Short Communication, J Regen Med Vol: 6 Issue: 1
Expression of CD146 on Human Placenta-Derived Mesenchymal Stromal Cells and their Osteogenic Differentiation Capacity are Modulated by Factors Contained in Platelet Lysate
Verpoorten A1, Abruzzese T1, Pils A1, Abele H2, Hart ML1,3 and Aicher WK1*
1Department of Urology, University of Tübingen Hospital, Tübingen, Germany
2Department of Gynaecology and Obstetrics, University of Tübingen Hospital, Tübingen, Germany
3Department of Orthopedics and Trauma Surgery, University of Freiburg, Freiburg, Germany
*Corresponding author : Wilhelm K. Aicher
Department of Urology, University of Tübingen Hospital, Tübingen, Germany
Tel: +49 7071 298 7020
E-mail: aicher@uni-tuebingen.de
Received: February 15, 2017 Accepted: April 06, 2017 Published: April 13, 2017
Citation: Verpoorten A, Abruzzese T, Pils A, Abele H, Hart ML, et al. (2017) Expression of CD146 on Human Placenta-Derived Mesenchymal Stromal Cells and their Osteogenic Differentiation Capacity are Modulated by Factors Contained in Platelet Lysate. J Regen Med 6:1. doi: 10.4172/2325-9620.1000133
Abstract
Expression of CD146 on bone marrow-derived mesenchymal stromal cells (bmMSCs) is considered a marker for their multipotent differentiation potential and in situ localisation in the stem cell niche. We recently observed an elevated expression of CD146 on human bmMSCs upon expansion in medium enriched with platelet lysate. We therefore investigated if CD146 expression followed comparable paths on human placenta-derived mesenchymal stromal cells (pMSCs). In contrast to bmMSCs, pMSCs expanded in medium with platelet lysate expressed significantly less CD146 and failed to undergo osteogenic differentiation. We conclude that the expression of the stem cell marker CD146 on pMSCs and their osteogenic differentiation potential are modulated by factors contained in platelet lysate.
Keywords: Mesenchymal stromal cells; CD146; Osteogenenic differentiation; Expansion media; GMP-compliant media
Introduction
Placenta is a rich source for mesenchymal stromal cells (MSCs) [1,2]. Placenta-derived MSCs (pMSCs) share many features with MSCs from bone marrow (bmMSCs) or other sources [3-5]. However pMSCs have a significantly different transcriptome than bmMSCs, highlighted by a significant decrease in expression of osteogenic factors [6-8]. Osteogenic differences correlate with expression of the adhesion molecule CD146 [9-13]. Hence, separating CD146+ from CD146- pMSCs produces pro-ostegenic pMSCs vs. cells having a significantly reduced ability to undergo osteogenesis, respectively [9]. This is contrast to CD146- bmMSCs which demonstrate a proosteogenic potential by generation of bone in vivo [13].
Expression of CD146 on other types of MSCs or MSC-like cells has also resulted in conflicting results with respect to its expression and function. In contrast to adipose tissue-derived (adMSCs) and umbilical cord-derived MSCs, CD146low dental pulp-derived MSC-like cells grew significantly faster compared to the CD146high population [14-16]. Moreover, loss of CD146 on MSCs has been associated with senescence [14,17]. In short term cultures expression of CD146 on pMSCs did not change considerably, but CD146 was reduced after prolonged expansion of umbilical cord-derives MSCs [9,14,18].
Part of the reason for these contradictory results may be due to the MSC source (e.g., bone marrow vs. adipose tissue), but a key elemental difference can be highlighted by a difference in the methods for isolation and/or expansion of these cells. Adipose tissue-derived MSCs (atMSCs) express significantly less CD146 ex vivo compared to cells after expansion in media enriched with 10% FBS, suggesting that expression of CD146 can be modulated in vitro by the serum source [19].
In many studies MSCs were expanded in media containing foetal bovine serum (FBS) [1,5]. In some studies, especially in (pre-) clinical investigations, FBS was replaced by human serum (HS) and sometimes enriched with platelet lysate (PL) [20-22]. Hence different laboratories used quite diverse culture conditions when investigating the expression and function of CD146 on MSCs [9,13,23,24]. The importance of phenotypic changes in MSCs during in vitro GMP expansion and their clinical implications have been recently reviewed [18]. But the effect of a GMP-compliant medium on expression of CD146 by pMSCs was not investigated. Therefore in the present study we investigated if some of the differences in the expression of CD146 on pMSCs and their osteogenic differentiation potential properties could be attributed to PL utilized to enrich MSC growth media [20,25].
Materials and Methods
Human pMSCs were isolated from full-term placenta from elective caesareans that were free of any medical/obstetrical complications (n=5, ethics #453/2011B02) [7-9]. Cells from the same donor were expanded either in a GMP-compliant medium enriched with 5% human serum plus 5% human platelet lysate (HS+PL medium) [22], or in commercially available MSC medium containing FBS (Lonza, MSCGM). Second passage cells were harvested to confirm their MSC phenotype [1,26]. The expression of CD146 (R&D Systems) was determined by flow cytometry as described recently [7]. Expression of CD73 (BD Pharmingen) and CD90 (R&D Systems) served as controls. Statistics were computed by a two-sided t-test. Osteogenesis was induced by incubation of the pMSCs in 6-well plates in differentiation media over 28 days (DMEM-HG, 10% FBS, enriched by 1 μmol/l dexamethasone, 10 mmol/l β-glycerophosphate, 170 μmol/l ascorbate). Osteogenic differentiation was visualized by von Kossa staining [5]. Cells in media without osteogenic stimuli served as controls (DMEM-HG, 10% FBS).
Results
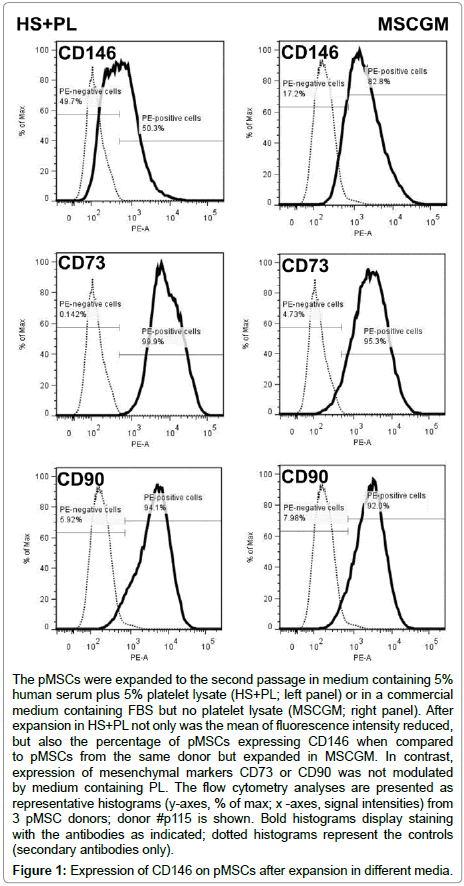
Modulation of expression of CD146 by MSC expansion media
All pMSCs were characterized as described in our recent studies [7,9,27] (not shown). As the next step we investigated if CD146 was regulated by PL. When expanded in HS+PL medium, 58.8% of pMSCs expressed moderate levels of CD146 with a mean of fluorescence intensities (MIF) of 779 Figure 1. When expanded in MSCGM complemented by FBS, significantly more pMSCs expressed CD146 (average 75.1%; p<0.028) with significantly higher signal intensities (MIF 3622; p<0.004). In contrast, expression of CD73 and CD90 was not modulated by PL Figure 1. This confirmed that human pMSCs display a variable expression of CD146 [7]. We conclude that expression of CD146 is significantly reduced by complementing the expansion medium with PL (Figure 1).
Osteogenic differentiation of pMSCs
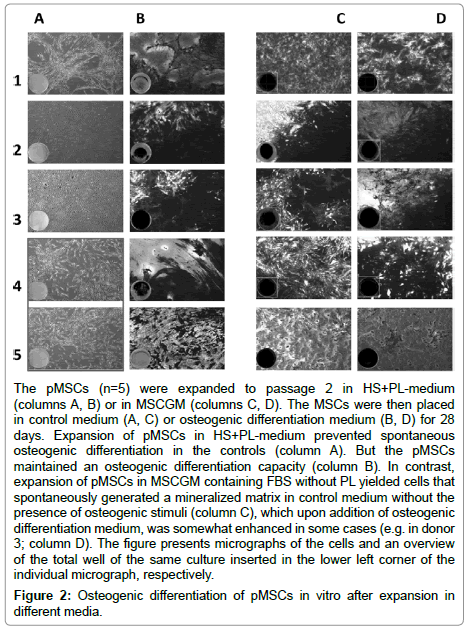
Next we investigated if PL influenced the osteogenic differentiation capacity of pMSCs (Figure 2). Human pMSCs incubated in HS+PL medium did not undergo spontaneous osteogenic differentiation in vitro, but a weak (2/5 populations), a moderate (2/5 populations), or a robust mineralization (1/5 population) was observed after 28 days of differentiation. In contrast, the same pMSCs spontaneously produced a mineralized extracellular matrix after incubation in MSCGM (5/5 populations), and osteogenesis was somewhat enhanced by addition of the differentiation cocktail to the pMSCs in all samples investigates (Figure 2). We conclude that osteogenic differentiation capacity of human pMSCs can be modulated by addition of PL to the expansion media.
Discussion
CD146 expression has been attributed to osteogenic differentiation and bone formation and is considered a marker for MSC “stemness” and multipotent differentiation potential capacity [10,14,17,24]. In contrast to these studies, others reported no clearcut difference in the osteogenic potential of CD146high versus CD146low bmMSCs [10,12,13,24,28]. We showed that CD146 was associated with osteogenesis of pMSCs [7-9]. Taken together this suggests that the role of CD146 in osteogenic differentiation of human MSCs seems a matter of debate [13,24,28]. Reasons for these seemingly conflicting findings may include differences in the MSC sources and in the methods for isolation and/or expansion of the cells.
In this short communication we show that expression of CD146 and osteogenesis are reduced in pMSCs after expansion in HS+PL medium compared to the same cells expanded in a commercial standard MSC medium enriched by FBS without PL. This data shows that the expression of CD146 on pMSCs and its effect on osteogenic differentiation is therefore modulated by factors contained in PL.
This is in sharp contrast to another of our recent publications investigating human bmMSCs upon expansion in GMP-compliant media [22]. In contrast to pMSCs, bmMSCs showed an enhanced the expression of CD146 upon expansion in media containing PL. In another study we showed that factors contained in PL did not compromise the osteogenic differentiation of bmMSCs nor the expression of CD146, but it facilitated their smooth muscle differentiation [22,29]. To the best of our knowledge, this is the first study to demonstrate how platelet lysate regulates osteogenesis in placenta-derived MSCs.
In contrast to bmMSCs [22,29], pMSCs may respond in a different way to factors contained in PL. Key components of PL include PDGF, bFGF and TGF-β1 [25] and MSCs express the corresponding receptors [30]. bFGF accelerated proliferation and reduced expression of CD146 in bmMSCs, whereas TGF-β1 retarded proliferation and enhanced CD146 [12]. Overexpression of PDGF-BB and bFGF enhanced proliferation of bmMSCs and facilitated osteogenesis, whereas TGF-β1 blocked osteogenesis but had no effect on proliferation [30]. Based on these studies we hypothesize that, the amount of TGF-β1 in HS+PL was possibly not sufficient to activate expression of CD146 on pMSCs, which may have been reduced by bFGF, but that TGF-β1 may be adequate to block osteogenesis of pMSCs. Alternatively, processing of TGF-β1 signals may follow different routes in pMSCs than in bmMSCs. Expression of CD146 on bmMSCs is reduced in microsphere 3D pellet cultures, but not in monolayer cultures, indicating that a 3D environment may also regulate this adhesion molecule [31]. Differences in expression of CD146 on MSCs from distinct sources and variations reported with respect to the differentiation potential of MSCs could therefore be in part explained by deviations in the blend of PDGF, bFGF and TGF-β1 in the media employed in the different studies.
These results may be important for studies that use PL for regenerative medicine with potential for clinical applications. Because PL is more commonly being used as an alternative growth supplement as opposed to xenogenic (e.g. FBS) supplements in MSC expansion medium, this finding bears considerable importance for physicians and their partners in research and health industry using PL or platelet extract to produce MSCs for clinical studies or cellular therapies. It has been noted, for instance, that MSCs expanded in media complemented with FBS displayed in about 30% of the tissue samples a spontaneous osteogenesis and mineralization in vivo when injected in a pre-clinical animal study in healthy or infarcted hearts [32]. In other studies MSCs generated bone and bone marrow upon subcutaneous transplantation in vivo [24]. Therefore, complementing the MSC expansion media with PL could ameliorate in a clinical situation the risk of in situ mineralization.
We conclude that in contrast to bmMSCs, pMSCs respond in a different way to factors contained in platelet lysate: In pMSCs the osteogenic differentiation capacity and expression of CD146 are reduced by expansion of the cells in GMP-compliant media enriched by PL [22].
Acknowledgments
The authors thank the midwives and nurses at the UKT women’s hospital for providing placenta samples and Chaim Goziga for help with preparing the artwork. This project was supported in part by grants from the DFG (KFO273) and the BMBF (Multimorb-INKO).
References
- In 't Anker PS, Scherjon SA, Kleijburg-van der Keur C (2004) Isolation of mesenchymal stem cells of fetal or maternal origin from human placenta. Stem Cells 22: 1338-1345.
- Parolini O, Alviano F, Bagnara GP, Bilic G, Bühring HJ, et al. (2008) Concise review: isolation and characterization of cells from human term placenta: outcome of the first international Workshop on Placenta Derived Stem Cells. Stem Cells 26: 300-311.
- Miao Z, Jin J, Chen L, Zhu J, Huang W, et al. (2006) Isolation of mesenchymal stem cells from human placenta: Comparison with human bone marrow mesenchymal stem cells. Cell Biol Int 30: 681-687.
- Hass R, Kasper C, Böhm S, Jacobs R (2011) Different populations and sources of human mesenchymal stem cells (MSC): A comparison of adult and neonatal tissue-derived MSC. Cell Commun Signal 9: 12.
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, et al. (1999) Multilineage potential of adult human mesenchymal stem cells. Science 284: 143-147.
- Maerz JK, Roncoroni LP, Goldeck D, Abruzzese T, Kalbacher H, et al. (2016) Bone marrow-derived mesenchymal stromal cells differ in their attachment to fibronectin-derived peptides from term placenta-derived mesenchymal stromal cells. Stem Cell Res Ther 7: 29.
- Pilz GA, Ulrich C, Ruh M, Abele H, Schäfer R, et al. (2011) Human term placenta-derived mesenchymal stromal cells are less prone to osteogenic differentiation than bone marrow-derived mesenchymal stromal cells. Stem Cells Dev 20: 635-646.
- Ulrich C, Rolauffs B, Abele H, Bonin M, Nieselt K, et al. (2013) Low osteogenic differentiation potential of placenta-derived mesenchymal stromal cells correlates with low expression of the transcription factors Runx2 and Twist2. Stem Cells Dev 22: 2859-2872.
- Ulrich C, Abruzzese T, Maerz JK, Ruh M, Amend B, et al. (2015) Human Placenta-Derived CD146-Positive Mesenchymal Stromal Cells Display a Distinct Osteogenic Differentiation Potential. Stem Cells Dev 24: 1558-1569.
- Sacchetti B, Funari A, Michienzi S (2007) Self-renewing osteoprogenitors in bone marrow sinusoids can organize a hematopoietic microenvironment. Cell 131: 324-336.
- Sacchetti B, Funari A, Remoli C, Giannicola G, Kogler G, et al. (2016) No identical "mesenchymal stem cells" at different times and sites: Human committed progenitors of distinct origin and differentiation potential are incorporated as adventitial cells in microvessels. Stem Cell Reports 6: 897-913.
- Espagnolle N, Guilloton F, Deschaseaux F, Gadelorge M, Sensébé L, et al. (2014) CD146 expression on mesenchymal stem cells is associated with their vascular smooth muscle commitment. J Cell Mol Med 18: 104 - 114.
- Harkness L, Zaher W, Ditzel N, Isa A, Kassem M (2016) CD146/MCAM defines functionality of human bone marrow stromal stem cell populations. Stem Cell Res Ther 7: 4.
- Jin HJ, Kwon JH, Kim M, Bae YK, Choi SJ, et al. (2016) Downregulation of Melanoma Cell Adhesion Molecule (MCAM/CD146) Accelerates Cellular Senescence in Human Umbilical Cord Blood-Derived Mesenchymal Stem Cells. Stem Cells Transl Med 5: 427-439.
- Lee NE, Kim SJ, Yang SJ, Joo SY, Park H, et al. (2017) Comparative characterization of mesenchymal stromal cells from multiple abdominal adipose tissues and enrichment of angiogenic ability via CD146 molecule. Cytotherapy 19: 170-180.
- Paduano F, Marrelli M, Palmieri F, Tatullo M (2016) CD146 expression influences periapical cyst mesenchymal stem cell properties. Stem Cell Rev 12: 592-603.
- Russell KC, Phinney DG, Lacey MR, Barrilleaux BL, Meyertholen KE, et al. (2010) In vitro high-capacity assay to quantify the clonal heterogeneity in trilineage potential of mesenchymal stem cells reveals a complex hierarchy of lineage commitment. Stem Cells 28: 788-798.
- Bara JJ, Richards RG, Alini M, Stoddart MJ (2014) Concise review: Bone marrow-derived mesenchymal stem cells change phenotype following in vitro culture: implications for basic research and the clinic. Stem Cells 32: 1713-1723.
- da Silva Meirelles L, de Deus Wagatsuma VnM, Malta TM (2016) The gene expression profile of non-cultured, highly purified human adipose tissue pericytes: Transcriptomic evidence that pericytes are stem cells in human adipose tissue. Ex Cell Res 349: 239-54.
- Muller I, Kordowich S, Holzwarth C, Spano MC, Isensee G, et al. (2006) Animal serum-free culture conditions for isolation and expansion of multipotent mesenchymal stromal cells from human BM. Cytotherapy 8: 437-444.
- Oikonomopoulos A, van Deen WK, Manansala A-R (2015) Optimization of human mesenchymal stem cell manufacturing: the effects of animal/xeno-free media. Sci Rep 5: 16570.
- Brun J, Abruzzese T, Rolauffs B, Aicher WK, Hart ML (2016) Choice of xenogenic-free expansion media significantly influences the myogenic differentiation potential of human bone marrow-derived mesenchymal stromal cells. Cytotherapy 18: 344-359.
- Seeger FH, Tonn T, Krzossok N, Zeiher AM, Dimmeler S (2007) Cell isolation procedures matter: a comparison of different isolation protocols of bone marrow mononuclear cells used for cell therapy in patients with acute myocardial infarction. Eur Heart J 28: 766-772.
- Bianco P, Cao X, Frenette PS, Mao JJ, Robey PG, et al. (2013) The meaning, the sense and the significance: translating the science of mesenchymal stem cells into medicine. Nat Med 19: 35-42.
- Doucet C, Ernou I, Zhang Y, Llense JR, Begot L, et al. (2005) Platelet lysates promote mesenchymal stem cell expansion: A safety substitute for animal serum in cell-based therapy applications. J Cell Physiol 205: 228-236.
- Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, et al. (2006) Minimal criteria for defining multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position statement. Cytotherapy 8: 315-317.
- Pilz GA, Braun J, Ulrich C, Felka T, Warstat K, et al. (2011) Human mesenchymal stromal cells express CD14 cross-reactive epitopes. Cytometry Part A 79: 635-645.
- Tormin A, Li O, Brune JC, Walsh S, Schütz B, et al. (2011) CD146 expression on primary nonhematopoietic bone marrow stem cells is correlated with in situ localization. Blood 117: 5067-5077.
- Felka T, Schafer R, De Zwart P, Aicher WK (2010) Animal serum-free expansion and differentiation of human mesenchymal stromal cells. Cytotherapy 12: 143-153.
- Fierro FA, Kalomoiris S, Sondergaard CS, Nolta JA (2011) Effects on proliferation and differentiation of multipotent bone marrow stromal cells engineered to express growth factors for combined cell and gene therapy. Stem Cells 29: 1727-1737.
- Bartosh TJ, Ylostalo JH, Mohammadipoor A, Bazhanov N, Coble K, et al. (2010) Aggregation of human mesenchymal stromal cells (MSCs) into 3D spheroids enhances their antiinflammatory properties. Proc Natl Acad Sci U S A 107: 13724-13729.
- Breitbach M, Bostani T, Roell W, Xia Y, Dewald O, et al. (2007) Potential risks of bone marrow cell transplantation into infarcted hearts. Blood 110: 1362-1369.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi