Research Article, J Soil Sci Plant Health Vol: 3 Issue: 1
Effect of Ammonium Sulfate Fertilizer Additions on Hydrogen Sulfide Toxicity
Julia M. Fryer1, Trenton L. Roberts1*, Yeshi Wamishe2, Jarrod T. Hardke2 and David M. Miller1
1Department of Crop, Soil, and Environmental Sciences at the University of Arkansas, Fayetteville, AR, USA
2Rice Research and Extension Center, University of Arkansas Division of Agriculture, Stuttgart, AR, USA
*Corresponding Author : Trenton Roberts
Department of Crop, Soil, and Environmental Sciences, University of Arkansas, 115 Plant Science, Fayetteville, AR, USA
Tel: 479-575-6752
E-mail: tlrobert@uark.edu
Received: July 26, 2018 Accepted: March 9, 2019 Published: April 10, 2019
Citation: Fryer JM, Roberts TL, Wamishe Y, Hardke JT, Miller DM (2019) Effect of Ammonium Sulfate Fertilizer Additions on Hydrogen Sulfide Toxicity. J Soil Sci Plant Health 3:1.
Abstract
Abstract: Hydrogen sulfide (H2S) toxicity is an increasingly problematic physiological disorder reported in certain Arkansas rice (Oryza sativa L.) fields. Though the exact causes of this disorder are unknown, one contributing suspect is use of ammonium sulfate (NH4)2SO4 fertilizer. A greenhouse study was designed to investigate the physical and chemical differences in four soils in Arkansas where this disorder regularly occurs (H and HR-Y) and never has been reported (HR-N and PTRS) and to investigate the effects of different rates of (NH4)2SO4 fertilizer on those soils. According to the Mehlich 3 soil test report, few major differences were present between soils prone to H2S toxicity and those that are not. With Zn deficiencies appearing in PTRS and HR-N, several factors likely influenced this deficiency including P fertilization and flooding effects. Significant differences in soluble SO42- concentration between soil locations occurred for the first 21 days after flooding. HR-N contained significantly more SO42- and levels did not change over time. However, the other locations contained less SO42and concentrations increased, particularly during the third week. Differences between fertilizer treatments were also significant from days 2-21 after flooding. The highest treatment of (NH4)2SO4 contained the highest concentration of SO42- in solution, followed by the low treatment of (NH4)2SO4, with the 0 kg control having the lowest concentration. Concentrations of SO42- increased in each location over time. The increase in SO42- was likely caused decomposition of soil organic matter (SOM). Lack of evidence of H2S toxicity in the root examination and in the above ground biomass nutrient content, along with the soil solution results; indicate that H2S toxicity is influenced by more than the SO42- content in the soil. A combination of other factors such as Eh, microorganisms, SOM content, and environmental conditions are likely major contributors to the occurrence of H2S toxicity.
Keywords: Soil solution; Fertilizer treatments; Flooding effects
Introduction
Approximately half of the rice produced in the United States is grown in Arkansas [1]. As the leader of rice production in the country, rice significantly contributes to Arkansas’s economy, contributing over 6 billion dollars annually [1] and providing over 25,000 jobs [2]. With an average yield of 8,221 kg ha-1 in 2015, Arkansas producers achieve the second highest yield in the United States [3]. As a vital part to Arkansas’s economy, researches must stay proactive in developing solutions for current and potential production problems.
One challenge that is becoming more problematic in Arkansas is hydrogen sulfide (H2S) toxicity. Also referred to as autumn decline or akiochi (Japanese for ‘autumn decline’), H2S toxicity is thought to be caused by excessive reduction of SO4 2- to H2S in the soil. Under anaerobic conditions, SO4 2- reduces to H2S, a toxic gas, but then typically reacts with active Fe in the soil and precipitates out as insoluble FeS, preventing the buildup and toxicity of H2S [4]. This phenomena is difficult to classify and has been termed a “physiological disease” [5] and was later classified as a “nutritional disorder” which is a more accurate term [4]. Though often classified as a disease, H2S toxicity is not caused by a pathogen and therefore is not truly a disease. However, H2S toxicity does weaken the plant by interfering with water and nutrient uptake, making plants more susceptible to invasion of opportunistic diseases such as brown spot (historically referred to as Helminthosporium leaf spot), rice blast, caused by Pyricularia oryzae , and crown rot, caused by the fungus Gaeumannomyces graminis vs. Graminis [6-8].
Once symptoms of H2S toxicity are identified in the field, only one rescue technique is currently available: draining and drying the field. By removing the flood from the field, the surface soil is able to reoxygenate which stops the reduction reactions occurring in the anaerobic soils [9]. Once oxygen reenters the root zone, new roots form and then the field can be flooded again [7]. If this rescue technique is performed early enough in the growth cycle of the rice, the majority of the rice is likely to recover, though yield loss may still occur [1]. However, rice is not a drought tolerant plant, and aerobic conditions increase the potential for other disease to take over, such as blast, and may influence yield [8]. In Arkansas, fields should be drained at the time recommended to drain for straight head using the DD-50 program [8].
Though the exact causes of H2S toxicity are unknown, there are two major management practices that likely impact the occurrence and severity of H2S toxicity: irrigation water source and fertilizer choice. In Arkansas, 76.4% of the rice acreage is irrigated using ground water [3]. With SO4 2- concentrations ranging from 2 to over 100 mg L-1 in the ground water, this can be a major source of SO4 2- regularly added to the field throughout the season [10]. In soils already high in SO4 2- or SOM, additional SO4 2- from irrigation water could likely increase the chance of H2S toxicity occurring. Irrigation water is a suspected contributor to the occurrence of H2S toxicity in Arkansas [9].
In addition to irrigation water potentially influencing this disorder, fertilizer choice can also increase the chance of H2S toxicity occurring, particularly (NH4)2SO4 fertilizer. Though (NH4)2SO4 is a more expensive fertilizer, this fertilizer is recommended as a pre-flood N source particularly on sandy soils that tend to be deficient in SO4 2- [10]. Based on data collected by the Soil Testing and Research Laboratory in Marianna, AR, 31%, 35%, and 52% of all soils tested in Cross County, Woodruff County and St. Francis County respectively were below optimal in SO4 2- in 2014 [11]. Ammonium sulfate is a practical choice for fertilization to increase both S and N, however, application to soils sufficient in SO4 2- may increase the likelihood of H2S toxicity occurring.
In Arkansas, N, P, and K are typically required to maximize rice yield potential [12]. Recommended fertilizer application rates depend upon soil test report information. Proper N fertilization and management are vital for excellent rice grain yield [10]. The two typical N sources for rice used in Arkansas are urea and (NH4)2SO4 [12]. Due to the higher cost of (NH4)2SO4, this fertilizer is only recommended when there is potential for S deficiencies [10]. Since sandy soils are typically low in SOM and SO4 2- is easily lost from the soil through leaching, S fertilizer is usually only required on sandy soils [10].
The objective of this study is to evaluate the effects of (NH4)2SO4 fertilizer on H2S toxicity. Three rates of (NH4)2SO4 will be utilized and soluble SO4 2- and Eh monitored throughout the experiment. The goal for the highest (NH4)2SO4 rate is to induce H2S toxicity, particularly in the soils prone to H2S toxicity.
Materials and Methods
Soil was collected from three locations in Arkansas during 2016. Surface soil was collected from the Pine Tree Research Station (PTRS) in Pine Tree, AR where H2S toxicity has never been reported, from a producer field in Hunter, AR (H) where H2S toxicity occurs every time when planted to rice, and two samples were taken from two locations in a producer’s field in Hickory Ridge, AR where H2S toxicity always occurs in one location when planted to rice (HR-Y) and another location within the same field where H2S does not occurred when planted to rice (HR-N). Surface soil was collected from each location and brought back to Fayetteville, AR for the greenhouse experiment. A soil test was conducted to assess pH (1:2 v:v soil: water ratio) [13], soil texture [14], total nitrogen (TN) [15], total carbon (TC) [16], soil OM via weight loss on ignition (LOI) [17], and Mehlich 3 extractable nutrients, P, K, and S [18]. Detailed soil and agronomic information is listed in Table 1.
| Location ID | County | Soil Series | Soil Classification | Soil Texture† | pH‡ | LOI§ | TN¶ | TC¶ | P# | K# | S# | Zn | ||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Sand | Silt | Clay | ||||||||||||
| ---------%--------- | ------------%------------ | -------mg kg-1------- | ||||||||||||
| HR-Y | Cross | Henry Silt Loam | Coarse-silty, mixed, active, thermic Typic Fragiaqualfs | 7 | 84 | 9 | 7.9 | 1.54 | 0.0547 | 0.6703 | 42 | 91 | 8 | 7 |
| HR-N | Cross | Henry Silt Loam | Coarse-silty, mixed, active, thermic Typic Fragiaqualfs | 10 | 76 | 14 | 7.9 | 2.09 | 0.0805 | 1.0254 | 54 | 116 | 92 | 3 |
| H | Woodruff | Hillemann Silt Loam | Albic Glossic Natraqualfs | 33 | 55 | 12 | 7.3 | 2.50 | 0.0819 | 1.1730 | 30 | 147 | 21 | 5 |
| PTRS | St. Francis | Calloway Silt Loam | Aquic Fraglossudalfs | 6 | 78 | 16 | 8.3 | 1.85 | 0.0455 | 0.6794 | 28 | 93 | 9 | 15 |
Table 1: Selected soil chemical and physical properties from locations used in greenhouse experiment.
Soil was passed through a 1 cm sieve to remove clods and large pieces of crop residue. A volume of 3.79 L of each soil was placed into a 7.57 L bucket. Each location had nine buckets with 3.79 L of soil per bucket. Three (NH4)2SO4 fertilizer treatments were applied for this experiment: 0 kg ha-1, 115 kg ha-1, and 230 kg ha-1. Buckets were arranged randomly on two benches in the greenhouse with 18 buckets on each bench. Fertilizer was incorporated in the top few centimeters of the soil at rates of 89.74 kg P2O5 ha-1 and 134.62 kg K2O ha-1. The P and K fertilizer rates were doubled for this experiment to avoid deficiency symptoms that occurred in prior trials with these soils. Deionized water was used in this experiment to eliminate excess SO4 2- being added through the water source. Cultivar ‘CL151’ seeds were germinated in a damp paper towel to ensure viable seeds was used. Soil was wetted with deionized water and 24h later, 10 rice seedlings were transplanted into each bucket. Deionized water was misted heavily in each bucket then the buckets were covered with plastic wrap to trap humidity and promote plant establishment and growth. Deionized water was misted in each bucket daily until seedlings were established. Buckets were hand weeded as needed. Six days later, each bucket was thinned to 5 uniform plants per bucket.
At the 1-2 leaf stage, (NH4)2SO4 fertilizer treatments were surface applied to their respective buckets and the soil was allowed to dry before the next watering. Once plants reached the 5 leaf stage, supplemental urea fertilizer was added to the soil: 352 kg urea ha-1 for the 115 kg ammonium sulfate ha-1 treatment, 300 kg urea per ha-1 for the 230 kg ammonium sulfate ha-1, and 692 kg urea ha-1 for the 0 kg ammonium sulfate ha-1 treatment to give each treatment the same total N application rate of 319 kg N ha-1. Urea rates are double what are recommended for rice growth in Arkansas to account for the closed system in the greenhouse. One porous ceramic cup sampler (IRROMETER® Soil Solution Access Tube – Model SSAT, Riverside, CA) was placed in each bucket and one platinum electrode redox sensor (Sensorex® electrochemical ORP sensor) was placed throughout 1 replication of the experiment for a total of 18 redox sensors [19]. Twenty four hours after the addition of urea, all buckets were flooded with deionized water to approximately 10 cm above the soil surface. This flood level was maintained for the duration of the experiment. Redox was continuously monitored by the electrodes and logged into a data logger, and soil solution extractions were taken 1, 2, 7, 14, 21, 28, 35, 42, 49, 63, 77, and 91 days after flooding, following a similar timeline to [20].
Before extracting soil solution, a 60 cc syringe was used to extract and discard all fluid in the porous ceramic cup sampler. To extract soil solution samples, pressure was drawn to 60 centibars to create a vacuum in each porous ceramic cup sampler using a hand vacuum pump. The vacuum was maintained for 3h before the solution was collected. A clean 60 cc syringe was then double rinsed with deionized water, and then used to extract the solution which was then placed in a scintillation vial. All scintillation vials contained two drops of concentrated HCl (37%) to acidify the solution to prevent precipitation of nutrients and to reduce microbial activity. The syringe was double rinsed with deionized water between each sample. Samples were stored at room temperature until analyzed using an inductively coupled argon plasma spectrophotometers (ICAP). Soil solution samples were analyzed for P, K, Ca, Mg, Na, S, Fe, Mn, Zn, CU, and B. After the final sampling was performed on day 91, root of all plants were washed and examined for any buildup of FeS. Above ground biomass was removed, dried, and analyzed for nutrient contents using a nitric acid digest.
On the first day of flooding, one sixth of a mosquito dunk was added to each bucket as a control for fungus gnats. This method was ineffective, so Fury® Insecticide was applied to all plants two days after flooding. Four days after the application of Fury®, all plants grown in the PTRS and HR-N soils were notable pale green to yellow. Plants were monitored but new leaves remained yellow and leaf tips became necrotic and growth was stunted. Thirteen days after flooding, a foliar treatment of zinc sulfate (ZnSO4) was applied to all plants. After a few days, leaf tissue began to darken again. One more application of zinc sulfate was applied as a foliar treatment to only PTRS and HR-N plants one week later. The application of ZnSO4 corrected nutrient deficiencies in the plants, but unfortunately a few plants were lost, giving a smaller plant stands in a few of the buckets.
Statistical Analysis
This experiment contained four soil locations and three treatments: 0, 115 and 230 kg (NH4)2SO4 ha-1. Each treatment was replicated three times totaling 36 individual buckets. Buckets were arranged in a randomized complete block design with three blocks. This experiment was designed so that one replication appeared within each block. Redox sensors were placed at random within one replication. Soil solution samples were collected from each individual bucket on the designated sampling days and redox potential was monitored constantly. An analysis of variance (ANOVA) was conducted for each day of soil solution data using JMP® Pro 12 (SAS Institute, Inc., Cary, NC). Comparisons were made at the p ≤ 0.05 significance level to evaluate if the different rates of (NH4)2SO4 impacted SO4 2- reduction and if SO4 2- reduction differed between soils from different locations. Student’s T Test was used to separate significant means.
Our statistical hypotheses were that SO4 2- levels be elevated in treatments with added ammonium sulfate and that SO4 2- levels would decrease more rapidly in H and HR-Y soils than the HR-N and PTRS soils. Soil solution data were analyzed by an ANOVA, and redox data were used to support the findings of the soil solution ANOVA.
Results and Discussion
Physical and chemical soil characteristics
After soils were collected for this experiment, a subsample from each location was analyzed for physical and chemical properties. Differences between locations prone to H2S toxicity and those not prone to the disorder were not major. Oddly, H and HR-N were similar to each other overall. However, a few differences may be important to note.
Mehlich 3 extractable S and Fe both varied between locations. Soluble S in HR-N exceeded all other locations by 70-80 mg SO4 2- kg-1 soil, and Fe concentrations were highest in H and HR-N. Again, H and HR-N were similar in TC, TN, and LOI. Though soil test report information can be useful, this data does not appear to point to any major differences between locations that could be the cause of H2S toxicity.
One major nutrient that proved problematic during this experiment was Zn. Deficiency symptoms occurred eight days after flooding though not severe. Thirteen days after flooding, Zn deficiency symptoms had worsened with pale green and yellow leaves, necrotic leaf tips, very little internode elongation, minimal tillering, and even death of a few plants. Zinc deficiencies are fairly common in lowland rice soils after flooding though sometimes plants can recover on their own after 6-8 weeks [4]. In Arkansas, Zn fertilizer is only recommended on silt and sandy silt loam textures [21]. All soils used in this experiment fall into that category (Table 1). Deficiency may have occurred for several reasons. Though PTRS contained the highest amount of Zn (14.82 mg SO4 2- kg-1 soil) symptoms occurred most severely in PTRS and HR-N soils regardless of (NH4)2SO4 treatment. PTRS contained Zn levels well above the critical level; however HR-N was below the critical level of 3.5 mg Zn kg-1 soil [21]. Keeping in mind that the Mehlich 3 data shows the nutrient status of the soil before flooding, once flooded chemical changes take place [22]. After flooding, pH rises in acidic soils and lowers in alkaline soils to settle in the range of 6.8-7.2 [23]. Solubility of Zn decreases by two orders of magnitude for every one unit of increase in pH [24]. With the soil pH ranging from 7.26-8.27, pH likely decreased up to 1 order of magnitude. This change in pH may have influence availability of Zn, though it is not likely. However, flooding increases availability of Fe, Ca, Mg, Cu, Mn and P and therefore suppresses Zn uptake [24]. Both HR-N and PTRS contained higher concentrations of Ca, Mg, and Mn. Decline in Eh is another chemical factor that changes after flooding. Under low Eh conditions, sulfides of Zn can form and cause Zn deficiencies [24]. Additionally, after high rates of P fertilizer are applied to the soil, Zn-phosphate commonly forms and reduces Zn availability [24]. Since the same P rate was applied to each of the soil locations, all locations showed P levels above the critical level, and P becomes more plant available after flooding [23], Zn likely reacted with P in all soil location to form Zn-phosphate to some extent. With the greatest pH change likely occurring in HR-N and PTRS, higher concentration so nutrients that can suppress Zn uptake and the addition of high levels of P fertilizer added to the soils, the severe Zn deficiencies in these two locations can be rationalized.
Along with differences in nutrient concentrations, texture differed between locations. H contained the highest percentage of sand with all other soil locations containing less than 10% sand. Since surface soil was collected for this experiment, accuracy of the subsample tested for nutrients, SOM, and texture is likely not high since soils vary greatly within fields [10]. In order to get a true index of nutrients and texture of a field, soil cores should be taken across the field in a zigzag pattern at a depth of 10 cm [10]. However, since surface soil was collected and utilized for this experiment, the soil collection method was appropriate, but proper sampling could have been used to support findings.
Flooding effect on sulfate concentration and redox potential
Although new redox sensors were used for this experiment, data from eight sensors were lost due to a malfunction with the data logger. One sensor from the working data logger appeared to malfunction as Eh increased over 1000 mV over time. Data from that sensor was discarded, but data from the other seven sensors will be used to support soil solution data.
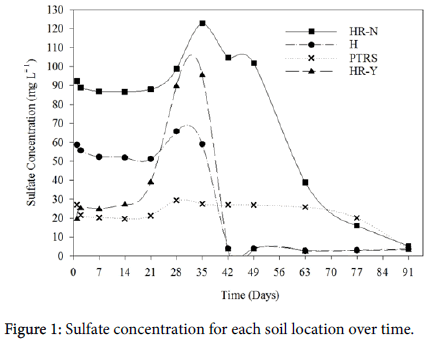
Days 1-21: Despite the different fertilizer treatments, significant differences between soil locations were present for the first three weeks after flooding (Table 2). With higher concentrations of SO4 2- in solution, approximately 51-57 mg SO4 2- L-1, HR-N had significantly higher concentrations of SO4 2- compared to all other soil locations, likely due to the high concentration of SO4 2- already present in the soil (Table 1). For the duration of the experiment, no statistical difference between PTRS and HR-Y were found. These results were unexpected since, in the field, H2S toxicity has never been a reported problem on HR-N soil but has been reported on HR-Y soils. Based on field reports, we would expect SO4 2- concentrations to be similar between HR-N and PTRS, not HR-Y and PTRS. Though our soil tests are representative of the samples used in this study, reports from whole field sampling would be interesting to compare to the samples used. If the samples used in this experiment are indeed representative of the whole field, results from this study indicate that H2S toxicity is not driven by SO4 2- concentrations alone. Other factors may contribute to the occurrence of H2S toxicity such as microbial populations and concentrations of substrates in the soil. Microorganisms are vital in the chemical changes that take place in soil. The reduction of compounds, such as SO4 2-, and changes in Eh would occur at a much slower rate without microbes [23,25]. The different species of microorganisms also affect what reactions take place in the soil. With certain species of microbes responsible for the majority of the SO4 2- reduction, Desulfovibrio and Desulfotomacuclum [26,27], these species are likely present in soils prone to H2S toxicity. Of course, concentrations of substrates in the soil greatly affect chemical changes as well. With high concentrations of SO4 2- in the soil and low concentrations of Fe2+ to react with H2S as the SO42- reduces, H2S toxicity could be an expected result.
| Effects | DF | 1 | 2 | 7 | 14 | 21 | 28 | 35 | 42 | 49 | 63 | 77 | 91 |
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| -------------------------------------------------------------------Days------------------------------------------------------------------- | |||||||||||||
| Location | 3 | 0.0051 | <0.0001 | <0.0001 | <0.0001 | 0.0183 | 0.4468 | 0.4012 | 0.2513 | 0.2513 | 0.6250 | 0.8862 | 0.9994 |
| Fertilizer Treatment | 2 | 0.5488 | 0.0275 | 0.0140 | <0.0001 | <0.0001 | 0.1461 | 0.1166 | 0.9923 | 0.9923 | 0.9997 | 0.9999 | 0.9977 |
| Location x Fertilizer Treatment | 6 | 0.3641 | 0.6209 | 0.4796 | 0.7415 | 0.6645 | 0.5661 | 0.2321 | 0.6386 | 0.6386 | 0.0016 | 0.0197 | 0.2322 |
Table 2: Analysis of variance with significant level a=0.05.
Another possible contributing factor is additional SO4 2- being added throughout the growing season by well water [9]. Though this factor was controlled in this greenhouse experiment by using deionized water, well water may contribute to the problem in the field, causing different changes in soil solution in the field compared to the controlled greenhouse environment.
Unlike the first greenhouse study and results found in the literature, SO4 2- did not rapidly decrease after flooding [28-31]. Approximately two weeks passed after flooding with SO4 2- levels remaining fairly constant in all soil locations. Though Eh began to drop immediately after flooding in all recovered data, all soils were above -100 mV except HR-N with 0 kg (NH4)2SO4 applied. Since Eh had not reached the SO4 2- reducing potential in the majority of the soils by two weeks after flooding, this could explain why SO4 2- concentrations remained steady. However, after two weeks of flooding, SO4 2- concentrations increased in all locations except HR-N rather than decreasing (Figure 1). Though SO4 2- may have been added to the system in low quantities due to the foliar application of ZnSO4 2- to correct the Zn deficiency, this increase in soluble SO4 2- was likely due to SOM mineralization rather than an outside source. Since mineralization occurs most rapidly in a soil pH of 6.5-7, this increase in SO4 2- was likely due to microbial activity [26].
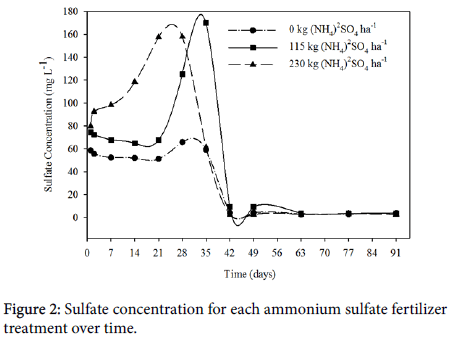
Days 2-21: Regardless of soil location, soluble SO4 2- concentrations differed significantly between fertilizer treatments for the first three weeks after flooding (Table 2). As expected, the control treatment with 0 kg (NH4)2SO4 ha-1 had the least SO4 2- in solution, the medium treatment of 115 kg (NH4)2SO4 ha-1 contained more, and the highest treatment of 230 kg (NH4)2SO4 ha-1 contained the highest concentration of soluble SO4 2- (Figure 2). With the addition of sulfate to the soil via (NH4)2SO4, soluble SO4 2- in the soil solution is expected to be elevated according to the addition of (NH4)2SO4. Though there was a numerical increase in soluble SO4 2- when 115 kg (NH4)2SO4 ha-1 was added to the soil, this was not enough to raise the soluble SO4 2- concentrations in solution significantly compared to the 0 kg (NH4)2SO4 ha-1 rate.
For the first three weeks after flooding, SO4 2- concentrations in the 0 kg (NH4)2SO4 ha-1 and 115 kg (NH4)2SO4 ha-1 treatment remained fairly steady, beginning to increase around the third week after flooding. However, SO4 2- concentrations in the 230 kg (NH4)2SO4 ha-1 treatment gradually increased during that time. Mineralization of organic matter is promoted when the pH of the soil solution is between 6.5 and 7 [26]. Microbial activity increasing due to the ideal pH when flooded and from the warm environment are likely causing an increase in mineralization of organic S, causing the increase in soluble SO4 2-. Based on the recovered Eh data, soils with 230 kg (NH4)2SO4 ha-1 did not go below -100 mV for the duration of the experiment, but the 0 kg (NH4)2SO4 ha-1 and 115 kg (NH4)2SO4 ha-1 treatments achieved -100 mV around 19 days after flooding.
After the termination of the experiment, plants were removed from buckets to wash and examine the roots. As expected due to the above ground appearance, root blackening signature of H2S toxicity did not appear in any of the rice roots. A few black streaks were noted in the 230 kg (NH4)2SO4 L-1 treatment in the H soils. According to the suggested protocol to identify if the blackening is due to H2S toxicity, these roots were left in the sun for an hour, and then examined again for signs of blackening [9].
Though there was sufficient (NH4)2SO4 applied in the treated buckets to intensify SO4 2- reduction and induce H2S toxicity symptoms, there are several reasons that may be responsible for the lack of symptomology. First, rice roots are known to oxidize the rhizosphere which prevents toxic substances produced by the reduced conditions from affecting the roots [31]. The large volume of roots confined in a relatively small bucket likely produced enough O2 to effectively oxidize any H2S that was produced, therefore avoiding the buildup and toxicity of H2S. Even though a straight head susceptible cultivar was used to improve chances of H2S toxicity occurring [33], these sheer volume of roots in each bucket appeared to produce ample O2 to prevent H2S toxicity from occurring. Another possible explanation for the lack of symptoms in the roots is the high temperature in the greenhouse during this experiment which was near 40°C during the day. According to Vamos [34], H2S toxicity occurred in rice fields under cool, cloudy conditions with low atmospheric pressure, but H2S toxicity did not occur under hot conditions with higher atmospheric pressure. In a study to investigate if temperature influenced the occurrence of H2S toxicity, [34] found that H2S toxicity occurred when temperatures were dropped from 30 to 6°C, but no symptoms appeared when 30°C temperatures were maintained. Severe symptoms of H2S toxicity near cold well water inlets in Arkansas have also made cold temperature a suspect in causing this disorder [7]. However, field reports in Arkansas show that H2S toxicity has been most severe in hot, dry years compared to more mild weather years [7].
Even though symptoms did not appear above or below ground, all above ground biomass was analyzed for nutrient content (Table 3). With Zn concentrations in the leaf tissue all above the critical content, we can conclude that our foliar application of ZnSO4 was effective in correcting the deficiency [6]. The major effect of H2S toxicity is the root damage that prevents nutrient and water uptake [6]. With this in mind, nutrient content would be expected to be below optimal in plants affected by H2S toxicity. However, P was above optimal in all plants as was K except in HR-N soils with all fertilizer treatments and PTRS with the 0 kg (NH4)2SO4 ha-1 treatment where K was below optimal. For S content, above ground biomass for all treatments was above the critical deficiency level [24]. Interestingly, Fe content in all plants except those with the 0 kg (NH4)2SO4 ha-1 and 115 (NH4)2SO4 ha-1 treatment grown in HR-Y soils had concentrations of Fe above the toxicity level [24]. Typically in plants affected by H2S toxicity, Fe concentrations in the leaf tissue are extremely high [35]. Despite the high Fe contents, symptoms of H2S toxicity were not present. Based on the leaf tissue report with the majority of nutrients above the critical content, we can conclude that the roots were not damaged by H2S and were able to take up sufficient nutrients from the soil.
| Location | Fertilizer Rate | P | K | Ca | Mg | S | Na | Fe | Mn | Zn | Cu | B | |||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| kg ha-1 | --------------------%-------------- | --------------------mg kg-1--------------- | |||||||||||||||
| H | 0 | 0.20 | 0.98 | 0.47 | 0.23 | 0.09 | 461 | 1200 | 903 | 32.6 | 1.15 | 3.87 | |||||
| H | 115 | 0.19 | 1.06 | 0.49 | 0.20 | 0.09 | 293 | 878 | 952 | 35.8 | 1.03 | 4.20 | |||||
| H | 230 | 0.20 | 1.03 | 0.49 | 0.23 | 0.10 | 441 | 556 | 996 | 37.5 | 1.14 | 4.25 | |||||
| HR-Y | 0 | 0.19 | 1.00 | 0.37 | 0.22 | 0.08 | 146 | 265 | 751 | 36.5 | 0.82 | 4.75 | |||||
| HR-Y | 115 | 0.19 | 0.96 | 0.43 | 0.23 | 0.09 | 144 | 210 | 747 | 39.0 | 0.95 | 5.22 | |||||
| HR-Y | 230 | 0.18 | 1.06 | 0.44 | 0.22 | 0.40 | 193 | 465 | 766 | 34.8 | 122 | 5.08 | |||||
| HR-N | 0 | 0.22 | 1.51 | 0.32 | 0.25 | 0.09 | 1050 | 285 | 619 | 21.6 | 0.83 | 6.09 | |||||
| HR-N | 115 | 0.23 | 1.52 | 0.26 | 0.23 | 0.09 | 946 | 367 | 574 | 23.2 | 0.83 | 5.76 | |||||
| HR-N | 230 | 0.21 | 1.58 | 0.32 | 0.25 | 0.11 | 730 | 1010 | 645 | 24.0 | 1.50 | 5.65 | |||||
| PTRS | 0 | 0.22 | 1.62 | 0.31 | 0.34 | 0.12 | 677 | 469 | 1136 | 48.0 | 1.95 | 6.89 | |||||
| PTRS | 115 | 0.22 | 1.42 | 0.30 | 0.32 | 0.13 | 663 | 241 | 905 | 53.9 | 2.34 | 4.86 | |||||
| PTRS | 230 | 0.19 | 1.46 | 0.35 | 0.29 | 0.12 | 448 | 1753 | 1068 | 48.7 | 3.10 | 4.90 | |||||
Table 3: Mean plant tissue elemental concentration.
Summary
The objective of this study was to evaluate the effect of (NH4)2SO4 fertilizer additions on soils prone to H2S toxicity compared to soils where this disorder has never been reported. Our hypothesis was that symptoms of H2S toxicity would occur in the treatments with the highest rate of (NH4)2SO4 fertilizer, 230 kg ha-1, in the soils most prone to H2S toxicity, H and HR-Y. A straight head susceptible cultivar of rice (CL 151) was used in this experiment since these cultivars also tend to be susceptible to H2S toxicity. In order to evaluate the effects of (NH4)2SO4 and the different soil locations, soluble SO4 2- and Eh were monitored. At the end of the experiment, all roots were washed and evaluated for the blackening symptom. Tissue analysis was performed by a nitric acid digestion for total S.
An initial soil test was performed on the soils collected and used for this experiment. Unexpectedly, the two soils prone to H2S toxicity, H and HR-N, were not similar to each other. Instead, H and HR-Y were very similar in their physical and chemical attributes as were PTRS and HR-N.
After these soils were flooded, one major problem occurred; severe Zn deficiencies appeared in the rice grown primarily in PTRS and HRN soils. Since flooding increases the availability of certain nutrients, particularly P, increased uptake of P likely suppressed the uptake of any available Zn causing plant deficiency [24]. This is most likely the major cause due to the over application of P in this experiment to avoid P deficiencies.
Results from the soil solution collected throughout the experiment showed a significant difference between soil locations for the first three weeks after flooding (p values ranged from 0.0183 to<0.0001). H and HR-N were not statistically different from each other but were different from HR-Y and PTRS. Since the results from HR-N and HR-Y were opposite of what was expected, this likely means that H2S toxicity is driven by a multitude of other factors and conditions such as microbial populations, Eh, OM content, as well as SO4 2- content.
Rather than SO4 2- concentrations immediately declining after flooding, as seen in the literature [28,29,30,31], SO4 2- concentrations remained fairly constant in all soil locations for the first two weeks after flooding, then began to increase. While Eh did decline immediately in all locations, only HR-N dropped below the SO4 2- reducing potential by the end of three weeks.
Along with the differences between locations, fertilizer treatments were significantly different for the first three weeks after flooding (p values range from 0.0275 to<0.0001). As expected, SO4 2- concentrations were highest in soils with the highest rate of (NH4)2SO4 applied, and the lowest SO4 2- concentration was in the soils with no (NH4)2SO4 added, while soils with the middle rate of (NH4)2SO4 had concentrations in between. For the 0 kg (NH4)2SO4 ha-1 and 115 kg (NH4)2SO4 ha-1 rates, concentrations of SO4 2- in solution remained steady for the first two weeks after flooding then began to increase. In the 230 kg (NH4)2SO4 ha-1, SO4 2- concentrations slowly increased for the first three weeks after flooding. The increase in soluble SO4 2- concentrations after two weeks was, again, likely due to the foliar application of ZnSO4. However, the increase in the 230 kg (NH4)2SO4 ha-1 treatment starting at day two after flooding was possibly due to the mineralization of OM [26], but is unclear.
After terminating the experiment, washing all rice roots revealed minimal root blackening, which is the most predominant symptom of H2S toxicity. Due to the small area that the roots were able to inhabit, the oxidation via the aerenchyma likely prevented H2S toxicity [32].
Nutrient content of the above ground biomass collected at the end of the experiment also indicated that H2S toxicity did not occur. With the majority of nutrients at or above the optimal levels, roots did not appear to be damaged by H2S [6].
Though symptoms of H2S toxicity were not present in this experiment, we can conclude from the different soils used and (NH4)2SO4 treatments used that H2S toxicity is driven by more than soil SO4 2-content. This disorder is multifaceted, and further study should be aimed at understanding how SO4 2- content, Eh, microbial populations, OM content, and water temperature influence the occurrence of H2S toxicity.
References
- Hardke JT (2015) Trends in Arkansas rice production, 2014. Wells rice research studies 2014, Fayetteville, USA.
- Richardson JW, Outlaw JL (2010) Economic contributions of the US rice industry to the US economy. Texas A&M University, College Station, USA.
- Hardke JT (2016) Trends in Arkansas rice production, 2015. Wells rice research studies 2014, Fayetteville, USA.
- Yoshida S (1981) Fundamentals of rice crop science. Int. Rice Res. Inst. Los Banos, Laguna, Philippines.
- Baba I, Inada K, Tajima K (1964) Mineral nutrition of the occurrence of physiological diseases. The mineral nutrition of the rice plant; proceedings. John Hopkins Press, Baltimore, USA.
- Tanaka A, Yoshida S (1966) Nutritional disorders of the rice plant. Technical Bulletin 7. IRRI, Los Banos, Philippines.
- Wamishe Y (2015) Weather and Akiochi disease of rice-is there a link? Arkansas Row Crops. University of Arkansas Division of Agriculture Research and Extension, USA.
- Wamishe Y, Cartwright R, Lee F (2013) Management of rice disease. Arkansas Rice Production Handbook. University of Arkansas, USA.
- Hardke JT, Wamishe Y (2015) Arkansas rice update 2-7-15. Arkansas Row Crops. University of Arkansas Division of Agriculture Research and Extension, USA.
- Norman R, Slaton N, Roberts T (2013) Soil fertility. Arkansas rice production handbook. University of Arkansas, USA.
- DeLong RE, Carroll SD, Slaton NA, Mozaffari M, Herron C (2016) Soil-test and fertilizer sales data: summary for the 2015 growing season. Arkansas soil fertility studies 2015. Fayetteville, USA.
- Hardke JT, Mazzanti R, Baker R (2017) 2017 Arkansas rice quick facts. University of Arkansas Division of Agriculture Research and Extension, USA.
- Thomas GW (1996) Soil pH and soil acidity. Methods of soil analysis. SSSA Book Series. Madison, WI, USA.
- Gavlak RG, Horneck DA, Miller RO, Kotuby-Amacher J (2003) Soil, plant, and water reference methods for the western region. Colorado State University, USA.
- Bremner JM (1996) Nitrogen-total. Methods of soil analysis. SSSA Book Series 5, Madison, USA.
- Nelson DW, Sommers LE (1996) Total carbon, organic carbon, and organic matter. Methods of soil analysis, Madison, USA.
- Schulte EE, Hopkins BG (1996) Estimation of soil organic matter by weight-loss-on-ignition. Soil organic matter: analysis and interpretation, Madison, USA.
- Helmke PA, Sparks DL (1996) Lithium, sodium, potassium, rubidium, and cesium. Methods of soil analysis, Madison, WI, USA.
- Patrick WH, Gambrell RP, Faulkner SP (1996) Redox measurements of soil. Methods of soil analysis. Madison, WI, USA.
- Gao S, Tanji KK, Scardaci SC, Chow AT (2002) Comparison of redox indicators in a paddy soil during rice-growing season. Soil Sci Soc Am J 66: 805-817.
- Slaton NA, Wilson Jr CE, Norman RJ, Gbur Jr EE (2002) Development of a critical Mehlich 3 soil zinc concentration for rice in Arkansas. Commun Soil Sci Plant Anal 33: 2759-2770.
- Slaton NA, Wilson Jr CE, Norman RJ, DeLong RE (2006) Correlation of soil pH and Mehlich-3 phosphorus with postflood rice phosphorus concentrations in Arkansas. Commun Soil Sci Plant Anal 37: 2819-2831.
- Ponnamperuma FN (1972) The chemistry of submerged soils. Adv Agron 24: 29-96.
- Dobermann K, Fairhurst T (2000) Mineral Deficiencies. Rice: nutrient disorders and nutrient management, Singapore.
- Strawn DG, Bohn HL, O’Connor GA (2015) Redox reactions in soil. Soil chemistry 4th ed. John Wiley and Sons, Hoboken, USA.
- Ponnamperuma FN (1984) Effects of flooding on soils. Flooding and plant growth. Academic Press, Orland, USA.
- Reddy KR, Feijtel TC, Patrick Jr WH (1986) Effect of soil redox conditions on microbial oxidation of organic matter. The Role of Organic Matter in Modern Agriculture. Martinus Nijhoff Publishers, Dordrecht, Netherlands.
- Ogata G, Bower C (1965) Significance of biological sulfate reduction in soil salinity. Soil Sci Soc Am Proc 29: 23-25.
- Connell W, Patrick W (1969) Reduction of sulfate to sulfide in waterlogged soil. Soil Sci Soc Am J 33: 711-715.
- Ponnamperuma FN (1981) Some aspects of the physical chemistry of paddy soils. Academia Sinica, Proceedings of symposium on paddy soils. Science Press, Springer, Beijing, China.
- Gao S, Tanji KK, Scardaci SC (2004) Impact of rice straw incorporation on soil redox status and sulfide toxicity. Agron J 96: 70-76.
- Ando T, Yoshida S, Nishiyama I (1983) Nature of oxidizing power of rice roots. Plant Soil 72: 57-71.
- Joshi MM, Ibrahim IKA, Hollis JP (1975) Hydrogen sulfide: effects on the physiology of rice plants and relation to straighthead disease. Phytopathology 65: 1165-1170.
- Vamos R (1964) The release of hydrogen sulphide from mud. Soil Sci 15: 103-109.
- Tanaka A, Mulleriyawa R, Yasu T (1986) Possibility of hydrogen sulfide induced iron toxicity of the rice plant. Soil Sci Plant Nutr 14: 1-6.
- Combs SM, Nathan MV (1998) Soil organic matter. Recommended chemical soil test procedures. North Central Region Research Publication, Columbia, USA.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi