Expert Review, J Virol Antivir Res Vol: 10 Issue: 2
Coronaviruses: A Mini Review on the Emerging Pathogens
Arsalan Rasheed1,2* and Tahir Usman3
1Department of Zoology, Faculty of Chemical and Life Sciences, Abdul Wali Khan University Mardan, Pakistan
2Department of SST Bio-Chemistry, KPESED, Pakistan
3Department of Veterinary Sciences and Animal Husbandry, Abdul Wali Khan University, Pakistan
- Corresponding Author:
- Arsalan Rasheed
Department of Zoology
Abdul Wali Khan University Mardan 23200, Pakistan
E-mail: arsalanrrasheed@gmail.com
Received: December 05, 2020 Accepted: March 19, 2021 Published: March 26, 2021
Citation: Rasheed A, Usman T (2021) Coronaviruses: A Mini Review on the Emerging Pathogens. J Virol Antivir Res 10:2.
Abstract
Coronaviruses having a single large and enveloped positive-stranded “+RNA” belongs to order Nidovirales, family Coronaviridae and include about 30 species. In order to cause a number of important socioeconomic diseases that result from the cold or flu to more chronic diseases such as MERS, SARS and SARS 2 or COVID-19, coronaviruses have both medical and veterinary significance. As a result, in the past few years, interest in this viral family has intensified. The reservoir host for these coronaviruses was bat, while the intermediate host was SARS COV civet cats, MERS COV dromedary camel, and SARS COV 2 bat or pangolin. Coronaviruses causing human respiratory diseases were first identified in the 1960s, but were not considered to be highly pathogenic to humans until China's SARS outbreak in 2003. As a result of the recent COVID-19 outbreak (which is a new strain caused by SARS COV-2 that was discovered in late December 2019 in the city of Wuhan, China.), coronavirus research is necessary to control its future host adaptation, viral evolution and load, transmissibility, pathogenicity, vaccine strategy development, and animal and human virus antiviral therapies. Vaccination is the most effective way to prevent coronavirus infections, but there is still no confirmed cure or vaccine for the coronavirus. Therefore, only through proper preventive measures to protect the population can coronaviruses be contained. We will analyze the knowledge in this review to understand the biology of coronaviruses
Keywords: Coronavirus; Severe acute respiratory syndrome (SARS); Middle east respiratory syndrome (MERS); SARS COV-2; COVID-19
Keywords
Coronavirus; Severe acute respiratory syndrome (SARS); Middle east respiratory syndrome (MERS); SARS COV-2; COVID-19
Introduction
Research background of coronavirus
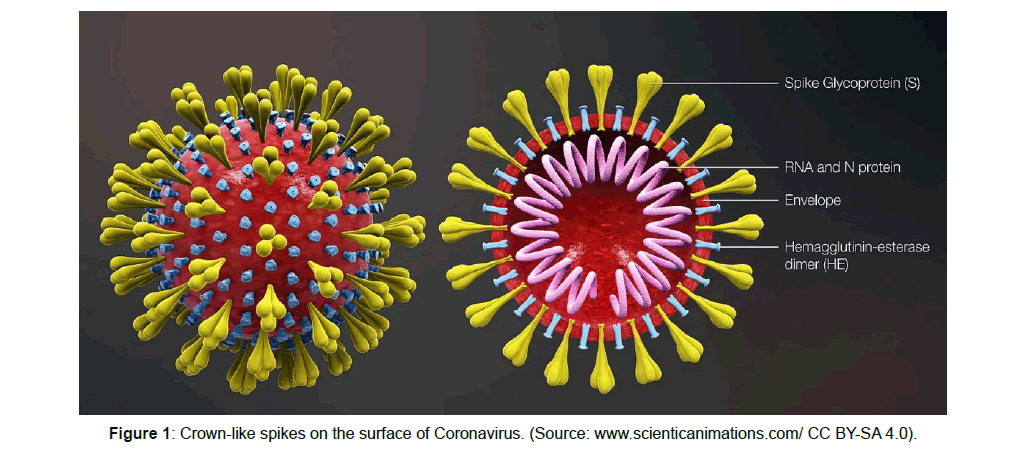
The word “Coronavirus” (CoV) was coined in 1968 for the first time and is derived from the Latin word corona which means “crown” due to the appearance of crown-like spikes on the virus surface (Figure 1). When properly prepared, coronavirus particles appear mediumsized, round, and moderately pleomorphic, and bear characteristic widely-spaced club-shaped surface projections. CoV belong to the Coronaviridae family, a family of enveloped RNA viruses that are distributed widely among mammals and birds. CoVs naturally infect man, chickens, pigs, mice and rats, causing a wide variety of disorders involving a number of different organ systems. The most distinctive feature of this viral family is genome size having the largest genomes among all RNA viruses. The genome have +RNA which can be translated by the host cells in their cytoplasm [1] (Figure 1).
Attachment to host epithelial cells is done by the spike (S) envelope proteins. Fusion occurs at the plasma membrane which is followed by endocytosis [2]. The genome is translated into a polyprotein, which is then posttranslational processed into structural proteins and form viral particles and nonstructural proteins. The Viral particles and nonstructural proteins are used in the replication of genome. Usually COV cause respiratory or enteric diseases but in some cases they cause neurologic illness or hepatitis [3]. CoVs have attracted abundant attention due to variety of highly pathogenic CoVs of livestock and three important human CoVs. This was exemplified by the 2003 outbreak of severe acute respiratory syndrome-coronavirus (SARS COV) in Southeast Asia and the ongoing transmission since 2012 of the Middle East respiratory syndrome coronavirus (MERS COV), which causes 35% mortality among patients seeking medical attention. Both these viruses are closely related to CoVs that are circulating in bats [4, 5] and other potential reservoir species. They may be transmitted to humans either directly or through intermediate hosts, like civet cats for SARS COV and dromedary camels for MERS COV [6]. Although most human coronavirus infections are mild, the epidemics of the three beta CoVs, MERS COV and SARS COV have caused more than 10 000 cumulative cases in the past two decades, with mortality rates of 10% for SARS COV and 37% for MERS COV while the confirmed cases of COVID-19 surpass both of them. As of 7th August, 2020 there have been 18,561,051 confirmed cases of COVID-19 and 701,263 deaths globally reported to WHO.
History of coronavirus
The recognition of CoVs as a separate virus family occurred in the 1960s, in the wake of the discovery of several new human respiratory pathogens [7]. These viruses had a characteristic morphology in negative-stained electron microscopy, marked by a “fringe” of surface structures described as “spikes” or “club-like” projections. Such structures were less densely distributed and differently shaped than those of the myxoviruses. To some, the fringe resembled the solar corona, giving rise to the name that was ultimately assigned to the group. The 1970s and early 1980s was the period in which coronavirus virion proteins and nested-set arrangements of mRNAs were identified and the discontinuous nature of coronavirus transcription was initially demonstrated. The first published sequence of a coronavirus gene appeared in 1983, starting an era in which the whole of the genomes of four CoVs were cloned in pieces and sequenced. This decade has seen the manipulation of these clones and of complementary DNAs (cDNAs) of defectiveinterfering RNAs to study coronavirus RNA replication, transcription, recombination, processing and transport of proteins, virion assembly, identification of cell receptors for CoVs, and processing of the polymerase.
The first human coronaviruses (HCOV) to be recognized as significant respiratory pathogens, HCOV-229E and HCOVVOC43, were identified in the 1960s [8]. Almost four decades later, recognition of the same characteristic virion morphology alerted the world to the emergence of another new human respiratory pathogen the coronavirus responsible for the devastating outbreak of severe acute respiratory syndrome (SARS) in 2002–2003 [9-11]. The SARS COV emerged in the Guangdong province of China in November 2002 and spread to 32 countries, leading to 8096 cases and 774 deaths worldwide by the time the outbreak was brought under control in June 2003 (WHO, 2004). Subsequently, heightened international surveillance for CoVs led to the identification of the strains HCOVNL63, NH, HKU1 in 2004 to 2005, SARS COV in 2003, MERS COV in 2012 [12] and SARS COV-2 in 2019. The sudden appearance of SARS has stimulated a burst of new research to understand the basic replication mechanisms of members of this family in order to their control and prophylaxis.
Taxonomy of coronavirus
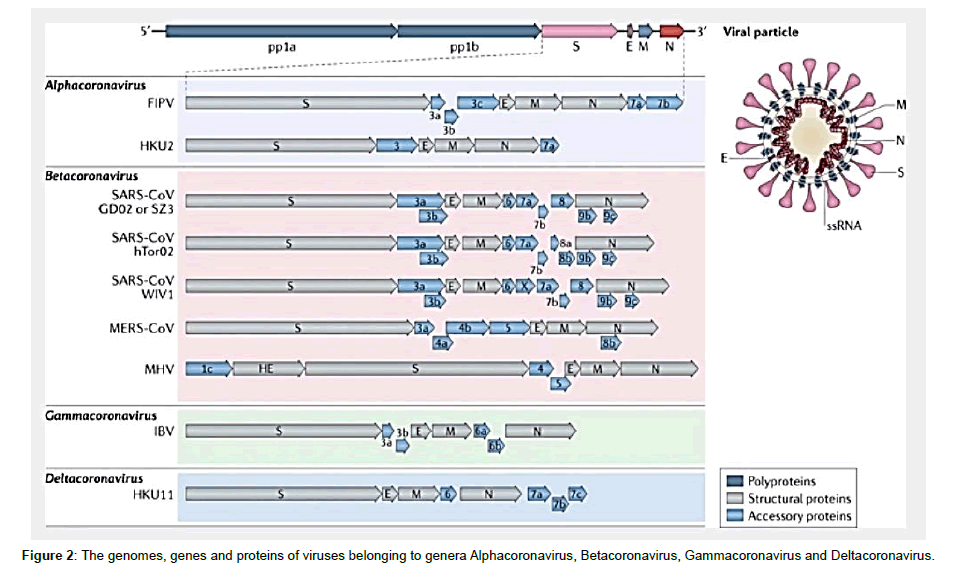
Coronaviruses are members of the family Coronaviridae and the order Nidovirales which was established by the International Committee on the Taxonomy of Viruses (ICTV) in 1975. The Coronaviridae family, along with the Arteviridae and Roniviridae families, form the Nidovirales order. The Arteviridae family includes swine and equine pathogens, and the Roniviridae family is composed of invertebrate viruses [13,14]. In June 2005, the 10th International Nidovirus Symposium in Colorado Springs, Colo proposed that the Coronaviridae family be divided into two subfamilies, the Coronavirinae and the Torovirinae. The latter of which cause enteric diseases in cattle and possibly in humans. COV were initially sorted into these groups based on serology but are now divided by phylogenetic clustering. On the basis of phylogenetic relationships and genomic structures, the subfamily Coronavirinae consists of four genera i.e. Alphacoronavirus (αCoV), Betacoronavirus (βCoV), Gammacoronavirus (γCoV) and Deltacoronavirus (δCoV). The γCoV and δCoV infect birds but some of them can also infect mammals. The αCoV and βCoV infect only mammals [15] and usually cause respiratory illness in humans and gastroenteritis in animals. Human αCoV and βCoV viruses include SARS COV, MERS COV, SARS COV-2 the other four human CoVs (HCOV-NL63, HCOV-229E, HCOV-OC43 and HKU1) while animal Human αCoV and βCoV porcine transmissible gastroenteritis virus, porcine enteric diarrhoea virus (PEDV) and the recently emerged swine acute diarrhoea syndrome coronavirus (SADS COV) (Figure 2).
On the basis of serological cross-reactivity, COV are divided into three groups. This grouping is confirmed by recent genome sequence analysis [16]. Group I CoVs include animal pathogens, such as Transmissible Gastroenteritis Virus (TGEV) of the pig, PEDV, and feline infectious peritonitis virus (FIPV), as well as the human CoVs HCOV-229E and HKU1 which cause respiratory infections. Group II also includes pathogens of veterinary relevance, such as BCOV, porcine hemagglutinating encephalomyelitis virus, and equine coronavirus, as well as human CoVs viruses OC43 and NL63, which, like HCOV-229E, also cause respiratory infections. Group II also includes viruses that infect both mice and rats. SARS COV is listed in group II but there has been controversy about whether it defines a new group of COV or whether it is a distant member of group II [17,18]. Group III thus far includes only avian CoVs such as Infectious Bronchitis Virus (IBV), turkey coronavirus and pheasant coronavirus [19] (Table 1). Recently, using reverse transcription- PCR (RT-PCR), coronavirus sequences were detected in the graylag goose (Anser anser), feral pigeon (Columbia livia) and mallard (Anasplaty rhynchos) [20]. Phylogenetic analyses of the replicase and nucleocapsid (N) sequences suggest that these viruses are members of group III, but as yet they have not been isolated or characterized.
Table 1: Taxonomy of CoV along with their hosts, cellular receptors and pathogenicity.
| Group | Coronavirus | Host | Disease | Cellular receptor |
|---|---|---|---|---|
| I | HCoV-229E | Human | Respiratory infection | Human APN |
| TGEV | Pig | Respiratory and enteric infection | Porccine APN | |
| PRCoV | Pig | Respiratory infection | Porccine APN | |
| CCoV | Dog | Enteric infection | Canine APN | |
| FeCoV | Cat | Enteric infection | Feline APN | |
| FIPV | Cat | Hepatitis, Respiratory, enteric and neurologic infection | Feline APN | |
| HCoV-NL-63 | Human | Respiratory infection and croup | ACE2 | |
| II | HCoV-OC43 | Human | Respiratory and enteric infection | Neu5,9Ac2-containing moiety |
| MHV | Mouse | Hepatitis, enteric and neurologic infection | Murine CEACAM1 | |
| SDAV | Rat | Neurologic infection | - | |
| HEV | Pig | Respiratory, enteric and neurologic infection | Neu5,9Ac2-containing moiety | |
| BCoV | Cow | Enteric infection | Neu5,9Ac2-containing moiety | |
| HCoV-HKU1 | Human | Respiratory infection | - | |
| SARS-CoV | Human | Severe acute respiratory syndrome | ACE2 | |
| MERS-CoV | Human | Middle East respiratory syndrome | DPP4 | |
| SARS-CoV-2 | Human | COVID-19 | ACE2 | |
| III | IBV | Chicken | Hepatitis and respiratory infection | - |
| TCoV | Turkey | Respiratory and enteric infection | - |
*HCoV-229E Human Coronavirus Strain 229E,TGEV: Transmissible Gastroenteritis Virus, PRCoV Porcine respiratory coronavirus, CCoV Canine Coronavirus, FeCoV Feline enteric Coronavirus, FIPV Feline Infectious Peritonitis Virus, PEDV :Porcine Epidemic Diarrhea Virus, HCoV-NL63 :Human Coronavirus Strain NL63, HCoV-OC43 :Human Coronavirus Strain OC43, MHV :Mouse Hepatitis Virus, SDAV :Sialodacryoadenitis Virus,HEV Hemagglutinating encephalomyelitis virus, BCoV Bovine Coronavirus, HCoV-HKU1 Human Coronavirus Strain HKU1, SARS-CoVSevere Acute Respiratory Syndrome Coronavirus,MERS-CoV :Middle East Respiratory Syndrome Coronavirus, IBV :Infectious Bronchitis Virus, TCoVTurkey coronavirus.
Origin of coronavirus
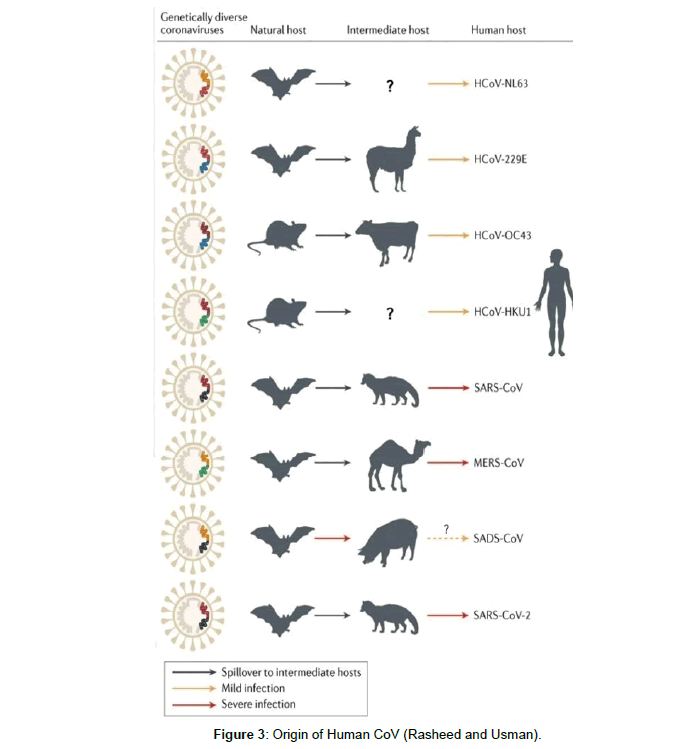
On the basis of current sequence databases, all human CoVs have animal origins. SARS COV, MERS COV, SARS COV-2, HCOV-NL63 and HCOV-229E are considered to have originated in bats while HCOV-OC43 and HKU1 likely originated from rodents. Domestic animals may have important roles as intermediate hosts that enable virus transmission from natural hosts to humans. In addition, domestic animals themselves can suffer from the disease caused by bat-borne or closely related CoVs. Genomic sequences highly similar to PEDV were detected in bats, and SADS-COV is a recent spillover from bats to pigs (Figure 3). Currently, 7 of 11 ICTV-assigned αCoV species and 4 of 9 βCoV species were identified only in bats. Thus, bats are likely the major natural reservoirs of αCoV and βCoV (Figure 3).
Molecular Biology of Coronavirus
Coronavirus have the largest RNA genome approximately 30 kb and 80 to 120 nm in diameter [21]. The genome RNA is completed with the basic N protein to form a helical capsid found within the viral membrane. The membranes of all CoVs contain at least three viral proteins. These are S, the type I glycoprotein that forms the peplomers on the virion surface, giving the virus its corona or crownlike morphology in the electron microscope; the membrane (M) protein, a protein that spans the membrane three times and has a short N-terminal ectodomain and a cytoplasmic tail; and small membrane protein (E), a highly hydrophobic protein. The E protein of IBV has a short ectodomain, a transmembrane domain, and a cytoplasmic tail [22] (Corse and Machamer, 2000). The E protein of MHV is reported to span the membrane twice, such that both N and C termini are on the interior of the virion [23]. Some group II CoVs have an additional membrane protein, hemagglutinin esterase (HE). While the function of HE is not known, it is not an essential protein, and it has been speculated to aid in viral entry and/or pathogenesis in vivo and will be discussed below. HE is not encoded in the SARS COV genome. There is an additional group II virion protein called I for internal, as it is encoded within the nucleocapsid open reading frame (ORF). This is a nonessential protein of unknown function. It has recently been shown that the ORF 3a-encoded SARS protein is an additional structural protein [24]. There may be other minor proteins, as yet undetected, included in virions.
The genomes of all CoVs have a similar structure (Figure 4). The genome consists of seven genes. The first 22 kb contains the replicase gene, which is organized into two overlapping open reading frames, ORFs 1a and 1b. These ORFs are translated into the ~400- kDa pp1a and the ~800-kDa pp1ab replicasepolyproteins. ORF 1b is translated via a translational frameshift encoded at the end of ORF 1a. The protein domains of the replicasepolyprotein are indicated by nonstructural protein numbers (nsp1 to 16) (Table 2) and by confirmed or predicted functions: PLP1 and PLP2, papain-like proteases; X, domain encoding predicted adenosine diphosphateribose 1”-phosphatase activity (ADRP); 3CLpro, 3C-like protease; RdRp, putative RNA-dependent RNA polymerase; Hel, helicase; ExoN, putative exonuclease; XendoU, putative poly(U)-specific endoribonuclease; 2`-O-MT, methyltransferase. Genes 2 to 7 are translated from subgenomic mRNA species (not shown). Relative locations of coding regions for the structural proteins HE, S, E, M, N, and I are shown, as are the coding region for the group-specific ORF 2a (encoding a predicted cyclic phosphodiesterase), 4 and 5a proteins. The 5` approximately 20 to 22 kb carries the replicase gene, which encodes multiple enzymatic activities. The replicase gene products are encoded within two very large open reading frames, ORFs 1a and 1b, which are translated into two large polypeptides, pp1a and pp1ab, via a frameshifting mechanism involving a pseudoknot structure formed by the genomic RNA [25,26,21]. The complete sequence (22 kilobases) of murine coronavirus gene 1 encoding the putative). The structural proteins are encoded within the 3` one-third of the genome, for all CoVs, in the order S-E-M-N. (When the HE protein is expressed, it is encoded 5` to S.) Each group of CoVs in addition encodes a group of unique small proteins; while these protein are nonessential and have been speculated to serve as accessory proteins and to interact or interfere with the host innate immune response, this has not been demonstrated for any of these proteins. There are untranslated regions (UTRs) on both the 5` and 3` ends of the genome, which are believed to interact with host and perhaps viral proteins to control RNA replication, which includes the synthesis of positive- and negative-strand genomic-length RNA. Likewise, there are conserved sequences at the beginning of the transcription sites for each of the multiple subgenomic mRNAs; these are called transcriptional regulatory sequences (previously known as intergenic sequences). Coronavirus transcription has been reviewed recently [27,28].
Table 2: Functions of coronavirus non-structural proteins (nsps).
| Protein | Function |
|---|---|
| Nsp1 | Promotes cellular mRNA degradation and blocks host cell translation, results in blocking innate immune response |
| Nsp2 | No known function, binds to prohibitin proteins |
| Nsp3 | Large, multi-domain transmembrane protein, activities include: • Ubl1 and Ac domains, interact with N protein • ADRP activity, promotes cytokine expression • PLPro/Deubiquitinase domain, cleaves viral polyprotein and blocks host innate immune response • Ubl2, NAB, G2M, SUD, Y domains, unknown functions |
| Nsp4 | Potential transmembrane scaffold protein, important for proper structure of DMVs |
| Nsp5 | Mpro, cleaves viral polyprotein |
| Nsp6 | Potential transmembrane scaffold protein |
| Nsp7 | Forms hexadecameric complex with nsp8, may act as processivity clamp for RNA polymerase |
| Nsp8 | Forms hexadecameric complex with nsp7, may act as processivity clamp for RNA polymerase; may act as primase |
| Nsp9 | RNA binding protein |
| Nsp10 | Cofactor for nsp16 and nsp14, forms heterodimer with both and stimulates ExoN and 2-O-MT activity |
| Nsp12 | RdRp |
| Nsp13 | RNA helicase, 5′ triphosphatase |
| Nsp14 | N7 MTase) and 3′-5′ exoribonuclease, ExoN; N7 MTase adds 5′ cap to viral RNAs, ExoN activity is important for proofreading of viral genome |
| Nsp15 | Viral endoribonuclease, NendoU |
| Nsp16 | 2′-O-MT; shields viral RNA from MDA5 recognition |
* Ubl, ubiquitin-like; Ac, acidic; ADRP, ADP-ribose-1’-phosphate; PLPro, papain-like protease; NAB, nucleic acid binding; SUD, SARS-unique domain; DMVs: Double-Membrane Vesicles; Mpro: Main Protease; RdRp: RNA-dependent RNA polymerase; MTase, methyltransferase; Viral exoribonuclease, ExoN; Viral endoribonuclease, NendoU; 2’-O-MT, 2’-O-Methyltransferase; MDA5, Melanoma differentiation associated protein 5.
Replication cycle of coronavirus
Weiss and Martin [28] designed a model (Figure 4) and summarized that COV attach to specific cellular receptors via the spike protein. This triggers a conformational change in spike which then mediates fusion between the viral and cell membranes which results in the release of the nucleocapsid into the cell. Upon entry into the cell, the 5` end of the genome RNA, ORFs 1a and 1b, are translated into pp1a and pp1ab; pp1ab is translated via a frame shift mechanism, which occurs at high frequency (25 to 30%) [29]. ORF 1a encodes one or two papain-like proteases (PLpro or PLP) and a picornavirus 3C-like protease (3CLpro), which functions to process pp1a and pp1ab into the mature replicase proteins. Also, encoded in the X domain of ORF 1a is a (putative) ADP-ribose 1”-phosphatase activity. Encoded in ORF 1b and processed from pp1ab are an RNAdependent RNA polymerase (RdRp) and a helicase as well as other enzymatic activities, including a (putative) 3`-to-5`exonuclease (ExoN), poly(U)-specific endoribonuclease (XendoU), and (putative) S-adenosylmethionine-dependent ribose 2`-O-methyltranferase [30]. An additional putative enzymatic activity, cyclic phosphodiesterase, is encoded downstream in ORF 2a. These multiple enzymatic activities are speculated to play roles in metabolism of coronavirus RNA and interfering in the processes of host cell [31,32] (Figure 4).
Pathogenicity of coronavirus
CoVs cause a variety of diseases in mammals and birds ranging from enteritis in cows and pigs and upper respiratory disease chickens to potentially lethal human respiratory infections. CoVs cause a large variety of diseases in animals, and their ability to cause severe disease in livestock and companion animals such as pigs, cows, chickens, dogs and cats led to significant research on these viruses in the last half of the 20th century. Prior to the SARS COV outbreaks, CoVs were only thought to cause mild, self-limiting respiratory infections in humans. Some of coronaviruses and their pathogenicity are summarized in (Table 3).
Table 3: Pathogenicity of different animal and human CoV.
| CoV | Host | Disease |
|---|---|---|
| TGEV | Young piglets | Severe gastroenteritis |
| PEDV | Young piglets | Severe gastroenteritis |
| PHEV | Pigs | Enteric infection, infect the nervous system, causing encephalitis, vomiting |
| FCoV | Domestic cats | Mild or asymptomatic infection |
| FIPV | Domestic cats | Lethal disease called feline infectious peritonitis |
| Bovine CoV | Cattle, elk, deer, camels and mice | Mild to severe respiratory tract infections and diarrhea (‘winter dysentery’ and ‘shipping fever’) leading to weight loss, dehydration, decreased milk production, |
| Rat CoV | Rats | Mild to severe respiratory tract infections |
| IBV | Chickens | Mild to severe respiratory tract infections and decrease egg production. |
| SW1 | Whale | Respiratory disease and acute liver failure |
| Novel group of nidoviruses (Mesoniviridae) | Insects | Not known |
| MHV | Mice | Multiple sclerosis, respiratory, enteric, hepatic, and neurologic infections |
| HCoV-229E, HCoV-OC43 and HCoV-HKU1 | Human | Respiratory tract infections (cause more severe disease in neonates, the elderly, and in individuals with underlying illnesses, with a greater incidence of lower respiratory tract infection) |
| HCoV-NL63 | Human | Acute Laryngotracheitis (croup), respiratory tract infections (cause more severe disease in neonates, the elderly, and in individuals with underlying illnesses, with a greater incidence of lower respiratory tract infection) |
| SARS-CoV | Human | Severe Acute Respiratory Syndrome |
| MERS-CoV | Human | Series of highly pathogenic respiratory tract infections |
| Novel SARS-CoV-2 | Human | COVID-19 |
* CoV: Coronavirus, TGEV: Transmissible Gastroenteritis Virus,PEDV Porcine Epidemic Diarrhea Virus, PHEV Porcine hemagglutinating encephalomyelitis virus, FCoV Feline enteric coronavirus, FIPV Feline Infectious Peritonitis Virus, IBV Infectious Bronchitis Virus, SW1, MHV murine hepatitis virus, HCoV-229E, HCoV-OC43, HCoV-HKU1 and HCoV-NL63: Different strains of Human CoronaVirus.
Conclusion
Over the past 50 years the emergence of many different coronaviruses that cause a wide variety of human and veterinary diseases has occurred. It is likely that these viruses will continue to emerge and to evolve and cause both human and veterinary outbreaks owing to their ability to recombine, mutate, and infect multiple species and cell types. A considerable progress has been achieved with respect to seventeen years ago when the world had to face a completely unprepared SARS pandemics. Recently, the CDC networks identified a new strain of coronavirus SARS COV-2 and confirmed it as the pathogen new epidemic COVID-19 within a few weeks after the first case was noticed. The Chinese health authorities communicated the detail information of the epidemic with the public, WHO and international community in a timely manner and shared the full genome sequence of the new virus with the global scientific community. Nonetheless, there is more hope for the better control of coronaviruses, since the viruses are facing a much better prepared epidemic control forces globally. The most effective way to prevent viral zoonosis is to maintain the barriers between natural reservoirs and human society.
Authors’ Contributions
All authors discussed and conceived the study, drafted the manuscript, critically read and revised the manuscript and gave final approval for publication.
References
- Kennedy M, Boedeker N, Gibbs P, Kania S, (2001) Deletions in the 7a ORF of feline coronavirus associated with an epidemic of feline infectious peritonitis. Vet Microbiol, 81: 227-234.
- Blau DM, Holmes KV, (2001) Human coronavirus HCoV-229E enters susceptible cells via the endocytic pathway. InThe Nidoviruses Springer, Boston, MA, 193-198.
- Lai MC, Holmes KC, (2001) Coronaviridae: the viruses and their replication. In Fields Virology, Fourth Edition (Knipe DM, Howley PM, eds). Lippincott Williams and Wilkins, Philadelphia: 1163-1185.
- Ge Xy, Li JL, Yang XL, Chmura AA, Zhu G, et al.( 2013) Isolation and characterization of a bat SARS-like coronavirus that uses the ACE2 receptor. Nature, 503(7477): 535-538.
- Menachery VD, Yount BL, Debbink K, Agnihothram S, Gralinski LE, et al. ( 2015) A SARS-like cluster of circulating bat coronaviruses shows potential for human emergence. Nature medicine, 21(12): 1508-1513.
- Reusken CB, Haagmans BL, Müller MA, Gutierrez C, Godeke GJ, et al. (2013) Middle East respiratory syndrome coronavirus neutralising serum antibodies in dromedary camels: a comparative serological study. Lancet Infect Dis,13: 859-866.
- Lai MM, Cavanagh D, (1997) The molecular biology of coronaviruses. In Advances in virus research, 48: 1-100.
- Greenberg SB, ( 2011) Update on rhinovirus and coronavirus infections. Semin Respir Crit Care Med, 32(4): 433-446.
- Ksiazek TG, Erdman D, Goldsmith CS, (2003) A novel coronavirus associated with severe acute respiratory syndrome. N Engl J Med, 348(20): 1953-1966.
- Peiris JS, Yuen KY, Osterhaus AD, Stöhr K, (2003) The severe acute respiratory syndrome. New England Journal of Medicine, 349(25): 2431-2441.
- Peiris JSM, Chu CM, Cheng VCC, Chan KS, Hung IFN, et al. (2003) Clinical progression and viral load in a community outbreak of coronavirus-associated SARS pneumonia: a prospective study. The Lancet, 361(9371): 1767-1772.
- Zaki AM, van Boheemen S, Bestebroer TM, Osterhaus AD, Fouchier RA, et al. (2012) Isolation of a novel coronavirus from a man with pneumonia in Saudi Arabia. N Engl J Med, 367(19): 1814-1820.
- Cowley JA, Dimmock CM, Spann KM, Walker PJ, ( 2000) Gill-associated virus of Penaeus monodon prawns: an invertebrate virus with ORF1a and ORF1b genes related to arteri and CoVs. Journal of General Virology, 81(6): 1473-1484.
- Enjuanes L, Cavanagh D, Holmes K, Lai MC, Laude H, et.al (2000) Coronaviridae, 835-849.
- Woo PC, Lau SK, Lam CS, Lau CC, Tsang AK, et al. (2012) Discovery of seven novel Mammalian and avian CoVs in the genus deltacoronavirus supports bat CoVs as the gene source of alphacoronavirus and betacoronavirus and avian CoVs as the gene source of gammacoronavirus and deltacoronavirus. Journal of virology, 86(7): 3995-4008.
- Gonzalez JM, Gomez-Puertas P, Cavanagh D, Gorbalenya AE, Enjuanes L, et al. (2003) A comparative sequence analysis to revise the current taxonomy of the family Coronaviridae. Archives of virology 148(11): 2207-2235.
- Goebel SJ, Taylor J, Masters PS, (2004) The 3′ cis-acting genomic replication element of the severe acute respiratory syndrome coronavirus can function in the murine coronavirus genome. Journal of virology, 78(14): 7846-7851.
- Gorbalenya AE, Snijder EJ, Spaan WJ, (2004) Severe acute respiratory syndrome coronavirus phylogeny: toward consensus. Journal of virology, 78(15): 7863-7866.
- Cavanagh D, Mawditt K, Welchman DDB, Britton P, et.al (2002) CoVs from pheasants (Phasianus colchicus) are genetically closely related to CoVs of domestic fowl (infectious bronchitis virus) and turkeys. Avian pathology, 31(1): 81-93.
- Jonassen CM, Kofstad T, Larsen IL, Løvland A, Handeland K, et al. (2005) Molecular identification and characterization of novel CoVs infecting graylag geese (Anser anser), feral pigeons (Columbia livia) and mallards (Anas platyrhynchos). Journal of General Virology, 86(6): 1597-1607.
- Lee HJ, Shieh CK, Gorbalenya AE, Koonin EV, La Monica, et al. ( 1991) The complete sequence (22 kilobases) of murine coronavirus gene 1 encoding the putative proteases and RNA polymerase. Virology, 180(2): 567-582.
- Maeda J, Repass JF, Maeda A, Makino S, (2001) Membrane topology of coronavirus E protein. Virology, 281(2): 163-169.
- Corse E, Machamer CE, (2000) Infectious bronchitis virus E protein is targeted to the Golgi complex and directs release of virus-like particles. Journal of virology,74(9): 4319-4326.
- Ito N, Mossel EC, Narayanan K, Popov VL, Huang C, et al. (2005) Severe acute respiratory syndrome coronavirus 3a protein is a viral structural protein. Journal of virology, 79(5): 3182-6.
- Gorbalenya AE. Big nidovirus genome, (2001) when count and order of domains matter. Adv. Exp. Med. Biol, 494: 1-17.
- Bredenbeek PJ, Pachuk CJ, Noten AF, Charité J, Luytjes W, et al.( 1990) The primary structure and expression of the second open reading frame of the polymerase gene of the coronavirus MHV-A59; a highly conserved polymerase is expressed by an efficient ribosomal frameshifting mechanism. Nucleic acids research, 18(7): 1825-1832.
- Brian DA, Baric RS, (2005) Coronavirus genome structure and replication. Curr Top Microbiol Immunol, 287: 1-30.
- Weiss SR, Navas-Martin S, (2005) Coronavirus pathogenesis and the emerging pathogen severe acute respiratory syndrome coronavirus. Microbiol Mol Biol Rev ,69(4): 635-64.
- Snijder EJ, Bredenbeek PJ, Dobbe JC, Thiel V, Ziebuhr J, et al. (2003) Unique and conserved features of genome and proteome of SARS-coronavirus, an early split-off from the coronavirus group 2 lineage. Journal of molecular biology, 331(5): 991-1004.
- Ziebuhr J, (2005)The coronavirus replicase. In Coronavirus replication and reverse genetics. Springer, Berlin, Heidelberg, 57-94.
- Fehr AR, Perlman S, (2015) CoVs: an overview of their replication and pathogenesis. In CoVs. Humana Press, New York, NY: 1-23.
- Cui J, Li F, Shi Z, (2019) Origin and evolution of pathogenic coronaviruses. Nature Reviews Microbiology, 17: 181-192.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi