so far there has been no study focusing on complex real-life related PM in soldiers with PTSD in comparison with non-PTSD soldiers and healthy controls. In these times, it is of particular interest to scrutinize the consequences of combat-related trauma on a memory function that is of such an importance for everyday routine as PM is. Additionally, the study may give better insights in occupational rehabilitation and therapy of PTSD. Referring to the findings of prior research we hypothized that soldiers with PTSD show poorer complex PM especially during the retrieval and execution phase, compared to non-traumatized soldiers and healthy controls. Since the present study applies a highly complex real life- related PM task that requires a high PM capacity for both time- and event-based PM it is reasonable to assume that PTSD may alter both domains of PM.
Materials and Methods
Participants
13 male soldiers who have a combat related PTSD (PTSD group; mean age 35.25 years, standard deviation (SD) ± 7.54) and a trauma exposed comparison group (military control group; MC group) of 12 male soldiers who were also exposed to combat but do not meet criteria for PTSD (mean age 31.02 years, SD ± 10.95) were tested. All soldiers were recruited from the hospital of German Armed Forces Berlin. Additionally, 21 male non-traumatized and non-military control participants (non-MC group; mean age 27.67 years, SD ± 10.32) were recruited from Witten/Herdecke University, Bochum University, and Dortmund University. The groups did not differ in age. It was ensured, that the participants IQ raged within one standard deviation. One participant of the PTSD group and one participant of the MC group had an IQ between 80 and 85. We decided to include the data of those participants, because the general cognitive performance did not significantly differ from the other participants of their group. All volunteers were controlled for a normal or correctedto- normal vision. Exclusion criteria were severe psychiatric illnesses (e.g., psychoses), neurological diseases (including head injury with a loss of consciousness), severe medical diseases (e.g. thyroid disease, cancer etc.), neuroleptic medication, and alcohol or substance abuse. Each participant gave written informed consent, and the study was approved by the ethics committee of Witten/Herdecke University.
Psychiatric assessment
Before the testing, all soldiers were administered to the “Strukturiertes Klinisches Interview für DSM-IV” (SKID) by extensively trained clinicians [40]. PTSD was diagnosed according to DSM-IV criteria. Additionally, combat soldiers that suffered from trauma also completed the “PTSD Diagnostic Scale for DSM-5” (PDS) prior to the actual test day [41]. Non-traumatized participants were screened for psychological and physiological disorders with screening interview before the first testing. To assess putative depressive symptomatology, all participants completed the Beck Depression Inventory II [42]. Former research has shown, that in patients with PTSD, the physiological stress response is mainly characterized by diminished levels of cortisol [20,43,44]. Therefore, we also gathered five saliva cortisol samples across the testing as an endocrinological marker in all participants.
Procedure and experimental paradigm
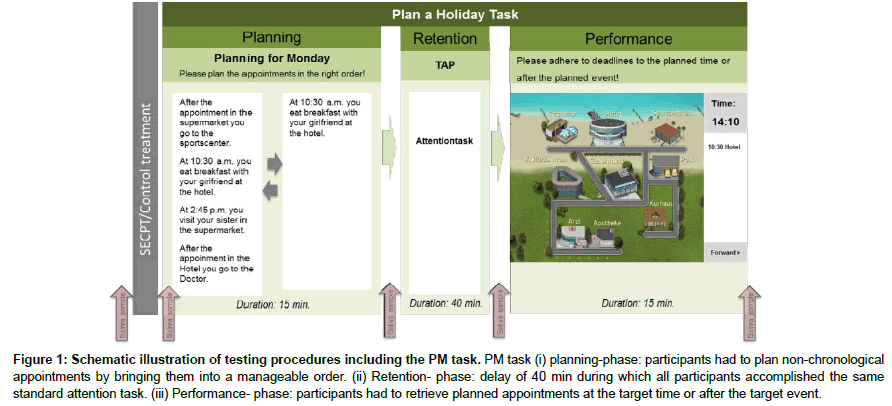
Participants of the study were informed about the aims of the study and the experimental procedures. They gave their first saliva sample (baseline-sample) before the beginning of the planning phase of the complex and real life-related PM paradigm “Plan a Holiday” (Figure 1). The paradigm requires the planning, retention, and retrieval of a fictional holiday week. On each of the 4 holidays, 4 appointments had to be planned, remembered and retrieved, resulting in a total of 16 appointments for the whole holiday. Half of the items require time-dependent, the other half event-dependent PM (for details of the paradigm see Glienke and Piefke, the authors developed the “Plan a Holiday” PM paradigm together with the Hasomed Company, Magdeburg, Germany; www.hasomed.de) [45]. After completion of the planning phase, participants gave their second saliva sample (postplanning). Then, a retention phase of 40 min was implemented. In everyday life, distracting events are likely to occur during the retention phase of PM. To experimentally model and control distractors during the retention phase, all volunteers completed an attention task for 15 min (for detail see [45]). Participants were not allowed to write anything down or use smartphones during the retention phase. At the end of the retention interval, participants gave their third saliva sample (post-retention). Participants then retrieved and executed their appointments at four days of a fictional holiday week. After the retrieval phase, subjects gave their fourth saliva sample (postretrieval). The PM experiment had duration of 1.5 h.
Figure 1: Schematic illustration of testing procedures including the PM task. PM task (i) planning-phase: participants had to plan non-chronological appointments by bringing them into a manageable order. (ii) Retention- phase: delay of 40 min during which all participants accomplished the same standard attention task. (iii) Performance- phase: participants had to retrieve planned appointments at the target time or after the target event.
Neuropsychological assessment
Following the PM experiment cognitive capacities that are assumed to be relevant for PM performance were tested. They contain retrospective memory, working memory and executive functions [26,46-48]. We applied a neuropsychological testing battery including verbal and non-verbal working memory tasks (digit and block span, Wechsler Adult Intelligence Scale- Fifth edition) [49,50], a verbal retrospective long-term memory task (Wechsler Memory Scale IV) [51], and a test of cognitive speed and flexibility (Trail Making Test A and B) [52,53]. To assess intelligence, we applied a short version of the Leistungsprüfsystem [54]. The neuropsychological assessment lasted about 45 min.
Statistical analysis
For the clinical indicator saliva cortisol a Kolmogorov-Smirnov test revealed a normal distribution of data. The stress hormone cortisol measures were analysed by a univariate analysis of variance (ANOVA) with the factor GROUP (PTSD group vs. MC group vs. non-MC group). Between-group comparisons of cortisol were conducted by using two- sample t-tests. A Kolmogorov-Smirnov test for the results of the BDI and PM indicated that data was not normally distributed. In consequence, for the following analysis of the BDI and PM data we applied non-parametric test statistics. To identify differences in the BDI scores between the groups, we employed Kruskal–Wallis one-way ANOVA with the three levels between subject factors GROUP (PTSD group vs. MC group vs. non- MC group). For further analysis, the Mann-Whitney U- tests were applied. To compare PM between the PTSD group, MC group and the non-MC group we consulted a Kruskal–Wallis one-way ANOVA with the three level between subject factor GROUP (see above) and the dependent variables OVERALL PM (defined as the number of correctly planned and remembered appointments), EVENT- and TIME- DEPENDENT PM (defined as the number of correctly planned and remembered time- or event- dependent appointments). For post-hoc between group comparisons Mann-Whitney U- tests were utilized. This analysis was consulted equivalently for the data of the planning phase and the performance phase of PM. We further calculated correlations between all PM modalities during planningand performance phase and cortisol, the PDS and duration of disease.
With respect to normal distribution of data, for neuropsychological assessment, parametric univariate ANOVAs were utilized to compare the performance in neuropsychological tests between the different GROUPS (i.e. PTSD group vs. MC group vs. non-MC group). Multiple post-hoc t-tests were consulted if necessary.
The threshold for significance was set to p<.05 in all statistical calculations. The Bonferroni approach was applied to correct for multiple comparisons in post-hoc t-tests.
Results
Clinical measures
Depression: Results show a significant difference between the total BDI II score of the PTSD group, the MC group and the non-MC group (H(2)= 27. 61; p<.001). The total BDI II scores of the soldiers with PTSD are significantly higher compared to those soldiers of the MC group (U=-4.17; p<.001) and the non-MC group (U=-5.02; p<.001). No difference was found between the MC group and the non-MC control group (U=-2.85; p=.80). Based on the test manual of the BDI II, both control groups do not show depressive symptoms, whereas the combat soldiers with PTSD show a severe depressive symptomatology.
Cortisol levels: An univariate ANOVA revealed significant differences in saliva cortisol levels between the three groups (F(2,43)= 6.67; p<.01). Post-hoc t-test analysis showed significantly lower total cortisol levels in combat soldiers with PTSD compared to MC group (t(23)=3.98; p<.01) and the non-MC group (t(32)=-2.76; p<.05). Between the MC group and the non-MC control participants no differences in saliva cortisol levels were detected (t(31)=1.24; p=.52) (Table 1).
| PTSD Group | MC group | Non-MC group | ||||
|---|---|---|---|---|---|---|
| M | ± SD | M | ± SD | M | ± SD | |
| BDI II total score | 27,83 | 9,54 | 2,13 | 3,13 | 1,95 | 2,92 |
| Cortisol total (nmol/l) | 2,43 | ,97 | 4,80 | 1,89 | 3,93 | 1,84 |
Table 1: Descriptive data of clinical indicators.
PM - Planning phase
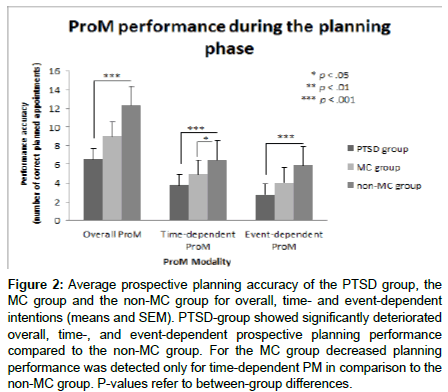
Figure 2 shows the number of correctly planned appointments for overall PM, and separately for time- and event-dependent PM for the PTSD group, the MC group and the non-MC participants. On average, PTSD group correctly planned M=6.54 (SD= ± 4.31) appointments. MC group correctly planned M= 9.00 (SD= ± 4.02), and non–MC participants M=12.33 (SD= ± 2.65) appointments. A Kruskal–Wallis one-way ANOVA showed a significant main effect of GROUP (H(2)=14.59; p<.01). Post-hoc Mann-Whitney U- tests indicated significantly deteriorated overall planning performance in PTSD group compared to non-MC group (U=-3.57; p<.001). Such differences were neither found between PTSD group and MC group (U=-1.34; p>.05), nor between MC group and non-MC group (U=- 2.41; p>.05).
Figure 2: Average prospective planning accuracy of the PTSD group, the MC group and the non-MC group for overall, time- and event-dependent intentions (means and SEM). PTSD-group showed significantly deteriorated overall, time-, and event-dependent prospective planning performance compared to the non-MC group. For the MC group decreased planning performance was detected only for time-dependent PM in comparison to the non-MC group. P-values refer to between-group differences.
Time-dependent prospective planning
As displayed in Figure 2, the average number of correctly planned time-dependent appointments was M= 3.77 (SD= ± 2.31) for PTSD group, M= 4.91 (SD= ± 1.88) for MC group, and M= 6.48 (SD= ± 1.44) for non-MC group. A Kruskal–Wallis one-way ANOVA revealed a significant main effect of GROUP (H(2)=13.89; p< .01). Post-hoc Mann-Whitney U- tests showed that the PTSD group performed significantly worse than participants of the non- MC group (U=-3.41; p<.001) and the MC group had poorer performance compared to the non-MC participants (U=-2.49; p<.05). No differences were found between PTDS group and MC group (U=-1.32; p>.05).
Event-dependent prospective planning
The average number of correctly planned event-dependent appointments was M=2.77 (SD= ± 2.13) for PTSD group, M=4.08 (SD= ± 2.23) for MC group and M= 5.90 (SD= ± 1.30) for non-MC group (Figure 2). A Kruskal–Wallis one-way ANOVA revealed a significant main effect of GROUP (H(2)= 15.58; p<.001). Post-hoc t-tests showed a significant poorer event-dependent prospective planning performance for PTSD patients compared to non-MC group (U=-3.81; p<.001). No differences were found between PTSD and MC group (U=-1.37; p>.05), and between MC group and non- MC group (U=-2.15; p>.05).
PM Retrieval
Overall PM
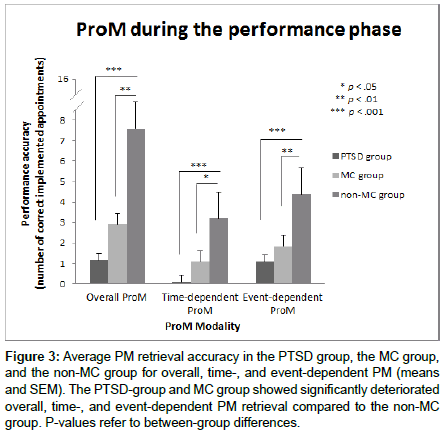
Figure 3 illustrates the average number of correctly remembered appointments for overall PM and separately for time- and eventdependent PM for each of the three groups (PTSD group, MC group, non-MC group). Participants with PTSD on average remembered M= 1.15 (SD= ± 1.14) appointments, participants of the MC group M= 2.92 (SD= ± 2.75) appointments, and participants of the non- MC group remembered M= 7.57 (SD= ± 4.01) appointments across the whole 4 days. A Kruskal–Wallis one-way ANOVA showed a significant main effect of GROUP (H(2)=21.06; p<.001). Post-hoc Mann-Whitney U- tests indicated significantly poorer overall PM for soldiers with PTSD compared to non-MC group (U=-4.17; p<.001), and for MC group compared to non-MC group (U=-3.09; p<.01). No difference was found between the MC group and the PTSD group (U=-1.53; p>.05).
Figure 3: Average PM retrieval accuracy in the PTSD group, the MC group, and the non-MC group for overall, time-, and event-dependent PM (means and SEM). The PTSD-group and MC group showed significantly deteriorated overall, time-, and event-dependent PM retrieval compared to the non-MC group. P-values refer to between-group differences.
Time-dependent PM
PTSD group remembered M= .08 (SD= ± .28), the MC group remembered M= 1.08 (SD= ± 1.68), and non-MC group remembered M= 3.19 (SD= ± 2.48) time-dependent appointments across the whole 4 days (Figure 3). A Kruskal–Wallis one-way ANOVA showed a significant main effect of GROUP (H(2)= 20,39; p<.001). Posthoc Mann-Whitney U- tests indicated significantly deteriorated time-dependent PM for the PTSD group compared to the non-MC group (U=-4.22; p<.001), and for MC group compared to non-MC participants (U=-2.61; p<.05). No difference in time-dependent PM was found between the PTSD group and the MC group (U=-2.04; p>.05).
Event-dependent PM
Soldiers with PTSD remembered M=1.08 (SD= ± 1.12), the MC group remembered M= 1.83 (SD= ± 1.53), and the non-MC group remembered M=4.38 (SD= ± 1.96) event-dependent appointments across the whole 4 days (Figure 3). A Kruskal–Wallis one-way ANOVA revealed a significant main effect for GROUP (H(2)=20.56; p<.001). Post-hoc Mann-Whitney U-tests showed that the PTSD group (U=-4.02; p<.001) and the MC group (U=-3.27; p<.01) had significantly poorer event-based PM compared to non-MC group. No difference in event-dependent PM was found between the PTSD group and the MC group (U=-1.35; p>.05).
Correlations in the PTSD group
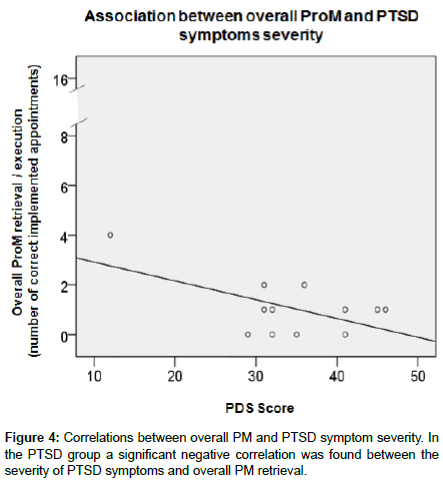
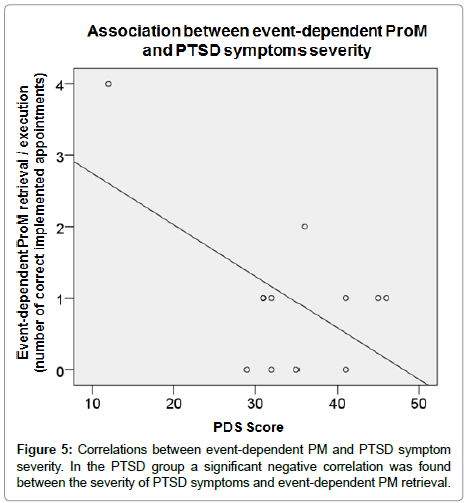
To assess putative associations between PM and clinical indicators (PDS, saliva cortisol or the BDI for PTSD patients, Spearman correlations) were calculated for the PTSD group. Significant negative correlations were found between the PDS score and overall PM retrieval (r=-.59; p<.05; Figure 4). Additionally, a significant negative correlation was found between the PDS score and event-dependent PM retrieval accuracy (r=-.58; p<.05; Figure 5). Significant correlations were found neither between cortisol and PM, nor between BDI scores and PM.
Neuropsychological assessment
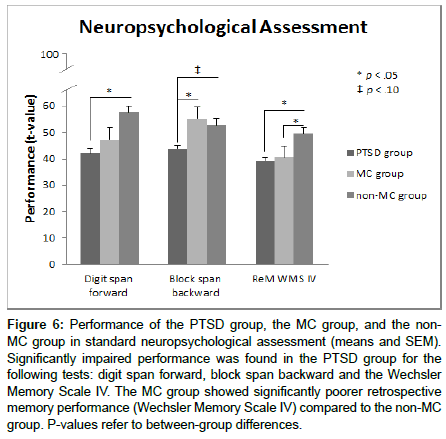
The parametric univariate ANOVAs for the neuropsychological assessment data resulted in significant main effects for GROUP for the digit span forward (F(2,40)=4.22; p<.05), the block span backward (F(2,41)=6.42; p<.01), and long-term memory of the Wechsler Memory Scale IV (F(2,43)=4.29; p<.05).
As depicted in Figure 6 a post-hoc independent t-test for digit span forward revealed a significantly poorer performance in the PTSD group compared to the non-MC group (t(30)=-2.94; p<.05). No differences were found between the PTSD group and the MC group (t(20)=-.75; p>.05), and between the MC group and the non- MC group (t(30)=-1.77; p>.05).
Figure 6: Performance of the PTSD group, the MC group, and the non- MC group in standard neuropsychological assessment (means and SEM). Significantly impaired performance was found in the PTSD group for the following tests: digit span forward, block span backward and the Wechsler Memory Scale IV. The MC group showed significantly poorer retrospective memory performance (Wechsler Memory Scale IV) compared to the non-MC group. P-values refer to between-group differences.
For the block span backwards test, independent post-hoc t-tests showed differences between the PTSD group and the MC group (t(20)=3.01; p<.05) (Figure 6). This difference indicated a significantly poorer performance for the PTSD group compared to MC group. Additionally, a trend towards a significant deteriorated performance was found for the PTSD group compared the non-MC group (t(31)=- 2.18; p=.07). No difference was detected between the MC group and the non-MC group (t(29)=.89; p>.05).
Figure 6 also displays the long-term RM of the WMS. For long term memory independent post-hoc t-tests yielded a significant difference between the PTSD group and the non-MC group (t(31)=- 3.16; p<.05), indicating poorer long term memory for soldiers with PTSD compared to the non-MC group. No differences were detected between the PTSD group and the MC group (t(22)=-.40; p>.05). However, retrospective memory of the MC group was significantly poorer compared to the non-MC group (t(31)=-2.63; p<.05).
Discussion
The aim of the study was to investigate (i) performance of combat soldiers with PTSD, compared to MC group, and non-MC group for the different phases of complex real life-related PM, (ii) the putative relevance of salivary cortisol levels for PM performance and (iii) the relationship between measures of PM and a standard neuropsychological assessment of different memory functions. We found that the PTSD group showed significantly poorer overall, time and event-dependent PM planning and retrieval compared to the non- MC group, but not the MC group. Except, time-dependent planning of the MC group was significantly inferior to the performance of the non-MC-group PM retrieval was also significantly declined in the MC group compared to the non-MC group. Moreover, in the PTSD group negative correlations were detected between PTSD symptom severity and overall as well as event-dependent PM retrieval. In salivary cortisol levels we found significantly decreased cortisol levels solely for the PTSD group compared to the MC group and the non-MC group. Neuropsychological assessment showed significantly poorer digit span (forward) and retrospective memory (logical memory) in the PTSD group compared to the non-MC group and significantly decreased performance for block span (backwards) compared to the MC group. Additionally, the MC group showed deteriorating performance in retrospective memory (logical memory) compared to the non-MC group. Our data demonstrates for the first time a deterioration of PM retrieval in combat soldiers with and without PTSD. Considering, that only the PTSD group had decreased cortisol levels, these results suggest that lowered cortisol levels may not be the exclusive factor for the induction of declined PM in combat soldiers. Rather, further neurobiological mechanisms associated with the exposure to combat-related stress need to be considered, which have a deteriorating effect on PM retrieval in combat soldiers who do not meet the criteria for a PTSD diagnosis.
The influence of PTSD on PM performance
It is well known from previous research, that RM is impaired in both combat soldiers and other patients with PTSD [10-13,55]. Although published data on the influence of PTSD on PM are rare, recent studies suggest that PM may be severely affected by the disorder. McFarland et al. demonstrated that event-dependent PM is preserved in combat soldiers with PTSD, and that PTSD symptom severity may even be negatively correlated with PM [36]. Other authors report that time-dependent PM declined in combat soldiers with PTSD compared to combat soldiers without PTSD [37]. As mentioned above, the current attempt extends these findings by investigating the impact of PTSD on the different phases of PM in a complex real life-related PM and including an additional control group that is free of combat or another trauma (non-MC group). While ongoing PM paradigms usually contain all phases of PM at once, the current task allows a more precise insight into the impact of PTSD on the different processing phases of PM. Moreover, to control for confounding factors it is of particular relevance to include a nontraumatized control group that was not exposed to combat because there are indices that combat-related stress already affects cognitive functions (i.e. Lieberman et al., 2005) [56].
As reported, the PTSD group showed significantly decreased overall, time- and event-based planning performance in the planning phase of PM in comparison to the non-MC group. Planning performance aside from PM is commonly assigned to the executive functions and often assessed with the Tower of London Task [57,58]. When measuring planning by the Tower of London Task, many studies report that planning behavior is mostly spared in PTSD [15,59]. However, the cognitive strategies used for the planning within the complex real-life related PM task of the current study may go beyond the simple planning, measured in PTSD before. The planning phase of PM in this study could more likely be interpreted as an encoding process known from RM, although the contents (i.e. future intentions) differ from the contents of RM encoding (e.g. current experiences, events, emotions). From RM research it is known that the memory encoding is frequently impaired in PTSD patients [60]. Although, PM intentions do not necessarily refer to RM contents, the encoding of future intentions may be comparably affected by PTSD, as it was found for RM memory encoding. Since cognitive strategies used for the planning of event- and time-dependent appointments are presumably comparable, it may explain why both types of PM were affected by PTSD in the current study.
In time-dependent planning and in the retrieval phase of PM, both the PTSD group and the MC group showed significantly poorer PM compared to the non-MC group, while there were no significant differences between the PTSD and the MC group. This result is unexpected, because only the PTSD group showed significantly decreased cortisol levels and higher BDI II scores compared to both control groups (see above). Given that both military groups were exposed to combat, it could be reasonable to assume that the MC group also suffered from combat-related stress, but did not develop typical PTSD symptoms in response to stress exposure. Nevertheless, the combat experience may have negatively influenced their cognitive capability, which leads to decreased performance in PM. Normal cortisol levels in the MC group suggest that stress-related neurobiological alterations, which may lead to cognitive decline, do not necessarily affect cortisol metabolism. Metabolism of other neurotransmitters – in particular, norepinephrine and serotonin - may be involved in these neurobiological processes [61-63]. On the behavioral level, a study of Lieberman et al. [56] is in line with these ideas. The authors simulated combat-related stress in their study and found a degradation of cognitive performance in response to the stress induction procedure. To generate high levels of stress comparable to military exercise, the authors asked U.S. army officers to attend to an intense training exercise conducted for 53 hours in the heat. The participants’ cortisol levels were higher before the training but did not differ between participants. With respect to the hypothesis that the impact of combat stress without PTSD can also have long-lasting effects on cognition a study investigated the mesofrontal circuit before and after deployment in Afghanistan in soldiers without PTSD [64]. A functional magnetic resonance imaging was recorded while soldiers completed a sustained attention task right before their first deployment, 1.5 months after their first deployment (short-term follow up) and about 1.6 years after their first deployment (long-term follow up). No stress symptoms, no significant differences in PTSD, state anxiety or positive and negative affect scores before and after combat exposure were observed. However, they found that combatrelated stress leads to a persistent reduction in functional connectivity between the midbrain and the prefrontal cortex. Given these findings of Van Wingen et al. [64], it is important to mention that especially the prefrontal and frontal areas (mid-ventrolateral prefrontal regions, anterior cingulate cortex) were reported to be relevant for PM [65]. Neurofunctional alterations of connectivity in frontal brain regions may thus play a key role for the degradation of PM planning and retrieval in the MC group in the present study. However, since we did not assess neural activation within this study, future research is needed to clarify this assumption.
Besides the impairment in the MC group, we also found impairment in all domains of PM retrieval for the PTSD group. This finding contradicts the data reported from previous studies. For instance, in a previous study it was found that only time-dependent PM is impaired in soldiers with PTSD compared to those without PTSD [37]. The authors discussed their results with respect to the issue of task complexity. They determined that time-dependent items were more demanding in terms of a higher cognitive load, than eventdependent items of the MIST used in their study. In consequence, combat soldiers with PTSD had greater difficulties with accurate PM retrieval when tasks require greater demands. Some theoretical models (i.e. the dynamic multiprocess framework) propose that demanding PM tasks absorb many cognitive resources and may therefore lead to more spontaneous retrieval processes [65-67]. According to the argumentation of Scott et al. [37], it may be also reasonable that the impairment of the PTSD group in the retrieval phase found in the current study was initiated by diminished automatic processes like spontaneous retrieval in response to PTSD.
Another study reported a negative correlation between symptom severity of combat soldiers with PTSD and the accuracy of eventdependent PM [36]. Since the authors did not include combat soldiers without PTSD as a control group or time-dependent PM in their study, the data can neither be directly compared to the data of Scott et al. [37], nor to our results. Nevertheless, in our study, we also found negative correlations between PTSD symptom severity and overall and event-dependent PM retrieval in the PTSD group. Moreover, our data show for the first time, that event-dependent PM is significantly diminished in soldiers with PTSD compared to MC and non-MC group. This finding leads to the assumption that eventdependent PM is specifically vulnerable to PTSD symptoms. Given that event-dependent PM is supposed to be externally prompted and thus less demanding than time-dependent PM, the underlying cognitive processes more likely refer to strategic monitoring [65-67]. With respect to the current knowledge of the cognitive decline that may be related to PTSD it is conceivable that cognitive resources in general are limited in combat soldiers with PTSD. This could have led to a shift from high demanding towards low demanding processing strategies during PM retrieval in the PTSD group [68]. This in turn resulted in a higher vulnerability towards impairment, based on a potential higher fragility of low demanding processing strategies like spontaneous retrieval.
Besides, the current study applied a PM task with a relatively high external validity and complexity for time- and event- dependent PM. With respect to the higher task demand for both time and eventdependent PM it is reasonable that both PM modalities might have an increased vulnerability for the physiological and psychological alterations in PTSD.
For memory retrieval prior research revealed similar neural processes underling prospective and retrospective memory [69]. In the current study PTSD group showed significantly poorer retrospective memory compared to non-MC group also. Certainly, memory retrieval and the execution of intentions in prospective memory proceeds self-initiated and are therefore beyond a simple storing, known from retrospective memory [70]. However, PTSD may have comparable impact on PM as it has on RM [10-13,55].
Relevance of the biological stress marker cortisol
Significantly decreased saliva cortisol of the PTSD group compared to the MC and non-MC groups indicates a dysfunctional HPA-axis reaction associated with the development of a PTSD in response to military trauma. This finding is in line with prior research, which also described lowered cortisol levels in PTSD patients [20,71,72]. However, there is a substantial overlap between the symptoms of PTSD and those of depression, which is corroborated by higher BDI scores in the PTSD group of the current study. Available data demonstrate inconsistent alterations of the HPA axis in patients with major depression (MD). For example, MD patients show a broader range and a more arbitrary cortisol production after the administration of dexamethasone and not necessarily a greater production of cortisol [20,73]. Therefore, it is conceivable that the lowered cortisol levels of the current study support the existence of a PTSD diagnosis and the depressive symptoms reported in the BDI by the PTSD group merge in the PTSD symptoms. So far, little is known about the direct impact of significantly lowered cortisol levels on PM. For RM in healthy participants it was described that lowered cortisol levels improve memory whereas increased impair memory [74,75]. In a study with healthy adults the impact of stress on timebased PM with and without a relaxation phase directly before the beginning of the PM task was examined [76]. They did not assess cortisol, but blood pressure which is also understood as a stress indicator and indirectly linked to cortisol. The authors found that the relaxation phase did not influence PM although blood pressure measures declined under relaxation [76]. However, so far it remains unclear how lowered cortisol levels directly affect PM. Nevertheless, lowered cortisol reaction of the PTSD group may have an indirect impact on the impairment in all domains of PM in the planning and retrieval phase. Via the so called negative feedback loop cortisol is needed to downregulate the production of epinephrine (E) and norepinephrine (NE) towards the initial homeostatic state [77-79]. Due to the low basal cortisol levels associated with the disorder, the negative feedback loop is interrupted in patients with PTSD. This in turn leads to increased levels of E and NE, which are known to induce hyperarousal symptoms in soldiers with PTSD [72]. It has been shown for the psychophysiology of PTSD that based on a dysfunctional HPA axis high levels of NE are released, which in turn impair prefrontal functions and lead to dendritic atrophy in the prefrontal cortex (PFC) [21,61,62,72,80]. According to the literature the PFC is especially relevant for the planning and retrieval of future actions [26,29,65]. Dendritic atrophy in the PFC caused by a decreased downregulation of NE in response to the chronically lowered cortisol levels in PTSD may thus at least in part explain the declined PM performance of the PTSD across both the planning and the retrieval phase [61,62].
Interestingly, decreased cortisol levels were found only for the PTSD group, but the MC group also showed impaired PM retrieval, but not planning (relative to the non-MC group). Given these data, it is reasonable to assume that cortisol is of particular relevance for PM planning rather than retrieval. We propose that PM retrieval may rely on neurobiological processes, in which cortisol is not a key player and that the exposure to combat-related stress may lead to alterations of these neurobiological bases, resulting in declined PM retrieval. As reported above, combat-related stress may induce both reversible and persistent alterations of brain structures associated with PM [56,64]. For instance, Van Wingen et al. [64] found neither general stress symptoms, nor PTSD symptoms in combat soldiers after their combat deployment. Nonetheless, the authors reported persistent morphological alterations of brain structures and cognitive changes in response to combat. It has been suggested that intense acute stress reactions during combat may cause persisting cognitive alterations beyond those that are related to chronic changes in cortisol metabolism. Interestingly, alterations in the plasma neuropeptide Y (NPY), which also acts as a neurotransmitter in the brain, was found to be a neurobiological marker of trauma exposure [81]. NPY is supposed to act as protective stress factor and is significantly and positively associated with the stress-related release of cortisol [82,83]. Some authors found reduced baseline plasma NPY levels in combat soldiers compared to combat free participants [81], while others detected higher NPY plasma levels in exposed soldiers without PTSD than in non-exposed but comparable levels in soldiers with current PTSD [84]. Although, prior research reveals a diverging pattern of results, all studies reported alterations in the NPY for combat exposed soldiers with and without PTSD, but not in non-exposed participants. Our findings of normal cortisol levels and declined PM retrieval in the MC group may thus result at least in part from combat traumarelated alterations of NPY metabolism. However, when considering the close relationship between cortisol and NPY metabolism, one can also suggest that impaired PM in the MC group found in the current study may not only be related to NPY, but also to other neuropeptides and neurohormones. Since, we only measured salivary cortisol levels in the present study, but not other putative neurobiological markers of stress, this suggestion remains speculative based on our data. Thus, future research is needed to investigate in detail the combatrelated neurobiological acute stress reactions and their impact on PM retrieval and other cognitive functions that go beyond influences of chronic changes in cortisol levels.
Relationship between neuropsychological assessment and PM
Neuropsychological assessment detected significant poorer digit span (forward) and retrospective memory (logical memory) in the PTSD group. Digit and block span are both measures for working memory. Moreover, working memory is known to be involved in PM [85,86]. This view is corroborated by our results of standard neuropsychological tests of working memory. It was to be expected, that when PTSD affects PM, it should also affect working memory. Additionally, prior research also reports abnormalities in working memory and its underlying neural functionality in patients with PTSD [87]. Since the current study applied a cognitively high demanding PM task while the working memory tasks were less demanding the lacking correlations might be a result of divergent work load. Future research with tasks more comparable in cognitive load would be needed to clarify more precisely any relationship between complex PM and working memory.
Previous research has extensively shown that that RM is impaired in both combat soldiers and other patients with PTSD [10-14]. Thus, the impairment in RM of the PTSD group in the current study is not a surprise. Interestingly RM was also negatively affected in both PTSD group and MC group, as it was similarly found for PM. This could again underline the assumption, that memory processes may be sensible for general combat stress.
Limitations of this study should be acknowledged and include the relatively broad range in the IQ across the groups. There is evidence that intelligence may interact with PM. However, we considered an IQ range between one standard deviation [88]. Two participants happened to be slightly under the cut off but did not differ in their general cognitive and PM performance compared to the rest of their group. Another limitation is that our paradigm not yet directly assesses underlying processing strategies of PM (i.e. spontaneous retrieval, strategic monitoring). Future research is needed to directly investigate makers of those cognitive strategies within the retrieval phase in complex prospective memory. Additionally, the number of participants in the PTSD group and the MC group was quite small. Despite the small sample size, the statistical effects of the current study were very strong.
Conclusion
Despite these limitations, the current study provides initial evidence of diminished time- and event-dependent prospective planning and PM retrieval for soldiers with PTSD in a real-life-related PM task. Data further suggest that already the exposure to combat related stress may have a deteriorating effect on real-life-related PM in the absence of PTSD. Moreover, our results indicate that lowered cortisol concentration in PTSD may lead to deterioration of performance particularly in the planning phase of PM, but does not exclusively justify impairments during retrieval. Future research is needed to investigate in detail the combat-related neurobiological acute stress reactions and their impact on PM retrieval and other cognitive functions that go beyond influences of chronic changes in cortisol levels.
Acknowledgements
We are grateful to the soldiers with and without PTSD who participated in the study, the staff of the Bundeswehrkrankenhaus Berlin, and our student co-worker Tasja Lutz who was involved in data collection.
References
- American Psychiatric Association (2013) Diagnostic and statistical manual of mental disorders: DSM-5. (5th edtn.), Washington DC: American Psychiatric Publishing, USA.
- Chivers-Wilson KA (2006) Sexual assault and posttraumatic stress disorder: a review of the biological, psychological and sociological factors and treatments. Mcgill J. Med 9:111–118.
- Heim C, DJ Newport, S Heit, YP Graham, M Wilcox, et al. (2000) Pituitary-Adrenal and Autonomic Responses to Stress in Women After Sexual. JAMA 284:592-597.
- Y Neria, A Nandi, S Galea (2008) Post-traumatic stress disorder following disasters: a systematic review. Psychol. Med 38:467–480.
- LK Richardson, BC Frueh, R Acierno (2010) Prevalence Estimates of Combat-Related Post-Traumatic Stress Disorder: Critical Review. Aust. N Z J Psychiatry 44: 4–19.
- HU Wittchen, S Schönfeld, C Kirschbaum, C Thurau, S Trautmann, et al. (2012) Traumatic Experiences and Posttraumatic Stress Disorder in Soldiers Following Deployment Abroad. Dtsch. Ärtztebl Int 109: 559–568.
- S Trautmann, S Schönfeld, M Höfler, A Heinrich, R Hauffa, et al. (2013) Posttraumtic stress disorder after deployment of German soldiers : does the risk increase with deployment duration. Bundesgesundheitsblatt Gesundheitsforsch. Gesundheitsschutz 56: 930–940.
- S Trautmann, L Goodwin, M Höfler, F Jacobi, J Strehle, et al. (2017) Prevalence and severity of mental disorders in military personnel: a standardised comparison with civilians. Epidemiol. Psychiatr. Sci 26: 199–208.
- S Trautmann, S Schönfeld, A Heinrich, J Schäfer, P Zimmermann, et al. (2015) Risk factors for common mental disorders in the context of military deployment: A longitudinal study. Eur. Psychiatry 30: 303.
- S Schuitevoerder, JW Rosen, EW Twamley, C R Ayers, H Sones, et al. (2013) A meta-analysis of cognitive functioning in older adults with PTSD. J. Anxiety Disord 27: 550–558.
- TC Buckley, EB Blanchard, WT Neill (2000) Information Processing and PTSD: A Review Of The Empirical Literature. Clin. Psychol. Rev 28: 1041–1065.
- Wingenfeld K, OT Wolf (2015) Effects of cortisol on cognition in major depressive disorder, posttraumatic stress disorder and borderline personality disorder - 2014 Curt Richter Award Winner. Psychoneuro- endocrinology 51: 282–295.
- Piefke M, Pestinger M, Arin T, Kohl B, Kastrau F, et al. (2008) The Neurofunctional Mechanisms of Traumatic and Non-Traumatic Memory in Patients with Acute PTSD Following Accident Trauma. Neurocase 13: 342–357.
- Driessen M, Beblo T, Mertens M, Piefke M, Rullkoetter N, et al. (2004) Posttraumatic stress disorder and fMRI activation patterns of traumatic memory in patients with borderline personality disorder. Biol. Psychiatry 55: 603–611.
- G Lagarde, J Doyon, A Brunet, (2010) Memory and executive dysfunctions associated with acute posttraumatic stress disorder. Psychiatry Res 177: 144–149.
- JJ Vasterling, LM Duke, K Brailey, JI Constans, AN Allain, et al. (2002) Attention, learning, and memory performances and intellectual resources in Vietnam veterans: PTSD and no disorder comparisons. Neuropsychology 16: 5–14.
- AM Hull (2002) Neuroimaging findings in post-traumatic stress disorder : Systematic review Neuroimaging findings in post-traumatic stress disorder Systematic review. Br. J. Psychiatry 181: 102–110.
- SL Rauch, LM Shin, EA Phelps (2006) Neurocircuitry Models of Posttraumatic Stress Disorder and Extinction: Human Neuroimaging Research-Past, Present, and Future. Biol. Psychiatry 60: 376–382.
- BM Elzinga (2002) Are the neural substrates of memory the final common pathway in posttraumatic stress disorder ( PTSD ). J. Affect. Disord 70:1–17.
- R Yehuda, MH Teicher, RL Trestman, RA Levengood, LJ Siever (1996) Cortisol Regulation in Posttraumatic Stress Disorder and Major Depression : A Chronobiological Analysis. Biol. Psychiatry 40: 79–88.
- R Yehuda, EL Giller, SM Southwick, MT Lowy, JW Mason (1991) Hypothalamic-Pituitary-Adrenal Dysfunction in Posttraumatic Stress Disorder. Biol. Psychiatry 30: 1031–1048.
- JA Ellis (1996) Prospective Memory or the realization of delayed intentions: A conceptual framework for research. Lawrence Erlbaum Associates Publishers, 1996: 1–22.
- M Kliegel, MA McDaniel, GO Einstein (2012) Prospective Memory Cognitive, Neuroscience, Developmental, and Applied Perspectives. New York: Psychology Press.
- M Kliegel, M Martin, M Mcdaniel, GO Einstein (2001) Varying the importance of a prospective memory task: Differential effects across time - and event-based prospective memory. Memory 9:1–11.
- G Cona, G Arcara, V Tarantino, PS Bisiacchi (2012) Electrophysiological correlates of strategic monitoring in event-based and time-based prospective memory. PLoS One.
- DR Addis, AT Wong, DL Schacter (2007) Remembering the past and imagining the future: common and distinct neural substrates during event construction and elaboration. Neuropsychologia 45: 1363–1377.
- J Okuda, T Fujii, H Ohtake, T Tsukiura, A Yamadori, et al. (2007) Differential involvement of regions of rostral prefrontal cortex (Brodmann area 10) in time- and event-based prospective memory. Int. J. Psychophysiol 64: 233–246.
- HD Cheng, K Wang, C Xi, C Niu, XM Fu (2008) Prefrontal cortex involvement in the event-based prospective memory: Evidence from patients with lesions in the prefrontal cortex. Brain Inj 22:697–704.
- DL Schacter, DR Addis, RL Buckner (2007) Remembering the past to imagine the future: the prospective brain. Nat. Rev. Neurosci 8: 657–661.
- Kleim B, Graham B, Fihosy S, Stott R, Ehlers R (2014) Reduced Specificity in Episodic Future Thinking in Posttraumatic Stress Disorder. Clin Psychol Sci 2: 165–173.
- Brown AD, Root JC, Romano TA, Chang LJ, Bryant RA, et al. (2013) Overgeneralized autobiographical memory and future thinking in combat veterans with posttraumatic stress disorder. J Behav Ther Exp Psychiatry 44: 129–134.
- Brown AD, Addis DR, Romano TA, Marmar CR, Bryant RA, et al. (2014) Episodic and semantic components of autobiographical memories and imagined future events in post-traumatic stress disorder. Memory 22: 595–604.
- Altgassen M, Kretschmer A, Schnitzspahn KM (2016) Future thinking instructions improve prospective memory performance in adolescents. Child Neuropsychol 7049: 1–18.
- Kliegel M, Mcdaniel MA, Einstein GO (2000) Plan formation, retention, and execution in prospective memory: A new approach and age-related effects. Mem Cognit 28: 1041–1049.
- Terrett G, Rose NS, Henry JD, Bailey PE, Altgassen M, et al. (2015) The relationship between prospective memory and episodic future thinking in younger and older adulthood. Q J Exp Psychol (Hove) 218e: 1–34.
- McFarland CP, Clark JB, Lee LO, Grande LJ, Marx BP, et al. (2016) Event-based prospective memory among veterans: The role of posttraumatic stress disorder symptom severity in executing intentions. J Clin Exp Neuropsychol 38: 251–260.
- Scott JS, Woods SP, Wrocklage KM, Schweinsburg BC, Southwick SM, et al. (2016) Prospective Memory in Posttraumatic Stress Disorder. J Int Neuropsychol Soc 22: 724–734.
- Kamat R, Weinborn M, Kellogg EJ, Bucks RS, Velnoweth A, et al. (2014) Construct Validity of the Memory for Intentions Screening Test (MIST) in Healthy Older Adults. Natl Institue Heal 21: 742–753.
- S. Raskin (2009) Memory for Intentions Screening Test: Psychometric Properties and Clinical Evidence. Brain Impair 10: 23–33.
- Wittchen HU, Zaudig M, Fydrich T (1997) Strukturiertes Klinisches Interview für DSM-IV. Göttingen: Hogrefe.
- Foa E (1996) Posttraumatic Diagnostic Scale (PDS) Manual. National Computer Systems, Minneapolis.
- Hautzinger M, Keller F, Kühner C (2006) BDI-II. Beck-Depressions-Inventar Revision - Manual, Frankfurt: Harcourt Test Services.
- Mason JW (1968) A review of psychoendocrine research on the sympathetic-adrenal medullary system. Psychosom Med 30: 631-653.
- Elzinga BM, Roelofs K, Tollenaar MS, Bakvis P, van Pelt J, et al. (2008) Diminished cortisol responses to psychosocial stress associated with lifetime adverse events a study among healthy young subjects. Psychoneuroendocrinology. 33: 227–37.
- Glienke K, Piefke M (2016) Acute social stress before the planning phase improves memory performance in a complex real life-related prospective memory task. Neurobiol Learn Mem 133: 171–181.
- Kidder D, Park D, Hertzog C, Morrell R (1997) Prospective memory and aging: The effects of working memory and prospective memory task load. Aging Neuropsychol. Cogn. 4: 93–112.
- West R, Bowry R, Krompinger J (2006) The effects of working memory demands on the neural correlates of prospective memory. Neuropsychologia 44: 197–207.
- Kliegel M, Ramuschkat G, Martin M (2003) Neuropsychologische Grundlagen komplexer prospektiver Gedächtnisleistung. Zeitschrift für Neuropsychol. 14: 293–301.
- DB Berch, R Krikorian, EM Huha (1998) The Corsi block-tapping task: methodological and theoretical considerations. Brain Cogn 38:317–338.
- N Benson, DM Hulac, JH Kranzler (2010) Independent examination of the Wechsler Adult Intelligence Scale—Fourth Edition (WAIS-IV): What does the WAIS-IV measure? Psychol. Assess 22: 121–130.
- F Petermann, AC Lepach (2009) Wechsler memory scale – (4th edtn.). Pearson Assessments.
- RM Reitan, D Wolfson (2004) The Trail Making Test as an initial screening procedure for neuropsychological impairment in older children. Arch. Clin. Neuropsychol 19: 281–288.
- AM Plohmann (2009) Zahlen-Verbindungs-Test (ZVT). Testrezension. 73–83.
- W Horn (1983) LPS Leistungsprüfsystem, 2nd ed. Göttingen: Hogrefe.
- M Driessen, T Beblo, M Mertens, M Piefke, N Rullkoetter, et al. (2004) Posttraumatic stress disorder and fMRI activation patterns of traumatic memory in patients with borderline personality disorder. Biol. Psychiatry 55: 603–611.
- HR Lieberman, GP Bathalon, CM Falco, FM Kramer, CA Morgan, et al. (2005) Severe decrements in cognition function and mood induced by sleep loss, heat, dehydration, and undernutrition during simulated combat. Biol. Psychiatry 57: 422–429.
- AM Owen (1997) Cognitive planning in humans: Neuropsychological, neuroanatomical and neuropharmacological perspective. Prog. Neurobiol 53: 431–450.
- T Shallice (1982) Specific impairments of planning. Philos. Trans. R. Soc. Lond. B. Biol. Sci 298: 199–209.
- RL Aupperle, AJ Melrose, MB Stein, MP Paulus (2012) Executive Function and PTSD: Disengaging from Trauma. Neuropharmacology 62: 686–694.
- EW Dickie, A Brunet, V Akerib, JL Armony (2008) An fMRI investigation of memory encoding in PTSD: Influence of symptom severity. Neuropsychologia 46: 1522–1531.
- AFT Arnsten (2015) Stress weakens prefrontal networks: molecular insults to higher cognition. Nat. Neurosci 18: 1376–1385.
- AFT Arnsten, MA Raskind, FB Taylor, DF Connor (2015) The effects of stress exposure on prefrontal cortex: Translating basic research into successful treatments for post-traumatic stress disorder. Neurobiol. Stress 1: 89–99.
- MC Buhot, S Martin, L Segu (2000) Role of serotonin in memory impairment. Ann. Med 32: 210–221.
- GA Van Wingen, E Geuze, MWA Caan, T Kozicz, SD Olabarriaga, et al. (2012) Persistent and reversible consequences of combat stress on the mesofrontal circuit and cognition. PNAS 109: 15508–15513.
- G Cona, C Scarpazza, G Sartori, M Moscovitch, PS Bisiacchi (2015) Neural bases of prospective memory: A meta-analysis and the ‘Attention to Delayed Intention’ (AtoDI) model. Neurosci. Biobehav. Rev 52: 21–37.
- MK Scullin, MA McDaniel, JT Shelton (2013) The Dynamic Multiprocess Framework: Evidence from Prospective Memory with Contextual Variability. Cogn. Psychol 67: 55–71.
- MA McDaniel, GO Einstein (2000) Strategic and automatic processes in prospective memory retrieval: A multiprocess framework. Appl. Cogn. Psychol 14: S127–S144.
- M Möschl, M Walser, F Plessow, T Goschke, R Fischer (2017) Acute stress shifts the balance between controlled and automatic processes in prospective memory. Neurobiol. Learn. Mem 144:53-67.
- R West, J Krompinger (2005) Neural correlates of prospective and retrospective memory. Neuropsychologia 43: 418–33.
- PW Burgess, T Shallice (1997) The relationship between prospective and retrospective memory: Neuropsychological evidence. London: Psychology Press 247–274.
- R Yehuda (2006) Advances in understanding neuroendocrine alterations in PTSD and their therapeutic implications. Ann. N. Y. Acad. Sci 1071: 137–166.
- R Yehuda (2002) Post-Traumatic Stress Disorder. N. Engl. J. Med 346:108–114.
- F Peeters, NA Nicolson, J Berkhof (2004) Levels and variability of daily life cortisol secretion in major depression. Psychiatry Res 126: 1–13.
- SJ Lupien, BS McEwen (1997) The acute effects of corticosteroids on cognition: integration of animal and human model studies. Brain Res. Rev 24: 1–27.
- OT Wolf (2009) Stress and memory in humans: twelve years of progress? Brain Res 1293:142–54.
- A Ihle, M Kliegel, A Hering, N Ballhausen, P Lagner, et al. (2014) Adult age differences in prospective memory in the laboratory: are they related to higher stress levels in the elderly. Front. Hum. Neurosci81021.
- ER de Kloet (2004) Hormones and the stressed brain. Ann. N. Y. Acad. Sci 1018:1–15.
- ER de Kloet, M Joëls, F Holsboer (2005) Stress and the brain: from adaptation to disease. Nat. Rev. Neurosci 6: 463–475.
- KJ Thiel, MN Dretsch (2011) The Basic of the Stress Response A Historical Context and Introduction. London: Blackwell Publishing Ltd.
- AFT Arnsten (2009) Stress signalling pathways that impair prefrontal cortex structure and function. Nat. Rev. Neurosci 10:410–422.
- CA Morgan, MA Rasmusson, B Winters, SM Southwick (2003) Trauma exposure rather than PTSD is associated with reduced baseline plasma neuropeptide-Y levels. Biol. Psychiatry 54: 1087–1091.
- CA Morgan, MA Rasmusson, S Wang, G Hoyt, R.L Hauger, et al. (2002) Neuropeptide-Y, cortisol, and subjective distress in humans exposed to acute stress: replication and extension of previous report. Biol. Psychiatry 52:136–142.
- CA Morgan, S Wang, MA Rasmusson, DS Charney (2001) Relationship Among Plasma Cortisol, Catecholamines, Neuropeptide Y, and Human Performance During Exposure to Uncontrollable Stress. Psychosom. Med 63: 412–22.
- R Yehuda, S Brand, RK Yang (2006) Plasma neuropeptide Y concentrations in combat exposed veterans: Relationship to trauma exposure, recovery from PTSD, and coping. Biol. Psychiatry 59: 660–663.
- Y Wang, Wy Cao, Jf Cui, DHK Shum, RCK Chan (2013) The relation between prospective memory and working memory: Evidence from event-related potential data. Psych J 2: 113–121.
- RE Smith, D Persyn, P Butler (2011) Prospective Memory, Personality, and Working Memory: A Formal Modeling Approach Rebekah. Z Psychol 219:108–116.
- C Galletly, CR Clark, MC McFarlane, DL Weber (2001) Working memory in posttraumatic stress disorder—an event-related potential study. J. Trauma. Stress 14: 295–309.
- J Cockburn, PT Smith (1991) The Relative Influence of Intelligence and Age on Everyday Memory. J. Gerontol 46: P31–P36.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi