Research Article, J Soil Sci Plant Health Vol: 6 Issue: 2
Economics of a Fungicide to Manage Rice Sheath Blight in Arkansas
Wamishe Y1*, Watkins KB2, Hardke JT3, Belmar S1, Gebremariam T1 and Kelsey C1
1Department of Plant Pathology, University of Arkansas system ,Rice Research and Extension Center, Stuttgart, Arkansas, United States
2Department of Agricultural Economics and Agribusiness, Rice Research and Extension Center, Stuttgart, Arkansas, United States
3Department of Crop, Soil, and Environmental Sciences, Rice Research and Extension Center, Stuttgart, Arkansas, United States
*Corresponding Author:Wamishe Y
Department of Plant Pathology, University of Arkansas system ,Rice Research and Extension Center, Stuttgart, Arkansas, United States
E-mail: ywamishe@uada.edu
Received date: 02 February, 2022, Manuscript No. JSPH-22-53149;
Editor assigned date: 04 February, 2022, Pre QC No. JSPH-22-53149 (PQ);
Reviewed date: 11 February, 2022, QC No JSPH-22-53149;
Revised date: 18 February, 2022, Manuscript No. JSPH-22-53149 (R);
Published date: 28 February, 2022, DOI: 10.4172/2325-9639.1000149
Citation: Wamishe Y, Watkins K, Hardke JT, Belmar S, Gebremariam T and Kelsey C (2022) Economics of a Fungicide to Manage Rice Sheath Blight in Arkansas. J Soil Sci Plant Health 6:2.
Keywords: Economics; Fungicide; sheath blight management; Rice; Arkansas
Keywords
Economics; Fungicide; sheath blight management; Rice; Arkansas
Introduction
In global food supply, rice (Oryza sativa L.) is a staple food for more than half of the world’s population [1,2]. The United States of America (USA), although it produces less than 2% of global rice, is a major exporter of rice for up to 6 % of the annual global rice trade [3]. Among southern rice producing states of the USA, Arkansas ranks first in rice production [4]. Arkansas produces close to 50% of the nation’s rice and more than 60% of the rice produced in Arkansas is exported [5].
Sheath blight (Rhizoctonia solani AG1-1A Kühn) is a major disease of rice worldwide and is the most widespread disease in Arkansas and other southern rice producing states of the USA [6-9]. Sheath blight in rice is caused by a soil-borne multinucleate and necrotrophic fungus. The fungus has several other hosts including soybeanGlycine max(L.) Merr), sorghum (sorghum bicolor), corn (Zea mays), sugarcane (Saccharum officinarum), several turf grasses, weed hosts such as barnyard grass (Echinochloa spp.), crabgrass (Digitaria spp.), and broadleaf signal grass (Brachiariaspp.) among others [10,11].
The fungus survives in soil as mycelia or mycelial masses known as sclerotia [12]. Sclerotia are mainly survival and overwintering structures [13]. Infection in the rice plant often starts either at soil or waterline depending on the production system. In flooded rice, sclerotia afloat and infect rice plants at the waterline. In row rice infection may start from the soil surface. Once infection starts, the disease primarily progresses upwards to the upper canopy. Later, mycelium spreads laterally to neighboring plants through leaf contacts and in favorable environmental conditions and susceptible rice cultivars, the fungus can develop and spread throughout the rice growing season [14].
Before 1970, sheath blight was rare in rice producing states of the USA. However, with the introduction of shorter and more susceptible cultivars and applications of excessive nitrogen (N) fertilization, often applied to maximize grain yield, the disease has expansively increased [15]. Grain yield losses of 50 percent and greater were reported in the 1980's on very susceptible varieties [16]. In recent years, with the development of more tolerant cultivars and improved cultural practices, actual yield loss to sheath blight has been reduced. However, yield losses from 5% to 15% have been common in several rice fields of Arkansas. About 62% of Arkansas rice fields receive fungicide applications [17] on an annual basis and the greatest proportion appeared to go to suppression sheath blight followed by rice blast, kernel smut and false smut.
Managing sheath blight with host plant resistance alone can be risky under conditions that favor disease development. There are variations in the quantitative inheritance of the multi-genic resistance available in rice for sheath blight [18]. Arkansas does not have commercial rice cultivars with complete resistance to the rice sheath blight pathogen, R. solani AG1-1A [19]. There is also considerable variability in sheath blight disease development due to variable weather conditions and management practices. The pathogen is favored more by warm and humid environmental conditions. Moreover, the pathogen has a wide host range that increase its chance of survival. In the last two decades, sheath blight management in Arkansas commonly receives at least one fungicide application (Cartwright, personal communication). Currently available fungicides labeled for sheath blight management in Arkansas rice are still active towards slowing the disease progress when properly applied following the recommended guidelines including treatment threshold and adequate canopy coverage [20]. Fungicide applications made when not required increase production cost. However, the greater concern is the imminent risk that the pathogen develops resistance to the limited commercial fungicides available for USA rice production. To date, there are no reported cases in Arkansas that R. solani AG1-1A is insensitive to azoxystrobin, but there are reported cases in Louisiana rice fields [21,22]. Accordingly, fungicides should only be used when management alternatives such as variety resistance and best cultural practices are not available. This helps to avoid continuous fungicide applications which can lead to fungicide resistant populations of R. solani [23]. The main objective of this study was to assess the monetary gains/losses of sheath blight management in rice with a onetime fungicide application under different seeding rates and chemical application timings.
Materials and Methods
Tests were conducted from 2016 to 2018 at two University of Arkansas system Division of Agriculture locations: Rice Research and Extension Center (RREC) and the Pine Tree Research Station (PTRS). Two pure-line rice cultivars, LaKast (long-grain) and Jupiter (medium grain) were selected representing tall and medium-short stature rice, respectively. Breeder’s recommended seeding rates, optimum and maximum were used for each cultivar. LaKast was planted at 81 kg/ha (72 lb/acre) and 122 kg/ha (109 lb/acre) and Jupiter at 82 kg/ha (73 lb/acre) and 124 kg/ha (111 lb/acre) as optimum and maximum seeding rates, respectively. Two fungicide application timing were selected based on Arkansas rice producers’ practice at Panicle Differentiation (PD) and Boot Split (BS). Seeds of both LaKast and Jupiter were drill-seeded using an 8-row ALMACO grain drill (Almaco, Nevada, IA). Plots at both RREC and PTRS received pre-flood N fertilizer at a rate of 118 kg N/ha (105 lb N/acre) in the form of urea at the 5-leaf stage, a day prior to permanent flood. A mid-season N application was made at Panicle Initiation (PI) at a rate of 50 kg N/ha (45 lb N/acre) both at RREC and PTRS. At RREC, pre-plant fertilizer was applied at a rate of 0-101-179-22-2.2 kg/ha (0-90-160-20-2 lb/acre) as N-P2O5-K2O-SO4-Zn. While at PTRS, pre-plant fertilizer was applied at a rate of 67-101-0-11 kg/ha (60-90-0-10 lb/acre). Treatments were randomized in factorial split-split plot design with four replications. Flood water was maintained on plots from the first application of nitrogen until pre-seed harvest.
All rice plots were artificially inoculated at (PI) with air dried R. solani AG1-1A inoculum made with the fungus colonizing a corn and rice seed mixture. For each 7m2 plot, 100 g of the inoculum was broadcasted by hand over the rice plants followed by a gentle sweep of foliage to deposit the inoculum onto the floodwater. A week after inoculation, plants were at PD, so 16 plots were treated with a fungicide containing azoxystrobin (Quadris 2.08 SC) at a rate of 0.9 L/ha (12.5 fl oz/acre). The fungicide was delivered using a MudMasterTM model MM2013 (Bowman Mfg., Newport, AR) in 187 L/ha (20 gal/acre) spray rate at 30 psi with Tee Jet XR11015 spray tips (Tee Jet Technologies, Wheaton, IL). Two weeks later, another sixteen other plots were treated with similar fungicide rate to correspond to BS application. The remaining 16 plots out of 48 were left untreated and used as unsprayed control.
Disease ratings at both locations were recorded twice within each season. The first rating was carried out on all plots 28 days after the PD fungicide application overlapped with 2 weeks after the BS application. The second disease rating on all plots was 2 to 3 weeks before harvest. Disease ratings included vertical and horizontal sheath blight disease progress. A 0 to 9 scale was used to estimate the vertical disease progress where 0 represented no disease and 9 represented disease reaching the flag leaf and rice panicles. Horizontal or lateral infection was estimated using percentage estimates of rice plants showing typical sheath blight symptoms from one-meter (3 ft) length of the middle row of each plot. Disease index was calculated by multiplying the vertical rating with the horizontal diseased rice score. Plots were harvested with a Wintersteiger classic plot combine (Wintersteiger, Austria). Disease indices, grain yield, and milling yield (percent whole kernel and total milled rice) were analyzed statistically using PROC MIXED procedure in SAS 9.4 (SAS Institute, Cary, NC).
Monetary gains or losses associated with sheath blight disease control were calculated as gross returns (rice price multiplied by yield) minus the cost of fungicide application and cost of seed. A rice price of $4.75, $5.22, and $5.70 per 20.41 kg (45 lb or a bushel of rice) was used in the analysis for 2016, 2017 and 2018, respectively. These rice prices represented the average U.S. farm prices for the months of August through October [24]. The cost of fungicide application included the cost of the fungicide and the cost to make a single aerial application. The fungicide cost was calculated at $69.6, $65.6, $63.5/liter ($2.06, $1.95, $1.88/fl oz)) of azoxystrobin multiplied by the fungicide application rate 0.9 L/ha (12.5 fl oz/acre) in 2016, 2017 and 2018, respectively. A cost of $17.28/ha ($7/acre) was considered for custom aerial application. The cost of seed was calculated as the product of the seeding rates used for each cultivar multiplied by a seed price of $0.95/kg ($0.43/lb) in 2016 and 2017, and $1.1/kg ($0.50/lb) in 2018. Costs per unit of fungicide, seed, and aerial application were obtained from Arkansas crop enterprise budgets in [25- 27]. Monetary gains to fungicide application were averaged across years by location, cultivar, seeding rate, and fungicide application. Monetary gains or losses of sheath blight control were also analyzed statistically using the PROC MIXED procedure in SAS 9.4.
Results
Sheath blight lesions appeared at the base of some rice plants above the flooded waterline between 7 to 10 days after inoculation in all the three years and at both locations. However, it slowed down when weather conditions changed from warm and humid to hot and dry. Although disease data were collected twice in each season, the 1st disease ratings from all three seasons at both locations were omitted from analysis. Data from the 1st reading did not show clear differences in sheath blight levels between plots that received the fungicide and the untreated control. Hence, the second disease ratings were used for the statistical analysis.
No interaction was observed between seeding rate and spray timings. Seeding rate had no significant impact on sheath blight disease index (Tables 1 and 2).
| Treatment | Sheath blight disease index | |||
|---|---|---|---|---|
| Jupiter | LaKast | |||
| RREC | PTRS | RREC | PTRS | |
| Optimum seeding rate | 2.6 | 2.5 | 2.3 | 2.5 |
| Maximum seeding rate | 2.9 | 2.4 | 2.5 | 2.3 |
| Pr>|t| | 0.4123 | 0.773 | 0.5 | 0.692 |
| No spray | 6.1 a | 4.0 a | 6.1 a | 3.9 a |
| Spray at PD | 1.6 b | 1.5 b | 0.4 b | 1.8 b |
| Spray at BS | 0.5 b | 1.9 b | 0.7 b | 1.4 b |
| Pr>|t| | <0.0001 | 9E-04 | <0.0001 | 0.002 |
Table 1: Rice sheath blight disease index on Jupiter and LaKast, two rice varieties across three years (2016-2018) at two locations--Rice Research and Extension Center (RREC) and Pine Tree Research Station (PTRS) tested at two seeding rates and three fungicide spray timings. No spray: No fungicide application; PD: Panicle Differentiation; BS: Boot Split; values in a column followed by the same letter are not significantly different.
| Effect | DF* | Jupiter | LaKast | ||||||
|---|---|---|---|---|---|---|---|---|---|
| RREC | PTRS | RREC | PTRS | ||||||
| F value | Pr>F | F value | Pr>F | F value | Pr>F | F value | Pr>F | ||
| SR | 1 | 0.68 | 0.4123 | 0.08 | 0.7734 | 0.57 | 0.4518 | 0.16 | 0.6915 |
| ST | 2 | 77.56 | <0.0001 | 7.89 | 0.0009 | 178.15 | <0.0001 | 6.96 | 0.0019 |
| SR × ST | 2 | 2.22 | 0.1177 | 1.06 | 0.3522 | 0.3 | 0.739 | 0.2 | 0.8191 |
Table 2: Tests of fixed effects of sheath blight index on Jupiter and LaKast, across three years at Rice Research and Extension Center (RREC) and Pine Tree Research Station (PTRS) tested at two seeding rates and three fungicide spray timings. *Number of degrees of freedom; SR: Seeding Rate; ST: Spray Timing.
Spray timing, however, showed significant differences on sheath blight disease levels between the plots that received fungicides and the unsprayed control plots for each variety at each location. Seeding rate had no significant impact on grain yield (Tables 3 and 4).
| Treatment | Jupiter (kg/ha) | LaKast (kg/ha) | ||
|---|---|---|---|---|
| RREC | PTRS | RREC | PTRS | |
| Optimum seeding rate | 10026 | 10095 | 9467 | 10034 |
| Maximum seeding rate | 10239 | 9977 | 9320 | 10074 |
| Pr>|t| | 0.5542 | 0.6303 | 0.5745 | 0.8615 |
| No spray | 9644 a | 9897 | 8831 a | 9972 |
| Spray at PD | 9968 a | 10123 | 9613 b | 10161 |
| Spray at BS | 10786 b | 10087 | 9736 b | 10028 |
| Pr>|t| | 0.033 | 0.7191 | 0.012 | 0.7829 |
Table 3: Yield of Jupiter and LaKast in kg/ha across three years at Rice Research and Extension Center (RREC) and Pine Tree Research Station (PTRS) tested at two seeding rates and three fungicide spray timings. No spray: No fungicide application; PD: Panicle Differentiation; BS: Boot Split; values in a column followed by the same letter are not significantly different.
| Effect | DF* | Jupiter | LaKast | ||||||
|---|---|---|---|---|---|---|---|---|---|
| RREC | PTRS | RREC | PTRS | ||||||
| F value | Pr>F | F value | Pr>F | F value | Pr>F | F value | Pr>F | ||
| SR | 1 | 0.35 | 0.5542 | 0.35 | 0.6303 | 0.32 | 0.5745 | 0.03 | 0.8615 |
| ST | 2 | 3.62 | 0.033 | 3.62 | 0.7191 | 4.78 | 0.012 | 25 | 0.7829 |
| SR × ST | 2 | 0.24 | 0.791 | 0.24 | 0.419 | 0.35 | 0.707 | 0.11 | 0.8965 |
Table 4: Tests of fixed effects of yield of Jupiter and LaKast, in Kg/ha across three years at Rice Research and Extension Center (RREC) and Pine Tree Research Station (PTRS) tested at two seeding rates and three fungicide spray timings. *Number of degrees of freedom; SR: Seeding Rate; ST: Spray Timing.
However, spray timing showed significant differences (p<0.05) on grain yields between the plots that received fungicides and the unsprayed control plots at RREC on both varieties in (Tables 3 and 4). Use of fungicides resulted in no grain yield differences at PTRS. Interactions between seeding rates and spray timings showed no significant difference (p=0.05) on grain yields. Two milling quality parameters of percent whole kernel and total milled rice were not affected by the sheath blight disease or fungicides applied (data not shown).
Based on the prices of seeds, fungicides and application expenses during the study period, there were no significant differences in the dollar ($) net return on for seeding rate or spray timing of either cultivar (Tables 5 and 6).
| Treatment | Jupiter | LaKast | ||
|---|---|---|---|---|
| RREC | PTRS | RREC | PTRS | |
| $ Net return | $Net return | $ Net return | $Net return | |
| Optimum seeding rate | 2460 | 2485 | 2321 | 2465 |
| Maximum seeding rate | 2469 | 2410 | 2243 | 2432 |
| Pr>|t| | 0.9256 | 0.2444 | 0.2667 | 0.583 |
| No spray | 2391 | 2469 | 2183 | 2482 |
| Spray at PD | 2388 | 2441 | 2315 | 2446 |
| Spray at BS | 2622 | 2433 | 2348 | 2418 |
| Pr>|t| | 0.1006 | 0.8896 | 0.1273 | 0.6794 |
Table 5: Dollar ($) Net return from Jupiter and LaKast, across three years at Rice Research and Extension Center (RREC) and Pine Tree Research Station (PTRS) tested at two seeding rates and three fungicide spray timings. No spray: No Fungicide Application; PD: Panicle Differentiation; BS: Boot Split.
| Effect | DF* | Jupiter | LaKast | ||||||
|---|---|---|---|---|---|---|---|---|---|
| RREC | PTRS | RREC | PTRS | ||||||
| F value | Pr>F | F value | Pr>F | F value | Pr>F | F value | Pr>F | ||
| SR | 1 | 0.01 | 0.9256 | 1.38 | 0.2444 | 1.26 | 0.2667 | 0.3 | 0.583 |
| ST | 2 | 2.39 | 0.1006 | 0.12 | 0.8896 | 2.14 | 0.1273 | 0.39 | 0.6794 |
| SR × ST | 2 | 0.19 | 0.8306 | 0.9 | 0.4122 | 0.38 | 0.686 | 0.11 | 0.8979 |
Table 6: Tests of fixed effects on dollar ($) net return from Jupiter and LaKast across three years at Rice Research and Extension Center (RREC) and Pine Tree Research Station (PTRS) tested at two seeding rates and three fungicide spray timings. *Number of Degrees of Freedom; Seeding Rate (SR) Spray Timing (ST).
Discussion
Both rice cultivars, LaKast, a tall stature Japonica with long-grain type, and Jupiter, a short stature medium-grain, were selected for their known susceptibility to sheath blight. Since both varieties have different yield potentials the data analysis was done separately. In agreement to their history, both varieties showed relatively higher levels of sheath blight in the unsprayed control plots at both locations in this study (Table 1).
The fungicide, containing azoxystrobin, was applied at the highest labeled rate and selected based on its high acceptance in Arkansas rice production to suppress sheath blight. Such a rate of azoxystrobin was shown to suppress sheath blight from four to six weeks.
The two seeding rates were selected based on agronomic recommendations for both locations. Maximum seeding rates normally are used in heavy clay Arkansas soils when rice is planted in late March or early April when the soil temperatures are below optimum [28]. However, in this study, maximum seeding rate was used to increase the crop stand density to create a more closed canopy micro-environment to encourage sheath blight disease development. The fungicide timing was selected based on recommendations for Arkansas rice production which range between PD to BS plant growth stage [29]. Although sheath blight infection in Arkansas-flooded-rice often starts at PI, rice plants are most susceptible at booting through flowering. According to [30], the effect of sheath blight on yield or yield components can be minimal when infection occurs early at tillering or later at grain filling. Yield losses were significant when sheath blight infection started at panicle initiation, booting, or flowering. Reproductive stages particularly at flowering to heading are most sensitive developmental stages that impact yield.
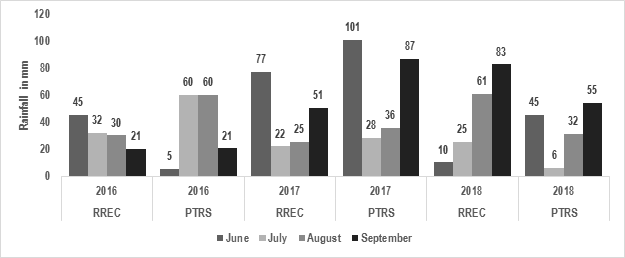
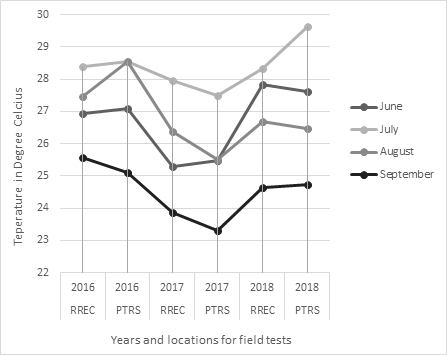
To obtain observable levels of damage, plots at RREC and PTRS were inoculated at PI. Normally, when sheath blight progresses, it invades from outer to inner sheath. The pathogen then spreads vertically from sheath to sheath and horizontally from tiller to tiller of the same plant or neighboring rice plants. Heavily infected rice produces immature panicles or poorly filled grains [31]. However, as the temperatures and rainfall amounts fluctuate (Figures 1 and 2)
During the rice growing seasons, sheath blight progression also fluctuates. Under favorable weather, the R. solani AG1-A fungus grows fast, but mycelial growth slows down when less favorable conditions prevail. Due to such influences of weather conditions, regular and frequent field monitoring is recommended before deciding to make a fungicide application. Rice producers may choose to delay a fungicide application for sheath blight if the benefit from fungicide application is low. Depending on the growing conditions, the crop may escape the disease particularly when a field has no history of the disease and the variety has some level of tolerance. Alternatively, fungicide application can be delayed in rice to target both sheath blight and blast. It is important to ensure that the upper three leaves including flag leaves are not threatened by sheath blight. Generally, sheath blight showed higher disease indices at RREC compared to PTRS (Table 1). Regardless of the disease level, yield advantages were not significant (Table 3). Similarly, there were no significant differences in dollar ($) net return at either location (Table 5). In this study, the late season sheath blight was not severe enough to reach to the panicles to cause damage on the kernels.
In Arkansas, multiple fungicide applications to manage sheath blight alone are not economical and simply add to production costs (Cartwright, personal communication). Cost control is essential to be successful in the USA rice industry. Research and experience have shown that if the upper three leaves including the flag leaf are protected from the disease, grain yield loss is minimal (Cartwright, personal communication). However, depending on the cultivar, without fungicide application, yield loss from 0.34 to 2.02 tones/hectare (5 to 30 bushels per acre) has been reported in severe sheath blight fields in Arkansas. Up to 16% grain yield advantage was shown in a nitrogen fertilization study using azoxystrobin. With appropriate field management and timely scouting, there is a likely chance that a fungicide application can be avoided, delayed, or limited to one application. Fungicide application for sheath blight is justified when the weather conditions are favorable for the disease pressure to increase during reproductive stages of high yielding susceptible varieties.
A more economical approach to using fungicides in rice is by pairing fungicide timings to target more than one rice disease. This can get complicated as fungicides need to be applied at the recommended times for each disease. For instance, in a field known to have a history kernel smut or false smut, mixed fungicides containing a triazole and an azoxystrobin can be applied at mid-boot to target sheath blight at the same time. Mid boot is the recommended timing for smuts and it is also within the range for the sheath blight fungicide application to be affective. If the field requires no fungicide application for smuts, and the upper three leaves are not threatened by sheath blight, fungicide application can be delayed further to full boot or Boot Split to coincide the 1st application timing for neck blast provided the field has a history of blast. In such a case, a fungicide containing azoxystrobin can be applied for sheath blight suppression and to protecting the rice from collar, panicle and neck blast. To protect rice from neck blast, two applications are recommended the 1st to protect primary tillers and the 2nd the secondary tillers. The pairing of fungicides, timing, and diseases present can at least reduce application costs and unnecessary multiple fungicide applications. Fungicides are important tools in modern crop production. However, excessive or unnecessary fungicide applications are discouraged because of possible selection pressure on the pathogen to result in loss of fungicide efficacy [32]. With continued use, pathogens develop resistance to fungicides.
Varietal selection for better disease resistance allows producers to use less fungicides and avoid significant reduction in grain yields. A loss up to 8% grain yield in moderately resistant rice as opposed to 40% loss in very susceptible cultivar under severe sheath blight situation demonstrated that breeding programs can have a major role in management of rice diseases [33]. Sheath blight also weakens stem strength and causes rice crop to lodge easily causing indirect damage to the crop.
Regardless of the significant differences shown in disease levels (Table 1) and yield (Table 3) mean monitory gains (Table 5) were not significant enough to show the benefit of using fungicides to suppress sheath blight in this study. This study is a good example of unnecessary use of a fungicide before the disease is at or beyond the recommended timing and disease levels. Moreover, sheath blight incidence or severity in susceptible rice can be reduced using cultural practices by using the recommended seeding and nitrogen fertilization rates. Crop rotation is another strategy recommended to reduce the inoculum source in the soil provided the crop is not soybean as the same strain of R. solani AG1-1A causes aerial web blight in soybeans. The fungus has a wide host range and destroying weeds narrows buildup of inoculum in the soil.
Overall, fungicide application either at PD or BS had shown adequate suppression of sheath blight which agreed with previous recommendations for Arkansas rice [30]. Adequate field management is vital particularly when susceptible or very susceptible rice varieties are planted. Although fungicide application at Boot Split appeared advantageous at RREC over PTRS, the results in this study suggested the current recommended fungicide application timing of PD through BS is generally most appropriate for use in Arkansas. According to this study, the important point is to remember that fungicide application may not be needed to all fields that show sheath blight. Application of a fungicide in some locations and years, where sheath blight is not severe, lowers mean monitory gain and makes the practice unnecessary.
Funding and Source
The authors appreciate the funding support from the rice growers of Arkansas administered by the Arkansas Rice Research and Promotion Board, as well as, support from the University of Arkansas system Division of Agriculture.
References
- Fairhurst TH and Doberman A (2002) Better crops international. IPNI Canada 16.
- Gnanamanickam SS (2009) An overview of progress in biological control. Biological Control of Rice Diseases 8: 43-51.
[Crossref], [Google Scholar]
- Better crops international
- An overview of progress in biological control
- Crossref
- Lee FN and Rush MC (1983) Rice sheath blight: A major rice disease. Plant Dis 67: 829-832.
[Crossref], [Google Scholar]
- Park DS, Sayler RJ, Hong YG, Nam MH, Yang Y (2008) A method for inoculation and evaluation of rice sheath blight disease. Plant Dis 92: 25-29.
[Crossref], [Google Scholar].
- Wamishe Y, Cartwright RD, Lee F (2018). Management of rice diseases In: 2018 MP192 Arkansas rice production handbook. Division of Agriculture. Hardke JT ed. pp: 120-143.
- Yellareddygari SKR, Reddy MS, Kloepper JW, Lawrence KS, Fadamiro H (2014) Rice sheath blight: A review of disease and pathogen management approaches. J Plant Pathol Microb 5: 241-257.
[Crossref], [Google Scholar]
- Google Scholar
- Uppala S, Zhou XG (2018) Rice Sheath Blight. Plant Health Instr.
- Bell DK, Sumner DR (1987) Survival of Rhizoctonia solani and other soilborne basidiomycetes in fallow soil. Plant Dis 71: 911–915.
[Crossref], [Google Scholar]
- Kimiharu I, Qingyuan G, Masao A (2004) Overwintering of rice sclerotial disease fungi, Rhizoctonia and Sclerotium spp. in paddy fields in Japan. Plant Pathol J 3: 81–87.
[Crossref], [Google Scholar]
- Groth DE, Bond JA (2006) Initiation of rice sheath blight epidemics and effect of application timing of azoxystrobin on disease incidence, severity, yield, and milling quality. Plant Dis 90:1073-1076.
[Crossref], [Google Scholar].
- Groth DE Rush MC Hollier CA (1992) Prediction of rice sheath blight severity and yield loss based on early season infection. Louisiana Agriculture 35: 20-23.
[Crossref], [Google Scholar].
- Slaton NA, Cartwright RD, Meng J, Gbur EE, Norman RJ (2003) Sheath blight severity and rice yield as affected by nitrogen fertilizer rate, application method, and fungicide. Agronomy Journal 95(6): 1489-1496.
[Crossref], [Google Scholar]
- Watkins B, Mane R (2016) Estimation of Fungicide Cost for Rice Production in the United States. University of Arkansas System Division of Agriculture Rice Research and Extension Center Stuttgart, Arkansas.
- Li Z, Pinson SR, Marchetti MA, Stansel JW, Park WD (1995). Characterization of Quantitative Trait Loci (QTLs) in cultivated rice contributing to field resistance to sheath blight (Rhizoctonia solani). Theor Appl Genet 91: 382-388.
[Crossref], [Google Scholar]
- Brooks SA (2007) Sensitivity to a Phytotoxin from Rhizoctonia solani correlates with sheath blight susceptibility in rice. Phytopathology 97: 1207 -1212.
[Crossref], [Google Scholar]
- Wamishe Y (2019) Rice- Fungicides In: 2019 MP154 Arkansas Plant Disease Control Products Guide. Division of Agriculture. Faske T ed. pp: 140-147.
- Lunos AG (2016) Geographic Distribution and Severity of Strobilurin Fungicide Resistance among Rhizoctonia solani on Rice in Southwestern Louisian. LSU Master's Theses.
- Olaya G, Sarmiento L, Edlebeck K, Buitrago C, Sierotzki H, et al. (2013) Azoxystrobin (QoI) resistance monitoring of Rhizoctoniasolani isolates causing rice sheath blight in Louisiana. The American Phytopathological Society.
- Brent KJ, Hollomon DW (2007) Fungicide resistance in crop pathogens: How can it be managed? Fungicide Resistance Action Committee. 2nd ed.
- https://www.ers.usda.gov/topics/crops/rice/rice-sector-at-a-glance/
- Flanders A, Watkins B (2016) Crop enterprise budgets for arkansas field crops planted in 2016.
- Flanders A, Watkins B (2017) Crop enterprise budgets for arkansas field crops planted in 2017.
- https://www.statista.com/statistics/190823/top-us-states-for-rice-production/
- Hardke JT (2015) Minimum, optimum, and maximum seeding rates for rice cultivars. Division of agriculture. research and extension.
- Wamishe A, Hardke J (2019) Match fungal disease to best fungicide application timing. Rice farming.
- Cu RM, Mew TW, Cassman KG, Teng PS (1996) Effect of sheath blight on yield in tropical intensive rice production system. Plant Dis 80: 1103-1108.
- Dasgupta MK (1992) Rice sheath blight: The challenge continues plant diseases of international importance: Diseases of cereals and pulses. Prentice hall, Englewood Chiffs.
- Molla KA, Subhasis K, Johiruddin M, Prasad Bajaj, Rajeev K, et al. (2020) Understanding sheath blight resistance in rice: The road behind and the road ahead. Plant Biotechnol J 18: 895-915.
[Crossref], [Google Scholar]
- Groth DE (2008) Effects of cultivar resistance and single fungicide application on rice sheath blight, yield, and quality. Crop Prot 27: 1125-1130.
[Crossref], [Google Scholar]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi