Research Article, J Spine Neurosurg Vol: 7 Issue: 2
Early Outcomes of Anterior Cervical Discectomy and Fusion Using a Porous PEEK Interbody Fusion Device
J Kenneth Burkus*
Spine Service, Hughston Clinic, Columbus, Georgia
*Corresponding Author : Dr. Kenneth Burkus J
The Hughston Clinic, 6262 Veterans Parkway, Columbus, Georgia, 31909
Tel: 706-326-6661
Fax: (706) 494-3102
E-mail: jkb66@knology.net
Received: March 16, 2018 Accepted: April 16, 2018 Published: April 25, 2018
Citation: Burkus JK (2018) Early Outcomes of Anterior Cervical Discectomy and Fusion Using a Porous PEEK Interbody Fusion Device. J Spine Neurosurg 7:2. doi: 10.4172/2325-9701.1000295
Abstract
Background: Recently, porous surface polyether-etherketone (PEEK) implants have been developed to interact with adjacent endplates. Our goal was to evaluate the efficacy of novel high-strength, porous PEEK interbody fusion devices for anterior cervical discectomy and fusion (ACDF) in patients with symptomatic single-level and multilevel degenerative cervical disc disease at 1 year. Methods: Fifty consecutive patients (31 women, 19 men; average age, 60 years) with degenerative cervical disc disease underwent ACDF using a porous PEEK interbody implant and plate. There were 11 1-level; 23 2-level, and 16 3-level fusions between C3 and C7. Patients were assessed at 1.5, 3, 6, and 12 months. Standardized outcome measures were used to evaluate the patient’s condition before and after surgery. Plain radiographs were used to assess fusion, bony in-growth, subsidence, and implant migration. Sagittal plane angulation was measured on neutral lateral radiographs and determined by Cobb’s criteria. Intradiscal distraction and subsidence were measured by assessing the vertical distance between the midpoints of the adjacent vertebral endplates. Results: At 12 months after surgery, all patients showed improvement in Oswestry Neck Disability Index and neck and arm pain scores. Similarly, at 12 months, all patients showed radiographic fusion. No patient demonstrated motion across the interspace on flexion-extension lateral radiographs. Sagittal plane angulation improved to an average of -6° (range, -2° to -8°) with no measurable evidence of implant migration or subsidence. Average disc space height increased more than 4 mm. No patient had measurable radiographic evidence of a pseudarthrosis or halo formation around the implant. Conclusion: One-year results in this prospective nonrandomized study show that porous-surface PEEK is a clinically viable alternative for improving osseointegration and fusion rates of interbody implants to treat degenerative cervical disc disease.
Keywords: Porous PEEK; Anterior cervical interbody fusion; Cervical arthrodesis; Degenerative disc disease
Introduction
Indications for surgery involving degenerative conditions in the cervical spine are progressive neurologic deficits and intractable painful symptoms following an adequate course of non-operative treatment modalities and therapy. Intractable or recurrent painful symptoms and motor weakness can be treated with surgical decompression. Anterior cervical discectomy and interbody fusion (ACDF) can effectively decompress the central spinal canal by direct removal of disc fragments, radial osteophytes and uncovertebral spurs [1]. The anterior approach can also enlarge canal and neuroforamina by restoring normal segmental lordosis and by reestablishing normal disc height [2,3]. This procedure has been found to be safe and reliable in achieving long-term improved functional outcomes, neurologic recovery and patient satisfaction [4-6].
After anterior cervical decompression, the goals of interbody fusion are to provide permanent segmental stabilization and to maintain segmental lordosis and anatomic disc space height. Numerous grafting and stabilization techniques have been described to meet these goals [7-9]. Allograft bone was introduced as an alternative to autograft; it was promoted to reduce the need for a second surgical site [9-13]. Allograft has been shown to be comparable to autograft with regard to efficacy when used for ACDF in onelevel instrumented procedures [12,13]. Importantly, reported fusion rates for allograft often fall below the fusion rate for autograft [14]. Additionally, reduced fusion rates have been reported with smokers diabetics, and multilevel and revision surgeries [15-18].
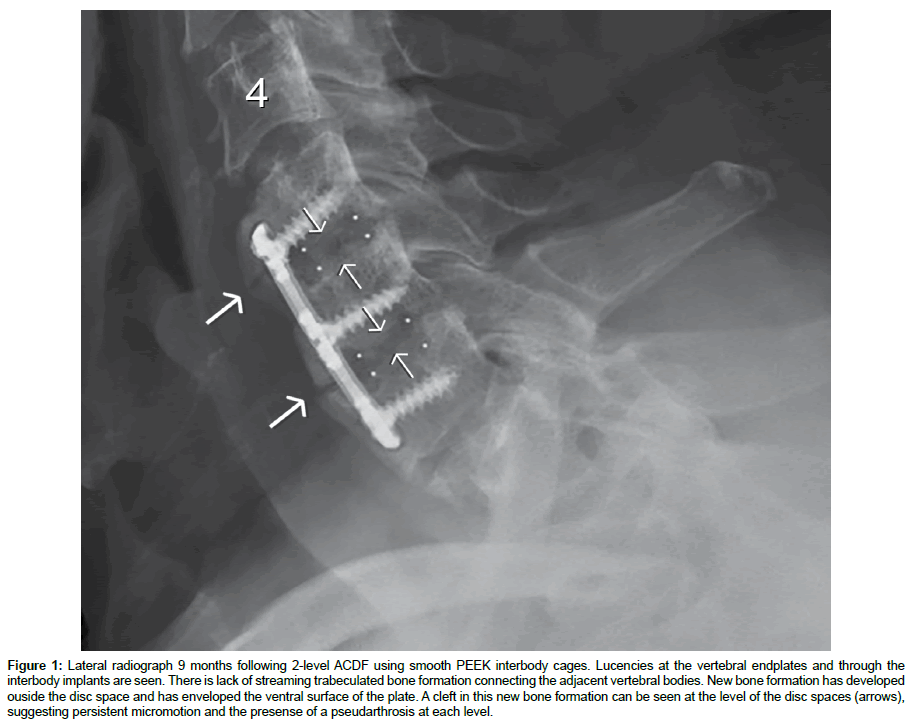
Synthetic cervical interbody spacers have been developed to enhance fusion rates and improve clinical outcomes while avoiding the complications associated with allograft constructs. Polyetheretherketone (PEEK) has been utilized in cervical interbody constructs because of its high strength, radiolucency and a modulus of elasticity comparable to bone [19,20]. However, smooth-surfaced PEEK implants have been associated with fibrous tissue formation, migration, and subsidence (Figure 1) [21]. Several other materials have been used to create synthetic interbody spacers, including rough and porous titanium spacers that promote better bone on-growth and in-growth directly surrounding the implant, creating an environment that reduces the potential of fibrous tissue formation. Unfortunately, most of these materials, especially titanium, are much stiffer than bone, causing stress-shielding and increased rates of subsidence [22].
Figure 1: Lateral radiograph 9 months following 2-level ACDF using smooth PEEK interbody cages. Lucencies at the vertebral endplates and through the interbody implants are seen. There is lack of streaming trabeculated bone formation connecting the adjacent vertebral bodies. New bone formation has developed ouside the disc space and has enveloped the ventral surface of the plate. A cleft in this new bone formation can be seen at the level of the disc spaces (arrows), suggesting persistent micromotion and the presense of a pseudarthrosis at each level.
Recently, porous surface PEEK implants have been developed with porosity on the inferior and superior sides of the device to interact with adjacent endplates. In vitro and in vivo data have demonstrated enhanced osteoconductivity and osseointegration, respectively, of the porous PEEK interbody fusion devices, and mechanical testing has shown the porous architecture to withstand significant clinically relevant stresses [23,24]. Our goal was to evaluate the clinical outcomes, radiographic outcomes, and fusion status in patients undergoing anterior cervical surgery in which a porous PEEK implant was used to treat degenerative cervical disc disease.
Materials and Methods
Fifty consecutive patients underwent anterior cervical discectomy and fusion (ACDF) using a porous PEEK interbody fusion device for degenerative cervical disc disease. All study subjects were adults (>18 years old) with symptomatic degenerative cervical disc disease between the C3 and C7 levels with intractable radiculopathy, myelopathy, or both. Patients had neck and arm pain for a minimum of 6 weeks before surgery that was recalcitrant to non-operative treatment modalities, such as physical therapy, reduced activities, and anti-inflammatory medications. All patients had radiographic evidence of cervical disc disease that was documented on dynamic radiographs and magnetic resonance imaging (MRI).
Data were collected preoperatively, intraoperatively, and at routine postoperative intervals of 1.5, 3, 6, and 12 months. Operative procedure details and adverse events were recorded. Both safety and effectiveness aspects of the treatments were assessed. Standardized outcome measures—the Neck Disability Index (NDI) and neck pain and arm pain numeric rating scales—were used to evaluate the patient’s condition before and after surgery [25,26]. Neurological status was also documented.
Plain radiographs were used to assess fusion, bony in-growth, subsidence and migration of the implant. Neutral anteroposterior and lateral radiographs and dynamic flexion-extension lateral radiographs were obtained at each study point. Sagittal plane angulation was measured on neutral lateral radiographs and determined by Cobb’s criteria. Intradiscal distraction and subsidence were measured by assessing the vertical distance between the midpoints of the adjacent vertebral endplates.
Fusion success was defined radiographically by meeting 3 criteria: 1) evidence of bridging bone (based on radiographic evidence of a continuous bony connection from the superior vertebral body to the inferior vertebral body; 2) evidence of radiolucency (no greater than 25% of the superior or inferior implant-vertebral interface); and 3) no evidence of motion (≤ 4° of sagittal angulation on flexion-extension radiographs). Thin-cut computed tomography (CT) scans with sagittal and coronal reconstructions were used to confirm the intradiscal fusion mass in patients with unresolved clinical symptoms. Independent radiographic reviewers were used to determine fusion status.
Results
The patient cohort included 31 women and 19 men with an average age of 60 years (range, 42 to 75 years) (Table 1). There were 11 1-level, 23 2-level and 16 3-level fusions between C3 and C7.
| Variable | Number of Patients (%) |
|---|---|
| Number of patients | 50 (100) |
| Mean age (years) | 60.1 |
| Sex Female Male |
19 (38%) 31 (62%) |
| Unresolved spinal litigation Yes No |
9 (18%) 41 (82%) |
| Tobacco use Yes No |
12 (24%) 38 (76%) |
| Alcohol use Yes No |
39 (78%) 11 (22%) |
| Preoperative work status Yes No |
31 (62%) 19 (38%) |
| Obesity (>30 BMI) Yes No |
20 (40%) 30 (60%) |
| Diabetes Yes No |
7 (14%) 43 (86%) |
Table 1: Patient demographic data.
Clinical outcomes
Numeric rating scales were used to assess neck and arm pain intensity. Patients rated their neck pain duration and intensity on a scale from 0-10, with a score of 0 representing “no pain” and a score of 10 representing “pain as bad as it could be”. After surgery, mean neck and arm pain scores showed improvement from preoperative scores. Average neck pain score improved from 6.7 to 3.4 points; average arm pain score improved from 7.1 to 2.9 points. Neurological success was defined as maintenance or improvement in three objective clinical findings (sensory, motor, and reflex testing). Neurological success was seen in all patients studied with no patients showing a loss in neurological functioning. The NDI questionnaire was used to measure the effects of neck pain associated with activities of daily living [25,26]. Preoperative NDI scores averaged 53.4 points. At last follow-up examination, NDI scores averaged 24.3 points, which was a mean overall improvement of 29.1 points compared with preoperative scores.
Radiographic outcomes
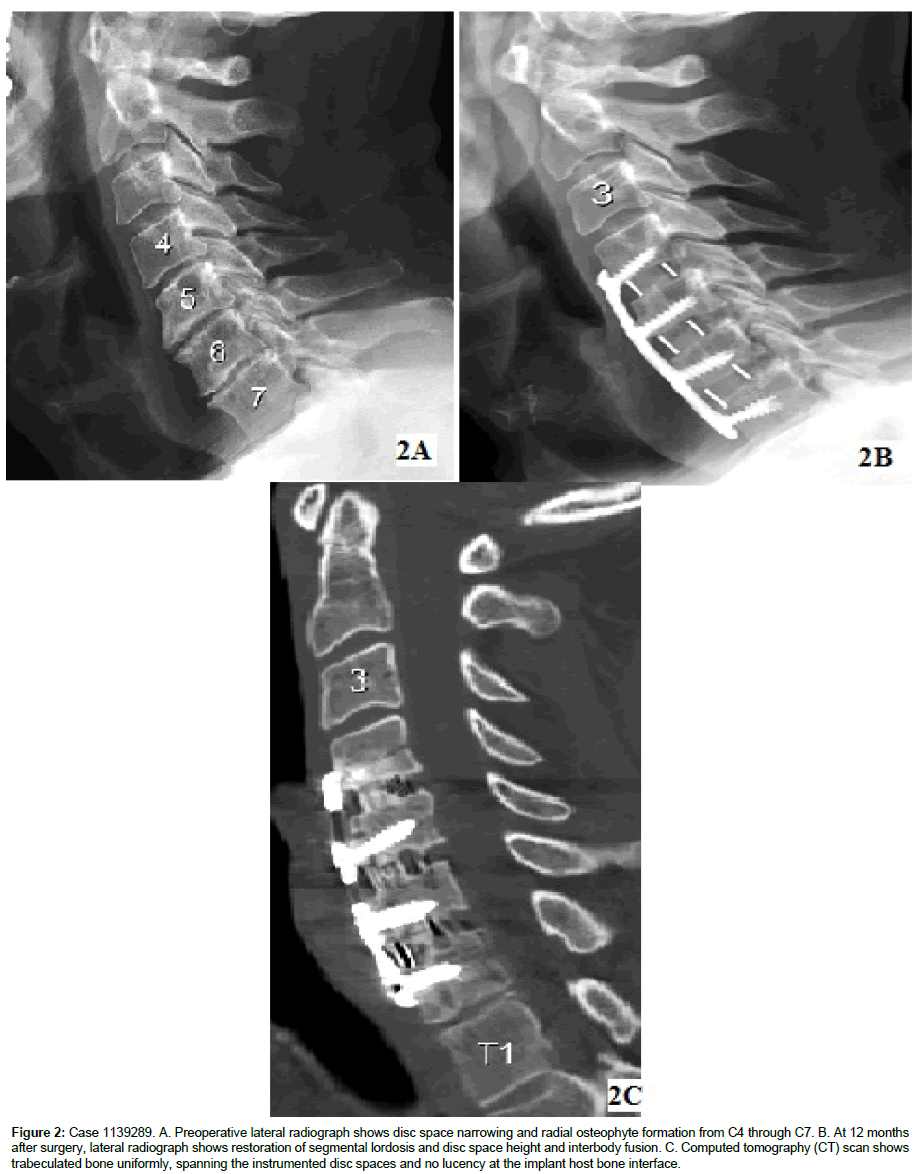
All patients showed successful radiographic fusion at 6 months after surgery with no motion across the interspace on lateral flexionextension radiographs and no deterioration in fusion status was seen between 6 and 12 months (Figure 2). At 12 months, the sagittal plane angulation improved to an average of -6° (range, -2° to -8°). There was no evidence of implant migration or subsidence. No patient showed radiographic evidence of a pseudarthrosis; no patient exhibited halo formation around the implant. The average disc space height increased more than 4 mm after surgery and was maintained through the last follow-up examination at 12 months.
Figure 2: Case 1139289. A. Preoperative lateral radiograph shows disc space narrowing and radial osteophyte formation from C4 through C7. B. At 12 months after surgery, lateral radiograph shows restoration of segmental lordosis and disc space height and interbody fusion. C. Computed tomography (CT) scan shows trabeculated bone uniformly, spanning the instrumented disc spaces and no lucency at the implant host bone interface.
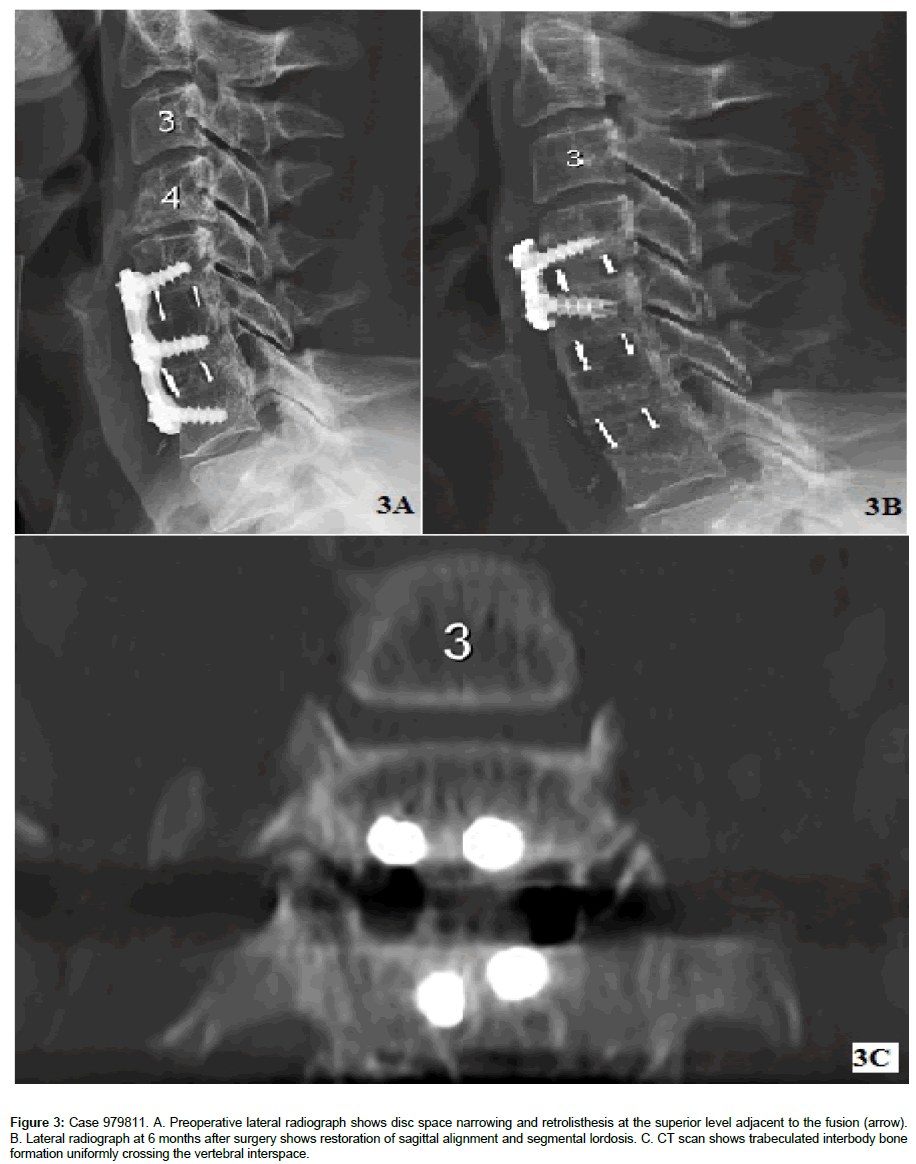
Subgroup analysis compared patients with risk factors for pseudarthrosis (current or former smokers, previous surgery, diabetes, or obesity) with those without risk factors and showed no differences in fusion rates [15,16,18]. Thirteen patients had undergone a previous anterior cervical fusion procedure. All patients who had revision surgery with porous PEEK implants went on to achieve fusion within 6 months of surgery (Figure 3).
Figure 3: Case 979811. A. Preoperative lateral radiograph shows disc space narrowing and retrolisthesis at the superior level adjacent to the fusion (arrow). B. Lateral radiograph at 6 months after surgery shows restoration of sagittal alignment and segmental lordosis. C. CT scan shows trabeculated interbody bone formation uniformly crossing the vertebral interspace.
Adverse events
No adverse events were identified at surgery or during the course of follow-up. No disruption of the porous surface was observed during intraoperative impaction of the interbody cages. No patient underwent additional surgical procedures; none had supplemental posterior fixation.
Discussion
Spine fusion, creating new bone formation across a spinal motion segment, occurs in a challenging healing environment. During the spinal fusion procedure, bioactive materials are placed in varying anatomic positions spanning an intervertebral motion segment. A successful fusion induces new bone formation that bridges an anatomic region of the spine that normally does not support viable bone. Fusion in the spine is a complex process that does not always heal successfully. This process is, in part, a race among resorption of the graft material, cellular apoptosis, and formation of new bone growing through the graft connecting the 2 adjacent mobile vertebral bodies.
Intradiscal spinal implants are used to maintain spinal alignment and encourage new bone formation and fusion. Metallic implants provide high strength but are associated with medical imaging artifacts and unwanted bone resorption due to their high modulus and corresponding stress shielding [22]. Bioresorbable polymers and composites facilitate osseointegration and implant resorption, but they are clinically limited to soft tissue reconstructions and are associated with high incidences of prolonged inflammation, migration, incomplete degradation, and implant breakage [27].
As a relatively new implant material, PEEK has gained widespread acceptance as a high-strength polymer used primarily in spinal fusions and soft tissue reconstructions. It has favorable imaging compatibility and stiffness that closely matches bone. However, PEEK suffers a key property tradeoff of poor osseointegration. Although it provides high strength and biocompatibility, its hydrophobic and chemically inert surface limits local bone attachment. The use of bioactive osteogenic proteins can overcome the short comings of smooth PEEK interbody implants in cervical spine fusions [28].
Recently, PEEK implants have been modified to enhance osseointegration by surface modification and bulk porosity [20,23]. Porous PEEK implants have exhibited increased osseointegration in animal models. Porous PEEK is associated with a more differentiated bone cell phenotype in vitro and greater implant fixation in vivo compared to Smooth PEEK.
Conclusion
These findings from the initial study of porous PEEK implants used in cervical fusions suggest this new material is a clinically viable alternative for improving osseointegration. The results of this interim study suggest that in the treatment of degenerative disc disease, porous PEEK interbody devices could become clinically viable implant alternatives for achieving successful clinical and radiological outcomes in ACDF surgery—particularly in multilevel or revision surgeries where fusion rates are lower.
Financial Disclosure
The author reports no conflict of interest concerning the materials or methods used in this study or the findings specified in this paper. Research funding was provided to the Hughston Foundation, a nonprofit organization, with which the author is affiliated.
References
- Southwick WO, Robinson RN (1959) Surgical approaches to the vertebral bodies in the cervical and lumbar regions. J Bone Joint Surg 39A: 631-643.
- Katsuura A, Hukuda S, Imanaka T, Miyamoto K, Kanemoto M (1996) Anterior cervical plate used in degenerative disease can maintain cervical lordosis. J Spinal Disorders 9: 470-476.
- Krag MH, Robertson RA, Johnson CC, Stein AC (1997) Anterior cervical fusion using a modified tricortical bone graft: a radiographic analysis of outcome. J Spinal Disord 10: 420-430.
- Bohlman HH, Emery SE, Goodfellow DB, Jones PK (1993) Robinson anterior cervical discectomy and arthrodesis for cervical radiculopathy: Long-term follow up of one hundred and twenty-two patients. J Bone Joint Surg 75A: 1298-1307.
- Emery SE, Bolesta MJ, Banks MA, Jones PK (1994) Robinson anterior cervical fusion: Comparison of the standard and modified techniques. Spine (Phila Pa 1976) 19: 660-663.
- Gore DR, Sepic SB (1984) Anterior cervical fusion for degenerated or protruded discs. A review of one-hundred forty-six patients. Spine (Phila Pa 1976) 9: 667-671.
- Brodke DS, Zdeblick TA (1992) Modified Smith-Robinson procedure for anterior cervical discectomy and Fusion. Spine (Phila Pa 1976) 17: S427-S430.
- Brown JA, Havel P, Ebraheim N, Greenblott SM, Jackson WT (1988) Cervical stabilization by plate and bone fusion. Spine (Phila Pa 1976) 13: 236-240.
- Connolly PJ, Esses SI, Kostuik JP (1996) Anterior cervical fusion: Outcome analysis of patients with and without anterior cervical plates. J Spinal Disord 9: 202-206.
- Arnold PM, Anderson KK, Selim A, Dryer RF, Burkus JK (2016) Heterotopic ossification following single-level anterior cervical discectomy and fusion: results from the prospective, multicenter, historically controlled trial comparing allograft to an optimized dose of rhBMP-2. J Neurosurg Spine 25: 1–11.
- Miller LE, Block JE (2011) Safety and effectiveness of bone allografts in anterior cervical discectomy and fusion surgery. Spine (Phila Pa 1976) 36: 2045-2050.
- Shapiro S (1996) Banked fibula and the locking anterior cervical plate in anterior cervical fusions following cervical discectomy. J Neurosurg 84: 161-165.
- Zdeblick TA, Ducker TB (1991) The use of freeze-dried allograft bone for anterior cervical fusions. Spine (Phila Pa 1976) 16: 726-729.
- Buttermann GR (2018) Anterior cervical discectomy and fusion outcomes over 10 Years: a prospective study. Spine (Phila Pa 1976) 43: 207-214.
- Berman D, Oren JH, Bendo J, Spivak J (2017) The effect of smoking on spinal fusion. Int J Spine Surg 11: 29.
- Brown CW, Orme TJ, Richardson HD (1986) The rate of pseudarthrosis in patients who are smokers and patients who are not smokers: A comparison study. Spine (Phila Pa 1976) 11: 942-943.
- Basques BA, Ondeck NT, Geiger EJ, Samuel AM, Lukasiewicz AM, et al. (2017) Differences in short-term outcomes between primary and revision anterior cervical discectomy and fusion. Spine (Phila Pa 1976) 42: 253-260.
- Veeravagu A, Cole T, Jiang B, Ratliff JK (2014) Revision rates and complication incidence in single- and multilevel anterior cervical discectomy and fusion procedures: an administrative database study. Spine J 14: 1125-1131.
- Liu JM, Xiong X, Peng AF, Xu M, Chen XY, et al. (2017) A comparison of local bone graft with PEEK cage versus iliac bone graft used in anterior cervical discectomy and fusion. Clin Neurol Neurosurg 155: 30-35.
- Tumialán LM, Pan J, Rodts GE, Mummaneni PV (2008) The safety and efficacy of anterior cervical discectomy and fusion with polyetheretherketone spacer and recombinant human bone morphogenetic protein-2: a review of 200 patients. J Neurosurg Spine 8: 529-535.
- Kersten RF, Gaalen SM, deGast A, Öner FC (2015) Polyetheretherketone (PEEK) cages in cervical applications: a systematic review. Spine J 15: 1446-1460.
- Seaman S, Kerezoudis P, Bydon M, Torner JC, Hitchon PW (2017) Titanium vs. polyetheretherketone (PEEK) interbody fusion: Meta-analysis and review of the literature. J Clin Neurosci 44: 23-29.
- Evans NT, Torstrick FB, Lee CS, Dupont KM, Safranski DL, et al. (2015) High-strength, surface-porous polyether-ether-ketone for load-bearing orthopedic implants. Acta Biomater 13: 159-167.
- Torstrick FB, Evans NT, Stevens HY, Gall K, Guldberg RE (2016) Do surface porosity and pore size influence mechanical properties and cellular response to PEEK? Clin Orthop Relat Res 474: 2373-2383.
- Vernon H, Mior S (1991) The Neck Disability Index: a study of reliability and validity. J Manipulative Physiol Ther 14: 409-415.
- Westaway M, Stratford P, Binkley J (1998) The patient-specific functional scale: validation of its use in persons with neck dysfunction. J Orthop Sports Phys Ther 27: 331-338.
- Torstrick FB, Safranski DL, Burkus JK, Chappuis JL, Lee CSD, et al. (2017) Getting PEEK to stick to bone: the development of porous PEEK for interbody fusion devices. Tech Orthop 32: 158-166.
- Burkus JK, Dryer RF, Arnold PM, Foley KT (2017) Clinical and radiographic outcomes in patients undergoing single-level anterior cervical arthrodesis: a prospective trial comparing allograft to a reduced dose of rhBMP-2. Clin Spine Surg 30: E1321-E1332.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi