Research Article, J Clin Exp Oncol Vol: 9 Issue: 1
Does Infiltrative Anesthesia Improve Post-operative Morbidity after Axillary Lymph Node Dissection in Patients of Breast Cancer? – A Prospective Study from a Tertiary Care Center in North India
Subramaniam Anand*
Department of Surgery, AFMC, Pune 40, India
*Corresponding Author: Subramaniam Anand Associate Professor, Department of Surgery, AFMC, Pune 40, India, Tel: 7080317133; E-mail: drandyaiyer@gmail. com
Received: September 10, 2019 Accepted: December 26, 2019 Published: January 02, 2020
Citation:Anand S (2020) Does Infiltrative Anesthesia Improve Post-operative Morbidity after Axillary Lymph Node Dissection in Patients of Breast Cancer? – A Prospective Study from a Tertiary Care Center in North India. J Clin Exp Oncol 9:1.
Abstract
Background: The role axillary lymph node dissection (ALND) in staging, prognostic assessment and local control in patients of breast cancer with clinically evident disease in the axilla is well established. ALND has its share of morbidity in the form of restriction of range of motion of arm, pain, paraesthesia and lymphedema influencing quality of life. This prospective study evaluates the effect of infiltrative anaesthesia injected along the nerve bundles exposed during ALND in reducing the restriction of range of motion of arm and pain scores following ALND.
Methods: A total of 60 ALND in patients of operable invasive breast cancer were randomized in to two groups:- Group 1: The control group where patients of ALND were managed in the postoperative period with standard analgesia of injection acetaminophen 1 gm 8 hourly and early arm and shoulder movements from the evening of the procedure. Group 2: The test group had 5 ml of 0.125% Bupivaccaine injected each around the three nerve bundles exposed during ALND along with early arm and shoulder exercises and standard analgesia similar to the control group. Medical records of patients were analysed for age, laterality, clinical staging, histopathological parameters and surgical complications. Range of motion of ipsilateral at the shoulder and pain scores as recorded by visual analog scale (VAS) were objectively noted on day 0 to day 28 postoperatively at various intervals.
Results: The range of motion at ipsilateral shoulder was measured by flexion, abduction and extension. The degrees of flexion and abduction were found to have statistically significant difference on Days 0, 3 and 7 in the test group in comparison to control group (p=0.001 for flexion on all Day 0,3 and 7 and p=0.009 for Day 0, p=0.001 for Day 3 and p<0.001 for Day 7 for abduction). Extension was better in the test group on Days 0 and 3 (p<0.001 and p=0.005 respectively). Pain scores assessed by VAS were significantly lower in test group when compared to control group on Days 0,1, 3 and 7 (p ≤ 0.01). There was no statistically significant difference in the restriction of flexion, extension and abduction and pain scores on Day 28 between the groups. Analgesia requirement was significantly reduced in the test group on days 0 and 1 (p ≤ 0.05).
Conclusion: Infiltrative anaesthesia with 0.125% bupivaccaine along the nerve bundles exposed during ALND improves range of motion at shoulder and pain scores in the immediate postoperative period in patients of breast cancer.
Keywords: Axillary Lymph Node dissection, Infiltrative anaesthesia, Range of motion restriction, Pain scores.
Keywords
Axillary Lymph Node dissection, Infiltrative anaesthesia, Range of motion restriction, Pain scores.
Introduction
Axillary lymph node dissection (ALND) remains the gold standard in patients of invasive breast cancer with axillary lymph node metastases in the staging and treatment of axilla. Though, an integral part of treatment specifically in patients staged as N1 or N2 according to the TNM staging, it has been largely replaced by sentinel lymph node biopsy (SLNB) in patients of breast cancer without axillary lymph node metastasis [1]. In India, only 2.5% - 22% of these cancers are confined to only breast and can be classified as early breast cancer and 40%- 85% fall in the locally advanced breast cancer category [2]. Hence a significant number of breast cancer patients in India undergo ALND.
Currently ALND involves removal of level I and II lymph nodes in the axilla, as described by Berg [3]. Though ALND is both therapeutic and prognostic in value, it is associated with sequelae of pain in the ipsilateral arm, paraesthesia, restricted range of motion and lymphedema. While various reconstructive procedures have evolved for partial or total reconstruction of the breast, very little has been done to correct these functional sequelae, [4]except that ALND has become increasingly conservative with surgeons avoiding dissection of Berg Level III lymph nodes [5]. Alternative approaches viz axillary sampling [6], limited level I dissection [7]and sentinel lymph node biopsy [8]have gained acceptance in last few decades.
A number of studies have objectively measured morbidity from ALND with contradictory results of reduced ALND aggressiveness [9]. However, no study has been done till date to evaluate any intervention at the time of ALND to reduce its morbidity. This study aims to establish the role of infiltrative anesthesia using 0.125% bupivaccaine along the nerve bundles exposed during axillary dissection and assessing the prevalence of pain relief and range of motion restriction of arm in patients of breast cancer undergoing ALND.
Materials and Methods
Patient recruitment
A prospective randomized study was performed at the malignant disease treatment centre of a tertiary care hospital in North India between July 2015 and June 2016 and the study was approved by Institutional Research and Ethics Committee. All patients were counselled about the study and written informed consent for infiltrative anaesthesia was obtained from them before randomization. A total of 60 ALND in patients of operable breast cancer were subjected to simple randomization using computer generated random numbers in to two groups-
Group 1: The control group where patients of ALND were managed in the postoperative period with standard analgesia of injection acetaminophen 1 gm 8 hourly and early arm and shoulder movements from the evening of the procedure.
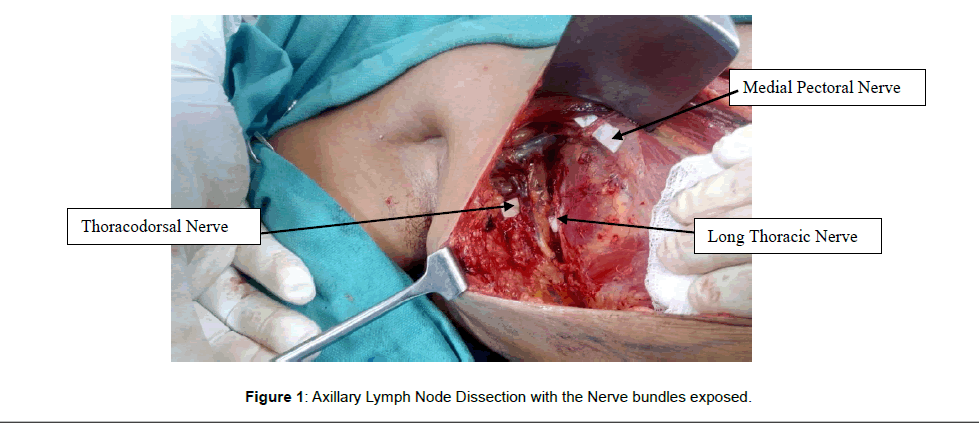
Group 2: The test group had 5 ml of 0.125% Bupivaccaine injected each around the three nerve bundles exposed during the ALND viz, Long thoracic nerve, Thoraco-dorsal nerve and Medial pectoral nerve (Figure 1). Postoperatively early arm and shoulder exercises and standard analgesia were started from the evening of the procedure, similar to the control group.
Inclusion and exclusion criteria
Patients between the age group 20 to 75 years with good Performance Status (PS-0 and PS-1) and histologically proven invasive breast cancer with palpable axillary nodes and without distant metastasis at diagnosis were included in the study. All the patients underwent ALND associated with breast conserving surgery (BCS) or mastectomy by same surgical oncologist, depending upon the site, tumour to breast ratio and patient’s desire. ALND was done up to level III in all cases. Modified Radical Mastectomy (MRM) was done by Auchincloss method using a transverse incision and ALND was done via a separate incision in patients undergoing BCS. Intercostobrachial nerves were divided in all cases. All patients were evaluated by a multidisciplinary team (MDT) of surgical, medical and radiation oncologists. Treatment protocols like Neoadjuvant chemotherapy and adjuvant treatment were decided and timed accordingly by the MDT and followed up after completion of adjuvant therapy.
Patients with recurrent cancer, age below 20 years and above 75 years, previous history of disorders or trauma to the arm or shoulder and neurological disorders like cerebero-vascular accidents were excluded.
Data collection
Medical records of patients were analysed for age, laterality, clinical staging, histopathological report (tumour size, number of lymph nodes harvested, number of lymph node positive for metastasis), preoperative chemotherapy, operation time, blood loss, complications of surgical site infection (SSIs) within 28 days, seroma formation, secondary procedures and any procedure related complications during infiltrative anesthesia in the test group.
Range of motion parameters: In the postoperative period, patients were taught to perform arm and shoulder exercises and the range of movements (flexion, extension and abduction at ipsilateral shoulder) were objectively recorded on day 0 (postoperative evening), days 3, 7 and 28, by the independent member of the research team, not involved in treatment of the patients, using an arm protractor for the purpose.
Pain parameters: Pain score was recorded using Visual Analog Scale (VAS) on Day 0 (postoperative evening), Day 1, 3, 7 & 28. Analgesia requirement from postoperative period till postoperative day 2 was also recorded.
Statistical analysis
The data were analysed using the IBM® SPSS version 25.0. The means and standard deviation were used to assess numerical values. The associations between the variables studied (range-of-motion and pain) and the other patient characteristics were assessed using Fisher’s exact test for the binary nominal variables and the Mann- Whitney test for continuous numeric variables. A p value <0.05 was considered significant.
Results
A total of 60 ALNDs performed in 59 patients of histologically proven breast carcinoma and clinically palpable ipsilateral axillary lymph nodes were included in the study. Mean age of patients was 53.3 ± 11.2 years and mean BMI was 24.4 ± 1.5 Kg/m2. The dominant arm was right in 54 patients and 5 patients were left arm dominant. Neoadjuvant chemotherapy (NACT) was given in 23 patients. Laterality of the cancer was found to be 29 each for right and left side and one patient underwent bilateral ALND for bilateral breast cancer.
ALND was performed as part of breast conserving surgery in 25 patients while the remaining were 35 modified radical mastectomies. The average dissected lymph nodes per ALND was 16.5 ± 5.8 (range 9-40 lymph nodes) and 30 ALNDs were positive for nodal metastases with a mean positive lymph nodes of 2.47 ± 4.37 (range 1-21 positive lymph nodes). Out of 30 ALNDs, which were negative for lymph node metastases, 12 patients had undergone NACT.
Ten axillae had complications of postoperative seroma with 06 in the control group and 04 from the test group. Surgical site infections (SSIs) were graded according to the Clavien-Dindo classification and noted in a total of 12 wounds. There were 04 grade I SSI (03 in control group and 01 in test group) and 08 grade II SSIs (04 in each group). All the SSIs were managed on outpatient basis, without any secondary procedures. There were no procedure related complications in the test group. The patient and their disease characteristics are shown in (Table 1).
| Variable | Number (%) |
|---|---|
| Age (yrs) | |
| Mean � SD | 53.3 � 12.26 |
| BMI (KG/M2) | |
| Mean � SD | 24.4 �1.5 |
| Dominant arm | |
| Right | 54 (90%) |
| Left | 05 (10%) |
| Surgery type | |
| Breast conserving | 25 (41.5%) |
| Mastectomy | 35 (58.5%) |
| Neo adjuvant chemotherapy | |
| Yes | 23 (38.3%) |
| No | 37 (61.7%) |
| Dissected axilla | |
| Right | 29 (49.1 %) |
| Left | 29 (49.1%) |
| B/L | 1 (1.8%) |
| PT stage | |
| 0 | 10 (16.7%) |
| 1 | 04 (6.7%) |
| 2 | 32 (53.3%) |
| 3 | 12 (20%) |
| 4 | 02 (3.3%) |
| PN stage | |
| 0 | 30 (50%) |
| 1 | 16 (26.7%) |
| 2 | 10 (16.6%) |
| 3 | 04 (6.7%) |
| Dissected alns (n) | |
| Mean � SD | 16.35 � 5.8 |
| Lymph nodes with metastasis | |
| Mean � SD | 2.47 � 4.37 |
| Post-operative seroma | |
| Yes | 10 (16.6%) |
| No | 50 (83.4%) |
| Surgical site infection | |
| Yes | 12 (20%) |
| No | 48 (80%) |
Table 1: Characteristics of ALNDs (n=60 ).
Range of motion parameters
The range of motion at the shoulder ipsilateral to ALND was measured by degree of flexion, extension and abduction on Day 0 (post-operative evening) and Days 3, 7 & 28 after surgery (Table 2). The degree of flexion and abduction was found to be with statistically significant difference on Days 0, 3 and 7 in the test group in comparison to control group (p = 0.001 for flexion on Days 0, 3 and 7 and p=0.009 for Day 0, p=0.001 for Day 3 and p ≤ 0.001 for Day 7 for abduction). Extension was better in the test group on Days 0 and 3 (p ≤ 0.001 and p=0.005 respectively) while there was no statistically significant difference in the restriction of flexion, extension and abduction on Day 28 between the groups.
| Arm Movement (in degrees) | Control group (mean � SD) | Test group (mean � SD) | p value |
|---|---|---|---|
| Flexion���������������� Day 0 | 121 � 12.63 | 134 � 15.69 | 0.001* |
| ���������������������������Day 3 | 133.7 � 16.12 | 150 � 19.29 | 0.001* |
| ���������������������������Day 7 | 152 � 11.14 | 164 � 15.22 | 0.001* |
| ���������������������������Day 28 | 178.9 � 5.57 | 178 � 7.61 | 0.603 |
| Extension����������� Day 0 | 30 � 0.20 | 38.5 � 7.56 | = 0.001* |
| ���������������������������Day 3 | 38.8 � 7.51 | 43.5 �4.57 | 0.005* |
| ���������������������������Day 7 | 45 � 0.40 | 45 � 0.40 | 1.000 |
| ���������������������������Day 28 | 45 � 0.40 | 45 � 0.40 | 1.000 |
| Abduction���������� Day 0 | 99.14 � 14.01 | 107.6 � 10.06 | 0.009* |
| ���������������������������Day 3 | 109.7 � 4.98 | 115.6 � 8.17 | 0.001* |
| ���������������������������Day 7 | 115.2 � 5.08 | 119.3 � 2.53 | = 0.001* |
| ���������������������������Day 28 | 119.7 � 1.86 | 120 � 2.02 | 0.552 |
| * statistically significant p value | |||
Table 2: Range of motion following ALND.
Pain parameters
Pain in the arm ipsilateral to ALND, assessed by VAS on Days 0, 1, 3, 7 & 28 and was significantly lower on Days 0, 1, 3 and 7 in the test group. On Day 28, pain scores were not significantly different between the two groups (Table 3).
| Study Variable | Control Group n= 30 | Test Group n=30 |
|---|---|---|
| Day 0 | 3 (4,3) | 3 (4,1)# |
| Day 1 | 3 (3,1) | 2 (3,0)# |
| Day 3 | 2 (3,1) | 1 (3,0)# |
| Day 7 | 1 (2,0) | 0 (1,0) # |
| Day 28 | 0 (0,0) | 0 (0,0) |
| Ordinal data are presented as medians and interquartile ranges (given in parenthesis) #p = 0.01 | ||
Table 3: Post-operative pain scores as per Visual Analog Scoring system (VAS).
Analgesia requirement was significantly reduced in the test group on Days 0 and 1 as compared to control group, while it was less also on Day 2, but with no statistically significant difference between the groups (Table 4). (Note: Pain scores for both axillae were recorded in two different scales for the patient with bilateral breast cancer).
| Study Variable | Control Group n= 30 | Test Group n=30 | p value |
|---|---|---|---|
| Day 0 | 2.9 � 0.30 | 2.4 � 0.49 | =0.001* |
| Day 1 | 2.4 � 0.56 | 1.7 � 0.79 | =0.001* |
| Day 2 | 1.45 � 0.57 | 1.2 � 0.55 | 0.089 |
| Values are presented as mean � SD,� *p = 0.05 | |||
Table 4: Post-operative rescue analgesic requirement.
Discussion
The role of ALND in staging, prognostic assessment and local control in patients of breast cancer with clinically evident disease in the axilla is well established [10]Though NSABP B-04 study concluded that there was no survival advantage with axillary dissection, the axillary recurrence rate was 19% in the no axillary treatment arm [11]On the contrary it has been reported that one life is saved for every four local recurrences avoided [12]and prophylactic axillary lymph node dissection had an average overall survival benefit of 5.4% [13]. In women presenting with clinically node negative axilla, the rate of axillary nodal metastasis is only 20-35% and hence a selective approach in the form of SLNB was developed and shown to be a safe and effective mode of staging the axilla in various trials [14]Husted Madesen and his colleagues in a prospective study established that in clinically node negative patients operated with SLNB have less arm morbidity compared to node negative patients operated with ALND. Further, they concluded that node positive patients who had secondary ALND after SLNB had no difference in either objective or subjective morbidity compared with node positive patients undergoing a one-step ALND [15]
Unfortunately, most of our patients present with either clinically node positive disease or locally advanced disease warranting ALND. ALND has its share of morbidity, in the form of functional sequelae including lymphoedema, paresthesia, restricted range-of-motion and pain in the arm ipsilateral to the lymph node dissection negatively influencing quality of life parameters of the affected patients. Ashikaga et al. reported shoulder abduction deficits >or=10% peaked at 1 week after ALND (75%), arm volume differences >or=10% at 36 months after ALND (14%), numbness and tingling peaked at 6 months after ALND (49%, 23%) [16]. Elizabeth Maunsell and her colleagues evaluated a cohort of 223 women with newly diagnosed breast cancer 3 months after operation and again 15 months later among 201 of these same women. At 3 months after operation, 182 (82%) patients reported at least one arm problem including swelling (24%), weakness (26%), some limitation in range of movement (32%), stiffness (40%), pain (55%) and numbness (58%), and these percentages had changed little 15 months later [17].
A complete ALND including clearance of level III nodes is warranted for patients with high axillary nodal burden breast cancer in developing countries like India [18]. The level III nodes are addressed by retracting the pectoralis minor muscle laterally with clearance upto the costoclavicular ligament of Halstead. Hence, during the complete ALND nerve bundles of large muscles like latissmus dorsi and serratus anterior and medial pectoral nerve along the lateral border of the pectoralis minor muscle are handled. Latissmus dorsi, one of the four muscles responsible for the thoraco-brachial motion, would cause limitation of range of motion of the ipsilateral arm when its neurovascular bundle is handled. Pain around the arm, shoulder and chest also, is contributed by dissection on serratus anterior, pectoral muscles and latissmus dorsi.
The present study hypothesized, that by infiltrating a local anesthetic around the nerve bundles of the muscles exposed during ALND, the symptoms of limited range of motion and pain parameters will show better results than the conventional treatment with early shoulder exercises and pain relief. In present study, it was found that by infiltrating 5 ml each of 0.125% bupivaccaine along the nerve bundles (Figure 1) exposed during ALND, the range of motion in the ipsilateral arm was less restricted compared to conventional exercises alone, specifically in the immediate post-operative week following ALND. Ranges of flexion and abduction were significantly improved in the infiltrative group as measured on Days 0, 3 and 7 and range of extension was found to be better on Days 0 and 3 (p<0.05). However by post-operative Day 28, and also on completion of treatment follow-ups, there was no difference in range of motion restriction between the groups.
In this study, pain score as recorded by VAS was found to be better in the infiltrative anesthesia group on days 0, 1, 3 and 7 (p<0.01) than the conventional treatment group, where only intermittent analgesia with 1 gm of Acetoaminophen was given in intervals of 8 hours. Statistically significant difference in pain scores was not noted between the two groups after the first post-operative week. Analgesia intake as surrogate marker for effectiveness of infiltrative anesthesia was found be lower in the test group (p<0.05) in the immediate postoperative period and the first postoperative day. There was no significant difference in the analgesia intake after Day 2 post-op between the groups. There was no infiltrative anesthesia related complication in the test group. Ten axillae had postoperative seroma, noticed from day 8 to day 21 after ALND which were managed by serial aspiration with none requiring any secondary procedures or delay in adjuvant therapy. A total of 12 wounds were labeled to have SSIs, of which 04 were grade I requiring anti-inflammatory agents and 08 had grade II infections requiring antibiotics. All of them were managed on outpatient basis and resolved within three weeks of surgery. Symptomatic seroma and increased arm temperature suggestive of early grades of SSIs are reported to have bivariate association in the formation of lymphoedema after ALND [19]. It is but logical that symptomatic seroma and SSIs would cause discomfort in the ipsilateral arm and breast, these symptoms have not been documented to be variables affecting range of motion restriction and long term pain after ALND in the literature.
To the best of our knowledge, this study is the first to evaluate the use of infiltrative analgesia in reducing the morbidity of restricted range of motion of arm and pain following ALND. The existing reports are based on self reported questionnaire or subjective in assessment. Though the sample size was limited, the study had variables in the test group measured against a control group in an objective manner. The limitation of the study includes that the range of motion prior to ALND was not recorded which could have categorically established that infiltrative anesthesia improved arm mobility in the postoperative period and in the long term. However it definitely improves the restriction of range of motion in the immediate postoperative period, facilitating early shoulder exercises involving large muscles around the arm which might promote lymphatic drainage and reduce the risk of lymphoedema following ALND [20]. The arm circumference was not measured as an indicator of lymphoedema, as this was not the objective of the study.
Conclusion
This study establishes that infiltrative anesthesia with commonly available local anesthetic bupivaccaine along the nerve bundles exposed during ALND, is effective in improving the range of motion restriction of shoulder and pain relief, the most prevalent morbidities in the immediate postoperative period in patients of breast cancer. Further, larger prospective studies are required to fully assess the impact of infiltrative anesthesia on lymphoedema, the most undesirable side effect and quality of life following ALND.
References
- Amin MB, Edge S, Greene F, Byrd DR, Brookland RK, et al. (Eds.). AJCC Cancer Staging Manual (8th edn). Springer International Publishing: American Joint Commission on Cancer; 2017: 589-633.
- NCRP - Consolidated Report of Hospital Based Cancer Registries 2007-2011, National Cancer Registry Programme (Indian Council of Medical Research), Bangalore, 2013. Chapter 10:120-125.
- Berg JW (2001) The significance of axillary node levels in the study of breast carcinoma. Cancer; 8: 776-778.
- Warmuth MA, Bown G, Prosnitz LR, Chu L, Broadwater G, etal. (1998) Complications of axillary lymph node dissection for carcinoma of the breast: a report based on a patient survey. Cancer 83: 1362-1368.
- Pesce C, Morrow M (2013) The need for lymph node dissection in nonmetastatic breast cancer. Annu Rev Med 64: 119-129.
- Kissin MW, Thompsone M, Price AB (1982) The inadequacy of axillary sampling in breast cancer. Lancet 29: 1210-1212.
- Seigel BM, Mayzel MD, Love SM (1990) Level I and II axillary dissection in the treatment of early stage breast cancer. Arch Surg 125: 1144-1147.
- Veronesi U, Giovanni P, Galimberti V, Viale G, Zurrida S, et al. (1997) Sentinel-node biopsy to avoid axillary dissection in breast cancer with clinically negative lymph nodes. Lancet 349: 1864-1867.
- Emerson S, Hildebrando N, Luis B, Ademar D, Reginaldo J et al. (2014) Morbidity after conventional dissection of axillary lymph nodes in breast cancer patients. World J Surg Oncol 12: 67-74.
- Moore MP, Kinne DW (1997) Axillary lymphadenectomy: A diagnostic and therapeutic procedure. J Surg Oncol 66: 2-6.
- Fisher B, Wolmark N, Redmond C, Deutsch M, Fisher ER et al. (1981) Findings from NSABP Protocol No. B-04: Comparison of radical mastectomy with alternative treatments. II. The clinical and biologic significance of medial-central breast cancers. Cancer 48: 1863-1872.
- McGale P, Taylor C, Correa C, Cutter D, Duane F et al. (2014) EBCTCG (Early Breast Cancer Trialists� Collaborative Group): Effect of radiotherapy after mastectomy and axillary surgery on 10-year recurrence and 20-year breast cancer mortality: Meta-analysis of individual patient data for 8135 women in 22 randomised trials. Lancet 383: 2127-2135.
- Orr RK (1999) The impact of prophylactic axillary node dissection on breast cancer survival--A Bayesian meta-analysis. Ann Surg Oncol 6:109-116.
- Krag DN, Anderson SJ, Julian TB, Brown AM, Harlow SP et al. (2007) Technical outcomes of sentinel-lymph node resection and conventional axillary-lymph node dissection in patients with clinically node-negative breast cancer. Results from the NSABP B-32 randomised phase III trial. Lancet Oncol 8: 881-8.
- Husted Madsen A, Haugaard K, Soerensen J, Bokmand S, Friis E et al. (2008) Arm morbidity following sentinel lymph node biopsy or axillary lymph node dissection: A study from the Danish Breast Cancer Cooperative Group. Breast 17(2): 138-47.
- Ashikaga T, Krag DN, Land SR, Julian TB, Anderson SJ, et al. (2010) Morbidity results from the NSABP B-32 trial comparing sentinel lymph node dissection versus axillary dissection. J Surg Oncol 102(2): 111-8.
- Maunsell E, Brisson J, Deschenes L (2016) Arm problems and psychological distress after surgery for breast cancer. Can J Surg 36(4): 315-320.
- Shalaka J, Jarin N, Rohini H, Kundgulwar G, Vanmali V et al. (2019) Merits of Level III Axillary Dissection in Node-Positive Brest Cancer: A Prospective Single-Instituion Study from India. J Glob Oncol 5: 1-8.
- Asli C, Emel E, Fatima C (2019) Early Detection and Treatment of Subclinical Lymphedema in Patients with Breast Cancer. Lymphat Res Biol:1-6.
- MR Fu (2014) Breast cancer-related lymphedema: Symptoms, diagnosis, risk reduction, and management. World J Clin Oncol 5 (3): 241-247.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi