Research Article, J Spine Neurosurg Vol: 6 Issue: 4
Early Decompressive Craniectomy in Conjunction with Medical Management in the Treatment of Severe Traumatic Brain Injury: A Case Report
Michael J DeRogatis*, Monika Shenouda, Gudata Hinika, Paul S. Issack, Ariel Loo and Ayman M. Salem
Department of Orthopaedics, Ross University School of Medicine, Dominica
*Corresponding Author : Dr. Michael J. DeRogatis
Medical student and PAC, Ross University School of Medicine, Dominica
Tel: 4849035655
E-mail: michael.derogatis@gmail.com
Received: September 01, 2017 Accepted: September 07, 2017 Published: September 12, 2017
Citation: DeRogatis MJ, Shenouda M, Hinika G, Issack PS, Loo A, et al. (2017) Early Decompressive Craniectomy in Conjunction with Medical Management in the Treatment of Severe Traumatic Brain Injury: A Case Report. J Spine Neurosurg 6:4. doi: 10.4172/2325-9701.1000277
Abstract
We present the case of a 20-year-old man with an initial Glasgow Coma Scale (GCS) of 4 secondary to traumatic brain injury. Computed tomography of the head demonstrated diffuse cerebral edema, an acute right frontal subdural hematoma with a right-toleft midline shift, and subarachnoid hemorrhage. Surgical treatment with early decompressive craniectomy (DC) and duraplasty improved the patient’s GCS score to 15 by postoperative day 3. While there are several reports on early DC for traumatic brain injury, there has been as to date no level 1 evidence proving it to be superior to medical management alone or other neurosurgical techniques including routine temporparietal craniectomy.
Keywords: Decompressive craniectomy; Traumatic brain injury; Hemicraniectomy; Decompressive craniotomy
Introduction
Traumatic brain injury is the leading cause of death between the age of 1 and 45 in North America resulting in an economic burden of $9.2 billion in medical costs and $51.2 billion in productivity losses [1]. It is the most common cause of intracranial hypertension, which is the most frequent cause of death and disability [2]. In particular, subdural hematomas have an overall mortality rate of 55%. Furthermore, patients who present initially with a Glasgow Coma Scale (GCS) score of 4-6 have mortality rate of 45 to 67% [3]. Poor outcomes have been associated with advanced age, low GCS scores, and computed tomography (CT) findings of diffuse edema with a midline shift.
Medical management of traumatic brain injuries includes hypothermia, hyperosmolar therapy, cerebrospinal fluid drainage, and hyperventilation, use of sedatives including barbiturates, corticosteroids, and seizure prophylaxis. Intracranial hypertension however, can be refractory to medical management, leaving decompressive craniectomy as the only feasible therapeutic option. Decompressive craniectomy is performed to reduce intracranial pressure, reduce swelling, and increase cerebral blood flow. However, there is a lack of level 1 evidence demonstrating clinical improvement with decompressive craniectomy. Furthermore, the literature varies with regards to degree of reduction of intracranial pressure, complications after surgery, and prognosis when compared to medical management alone. Currently, there is no clear protocol for the management of severe traumatic brain injury. There are no specific indications for decompressive craniectomy endorsed by the AANS. We present a case of a patient who underwent decompressive craniectomy for a large subdural hematoma with rapid improvement in neurologic function. We also review the current literature on decompressive craniectomy versus medical management for traumatic brain injury. The patient was informed that data concerning the case would be submitted for publication, and the patient agreed.
Case Report
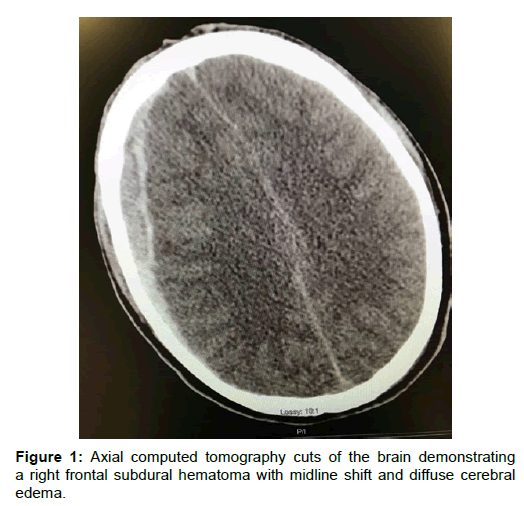
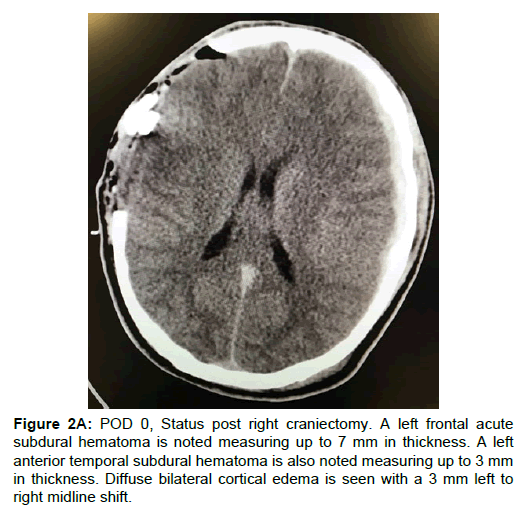
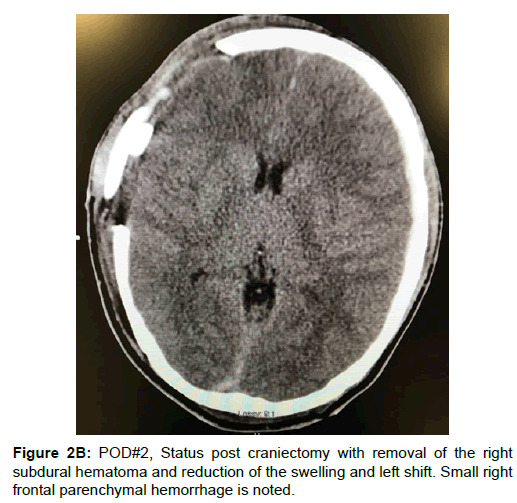
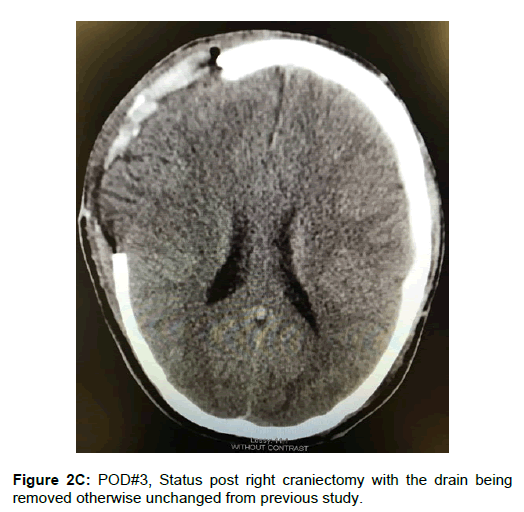
A twenty-year-old man struck by a motor vehicle while riding a bicycle was brought to our emergency department presenting with a severe head injury with Glasgow Coma Scale (GCS) score of 4. The patient was immediately intubated and stabilized following the Advanced Trauma Life Support (ATLS) protocol. After initial stabilization, a noncontrast head and neck CT was performed demonstrating diffuse cerebral edema, an acute right frontal subdural hematoma measuring 1.2 cm, a 6 mm right-to-left shift, and a subarachnoid hemorrhage (Figure 1). Upon noting these findings, immediate interventions were initiated including elevation of the head of the bed to 30 degrees, hyperosmolar therapy with mannitol, and anticonvulsant therapy with phenytoin, and hyperventilation. Given the patient’s young age, GCS score, and CT findings, neurosurgery was consulted for immediate decompressive craniectomy and hematoma evacuation. The patient was positioned supine with a shoulder roll, with the head was turned to the left side. The right frontotemporoparietal region was prepped and draped in the usual sterile fashion. Cefazolin was administered as prophylactic antibiotics. A U-shaped incision was made over the temporoparietal region of the skull. The right temporalis muscle and fascia were incised and elevated off the periosteum. Using a Midas-Rex drill, a free craniotomy bone flap was created in the right frontotemporoparietal region. The underlying dura was incised and the hematoma was evacuated. The subdural cavity was irrigated with an antibiotic based saline solution and hemostasis was achieved using bipolar cautery. Duraplasty was achieved using Suturable DuraGen™ (Integra Neuroscience). Two Jackson-Pratt drains were placed superficial to the duraplasty. The temporalis muscle was reapproximated loosely to the fascia. The bone flap was preserved for possible future reimplantation. The patient was then transferred to the ICU in a guarded condition. Immediately postoperative CT demonstrated right craniectomy with an acute left subdural hematoma, which was not visible on preoperative CT (Figure 2A). On postoperative day 1, the patient improved to a GCS score of 7 and the patient was extubated. On physical examination, the patient was awake, alert, and oriented to person and time. His pupils were 3mm, equal, round, reactive to light and accommodation. On postoperative day 2, the patient showed significant signs of improvement, a stable neurological exam and a GCS score of 13. Head CT at this time showed a stable right subdural hematoma and resolving left subdural hematoma without midline shift (Figure 2B). On postoperative day 3, the two Jackson-Pratt drains were removed without complication. CT showed resolution of the previous left subdural hematoma (Figure 2C). The patient was now alert and oriented to person, place and time. His GCS score improved to 15. On postoperative day 4, he was transferred to the surgical floor and was out of bed ambulating with physical therapy. Upon discharge on postoperative day 10, he maintained a GCS of 15, was neurologically intact, and able to ambulate without assistance.
Figure 2A: POD 0, Status post right craniectomy. A left frontal acute subdural hematoma is noted measuring up to 7 mm in thickness. A left anterior temporal subdural hematoma is also noted measuring up to 3 mm in thickness. Diffuse bilateral cortical edema is seen with a 3 mm left to right midline shift.
Discussion
There are few studies which compare decompressive craniectomy with medical management for traumatic brain injury. Cooper and colleagues compared bifrontotemporoparietal decompressive craniectomy (DC) to standard care without decompressive craniectomy in a randomized control study of 155 patients with traumatic brain injury. Fifty-eight percent (42) of the DC group had traumatic subarachnoid hemorrhage and 59% (48) in the standard care group; the diagnosis of the 65 patients was not mentioned. The median age was 24 years in the DC group and 25 years in the standard care group. All patients received standard medical care for the first 72-hours and then were randomly assigned to different groups. Seventy-three patients underwent DC and 82 received standard medical care with sedation, hyperventilation, mannitol, hypertonic saline, neuromuscular blockage, barbiturates, and external ventricular drainage. Patients who underwent DC had lower mean intracranial pressure (14.4 +/- 6.8) compared to those who underwent nonsurgical therapy (19.1 +/- 8.9, p<0.001). Patients who underwent DC spent a mean of 13 days (range, 10-18 days) in the intensive care unit compared to 18 days for patients who underwent medical treatment (range, 13-24) (p<0.001). However, patients who underwent DC demonstrated a worse Extended Glasgow Outcome Scale (GOS-E) at 6 months compared to patients who underwent medical management alone (p=0.03). A greater risk for unfavorable outcomes (p = 0.02) in the DC group was recorded, but after post hoc adjustment for pupil reactivity at baseline the differences between the groups were not significant. Six month mortality rate was similar in both groups (18% versus 19%). Limitations of this study were that it was not a blind study, one of the 15 centers received a third of participants, and the DC group had significantly more nonreactive pupils upon admission [4].
There are also few investigations comparing DC to other neurosurgical procedures for traumatic brain injury. Qiu and colleagues observed that larger unilateral frontotemporoparietal DC (>15 cm diameter) had a lower mortality rate at one month versus unilateral routine temporoparietal craniectomy (27% vs. 57%, p=0.01). They reported on 74 traumatic brain injuries with >5 mm midline shift. The mean age was 40 years in both groups. They reported 10 cases of traumatic cerebral hemisphere infarction, 13 cases of subdural hematoma, 19 cases with midline shift more than 10 mm, and 51 cases with compressed or obliterated basilar cistern and/or cisterna ambiens. All patients were medically managed with elevation of head 15-30 degrees, mannitol, hyperventilation, steroids, and sedation preoperatively for 24 hours prior to surgery and postoperatively [5]. Jiang et al. also reported similar findings with a mortality rate of 26% for 241 patients who underwent a larger DC compared to 36% for 245 patients treated with routine temporoparietal craniectomy (p=0.05). Patients included in this study had a GCS score < 9 with refractory intracranial hypertension. The mean age was 44 year in the larger DC group and 45 years in the routine temporoparietal craniectomy group. CT findings were consistent with 185 contusions, 297 contusions with hematoma, and 239 traumatic subarachnoid hemorrhages. All patients received medical care with mannitol, furosemide, and pressors when necessary to maintain a mean arterial pressure of 90-120 mm Hg. Both studies showed an improvement in overall neurological outcomes one year post injury measured by GOS-E. Jiang and colleagues concluded that 40% of larger DC had a GOS-E of 4 or 5 indicating good recovery vs 29% in smaller DC (p=0.05) [6]. Qiu et al. reported good neurological outcomes (GOS-E 4 or 5) in 57% larger DC versus 32% of smaller DC (p=0.035). Although there were no severe complications attributed to decompressive craniectomy, the incidence of delayed intracranial hematoma and subdural effusion were higher in the group that received a larger DC (21.6% and 10.8% versus 5.4% and 0%; respectively p=0.041 and p=0.040) [5]. These studies conclude that larger DC reduced mortality rate and improved neurological outcomes in patients with acute traumatic brain injury as compared to unilateral routine temporoparietal craniectomy. However, larger frontotemporoparietal DC had higher rates of complications compared to routine temporoparietal craniectomy.
Huang and colleagues retrospectively reviewed 38 DC with duraplasty and 16 craniotomies for treatment of patients with GCS scores < 9, hemorrhagic cerebral contusions >20 cm3, midline shift >4 mm, and/or cisternal compression on CT. Initial CT showed 34 (63%) subdural hematomas, 44 (82%) subarachnoid hemorrhage, and 24 (44%) brainstem compression. All patients received pre- and postoperative care including, mannitol, furosemide, hyperventilation, and anticonvulsants. The mean age for patients who underwent craniotomy was 46 years and 42 years in the DC group. Overall, there was no significant difference in mortality rate in DC vs craniotomy (13% vs 25%, respectively), but reoperation rates were higher in patients who had a craniotomy (38% vs 8%, p < 0.05). GOS-E scores were higher in patients who had DC compared to craniotomy (5.55 +/- 2.34 vs. 3.56 +/- 2.37, p<0.005). The limitation associated with this study was the small number of patients in the craniotomy group [7]. Soukiaasian et al. also reported no significant difference in mortality when comparing DC vs. craniotomy (52% vs 79%, p=0.08), however complications were more frequent in patients who underwent DC. Complications included collapse of basilar cisterns (30% vs 4%, p=0.0001) and herniation (17% vs. 5%, p=0.05) [8].
Akyuz and colleagues followed 76 patients with severe traumatic brain injury, in which 36 patients received late DC as a second tier treatment versus the remaining 40 patients who received early DC as a first tier treatment. Early DC resulted in a lower intracranial pressure postoperatively (17.2 +/- 3.5 vs. 23.3 +/- 3.5). Mortality was significantly higher in late DC (44% vs. 13%, p=0.0018). Early DC resulted in good neurological recovery (GOS of 4 or 5) in 50% of patients, compared to only 28% of late DC (p=0.047) [9]. However, Wen and colleagues followed 44 patients in a prospective cohort study in which 25 patients received early DC and 19 patients underwent late DC. The mean age for patients who underwent early DC was 47 years and 50 years in the late DC group. Early DC was performed within 24 hours and medical therapy of hyperventilation, barbiturates, hypothermia, and ventricular cerebrospinal fluid draining was not performed preoperatively. Late DC was performed after 24 hours as a second-tier treatment. This study demonstrated that there was no significant difference in mortality rate one month and six months or in the outcomes post-surgery [10].
Danish and colleagues performed a meta-analysis of 29 studies, consisting of 1422 cases of patients who underwent decompressive hemicraniectomy for traumatic brain injury in adults. They reported a pooled mean 6 month postoperative mortality rate of 28% (range 23.5–33%), pooled mean GOS score of 4 (moderate disability) in 59% of patients (range 0.526–0.658), and favorable outcomes (GOS score 4 or 5) noted in 46% of all patients (95% CI 34.5–57.5%) and 63% of survivors (95% CI 59.9–66.7%). These results cannot account for the heterogeneity of the patient demographics, indications for surgery, and surgical techniques. Also, they were unable to adjust the outcomes for patients’ age, timing of surgery, admission GCS score, or preoperative pupillary examination. However, given that the majority of the survivors fall in the moderate disability category, the authors conclude that patients with increased intracranial pressure in the setting of traumatic brain injury can have good restoration of cerebral function after decompressive hemicraniectomy [11].
In our patient, early decompressive craniectomy was performed because he developed a large subdural hematoma with midline shift and initial GCS of 4. In spite of the reportedly high mortality rates of an initial GCS of 4 in traumatic brain injuries, our patient rapidly improved to a GCS of 15 by postoperative day 3. This case suggests that medical management including hyperventilation, hyperosmotic therapy, anticonvulsant therapy, and head-of-bed elevation in conjunction with early decompressive craniectomy can result in good outcomes for severe traumatic brain injuries. Although this is only one case, the good outcomes with this approach may be the combined use of medical and surgical modalities, as many previously reported cases performed DC only when medical treatment failed or perform DC without medical management. Larger prospective studies are necessary to determine the effectiveness of early DC combined with medical management in restoring cerebral function in severe traumatic brain injury.
Declaration
Investigation performed at Dignity Health California Hospital Medical Center, 1401 S Grand Ave Los Angeles, CA 90015, Phone (213) 401-6505
References
- Rutland-Brown W, Langlois JA, Thomas KE, Xi YL (2006) Incidence of traumatic brain injury in the United States, 2003. J Head Trauma Rehabil 21: 544.
- Alvis-Miranda H, Castellar-Leones SM, Moscote-Salazar LR (2013) Decompressive Craniectomy and Traumatic Brain Injury: A Review. Bull Emerg Trauma 1: 60-68.
- Hatashita S, Koga N, Hosaka Y, Takagi S (1993) Acute subdural hematoma: severity of injury, surgical intervention, and mortality. Neurol Med Chir (Tokyo) 33: 13.
- Cooper DJ, Rosenfeld JV, Murray L (2011) Decompressive craniectomy in diffuse traumatic brain injury. N Engl J Med 365: 2040.
- Qiu W, Guo C, Shen H (2009) Effects of unilateral decompressive craniectomy on patients with unilateral acute post-traumatic brain swelling after severe traumatic brain injury. Crit Care 13: R185.
- Jiang JY, Xu W, Li WP (2005) Efficacy of standard trauma craniectomy for refractory intracranial hypertension with severe traumatic brain injury: a multicenter, prospective, randomized controlled study. J Neurotrauma 22: 623-628.
- Huang AP, Tu YK, Tsai YH (2008) Decompressive craniectomy as the primary surgical intervention for hemorrhagic contusion. J Neurotrauma 25: 1347-1354.
- Soukiasian HJ, Hui T, Avital I (2002) Decompressive craniectomy in trauma patients with severe brain injury. Am Surg 68: 1066-1071.
- Akyuz M, Ucar T, Acikbas C, Kazan S, Yilmaz M, et al. (2010) Effect of early bilateral decompressive craniectomy on outcome for severe traumatic brain injury. Turk Neurosurg. 20: 382-389.
- Wen L, Wang H, Wang F (2011) A prospective study of early versus late craniectomy after traumatic brain injury. Brain Inj 25: 1318-1324.
- Danish SF, Barone D, Lega BC, Stein SC (2009) Quality of life after hemicraniectomy for traumatic brain injury in adults. Neurosurg Focus 36: E2
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi