Research Article, J Vet Sci Med Diagn Vol: 8 Issue: 1
Excitatory and Inhibitory Amino Acids in Cerebrospinal Fluid of Familial Spontaneous Epileptic Cats
- *Corresponding Authors:
- Tomohiro Yonezawa
Department of Veterinary Clinical Pathobiology, Graduate School of Agricultural and Life Sciences
The University of Tokyo, Bunkyo-ku, Tokyo 113-8657, Japan
E-mail: ayone@mail.ecc.u-tokyo.ac.jp
Received: January 28, 2019 Accepted: February 15, 2019 Published: February 20, 2019
Abstract
Abstract Objective: To evaluate the excitatory and inhibitory amino acids in the cerebrospinal fluid (CSF) from cats with familial spontaneous epilepsy (FSEC). Animals: Thirteen epileptic cats (suspected homozygotes: Affected) and nine non-epileptic cats (suspected heterozygotes: Unaffected) from FSEC line, and six clinical healthy cats which were unrelated FSEC line (Healthy). Procedures: CSF samples were collected before and after vestibular stimulation, which induced generalized seizures only in the affected cats. Excitatory/inhibitory amino acids (aspartate, glutamate, serine, glycine taurine, and gamma-aminobutyric acid (GABA)) levels in the CSF were analyzed by high performance liquid chromatography. In addition, CSF samples from five Affected cats in the interictal phase, four Unaffected cats, and three Healthy cats were used for metabolomics. Results: There was no difference in CSF glutamate concentrations between Affected and Healthy groups, while significantly lower glutamate concentrations were detected in the unaffected group. Following vestibular stimulation, glutamate concentrations were significantly elevated in the Unaffected and Healthy groups, but not in the Affected group. Moreover, the affected group exhibited the lower glutamine concentrations than the Healthy group. There was no difference in GABA concentrations between the groups, although some Affected cats showed elevated GABA. Conclusions and clinical relevance: In FSEC, an imbalance between excitatory and inhibitory neurotransmitters may not be the direct cause of epileptic seizures, while alterations in the glutamate-glutamine cycle may be a causative mechanism. These findings provide new insight into the pathogenesis of FSEC and familial mesial temporal lobe epilepsy in human and animals.
Keywords: Cats; cerebrospinal fluid; Migration time; GABA
Introduction
Imbalance between excitatory and inhibitory neurotransmitters in the brain is considered a causative mechanism of epileptic seizures in humans and animal models [1–3]. In veterinary medicine, several reports have described the amino acid concentrations in the cerebrospinal fluid (CSF) from dogs with idiopathic epilepsy [4–8]. Glutamate is a major excitatory neurotransmitter in the mammalian central nervous system (CNS), and is key for higher brain functions such as learning and memory [9,10]. However, excess glutamate in the brain induces excitotoxicity, which causes numerous CNS pathogenesis including epilepsy [10,11]. For example, glutamate excitotoxicity plays an important role in the initiation, propagation, and continuance of epileptic activity [10–14]. By contrast, γ-aminobutyric acid (GABA) is a major inhibitory neurotransmitter in the CNS [15]. GABA is synthesized primarily from glutamate by glutamate decarboxylase in the soma and terminal of neurons [16]. In both in humans and animals models, GABA has also been shown to play a critical roles in the pathogenesis and treatment of epilepsy [5,17,18] and GABA concentrations in the CSF are reduced in human epileptic patients and animal seizure models [3,19]. In addition to glutamate and GABA, other amino acids such as aspartate, glycine, serine, and taurine also work as neurotransmitters and may modify the pathophysiology of epilepsy [20,21].
Cats have been widely utilized as experimental animals in the neurosciences, including epileptic seizure models. Neurotransmitters in the CSF and brain extracellular fluids were originally determined in feline seizure models [22,23]. However, the changes in neurotransmitters in cats with spontaneous (naturally occurring) epilepsy are poorly understood. We previously reported the model of FSECs, which show an autosomal recessive mode of inheritance [24]. FSEC represents a typical form of feline epilepsy [25] and is considered a natural model of familial mesial temporal lobe epilepsy [26,27].
Metabolomics is a tool that provides exhaustive analysis of metabolites in vivo. Metabolomic approaches have been widely used for identification of disease-related biomarkers and understanding the molecular mechanisms of disease [28–30]. Thus, we also employed a metabolomic approach to confirm our chromatographic analysis of CSF amino acids, and to assess changes in endogenous low molecular weight metabolites, in FSECs.
The purpose of this study reported here was to evaluate CSF levels of excitatory/inhibitory amino acids to further our understanding of the pathophysiology of FSEC. We hypothesized that the abnormal imbalance of excitatory and inhibitory amino acid concentrations in the CSF are the cause of epilepsy in FSEC. That is 1) higher excitatory amino acid and lower inhibitory amino acid during interictal phase, and then change into 2) lower excitatory amino acid and higher inhibitory amino acid during postictal phase.
Materials and Methods
Animals
This study was approved by the Animal Care and Use Committee of Nippon Veterinary and Life Science University (accession nos. 09-1, 10-3, 11-51, 12-42; representative researcher is D.H.). FSECs were maintained in the Nippon Veterinary and Life Science University and a commercial animal breeding facility (Chiba, Japan) [24]. These housings were conventional animal facilities. Cats were kept individually in the stainless clean cages (length × width × depth=around 1 × 1 × 1 m) with a water cup, bedding box and pet sheets. Light/dark cycle was loosely maintained 12L/12D, and room temperature was kept 23 ± 3°C by the air conditioner. Abundant tap water and necessary and sufficient amount of pouched cat food were served according to the manufacturer’s recommendation (Royal Canin, Aimargues, France). Welfare-related assessments were carried out before, during and after the experiment. From the family, thirteen epileptic cats (19–84 months old, 2.5–4.2 kg), which showed recurrent seizures and were predicted homozygote (Affected group) by pedigree analysis, and nine non-epileptic cats (69–114 months old, 2.6–4.5 kg), which were parents of the Affected cats and were predicted heterozygote (Unaffected group), were used in this study. In addition, six clinically healthy cats (9–15 months old, 2.2–3.8 kg), which were maintained in the same conditions and were unrelated family line to FSECs, were used as the Healthy group. All experiments were open studies and experimenters were not blinded while analyzing the samples.
Collection of CSF samples
CSF samples were collected by cisternal puncture with 23 G needles under sedation by 0.07–0.08 mg/kg body weight intramuscular injection of medetomidinea. In the Affected group, CSF samples were collected during the interictal phase, defined as a seizure-free time period of at least two weeks, and during the postictal phase, defined as the time period within 30 min after vestibular stimulation-induced generalized seizures. In all groups, vestibular stimulations were induced by manually swinging the carry cage up, down, right, and left for 2 min.24 In the Unaffected group and Healthy group, CSF samples were collected during the normal state and within 30 min after vestibular stimulation. CSF samples were stored −80°C until analysis.
High-performance liquid chromatography (HPLC) analysis of CSF amino acids
Excitatory/inhibitory amino acids (aspartate, glutamate, serine, glycine taurine, and GABA) levels in the CSF were analyzed by HPLC with o-phthalaldehyde precolumn derivatization. The HPLC system consisted of two pumpsb, an autosamplerc, a column ovend, a fluorescence detectore and an integratorf. Two Octa Decyl Silyl (ODS) columns (Pegasil ODS; 4.6 mm × 30 mmg and 4.6 mm × 250 mmh) were used in-line at 40°C. CSF samples were thawed immediately before use. Each of the CSF samples (40 μl) were deproteinized by mixing with 120 μl of 0.6 M HClO4 and 40 μl of 2 M K2CO3. The mixture was centrifuged at 10,000 × g for 5 min. The supernatant was used for amino acids analysis. For pre-column derivatization, 60 μl of sample was mixed with 15 μl of OPA solution. The OPA solution is prepared by adding 1 ml of o-phthalaldehyde (27 mg/1 ml methanol) and 25 μl of 2-mercaptoethanol in 4 ml of 0.1 M borate buffer (7.63g Na2B4O7•10H2O/200 ml ultra pure water). After 30 s reaction, 10 μl of sample solution was injected into a HPLC column. The resolution of the amino acids was accomplished by use of gradient elution. The mobile phase consisted of solvent A (50 mM CH3COONa, pH 5.80, 3.0% N,N-dimethylformamide) and solvent B (methanol), and was eluted at a flow rate of 1.0 ml/min. A gradient protocol was performed as follows: a linear gradient from 85% to 15% of solvent A for 55 min. Fluorescence of the eluent was monitored for 55 min with excitation wavelength set at 340 nm and emission wavelength set at 455 nm. To equilibrate the column between each run, the 85% solvent A was isocratically eluted for 9 min. Values of amino acids were calculated by analyzing amino acid standards with known levels.
Metabolomics of CSF
The CSF samples from five Affected cats in the interictal phase, and CSF from four Unaffected cats and three Healthy cats, were used for metabolomics. Metabolome measurements were performed through a facility service at Human Metabolome Technologies Inc. (Yamagata, Japan). CSF 100 μl was mixed with 900 μl of methanol including 10 μM of internal standard. Next, 1,000 μl of chloroform and 400 μl of ultra-pure water were added to the mixture, and were mixed thoroughly and centrifuged (2,300 × g, 4°C,5 min). The 400 μl of the supernatant was centrifugally filtered through a cutoff filteri to remove proteins. The filtrate was lyophilized, resuspended in 50 μl ultra-pure water, and analyzed by capillary electrophoresis timeof- flight mass spectrometry (CE-TOFMS)j. Peaks were extracted using automatic integration softwarek to obtain peak information including m/z, migration time for CE-TOFMS measurement (MT), and peak area. Signal peaks corresponding to isotopomers, adduct ions, and other product ions of known metabolites were excluded, and remaining peaks were annotated with putative metabolites from the Human Metabolome Technologies metabolite database based on their MTs and m/z values determined by TOFMS. The tolerance range for peak annotation was configured at ± 0.5 min for MT and ± 10 ppm for m/z. In addition, peak areas were normalized against those of the internal standards, and the resultant relative area values were then further normalized by sample amount.
Statistical analysis
Differences of amino acid levels in the CSF among groups were compared using Kruskal-Wallis test followed by post hoc test. Paired t-test was used to compare the CSF levels of amino acids between the interictal (normal) state and the postictal (after vestibular stimulation) in each group. Results of metabolomics were tested by Kruskal-Wallis test, followed by post hoc test. Statistical analysis was carried out using software programl. P< 0.05 was considered statistically significant.
Results
HPLC analysis of CSF amino acids
Figure 1 shows CSF concentrations of aspartate, glutamate, serine, glycine, taurine, and GABA during the interictal (Affected) or normal (Unaffected and Healthy) state. Glutamate level in the Unaffected group was significantly lower than in the Healthy group, while there was no significant differences in the CSF levels of aspartate, serine, glycine, or taurine. Several cats in the affected group also showed trends for elevated GABA concentrations.
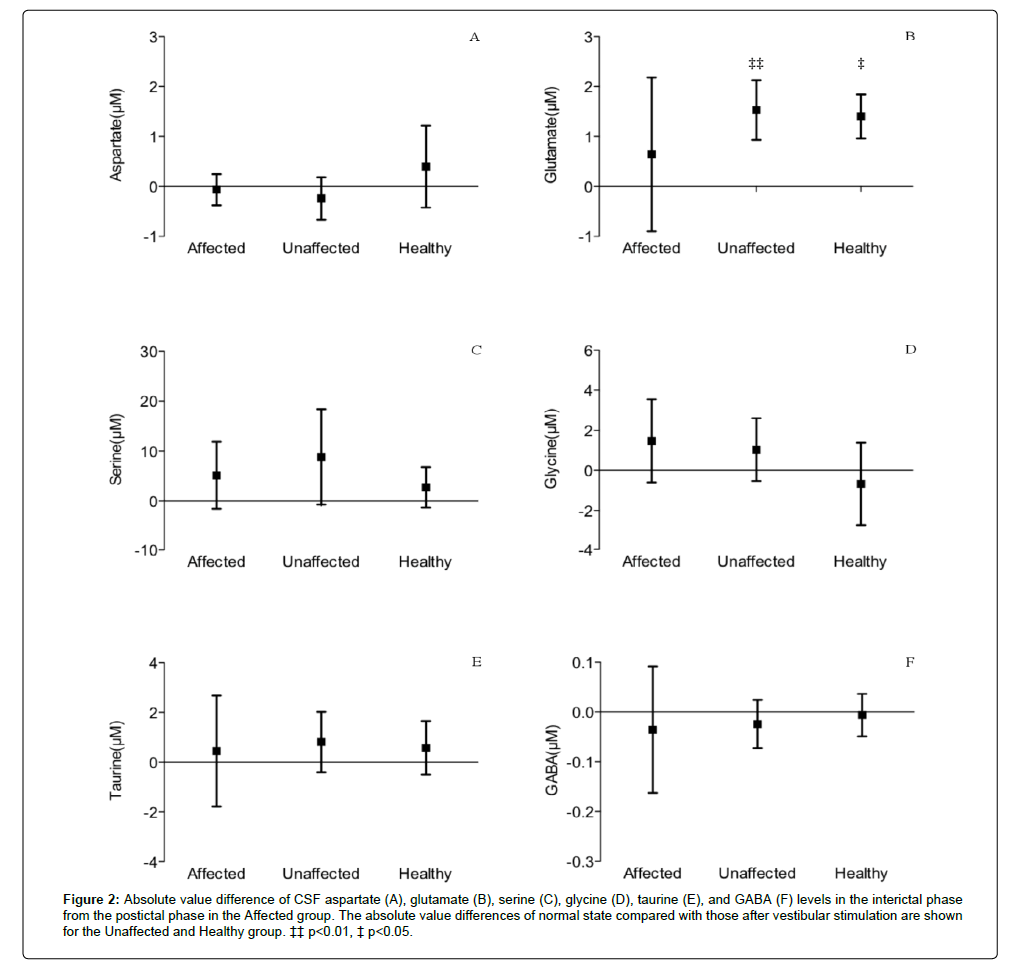
Figure 2 shows the changes in CSF amino acid levels following vestibular stimulation. Data are shown as absolute value difference between interictal (normal) and postictal (after vestibular stimulation). As vestibular stimulation induced epileptic seizures in five of 13 cats in the affected group, CSF samples of the five cats were collected immediately after the seizure, while the remaining eight cats were not included. CSF samples of eight of nine Unaffected cats and all of the six Healthy cats were collected after vestibular stimulation.
Figure 2:Absolute value difference of CSF aspartate (A), glutamate (B), serine (C), glycine (D), taurine (E), and GABA (F) levels in the interictal phase from the postictal phase in the Affected group. The absolute value differences of normal state compared with those after vestibular stimulation are shown for the Unaffected and Healthy group. ‡‡ p<0.01, ‡ p<0.05.
The CSF glutamate concentrations were significantly elevated after vestibular stimulation in the Unaffected and Healthy groups. By contrast, there was no significant change in glutamate concentrations in the Affected group. Further, there were no changes in other CSF amino acid following vestibular stimulation.
Metabolomics of CSF
Metabolomic analysis resulted in detection of 95 metabolites, which consisted of 21 amino acids and 74 low molecular weight compounds. The relative amounts of the 21 amino acids (Affected/ Healthy and Unaffected/Healthy) are shown in Table 1. CSF glutamine and 2-aminobutyric acid concentrations were significantly lower in the Affected group compared with the Healthy group. Among the 74 low molecular weight compounds, five compounds were significantly different between groups.
Table 1: Levels of metabolites and amino acids in the various groups.
| Ratio (Mean ± SD) | ||
|---|---|---|
| Affected/Healthy | Unaffected/Healthy | |
| Amino acids | ||
| Aspartate | 0.82 ± 0.11 | 0.95 ± 0.25 |
| Glutamate | 0.80 ± 0.12 | 0.85 ± 0.17 |
| Serine | 0.74 ± 0.13 | 0.90 ± 0.14 |
| Glycine | 0.90 ± 0.06 | 1.03 ± 0.13 |
| Methionine | 0.79 ± 0.14 | 1.12 ± 0.88 |
| Glutamine | 0.80 ± 0.05* | 0.86 ± 0.07 |
| Lysine | 2.10 ± 0.44 | 1.89 ± 0.67 |
| Arginine | 1.60 ± 0.40 | 1.68 ± 0.32 |
| Tryptophan | 1.17 ± 0.09 | 1.18 ± 0.19 |
| Phenylalanine | 1.02 ± 0.08 | 1.00 ± 0.16 |
| Leucine | 0.94 ± 0.10 | 1.15 ± 0.20 |
| Isoleucine | 0.91 ± 0.11 | 1.15 ± 0.18 |
| Histidine | 0.90 ± 0.14 | 1.00 ± 0.38 |
| Valine | 0.82 ± 0.10 | 0,83 ± 0.11 |
| Threonine | 0.74 ± 0.18 | 0,74 ± 0.12 |
| Asparagine | 0.88 ± 0.10 | 0.86 ± 0.14 |
| Tyrosine | 0.83 ± 0.12 | 0.78 ± 0.15 |
| Proline | 0.77 ± 0.11 | 0.77 ± 0.15 |
| Alanine | 0.70 ± 0.07 | 0.69 ± 0.19 |
| Ornithine | 1.36 ± 0.26 | 1.54 ± 0.27 |
| 2-Aminobutyric acid | 0.59 ± 0.11* | 0.93 ± 0.31 |
| Other metabolites | ||
| Pyridoxal | 1.35 ± 0.18† | 0.86 ± 0.12 |
| 2-Hydroxybutyric acid | 0.71 ± 0.11*† | 0.94 ± 0.12 |
| 3-Hydroxybutyric acid | 0.64 ± 0.12* | 0.76 ± 0.14 |
| Cyridine | 0.76 ± 0.08 | 0.66 ± 0.10* |
| Saccharopine | 0.61 ± 0.07* | 0.68 ± 0.06 |
* p<0.05, ** p<0.01, *** p<0.001, significantly different from Healthy group cats. † p<0.05, †† p<0.01, significantly different from Unaffected group cats.
Discussion
In the present study, we evaluated levels of excitatory/inhibitory amino acid in the CSF of FSECs. During the interictal state, the Affected group showed similar levels of CSF glutamate to those in the Healthy group. Moreover, during the normal (without vestibular stimulation) state, the Unaffected group showed significantly lower glutamate levels than the Healthy group. Several studies on spontaneous epilepsy in humans and animals reported increased levels of CSF glutamate during the interictal state and hypothesized that increased glutamate triggers epileptic seizures [6,18,19]. However, this hypothesis does not explain the CSF glutamate levels in our familial epileptic cats. In human patients with temporal lobe epilepsy, extracellular glutamate levels in the epileptogenic hippocampus are elevated before and during seizures, and then quickly return to basal levels after seizures resolution, Chapman et al.[13] suggesting that elevation of extracellular glutamate levels contributes to generation and evolution of seizures.
To evaluate the dynamic changes in CSF amino acids, we used vestibular stimulation to trigger epileptic seizures in affected cats.
In Unaffected and Healthy cats, CSF glutamate concentrations were significantly elevated by vestibular stimulation, suggesting that increased glutamate release and/or decreased glutamate uptake in the brain occurred after vestibular stimulation. By contrast, CSF glutamate in Affected cats remained at basal levels after epileptic seizures induced by vestibular stimulation. It is possible that glutamate levels in the CSF of Affected cats were elevated, but then quickly returned to the basal levels before CSF sampling. In rat kindling models, expression of excitatory amino acid transporters on glial cells (EAAT1/GLAST and EAAT2/GLT-1) was decreased by seizures 31. On the other hand, expression of the neuronal glutamate transporter EAAT3/EAAC1 was increased by seizures [31-33]. A similar mechanism may stabilize CSF glutamate levels after seizures in the Affected group in the present study.
We also found that CSF glutamine levels were significantly lower in the affected group than in the Healthy group. Further, in the unaffected group, glutamine levels tended to be lower than in the Healthy group. Glutamine is a major precursor of glutamate [34]. Glutamate released from presynaptic terminals is taken up by astrocytes and converted to glutamine by glutamine synthetase. The newly synthesized glutamine is transferred to neurons and hydrolyzed into glutamate by mitochondrial glutaminase. This cycle is called the ‘glutamate-glutamine cycle [35]. The low CSF glutamine levels in the Affected and Unaffected groups suggests that decreased substrates of the glutamate-glutamine cycle, which may result in the low CSF glutamate levels observed in the Unaffected group. In addition, increased glutamate uptake into neurons may lead to reduced glutamate uptake and glutamine synthesis by glial cells.
In the present study, elevated GABA levels were observed in several cats in the affected group. In the CNS, GABA is mainly synthesized from glutamate by glutamate decarboxylase in GABAergic neurons. GABA is an inhibitory neurotransmitter that reduces neural hyperexcitability and seizures [36–38]. In the Affected group, the elevated GABA suggests a compensatory reaction to epileptic activity in individual cats.
Our metabolomic analysis detected 21 CSF amino acids, with similar changes to that in the HPLC analysis. Additionally, there were significant differences in five low molecular weight compounds (pyridoxal, 2-hydrobutylic acid, 3-hydrobutylic acid, cyridine, and saccharopine) between the groups. However, our metabolic pathway analysis could not identfy the definite relationship to CNS metabolism. Further metabolomics studies are required to examine these compoundss in more detail.
The results obtained in this study were contrary to our initial hypothesis. Interestingly, the levels of excitatory amino acids in the affected group did not change during interictal or postictal phase, and the level of GABA, the main inhibitory amino acid, tended to be higher during interictal phase. In addition, in the unaffected group, the level of glutamate was lower during the interictal phase. These results may lead us to elucidation seizure mechanisms in FSECs.
Conclusion
In conclusion, we found characteristic changes in CSF glutamate, glutamine, and GABA levels in FSECs. These findings further our understanding of the pathogenesis of FSEC, as well as familial mesial temporal lobe epilepsy.
References
- Wilson CL, Maidment NT, Shomer MH, Behnke EJ, Ackerson L, et al. (1996) Comparison of seizure related amino acid release in human epileptic hippocampus versus a chronic, kainate rat model of hippocampal epilepsy. Epilepsy Res 26: 245–254.
- Rainesalo S, Keränen T, Palmio J, Peltola J, Oja SS, et al. (2004) Plasma and cerebrospinal fluid amino acids in epileptic patients. Neurochem Res 29: 319–324.
- Szyndler J, Maciejak P, TurzyÅ„ska D, Sobolewska A, Lehner M, et al. (2008) Changes in the concentration of amino acids in the hippocampus of pentylenetetrazole-kindled rats. Neurosci Lett 439: 245–249.
- Van Gelder NM, Edmonds HL, Hegreberg GA, Chatburn CC, Clemmonset RM, et al. (1980) Amino acid changes in a genetic strain of epileptic beagle dogs. J Neurochem 35: 1087–1091.
- Löscher W, Schwartz-Porsche D (1986) Low levels of gamma-aminobutyric acid in cerebrospinal fluid of dogs with epilepsy. J Neurochem 46: 1322–1325.
- Morita T, Shimada A, Takeuchi T, Hikasa Y, Sawadaet M, et al. (2002) Cliniconeuropathologic findings of familial frontal lobe epilepsy in Shetland sheepdogs. Can J Vet Res 66: 35–41.
- Ellenberger C, Mevissen M, Doherr M, Scholtysik G, Jaggy A (2004) Inhibitory and excitatory neurotransmitters in the cerebrospinal fluid of epileptic dogs. Am J Vet Res 65: 1108–1113.
- Creevy KE, Gagnepain JF, Platt SR, Edwards GL, Kent M (2013) Comparison of concentrations of γ-aminobutyric acid and glutamate in cerebrospinal fluid of dogs with idiopathic epilepsy with and without seizure-related magnetic resonance imaging hyperintense areas in the limbic system. Am J Vet Res 74: 1118–1125.
- Tzschentke TM (2002) Glutamatergic mechanisms in different disease states: overview and therapeutical implications -An introduction. Amino Acids 23: 147–152.
- Platt SR (2007) The role of glutamate in central nervous system health and disease-A review. Vet J 173: 278–286.
- Meldrum BS (1994) The role of glutamate in epilepsy and other CNS disorders. Neurology 44: S14–23.
- Treiman DM (2001) GABAergic Mechanisms in Epilepsy. Epilepsia 42: 8–12.
- Chapman AG, Elwes RD, Millan MH (1996) Role of glutamate and aspartate in epileptogenesis; contribution of microdialysis studies in animal and man. Epilepsy Res Suppl 12: 239–246.
- During MJ, Spencer DD (1993) Extracellular hippocampal glutamate and spontaneous seizure in the conscious human brain. Lancet 341: 1607–1610.
- Bradford HF (1995) Glutamate, GABA and epilepsy. Prog Neurobiol 47: 477–511.
- Mathews GC, Diamond JS (2003) Neuronal glutamate uptake Contributes to GABA synthesis and inhibitory synaptic strength. J Neurosci 23: 2040–2048.
- Petroff OAC, Rothman DL, Behar KL, Lamoureux D, Mattson RH (1996) The effect of gabapentin on brain gamma-aminobutyric acid in patients with epilepsy. Ann Neurol 39: 95–99.
- Podell M, Hadjiconstantinou M (1997) Cerebrospinal fluid gamma-aminobutyric acid and glutamate values in dogs with epilepsy. Am J Vet Res 58: 451–456.
- Málly J, Baranyi M, Vizi ES (1996) Change in the concentrations of amino acids in CSF and serum of patients with essential tremor. J Neural Transm 103: 555–560.
- Meldrum BS, Akbar MT, Chapman AG (1999) Glutamate receptors and transporters in genetic and acquired models of epilepsy. Epilepsy Res 36: 189–204.
- Sherwin AL (1999) Neuroactive amino acids in focally epileptic human brain: a review. Neurochem Res 24: 1387–1395.
- Griffith NC, Cunningham AM, Goldsmith R, Bandler R (1991) Interictal behavioral alterations and cerebrospinal fluid amino acid changes in a chronic seizure model of temporal lobe epilepsy. Epilepsia 32: 767–777.
- Qu Y, Arckens L, Vandesande F (2000) Sampling extracellular aspartate, glutamate and gamma-aminobutyric acid in striate cortex of awake cat by in vivo microdialysis: surgical and methodological aspects. Brain Res 866: 116–127.
- Kuwabara T, Hasegawa D, Ogawa F, Kobayashi M, Fujita M (2010) A familial spontaneous epileptic feline strain: a novel model of idiopathic/genetic epilepsy. Epilepsy Res 92: 85–88.
- Pakozdy A, Halasz P, Klang A (2014) Epilepsy in cats: theory and practice. J Vet Intern Med 28: 255–263.
- Hasegawa D, Mizoguchi S, Kuwabara T (2014) Electroencephalographic features of familial spontaneous epileptic cats. Epilepsy Res 108: 1018–1025.
- Mizoguchi S, Hasegawa D, Kuwabara T, Hamamoto Y1, Ogawa F, et al. (2014) Magnetic resonance volumetry of the hippocampus in familial spontaneous epileptic cats. Epilepsy Res 108: 1940–1944.
- Sugiura Y, Taguchi R, Setou M (2011) Visualization of spatiotemporal energy dynamics of hippocampal neurons by mass spectrometry during a kainate-induced seizure. PLoS One 6:e17952.
- Roux A, Lison D, Junot C, Heilier JF (2011) Applications of liquid chromatography coupled to mass spectrometry-based metabolomics in clinical chemistry and toxicology: A review Clin Biochem 44: 119–135.
- Wei C, Li Y, Yao H, Liu H, Zhang X, et al. (2012) A metabolomics study of epilepsy in patients using gas chromatography coupled with mass spectrometry. Mol Biosyst 8: 2197–2204.
- Miller HP, Levey AI, Rothstein JD, Tzingounis, AV, Conn PJ (1997) Alterations in glutamate transporter protein levels in kindling-induced epilepsy. J Neurochem 68: 1564–1570.
- Ross JR, Porter BE, Buckley PT, Eberwine JH, Robinson MB (2011) mRNA for the EAAC1 subtype of glutamate transporter is present in neuronal dendrites in vitro and dramatically increases in vivo after a seizure. Neurochem Int 58: 366–375.
- Medina-Ceja L, Sandoval-García F, Morales-Villagrán A, López-Pérez SJ (2012) Rapid compensatory changes in the expression of EAAT-3 and GAT-1 transporters during seizures in cells of the CA1 and dentate gyrus. J Biomed Sci 19: 78.
- Armano S, Coco S, Bacci A, Pravettoni E, Schenk U, et al. (2002) Localization and functional relevance of system a neutral amino acid transporters in cultured hippocampal neurons. J Biol Chem 277: 10467–10473.
- Bak LK, Schousboe A, Waagepetersen HS (2006) The glutamate/GABA-glutamine cycle: aspects of transport, neurotransmitter homeostasis and ammonia transfer. J Neurochem 98: 641–653.
- Richerson GB, Wu Y (2003) Dynamic equilibrium of neurotransmitter transporters: not just for reuptake anymore. J Neurophysiol 90: 1363–1374.
- Wu Y, Wang W, Díez-Sampedro A, Richerson GB (2007) Nonvesicular inhibitory neurotransmission via reversal of the GABA transporter GAT-1. Neuron 56: 851–865.
- Héja L, Nyitrai G, Kékesi O, Dobolyi A, Szabó P, et al. (2012) Astrocytes convert network excitation to tonic inhibition of neurons. BMC Biol 10: 26.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi