Research Article, J Spine Neurosurg Vol: 8 Issue: 3
Endovascular Treatment of Acute Ischemic Stroke beyond 6 Hours after Symptoms Onset: A Single Centre Experience
Orazio Buonomo1*, Antonio Armando Caragliano1, Rosario Papa1, Antonio Pitrone1, Mariano Velo1, Cristina Dell’Aera2, Angela Alibrandi3, Serena Giovanna Martorana4, Agostino Tessitore1, Marcello Longo1 and Sergio Lucio Vinci1
1Neuroradiology Unit-Biomedical Sciences and of Morphologic and Functional Images, University of Messina, Messina, Italy
2Department of Clinical and Experimental Medicine, UOSD Stroke Unit, University of Messina, Messina, Italy
3Department of Economics, Unit of Statistical and Mathematical Sciences, University of Messina, Messina, Italy
4Child and Adolescent Neurology and Psychiatry Public Service ASP Catania, Catania, Italy
- *Corresponding Authors:
- Orazio Buonomo
Neuroradiology Unit-Biomedical Sciences and of Morphologic and Functional Images
University of Messina, Messina, Italy
Tel: 0039-3465118088 E-mail:oraziobuonomo@gmail.com
Received:March 23, 2019Accepted: June 05, 2019 Published: June 11, 2019
Citation: Buonomo O, Caragliano AA, Papa R, Pitrone A, Velo M, et al. (2019) Endovascular Treatment of Acute Ischemic Stroke beyond 6 Hours after Symptoms Onset: A Single Centre Experience. J Spine Neurosurg 8:3.
Abstract
Background: Ischemic stroke is characterized by the sudden onset of neurologic symptoms related to the occlusion of cerebral vessels. Recently, some randomized clinical trials have demonstrated the effectiveness of mechanical arterial revascularization within 6 hours after the symptoms onset, getting through patient’s selection and the new endovascular devices. Purpose: Our aim is to characterize the factors that may influence the endovascular treatment, in order to extent the therapeutic time window. Results: 69 patients were treated beyond 6 hours from symptoms onset. The median time to treatment was 8.3 hours. Successful recanalization was achieved in the 85%. At discharge the median NIHSS score was 12. At 3 months the modified Ranking score 0-2 was achieved in the 33.3%, with a mortality rate of 26%. Conclusion: Our experience evaluates the role of endovascular therapy beyond 6 h after stroke onset secondary to a strict imaging guided selection of the patients, which is essential to achieve good clinical results.
Keywords: Ischemic stroke; Endovascular treatment; Time window; Patient selection
Introduction
The vast majority of cerebral stroke cases are caused by transient or permanent occlusion of a cerebral blood vessel, an ischemic stroke (IS), eventually leading to brain infarction [1]. The final infarct size and the neurological outcome depend on a multitude of factors such as the duration and severity of ischemia, the existence of collateral systems and an adequate systemic blood pressure, etiology and localization of the infarct, but also on age, sex, comorbidities with the respective multimedication and genetic background. Thus, Ischemic Stroke (IS) is a highly complex and heterogeneous disorder, characterized by the sudden onset of neurologic symptoms related to the occlusion site of cerebral vessels.
Stroke remains the leading cause of morbidity and mortality around the world, with an incidence rate of 95-290/100.000 people per year and a fatality rate at 1 month ranging from 13% to 35 [2,3]. Of all strokes, 85% are ischemic in nature [4].
Before the 2015, the main treatment for the IS in the acute phase (AIS) was the intravenous administration of pharmacologic thrombolytic agents, because the first randomized clinical trials (rCT) on endovascular therapy (EVT) were a failure [5-8].
Through the imaging-guided patients’ selection and the new endovascular devices, recent rCTs [9,10] demonstrated the great importance and effectiveness of mechanical arterial revascularization and the need to create realities able to offer the best therapeutic opportunities. These studies validate the EVT of ischemic stroke within 6 hour after the symptoms onset [11].
Recently two rCTs have been increased the therapeutic window, proving the effectiveness of EVT until 16 hours (DEFUSE 3) [12] or 24 hours (DAWN) [13], from the neurological status onset.
In the DEFUSE 3 and the DAWNTM trials the main role was played by the patient’s selection, performed with multimodality Computed Tomography (CT) or Magnetic Resonance Imaging (MRI), such as diffusion and perfusion weighted images [14].
In our single-centre experience, we analyzed all patients with AIS treated by endovascular procedure beyond 6 hours after the onset of symptoms, in order to identify which factors could influence the EVT results.
Materials and Methods
We retrospectively analyzed the clinical and radiological data from January 2014 to February 2018 of all patients suffering from AIS, regardless of the endovascular device used. The main inclusion criterion is the time window longer than 6 hours, from the stroke onset to the arrival in the angiographic room.
Other inclusion criterion is the presence of salvable eloquent brain areas on radiological images.
No other limitations were made regarding inclusion. No minimal “National Institute of Health Stroke Scale” (NIHSS) score was considered. We firmly believe that with the occlusion of a big vessel, it is better to treat the patient. Few neurological symptoms are a specific indicator of the presence of good collateral circulation [15], even if it is not easy to evaluate their efficacy on the long run.
Clinical examination
Every patient was evaluated by two expert neurologists belonging to our Stroke Unit, who calculated the NIHSS and the modified Rankin Scale (mRS) scores, the latter after a careful interview with the patient’s relatives.
Imaging protocol
Patients were initially exposed to a non-contrast CT scan (NCCT), to rule out intracranial haemorrhage, allowing the neurologist to eventually administer the intravenous (IV) thrombolysis).
Immediately after the MRI scan was performed with a few short sequences by default, such as Diffusion Weighted Imaging (DWI), Time of Flight (TOF-3D) for Magnetic Resonance Angiography (MRA) and Fluid attenuation Inversion Recovery (FLAIR). Additional sequences as Turbo Spin Echo-T2 Weighted Imaging (TSE-T2), Susceptibility Weighted Imaging (SWI), Pseudo- Continuous Arterial Spin Labelling (pcASL) could be added depending on the case.
If the patient could not be subjected to MRI, a multiphase CT angiography (CTA) was performed to assess the leptomeningeal collateral flow.
Multiphase CTA is an imaging tool that provides three timeresolved cerebral angiograms of pial arterial filling in the whole brain, from the skull base to the vertex after contrast material injection, unlike conventional single phase CTA [16].
The first phase of multiphase CTA is conventional arch to vertex scan; the next two phases are sequential skull base to vertex acquisitions performed in the mid and late venous phases.
The collateral flow in pial artery is evaluated on a 6 point scale (0 to 5, better scores indicates greater collateral flow); besides multiphase CTA doesn’t need any mathematical algorithm or complex post processing and it has an excellent inter-rater reliability, that can be used to predict clinical outcomes in patient with AIS.
Routinely we used to include patients for treatment evaluating the volume of ischemic core on DWI sequence applying the Alberta Stroke Program of Early CT score (ASPECTs) [17].
When the MRI shows an ASPECT score suitable for the EVT (>4), according to the general clinical condition of the patients, were transferred to the angiographic room quickly.
Endovascular procedure
The first approach in our experience is the so-called “a direct aspiration first pass technique” (ADAPT) [18,19], unless change the technique in case of failure usually switching into the “Solumbra Technique” (thromboaspiration and stent-retriever performed together) [20].
The procedure begins with the groin puncture to the mid-portion of the common femoral artery, putting in a short sheath of 8 French.
We typically use an 8 French guiding catheter (GC), placed into the internal carotid artery (ICA) or in the vertebral artery (VA) through 5 a French 125 cm length angiography catheter, and advanced over a 0.035 inches guidewire.
Through the GC, a large-bore aspiration catheter and a microcatheter with a 0.014 inches microguide wire are introduced, getting a triaxial system.
All the system is advanced into the proximal end of the thrombus.
In our opinion it’s recommended to avoid going across the thrombus with the system, because it could cause the clot disruption and its migration in a distal direction. Then, the aspiration catheter is advanced over the thrombus to promote its engagement.
Once the large-bore aspiration catheter is in place, the inner micro-catheter and micro-guidewire are removed. Thereafter, the proximal hub of the aspiration catheter is connected to the aspiration pump system through the aspiration tubing.
A negative pressure is necessary to create a vacuum state between the tip of the large-bore aspiration catheter and the clot. Absence of backflow mostly indicates that the thrombus is trapped in the catheter.
After 2 min, the aspiration catheter is slowly withdrawn under a continuous aspiration.
In the meanwhile the GC is subjected to a negative pressure connecting itself to a 50 ml syringe in a blocked aspiration position, to remove any residual thrombus fragments. This process can be repeated until successful reperfusion is achieved. However, usually after three attempts, the “Solumbra Technique” is recommended.
In this case the microcatheter is gently navigated beyond the site of occlusion using a 0.014 inches micro-guidewire. Simultaneously, a selective contrast injection through the micro-catheter and the aspiration catheter is performed to confirm the correct position within the occluded parent artery and the thrombus length at the site of occlusion. An appropriate-sized stent retriever is so deployed along the clot. After waiting 3-5 minutes to favourite the thrombus engagement, the stent retriever and the micro-catheter are retrieved simultaneously; the large bore aspiration catheter is advanced to the proximal part of the stent, aspirating eventual thrombus fragments and preventing any distal embolization.
At the end of procedure, a final angiogram is performed to verify the recanalization rate.
Endpoints
The first primary endpoint is the evaluation of clinical improvement, considering the average difference of the NIHSS scores at admission and discharge. The clinical status is also evaluated by the mRS scores at admission and at 90 days after the onset of stroke.
The main safety endpoint was stroke-related death at 90 days and symptomatic intracranial haemorrhage (sICH). The sICH is defined, according to European Cooperative Acute Stroke Study III criteria, as the presence parenchymal hemorrhage with a neurological decline NIHSS ≥ 4 or death, judged to be the predominant cause of neurologic deterioration during recovery, having furthermore a consistency correlation with mortality rates [21].
Results
On 404 endovascular procedures performed for AIS, 69 patients were treated after 6 hours from stroke symptoms onset, out of the therapeutic time window.
The majority of these patients were treated before the publication of the new trials DEFUSE 3 and DAWN, so out of the previous guidelines. Patients are 38 females and 31 males with a median age of 74 years old (minimum 39, maximum 93)
At baseline the median admission NIHSS score was 16.5 with a minimum score of 2 and a maximum of 36.
Moreover the median mRS was 1 (minimum 0, maximum 3).
According to the occlusion site, patients were divided in 2 groups: AIS of anterior circulation (84.1%) and AIS of the posterior circulation (15.9%).
Every patient treated had an ASPECT score on MRI diffusionweighted images more than 4, representing an acceptable amount of salvable brain tissue.
The median time from onset to groin puncture (time to treatment) was 8.3 hours with a minimum time delay of 7.3 hours and a maximum of 16 h.
On every first angiogram acquired, we estimated the collateral circulation both to confirm the presence of cerebral tissue salvable and looking for a correspondence between the involved areas on DWI and the collateral flow.
Successful recanalization, mTICI score 2B-3, was achieved in the 85% of all cases.
At the discharge, the median NIHSS score was 12, with a minimum score of 2 and a maximum score of 40; higher values were detected in the posterior circulation group.
At 3 months a good clinical status, mRS 0-2, was achieved in the 33.3% of all patients, with a median value of 3.
The mortality rate at 3 months was 26% and it is more evident in patients affected by posterior AIS.
No sICH or other complications, secondary to the procedure, occurred (Figures 1a and 1b).
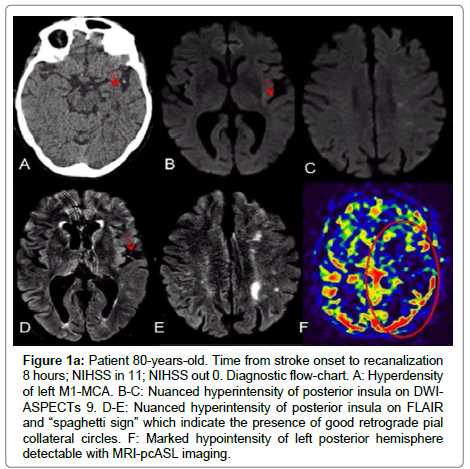
Figure 1a:Patient 80-years-old. Time from stroke onset to recanalization 8 hours; NIHSS in 11; NIHSS out 0. Diagnostic flow-chart. A: Hyperdensity of left M1-MCA. B-C: Nuanced hyperintensity of posterior insula on DWIASPECTs 9. D-E: Nuanced hyperintensity of posterior insula on FLAIR and “spaghetti sign” which indicate the presence of good retrograde pial collateral circles. F: Marked hypointensity of left posterior hemisphere detectable with MRI-pcASL imaging.
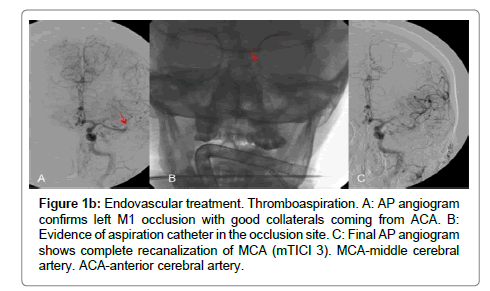
Figure 1b:Endovascular treatment. Thromboaspiration. A: AP angiogram confirms left M1 occlusion with good collaterals coming from ACA. B: Evidence of aspiration catheter in the occlusion site. C: Final AP angiogram shows complete recanalization of MCA (mTICI 3). MCA-middle cerebral artery. ACA-anterior cerebral artery.
The statistical analysis, made by the Wilcoxon test, showed a significant difference between the NIHSS score at admission and at discharge, with lower value in the latter case (p<0.001).
Moreover the non-parametric evaluation demonstrates that the NIHSS score at discharge is inversely proportional to mTICI score (p<0.012), so optimal recanalization are correlated to better neurological improvement.
The mTICI score influences also the mRS value at 3 months, indeed inadequate recanalizations are associated with lower scores on the mRS and the other way around.
Besides NIHSS score is directly proportional to the mRS values at 3 months (p<0.001), so higher NIHSS scores are correlated to worst clinical conditions.
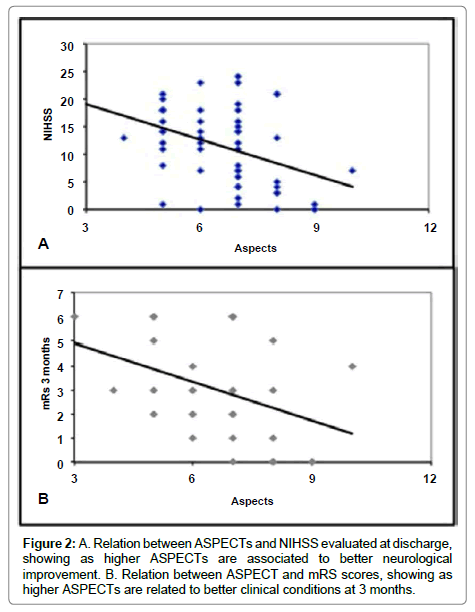
Another non-parametric analysis showed the main role of patient selection, so it has been demonstrate that higher ASPECTs values on MRI are correlated with lower NIHSS score at discharge and so with best clinical results, also at 3 months.
It is necessary to specify that the therapeutic benefit has been maintained along the whole spectrum of the aspect scores, as the successful treatment has allowed us to preserve the spared areas.
Discussion
Until few years ago, IV administration of recombinant tissue plasminogen activator (rt-PA) was the standard care in AIS. IV rt-PA was Food and Drug Administration (FDA) approved and endorsed by consensus guidelines for the treatment of select patients with ischemic stroke within 3 hours of symptoms onset in the United States (Class I, Level A evidence), Canada, and Europe [22-25]. More recent evidence from the European Cooperative Acute Stroke Study (ECASS 3) [21] suggested clinical benefit for IV rt-PA administration up to 4.5 hours after symptoms onset within a subgroup of stroke patients.
In the National Institute of Neurological Disorders and Stroke rt-PA Stroke Study Group (NINDS) [19] trial, patients treated with rt-PA were at least 30% more likely than patients given placebo to achieve a mRS value ≤ 1.
The NINDS study showed an 11-13% absolute increase in favorable outcomes at 3 months vs. placebo without mortality benefit [26].
However the location and size of thrombus are important variables to determine the recanalization rates after IV therapy [27].
Subsequently Intra-Arterial (IA) thrombolysis has emerged as a promising intervention, especially for AIS patients with contraindication for IV rt-PA [28].
Prolyse in Acute Cerebral Thromboembolism (PROACT) [29] was the first randomized trial that evaluated the administration of recombinant pro-urokinase (rproUK) in patients with angiographically documented proximal middle cerebral artery (MCA) occlusion.
The aim of the latter study was to test the safety and clinical efficacy of IA local infusion of plasminogen activators in patients with AIS due to MCA occlusion of less than 6 hours.
Investigators found a significant association of local rpro-UK infusion with greater recanalization rates in acute stroke patients with M1 or M2 occlusions compared with placebo (57.7% vs. 14.3%).
Contrary to the positive findings of the Prolyse in Acute Cerebral Thromboembolism (PROACT), a large open-label trial “Local Versus Systemic Thrombolysis for Acute Ischemic Stroke (SYNTHESIS) Expansion” [8], found that IA thrombolytic therapy was not superior to standard treatment with IV rt-PA. Disability-free outcome at 90 days was not significantly different between the IA thrombolytic and IV rt-PA groups (30.4% vs. 34.8%).
As the SYNTHESIS Expansion, other trials were not able to demonstrate the efficacy of the EVT like the “A Trial of Imaging Selection and Endovascular Treatment for Ischemic Stroke” (MR RESCUE) [7] and the “Endovascular Therapy after Intravenous t-PA versus t-PA Alone for Stroke” (IMS III) [6].
In the 2015, the successful five multicentre open-label randomized trials, including a multicenter randomized clinical trial of endovascular treatment for acute ischemic stroke in the Netherlands (MR CLEAN) [9], the Endovascular Treatment for Small Core and Anterior Circulation Proximal Occlusion with Emphasis on Minimizing CT to Recanalization Times (ESCAPE) [10], the Solitaire™ With the Intention For Thrombectomy as PRIMary Endovascular Treatment trial (SWIFT PRIME) [30], the Extending the Time for Thrombolysis in Emergency Neurological Deficits-Intra-Arterial trial (EXTEND-IA) [31] and the Endovascular Revascularization With Solitaire Device Versus Best Medical Therapy in Anterior Circulation Stroke Within 8 hours trial (REVASCAT) [32], demonstrated that the combined treatment with endovascular devices and IV rt-PA within 6 hours after stroke onset is superior to standard medical therapy (IV rt-PA alone) for AIS caused by a proximal large artery occlusion of the anterior circulation (Figure 2).
Immediately after the announcement of the MR CLEAN results, the other four trials were terminated prematurely for interim analysis. The results of these trials were then published in quick succession in late 2014 and early 2015. The number needed to treat (NNT) for these five trials ranged from a minimum of three (EXTEND-IA) to a maximum of 7.4 (MR CLEAN). The success of these trials has revolutionized the stroke therapy.
It is very important to underline that these studies are focused on the concept that stroke is a time-dependent disease, with best clinical results achieved when the EVT is performed within 6 hours from the onset.
To sum up, these trials make a convincing case for the administration of early EVT to limit post-stroke disability in patients with large vessel occlusion in the anterior circulation. They also reinforce the importance of early treatment based on an inverse association between time to endovascular reperfusion and better functional outcome.
A recent meta-analysis of Saver et al. has indicated the benefit of EVT plus medical therapy, showing that the odds of better disability outcomes at 90 days with the endovascular group declined with longer time from symptoms onset to arterial puncture retaining statistical significance through 7 hours and 18 minutes [32].
In the current year, two trials have been published trying to extend the time to endovascular reperfusion up to 24 hours, the DEFUSE 3 [12] and the DAWN [13] trial.
More and more authors discuss about the “DAWN of a new era” [5]; this means we are focusing on increasing the selection of the single patient and on its imaging parameters, allowing to break down the time barrier and to extend the therapeutic time window.
DEFUSE 3 selects AIS patients using penumbral mismatch on CTP or MRI who may benefit from EVT between 6 h and 16 h after onset.
Patients were eligible if they had an initial infarct volume (ischemic core) of less than 70 ml, a ratio of volume of ischemic tissue to initial infarct volume of 1.8 or more, and an absolute volume of potentially reversible ischemia (penumbra) of 15 ml or more.
The DAWN trial, which recruited patients treated with Trevo stent retriever within 6-24 hours after stroke onset, was terminated early after an interim analysis of the first 200 patients demonstrated a 73% relative risk reduction in disability (48.6% in MT group vs. 13.6% in control group). The DAWN study has confirmed that advanced imaging based patient selection outweighs time-based decision making in acute ischemic stroke.
Patients’ selection in the DAWN study and in the DEFUSE 3 study was very strict. In the DAWN Study, patients were eligible for inclusion in the trial if they had evidence of occlusion of the intracranial internal carotid artery, the first segment of the middle cerebral artery, or both on CT angiography or magnetic resonance angiography. In addition, patients had to have a mismatch between the severity of the clinical deficit and the infarct volume, which was defined according to the following criteria: those in Group A were 80 years of age or older, had a score of 10 or higher on the National Institutes of Health Stroke Scale (NIHSS; scores range from 0 to 42, with higher scores indicating a more severe deficit), and had an infarct volume of less than 21 ml; those in Group B were younger than 80 years of age, had a score of 10 or higher on the NIHSS, and had an infarct volume of less than 31 ml; and those in Group C were younger than 80 years of age, had a score of 20 or higher on the NIHSS, and had an infarct volume of 31 to less than 51 ml [13]. In the DEFUSE 3 trial, Patients were eligible if they had an initial infarct volume (ischemic core) of less than 70 ml, a ratio of volume of ischemic tissue to initial infarct volume of 1.8 or more, and an absolute volume of potentially reversible ischemia (penumbra) of 15 ml or more[11]. The results of these studies were also included in the 2018 American Guidelines for management of AIS [13].
Our data confirmed the EVT usefulness out of the traditional time window, showing a significant correspondence between the recanalization rate and the NIHSS score at discharge. The final NIHSS depends also by the ASPECTs calculated on the MRI in acute phase. Indeed higher ASPECT values, indicating more salvable cerebral tissue, are associated with lower NIHSS score.
The final NIHSS is also directly proportional to the mRS at 3 months, so better neurological conditions at discharge are related with good clinical status.
Therefore the ASPECT score and the recanalization rates modify the clinical outcome in the mid-term, being the main variables influencing the neurological status after the procedure.
The suffering brain parenchyma seen on the MRI was also related to the collateral flow on angiography. We found an inverse proportion between the involved areas on DWI and the collateral circulation; lower ASPECTs indicate poor collaterals so insignificant chance to clinical recovery.
Therefore, our analysis identify which issues determine the final clinical results, showing a direct correlation among the collateral flow, ASPECT and mTICI scores, that are able to modify the NIHSS at discharge and the mRS score at 3 months.
In our experience, we reported good clinical outcomes (mRS 0-2) in the 33, 3% of all cases against the 48,6% and the 45% of DAWN and DEFUSE 3 trials respectively.
Moreover, the mortality rate was slightly higher in our analysis 26% vs. 19% and 14% for the DAWN and the DEFUSE 3 respectively.
Probably, the difference in these results is due to patient selection; indeed as inclusion criteria we have not different selection according the age and we used an ASPECTs on MRI >4, lower than one used in the other two studies.
In our study, patients were enrolled even if they had lower ASPECT scores: however, the rate of mRS at 90 days and the clinical benefit is unchanged. So, our experience resulted to be more similar to the real daily clinical practice, rather than to the world of clinical trials.
Furthermore, it is also important to note that our experience does not rely on automatic software, but on the personal experience of the medical operator. This often happens in everyday practice, because not all Stroke Units can afford to have expensive automatic software.
Nevertheless, our data show the main role of EVT in appropriately selected patients with AIS, after 6 h from stroke symptoms onset, in the attempt to reach better clinical results compared to simple standard medical care.
Limitations
Our analysis includes a little sample of patients treated beyond 6 hours from stroke symptoms onset. We had not limitation about the patient’s age and we used the ASPECTs>4, on MRI scan, to select them. These inclusion criteria could have influenced the clinical results, slightly lower than those of literature.
Conclusion
Our experience, according with the main authoritative studies, validate EVT of AIS beyond 6 h after symptoms onset, provided that a strict selection of the patient is carried out on the basis of parameters obtained with second level imaging methods.
Therefore both good collateral circulation and great salvable brain tissue make patient’s eligible to the treatment, despite of the stroke onset time.
Informed Consent
Informed consent was obtained from all individual participants included in the study.
Conflicts of Interest
None of authors have any conflict of interest.
References
- Sommer CJ (2017) Ischemic stroke: experimental models and reality. Acta Neuropathol 133: 245-261.
- Norrving BL, Kissela B (2013) The global burden of stroke and need for a continuum of care. Neurology 15: 80-12.
- Feigin VL, Forouzanfar MH (2014) Global and regional burden of stroke during 1990-2010: findings from the Global Burden of Disease Study 2010. Lancet 383: 245-254.
- Mukherjee D, Patil CG (2011) Epidemiology and the global burden of stroke. World Neurosurgery 76: S85–S90.
- Bhaskar S, Stanwell P (2018) Reperfusion therapy in acute ischemic stroke: dawn of a new era? BMC Neurol 16: 8-18.
- Broderick JP, Palseh YY (2013) Endovascular therapy after intravenous t-PA versus t-PA alone for stroke. N Engl J Med 368:893-903.
- Kidwell CS, Jahan R (2013) A trial of imaging selection and endovascular treatment for ischemic stroke. N Engl J Med 368: 914-923.
- Ciccone A, Valvassori L (2013) Endovascular treatment for acute ischemic stroke. N Engl J Med 368: 904-913.
- Berkhemer OA, Fransen PS (2015) A randomized trial of intraarterial treatment for acute ischemic stroke. N Engl J Med 372: 11-20.
- Goyal M, Femchuk AM (2013) Randomized assessment of rapid endovascular treatment of ischemic stroke. N Engl J Med 372: 1019-1030.
- Malhotra K, Liebeskind DS (2015) Imaging in endovascular stroke trials. J Neuroimaging 25: 517-527.
- Albers GW, Marks MP (2018) Thrombectomy for stroke at 6 to 16 hours with selection by perfusion imaging. N Engl J Med 378: 708-718.
- Nogueira RG, Jadhav AP (2018) Thrombectomy 6 to 24 hours after stroke with a mismatch between deficit and infarct. N Engl J Med 378: 11-21.
- Kim BJ, Kang HG, Kim HJ, Ahn SH, Kim NY et al. (2014) Magnetic Resonance Imaging in Acute Ischemic Stroke Treatment. J Stroke 16: 131-145.
- Sarraj A, Hassan A, Savitz S, Grotta J, Cai C, et al. (2018) Endovascular thrombectomy for mild strokes: how low should we go? Stroke 49: 2398-2405.
- Menon BK, d’Esterre CD (2015) Multiphase CT angiography: a new tool for the imaging triage of patients with acute ischemic stroke. Radiology 275: 510-520.
- Barber PA, Demchuk AM (2000) Validity and reliability of a quantitative computed tomography score in predicting outcome of hyper acute stroke before thrombolytic therapy. ASPECTS Study Group. Alberta Stroke Programme Early CT Score. Lancet 355: 1670-1674.
- Turk AS, Frei D (2014) ADAPT FAST study: A direct aspiration first passes technique for acute stroke Thrombectomy. J Neurointerv Surg 6: 260-264.
- DG Romano, S Cioni, Leonini S, Gennari P, IM Vallone (2016) Manual thromboaspiration technique as a first approach for endovascular stroke treatment: A single-center experience. Interv Neuroradiol 22: 529-534.
- Kang DH, Park J (2017) Endovascular Stroke Therapy Focused on Stent Retriever Thrombectomy and Direct Clot Aspiration: Historical Review and Modern Application. J Korean Neurosurg Soc 60: 335-347.
- Hacke W, Kaste M, Bluhmki E, Brozman M, Dávalos A, et al. (2008) Thrombolysis with alteplase 3 to 4.5 hours after acute ischemic stroke. N Engl J Med 359: 1317-1329.
- Hacke W, Donnan G, Fieschi C, Kaste M, Von Kummer R, et al. (2004) Association of outcome with early stroke treatment: pooled analysis of ATLANTIS, ECASS, and NINDS rt-PA stroke trials. Lancet 363: 768-774.
- Lees KR, Bluhmki E, von Kummer R, Brott TG, Toni D, et al. (2010) Time to treatment with intravenous alteplase and outcome in stroke: an updated pooled analysis of ECASS, ATLANTIS, NINDS, and EPITHET trials. Lancet 375: 1695-1703.
- The National Institute of Neurological Disorders and Stroke rt-PA stroke study group (1995) NINDS-rtPA: Tissue plasminogen activator for acute ischemic stroke. N Engl J Med 333: 1581-1587.
- Kwiatkowski TG, Libman RB, Frankel M, Tilley BC, Morgenstern LB, et al. (1999) Effects of tissue plasminogen activator for acute ischemic stroke at one year. National Institute of Neurological Disorders and Stroke Recombinant Tissue Plasminogen Activator Stroke Study Group. N Engl J Med 340: 1781-1787.
- Riedel CH, Zimmermann P, Jensen-Kondering U, Stingele R, Deuschl G (2011) The importance of size: successful recanalization by intravenous thrombolysis in acute anterior stroke depends on thrombus length. Stroke 2011 42: 1775-1777.
- Rubiera M, Ribo M, Pagola J, Coscojuela P, Rodriguez-Luna D (2012) Systematic review of outcome after ischemic stroke due to anterior circulation occlusion treated with intravenous, intra-arterial, or combined intravenous intra-arterial thrombolysis. Stroke 43: 2350-2355.
- Furlan AJ, Abou-Chebl A (2002) The role of recombinant pro-urokinase (r-pro-UK) and intra-arterial thrombolysis in acuteischaemic stroke: the PROACT trials. Prolyse in Acute Cerebral Thromboembolism. Curr Med Res Opin 18: 44-47.
- Saver JL, Goyal M, Bonafe A, Diener HC, Levy EI (2015) Stent-retriever Thrombectomy after intravenous t-PA vs. t-PA alone in stroke. N Engl J Med 372: 2285-2295.
- Campbell BC, Mitchell PJ (2015) Endovascular therapy for ischemic stroke with perfusion-imaging selection. N Engl J Med 372: 1009-1018.
- Tudor GJ, Chamorro A, Cobo E, María A, Molina C (2015) Thrombectomy within 8 hours after symptom onset in ischemic stroke. N Engl J Med 372: 2296-2306.
- Saver JL, Goyal M, Lugt A, Menon BK, Majoie CB, et al. (2016) Time to treatment with endovascular thrombectomy and outcomes from ischemic stroke: A Meta-analysis. JAMA 316: 1279-1288.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi