Research Article, J Clin Exp Oncol Vol: 11 Issue: 1
Correlation of Ki67 Expression with Cyclin D1 Grading in Invasive Ductal Carcinoma of Breast and It's Relationship with other Biomarkers
Sumita Bhattacharyya Panda1*, Sudipta Chakrabarti2, Jayati Chakraborty1, Ranjan Bhattacharyya1
1Department of Pathology, ESI PGIMSR, Manicktala, Kolkata
2Department of Psychiatry, Murshidabad Medical College & Hospital
*Corresponding Author:Sumita Bhattacharyya Panda
Department of Pathology, ESI PGIMSR
Manicktala, Kolkata
Tel: 9433053389;
Email: drrbcal@gmail.com.
Received date: 27 September 2021, Manuscript No: JCEOG-21-30598;
Editor Assigned: 29 October 2021, PreQC No: JCEOG-21-30598 (PQ);
Reviewed Date: 12 November 2021, QC No: JCEOG-21-30598;
Revised Date: 26 November 2021, Revised Manuscript No: JCEOG-21-30598 (R);
Accepted Date: 27 January 2022, DOI: 10.4172/2324-9110.11.1.289
Citation: Panda SB, Chakrabarti S, Chakraborty J, Bhattacharyya R (2022) Correlation of Ki67 Expression with Cyclin D1 Grading in Invasive Ductal Carcinoma of Breast and It’s Relationship with Other Biomarkers. J Clin Exp Oncol 11:1.
Abstract
Introduction: Recent advancement in biomarkers and immunohistochemical markers with their response to chemotherapy, survival in breast carcinoma has increased significantly. With progression of new gene expression profiling technique, it has been evaluated that in comparison to traditional markers, molecular markers have important role.
Aims & objectives: The primary aim was to find out the correlation of Ki67 expression with Cyclin D1 grading in Invasive ductal carcinoma. The study will also examine the correlation with other variables like Estrogen Progesterone receptor status, Her2Neu status as secondary outcome measure.
Materials & Methods: The 57 cases with suspected breast carcinoma later proved to be intraductal carcinoma has been included in this study over a period of 18 months. Cyclin D1 expression was calculated semiquantitatively on the basis of positive nuclear staining fraction of tumour cells and their intensity Ki67 expression. It is expected that the IHC (Immunohistochemistry) markers especially the Expression of Cyclin D1 will be significantly correlated with different molecular
subtypes of invasive ductal carcinoma and Ki67 index. The data analysed by SPSS version 25.0.
Results: In this study 43(75.4%) patients had High Ki-67 Status and 14 (24.6%) patient had Low Ki-67 Status. High Ki 67 expression has been found more commonly in ER positive, PR negative, Her2Neu positive cases and in Cyclin D1 grade 4. Ki67 expression has been found to be significantly correlated in
ER positive & negative, PR positive & negative and Her2Neu positive groups. The correlation with Ki67 expression and Estrogen receptor status, Progesterone receptor status, Her2Neu status and Cyclin D1 Grading has been found statistically significant in chi square test.
Conclusion: There are only few studies on this subject in the Indian population. A longitudinal study involving more samples involving other biomarkers is the call for the day to extrapolate the present findings.
Keywords: Invasive ductal carcinoma, Ki67 expression, Cyclin D1 status, Emerging biomarkers, Her2Neu Status, Estrogen progesterone receptor.
Introduction
The International Agency for Research on Cancer (IARC) stated that USA, India, China collectively form one third of global breast cancer burden. India has maximum number of breast cancer death [1,2]. Dysregulation of the CDK4/6- cyclin D1 complex is an important step in the genesis of breast cancer, and several genetic alterations in cell cycle regulatory proteins have been described [3].
National Health Service Breast Screening Programme (NHSBSP) used single view mammography for screening women aged 50-60 years (1988). This was followed by the two view mammograph (2005) which was used to screen 1.3 million women aged 50-70 yrs. In 1940, Immunofluorescence was applied to detect pneumococcal antigens in frozen section. Later on in late 80s and 90s the technique of IHC (Immunohistochemistry) came into general application [4].
Ki67 is a nuclear protein of molecular mass 359kDa and commonly used for the detection and quantification of proliferating cells. Its expression is increased, associated with cell growth. It is commonly used as a diagnostic marker in various cancers because its expression reflects the cellular proliferation rate. Ki67 gene is located on long arm of chromosome 10. It plays a crucial role in cell division and thought to be required for maintaining cell proliferation, DNA metabolic process, cellular response to heat, meiosis and organ regeneration. Throughout the cell cycle, the expression of Ki67 varies in intensity. During G1 and early S phase, Ki67 level is low, during mitosis progressively increase to maximum level while, during anaphase and telophase , a rapid decrease in expression starts. The degree of expression can be used as a marker of different conditions of growth.
Ellis et al suggested from the reference of in the American College of Surgeons Oncology Group Z1031 Trial that the triage to chemotherapy with high Ki67 LI patients after neoadjuvant endocrine treatment is less effective. The new treatment methods–other than chemo-endocrine therapy–may be needed for the high-Ki67 group following neoadjuvant endocrine therapy [5].
The areas of higher proliferation are indicated by intense staining of Ki-67 also known as “hot spots.” Some may proceed with manual counting while others use Digital Image Analysis (DIA). The Ki-67 index may be recorded in continuous numbers or as categorical values which may lead to inter-observer variability [6]. Ellis suggested that chemotherapy after neoadjuvant endocrine therapy in the low-Ki67 LI group of patients could be avoided. The effectiveness of chemotherapy in the high-Ki67 LI group of patients after neoadjuvant endocrine therapy is not substantially proved [7]. Johnston showed that, in the PALLET trial involving ER-positive and HER2-negative breast cancer patients, the concomitant use of a CDK4/6 inhibitor and endocrine therapy significantly decreased Ki67 LI compared with endocrine therapy alone. But there was no clinical response difference was observed between patients treated with concomitant CDK4/6 inhibitor and endocrine therapy versus endocrine monotherapy [8].
The efficacy of post-neoadjuvant endocrine therapy with a CDK4/6 inhibitor in the residual high-Ki67LI group of patients with ER-positive and HER2-negative breast cancer are demands clinical trials using CDK 4/6 inhibitor in the neoadjuvant setting. The significant correlation between the expression of CCND1 and Ki67 LI following neoadjuvant endocrine therapy has been found. CCND1 acts as a stimulator in the cell cycle in breast cancer by binding to cyclin-dependent kinase 4/6 (CDK4/6). 55 Various recent research works have suggested the efficacy of CDK 4/6 inhibitor in endocrine therapy-resistant metastatic breast cancer. The concomitant use of a CDK4/6 inhibitor and endocrine therapy in neoadjuvant treatment is known to markedly decrease Ki67 LI [9].
Materials and Methods
The consecutive 57 samples who are meeting all over a period of 18 months have been included for the study in a tertiary care set up of a centralized institute. The written informed consent has been obtained from the patients or their legal guardians. Samples has been included which have been received by the department during the study period. Those who have been diagnosed as breast carcinoma clinically and have submitted either mastectomy /lumpectomy specimen to the department have been included in the study and after screening confirmed intraductal carcinoma cases are being analyzed. The cases with presence of fixation artefact, other types of benign / malignant breast tumor besides invasive duct carcinoma, patients who have received any kind of chemotherapy / radiotherapy /contemporary treatment and the cases where the written informed consent couldn’t be obtained are excluded from the study. The study has been approved by institutional review board.
The primary outcome measure was to evaluate the correlation between Cyclin D1 status of IDC (Intraductal carcinoma) with different biomarkers of carcinoma of breast. As secondary outcome measures, the correlation with other variables other variables like Estrogen Progesterone receptor status, Her2Neu status has been analyzed. For statistical analysis data were entered and analysed by SPSS (Statistical Package for the Social Sciences) software (version 25.0; SPSS Inc., Chicago, IL, USA) [10].
Results
Table 1: Relationship of different biomarkers and with Ki67 protein expression.
Table 2: Correlation between ER Pos & ER Neg cases with high & low Ki67 expression.
The χ2 (chi-square statistic) is 6.3629. The p-value is 0.011653. Significant at p<0.05.
Table 3: Correlation between PR positive & PR negative cases with high & low Ki67 expression.
Table 4: Correlation between Her2Neu positive, Her2Neu negative and Her2Neu equivocal with high & low Ki67 expression.
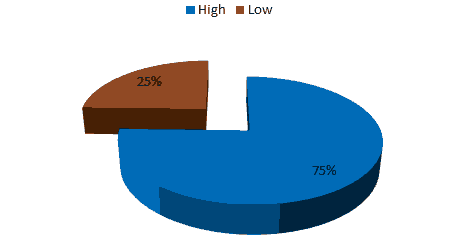
Figure 1: Break up of Ki67 status of the sample.
In the present study, 43 (75.4%) patients had High Ki-67 Status and 14 (24.6%) patient had Low Ki-67 Status Figure 1. High Ki 67 expression has been found more commonly in ER positive (n: 24, 42.1%), PR negative cases (n: 22, 38.5%), Her2Neu positive cases (n: 23, 40.3%), Cyclin D1 grade 4 (n: 22, 38.5%) cases Table 1. The correlation between ER positive and ER negative tumours with Ki67 High and Ki 67 low expression by chi square test has been done in this sample and it has been found statistically significant (0: 6.3629, p: 0.01*) Table 2. The Ki67 expression is high in n: 21 PR positive cases and n: 22, PR negative cases, whereas Ki67 expression is low in n: 13 PR positive cases and n: 1 PR negative cases. The chi square test shows significance with p <0.05 Table 3. High Ki67 biomarker expression has been found in n: 22 Her2Neu Positive group, n: 14 Her2Neu negative group and n: 6 Her2Neu equivocal groups. Low Ki67 expression has been observed in n: 1 Her2Neu Positive, 12 Her2Neu negative and 2 Her2Neu equivocal groups. The chi square test shows significance with p value 0.04 Table 4 [11,12].
Table 5: Correlation between different grades of Cyclin D1 with High & Low Ki67 expression.
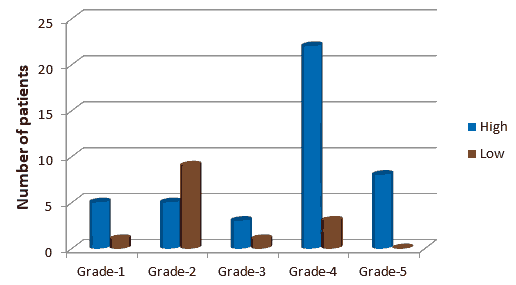
Figure 2: Break up different Grades of Cyclin D1 cases with Ki67 expression.
In Cyclin D1 Grade-1 Group, (n: 5, 83.3%) patients had High Ki-67 Status and (n: 1, 16.7%) patients had Low Ki-67 Status. In Grade-2 Group, (n: 5, 35.7%) patients had High Ki-67 Status and (n: 9, 64.3%) patients had Low Ki-67 Status. In Grade-3 Group, (n: 3, 75.0%) patients had High Ki-67 Status and (n: 1, 25.0%) patients had Low Ki-67 Status. In Grade-4 Group, (n: 22, 88.0%) patients had High Ki-67 Status and (n: 3, 12.0%) patients had Low Ki-67 Status. In Grade-5 Group, (n: 8,100.0%) patients had High Ki-67 Status. The association of Ki-67 Status vs group was statistically significant (p: 0.0021) Table 5, Figure 2.
Table 6: Association between Ki-67 Status: Molecular Classification.
In HER2 Group, (n: 11, 100.0%) patients had Ki-67 Status. In LA Group, (n: 1, 100.0%) patient had Ki-67 Status. In LB Group, (n: 23, 92.0%) patients had Ki-67 Status and (n: 2, 8.0%) patients had Ki-67 Status. In TN Group, (n: 9, 90.0%) patients had Ki-67 Status and (n: 1, 10.0%) patients had Ki-67 Status. The association of Ki-67 Status vs group was statistically significant (p<0.0001) Table 6, Figure 3.
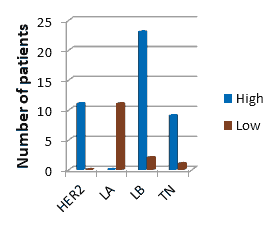
Figure 3: Ki67 status vs Molecular Classification.
Discussion
The pre-treatment Ki67 LI had been found to be significant prognostic and predictive factor for the efficacy of neoadjuvant chemo-endocrine therapy. The Ki67 LI is a useful biomarker for evaluating tumor proliferation Figure 4. 11,12 It has been reported in literature that Ki67 LI (Labelling index) after neoadjuvant endocrine therapy against ER-positive and HER2-negative breast cancer is a very important prognostic factor in carcinogenesis of breast cancer [13,14].
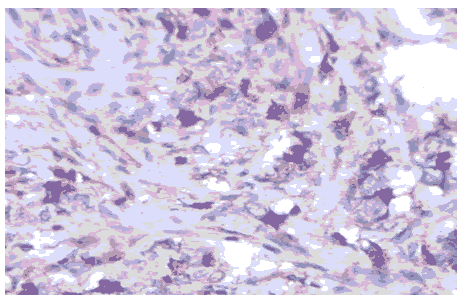
Figure 4: Ki67 with 70% expression (40X view).
Zhou CJ et al 2009 investigated the expression and association of ER, Ki-67 and cyclinD1 in Usual Ductal Hyperplasia (UDH), Atypical Ductal Hyperplasia (ADH) and Ductal Carcinoma In Situ (DCIS) in the breast. The sample included 56 cases of pre-cancerous lesions which were surgically excised and the Immuno Histo Chemistry (IHC) was used to determine the expression of ER, Ki-67 and cyclinD1. The double-labelling immunofluorescence technique was used to found co-expression of ER and Ki-67. The expression and distribution of ER-positive cells were significantly different in UDH, ADH and DCIS (Ductal Carcinoma In Situ). The ER-positive cells were much more in UDH than in normal TDLUs (Terminal Duct Lobular Units). The distribution of ER-positive cells interspersed amid ER-negative cells within UDH. However, the ER positive cells showed marked increases in ADH and low grade nuclear DCIS (P < 0.05), distributing in almost all constituent cells. The expression of ki-67 and cyclin D1 were significantly different between UDH and DCIS (P<0.05), and a positive correlation was found between expression of Ki-67 and morphological classification of pre-cancerous lesions (r: 0.3522, P<0.05) as well as cyclinD1 (r: 0.3901, P<0.05). Double-labelling immunofluorescence showed that there was no co-expression of ER and Ki-67 in normal breast tissue. The co-expression of the two markers was found in ADH and increased in DCIS. Overexpression of ER, Ki-67 and cyclinD1 significantly accompanies the transition of normal cells and UDH to ADH and DCIS [15].
The Cyclin D1 expression can be divided into five grades according to positive nuclear staining fraction of tumour cells (%). Grade 0 denotes no staining followed by Grade 1 (1-5%), Grade 2 (6-10%), Grade 3 (11-25%), Grade 4 (26-50%) and Grade 5 (51-100%) [16]. Expression of cyclin D1 has been found to be correlated with Ki-67 expression, but not correlated with pRb and p53 expression [17]. Reis-Filho JS et al. also noted that when MIB index is high for Ki67 expression, CCND1 gene is not getting amplified and Cyclin D1 value is low [18].
The present study revealed the most cases with High Ki67 expression are associated with ER positive, PR negative and Her2Neu positive cases which can be a significant decision making factor for formulating therapeutic strategy and predicting prognosis. Well differentiated DCIS usually have lower Ki67 level and vice versa. By automated image analysis, multiple DCIS samples have proved that the invasion is directly associated with Ki67 expression and inversely associated with ER, PR and Her2 expression [19].
Conclusion
Despite intensive studies on biomarkers to find out and predict the progression of DCIS as well as recurrence needs robust development of biomarkers and this phase is still in infancy [20]. The comprehensive systemic review over 10 years revealed that the therapeutic decision making process may be revolutionized with availability of biomarkers. The present study has some drawbacks. The other varieties of invasive breast carcinoma like lobular carcinoma, metaplastic, medullary carcinoma have not been studied. A follow up longitudinal study with higher sample size and more biomarkers will be helpful to extrapolate the findings of present study. The findings of the present study may help for future research, will add scientific values with current understandings and can be extrapolated with larger sample size. The determination of the biological markers will be helpful to predict responsiveness to therapy. Besides this emerging biomarker will enable subgrouping of breast carcinoma and will aid more effective treatment and ease up the therapeutic decision making process.
References
- Patil VW, Singhai R, Patil AV, Gurav PD (2011) Triple-negative (ER, PR, HER-2/neu) breast cancer in Indian women. Breast Cancer: Targets and Therapy 3: 9-19.
[Crossref] [Google Scholar] [Indexed]
- Siegel RL, Miller KD, Jemal A (2020) Cancer statistics 2020. CA Cancer J Clin 70: 7–30.
[Crossref] [Google Scholar] [Indexed]
- Lin H, Huang JF, Qiu JR, Zhang HL, Tang XJ, et al. (2013) Significantly up regulated TACSTD2 and Cyclin D1 correlate with poor prognosis of invasive ductal breast cancer. Exp Mol Pathol 94: 73-8.
[Crossref] [Google Scholar] [Indexed]
- Elmore JG, Armstrong K, Lehman CD, Fletcher SW (2005) Screening for breast cancer. JAMA 293: 1245-1256.
[Crossref] [Google Scholar] [Indexed]
- Ellis MJ, Suman VJ, Hoog J, Goncalves T Ki67 Proliferation index as a tool for chemotherapy decisions during and after neoadjuvant aromatase inhibitor treatment of breast cancer: Results from the american college of surgeons oncology group Z1031 trial. J Clin Oncol 35:1061-1069.
[Crossref] [Google Scholar] [Indexed]
- Koopman T, Buikema HJ, Hollema H, De-Bock GH, Van-Der-Veg B (2018) Digital image analysis of Ki67 proliferation index in breast cancer using virtual dual staining on whole tissue sections: clinical validation and inter-platform agreement. Breast Cancer Res Treat 169: 33–42.
[Crossref] [Google Scholar] [Indexed]
- Ellis MJ (2017) Lessons in precision oncology from neoadjuvant endocrine therapy trials in ER+ breast cancer. Breast 34: S104–S107. 10.1016/j.breast.2017.06.039.
[Crossref] [Google Scholar] [Indexed]
- Johnston S, Puhalla S, Wheatley D, Ring A, Barry P, et al. (2019) Randomized phase II study evaluating palbociclib in addition to letrozole as neoadjuvant therapy in estrogen receptor–positive early breast cancer: PALLET trial. J Clin Oncol 37: 178–189.
[Crossref] [Google Scholar] [Indexed]
- Chow LWC, Morita S, Chow CYC, Ng WK, Toi M (2018) Neoadjuvant palbociclib on ER+ breast cancer (N007): Clinical response and EndoPredict's value. Endocr Relat Cancer 25: 123–130.
[Crossref] [Google Scholar] [Indexed]
- SPSS (2020) SPSS Inc., Chicago, IL, USA.
- Mohamed S, Yasufuku K, Hiroshima K, Nakajima T, Yoshida S, et al. (2007) Prognostic implications of cell cycle-related proteins in primary resectable pathologic N2 nonsmall cell lung cancer. Cancer 109: 2506-14.
[Crossref] [Google Scholar] [Indexed]
- Shibuya R, Suzuki T, Miki Y, Yoshida K, Moriya T, et al. (2008) Intratumoral concentration of sex steroids and expression of sex steroid-producing enzymes in ductal carcinoma in situ of human breast. Endocr Relat Cancer 15: 113-24.
[Crossref] [Google Scholar] [Indexed]
- Cheang MC, Chia SK, Voduc D (2009) Ki67 index, HER2 status and prognosis of patients with luminal B breast cancer. J Natl Cancer Inst 101: 736–50.
[Crossref] [Google Scholar] [Indexed]
- Schymik B, Buerger H, Krämer A, Voss U, van der Groep P, et al. (2012) Is there 'progression through grade' in ductal invasive breast cancer? Breast Cancer Res Treat 135: 693-703.
[Crossref] [Google Scholar] [Indexed]
- Zhou CJ, Zhang QH, Zhang TG, Sun SZ, Li H, (2009) Expression of ER, Ki-67 and cylinD1 in the pre-cancerous breast of Chinese patients. Pathol Oncol Res 15: 153-8.
[Crossref] [Google Scholar] [Indexed]
- Ortiz AB, Garcia D, Vicente Y, Palka M, Bellas C, et al. (2017) Prognostic significance of cyclin D1 protein expression and gene amplification in invasive breast carcinoma.
[Crossref] [Google Scholar] [Indexed]
- Guo LL, Gao P, Wu YG, Jian WC, Hao CY, et al. (2007) Alteration of cyclin D1 in Chinese patients with breast carcinoma and its correlation with Ki-67, pRb, and p53. Arch Med Res 38: 846-52.
[Crossref] [Google Scholar] [Indexed]
- Reis-Filho JS, Savage K, Lambros M, James M, Steele D, et al. (2006) Cyclin D1 protein expression and CCND1 amplification in breast carcinomas: An immune histo-chemical and chromogenic in situ hybridisation analysis. Mod Pathol 19: 999-1009. [Crossref] [Google Scholar] [Indexed]
- Sarode VR, Han JS, Morris DH, Peng Y, Rao R (2011) A comparative analysis of biomarker expression and molecular subtypes of pure ductal carcinoma in situ and invasive breast carcinoma by image analysis: Relationship of the subtypes with histologic grade, Ki67, p53 overexpression, and DNA Ploidy. Int J Breast Cancer 217060.
[Crossref] [Google Scholar] [Indexed]
- Lari SA, Kuerer HM (2011) Biological markers in DCIS and risk of breast recurrence: A systematic review. J Cancer 1: 232-61.
[Crossref] [Google Scholar] [Indexed]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi 



