Research Article, J Vet Sci Med Diagn Vol: 6 Issue: 5
Feasibility, Within-Day and Between-Day Variability of Transthoracic Echocardiography in Sloths (Bradypus Variegatus and Choloepus Hoffmanni)
Valérie Chetboul1,2, Vassiliki Gouni1,2, Renaud Tissier2, Mauricio Jiménez Soto4, Minh Huynh3, Jean-Louis Pouchelon1,2 and Norin Chai5*
1Université Paris-Est, Ecole Nationale Vétérinaire d’Alfort, Unité de Cardiologie d’Alfort, Centre Hospitalier Universitaire Vétérinaire d’Alfort, 7 avenue du Général de Gaulle, 94704 Maisons-Alfort cedex, France
2INSERM, U955, Equipe 03, 51 avenue du Maréchal de Lattre de Tassigny, 94010 Créteil cedex, France
3Centre Hospitalier Vétérinaire Frégis, 43 Avenue Aristide Briand, 94110 Arcueil, France (Huynh)
4Hospital Especies Menoresy Silvestres, Escuela de Medicina Veterinaria, Universidad Nacional, Ulloa, Heredia, Costa Rica
5Muséum national d’Histoire naturelle, Ménagerie du Jardin des Plantes, 57 rue Cuvier 75005 Paris, France
*Corresponding Author : Norin Chai
Ménagerie du Jardin des Plantes, 57 rue Cuvier 75005 Paris, France
Tel: +33 -1 - 40 79 48 99
Fax: 33140793816
E-mail: norin.chai@mnhn.fr
Received: November 25, 2017 Accepted: December 13, 2017 Published: December 17, 2017
Citation: Chetboul V, Gouni V, Tissier R, Soto MJ, Huynh M, et al. (2017) Feasibility, Within-Day and Between-Day Variability of Transthoracic Echocardiography in Sloths (Bradypus Variegatus and Choloepus Hoffmanni). J Vet Sci Med Diagn 6:5. doi: 10.4172/2325-9590.1000245
Abstract
Bradypus variegatus (Xenarthra, Bradypodidae) and Choloepus hoffmanni (Xenarthra, Megalonychidae) are sloths from the tropical rain forest of the Caribbean and Pacific regions of Costa Rica. Because of their unique physiology and behavior, sloths have intrigued scientists, and several studies have already been conducted regarding blood pressure and heart rate variations. However, the use of transthoracic echocardiography (TTE) in these species has never been reported. The aim of the present study was therefore to determine the feasibility, and then the within-day (repeatability) and between-day (reproducibility) variability of TTE in sloths. A total of 36 TTE examinations (including a total of 1080 measurements) with continuous ECG monitoring were performed on 4 different days by a trained observer on 6 healthy, adult, sedated Choloepus hoffmanni (sexually intact females, age, mean ± SD [minmax] 5.1 ± 1.3 years [4.0-7.5]) from the Costa Rica Animal Rescue Centre. Standard transthoracic M-mode and two-dimensional mode measurements included left ventricular (LV) end-diastolic and endsystolic diameters and thicknesses, the LV fractional shortening, the E point-to-septal separation, the left atrium-to-aorta ratio, and the right atrial width-to-the left atrial width ratio. Pulsed-wave Doppler parameters included peak systolic aortic and pulmonary flow velocities as well as early and late diastolic mitral �?ow velocities. A general linear model was used to determine the within-day and between-day coefficients of variation (CV). The main results were the following: all measurements could be performed at each TTE examination. Most within- and between-day CV values (90%) were <15%, the lowest being observed for the right atrial width-to-left atrial width ratio (2.2%). These results suggest that TTE is feasible and reliable in sloths, and therefore can be part of the cardiovascular exploration in these species. Further studies are now required to determine the corresponding reference intervals.
Keywords: Sloth; Bradypus variegatus; Choloepus hoffmanni; Echocardiography; Repeatability; Reproducibility
Introduction
The brown-throated sloth (Bradypus variegatus) and the Hoffmann’s two-toed sloth (Choloepus hoffmanni) are species from the tropical rain forest of the Caribbean and Pacific regions of Costa Rica [1]. Due to this new proximity to humans, sloths are also becoming a popular “pet” for local people. Costa Rica maintains numerous captive sloths in zoos and rescue centers, primarily for educational purposes [2]. Sloths have intrigued scientists because of their unique anatomy, physiology and behavior specificities, characterized by a particular anatomy of the thoracic cage, a low metabolic rate, and a unique posture spending a large part of their time suspended from tree branches, head down. As a result, numerous studies have been carried out upon the respiratory and cardiovascular systems, and also anesthesia, blood chemistry, neuromuscular responses and system, sleeping and waking, water balance and kidney function and reproduction [3]. Regarding the cardiovascular system, few publications focusing on electrocardiography and systemic arterial blood pressure are found [3-11]. However, to the best of our knowledge, the use of transthoracic echocardiography (TTE) in these species has never been reported, although TTE is currently the most commonly used tool to non-invasively assess cardiovascular morphology and function of wild and companion animals. In order to introduce a new quantitative imaging technique in a given species, its reliability (i.e., if the technique is repeatable and reproducible, and therefore maybe used in a repeated manner) has to be evaluated. The aims of this prospective study were therefore 1) to assess the feasibility of TTE in sloths (optimal animal position and views), 2) to determine the intraobserver within-day (repeatability) and between-day (reproducibility) variability of the corresponding TTE measurements using M-mode, two-dimensional (2D) and pulsed-wave Doppler modes, and then, 3) report echocardiographic findings in a small population of clinically healthy young and adult sloths.
Materials and Methods
The care of the animals were in accordance with institutional and national guidelines (Commission de Genie Genetique, aDirection Departementale des Services Veterinaires”, European Union Directive 2010/63/EU, agreement decision No. C75-05-01-2 for the European Convention for vertebrate animals used for experimental and other scientific purposes). All protocols used in this study were approved under the reference number 68-037 by the Ethical Committee of the National Museum of Natural History (Paris).
Animals
Inclusion criteria for the study were a normal clinical history and the absence of any clinical signs apparent at a distance. Thirteen animals were evaluated: 6 adult, sexually intact females Choloepus hoffmanni kept in outdoor enclosures the Costa Rica Animal Rescue Centre were used to assess TTE feasibility and variability. Additionally, the clinical and echocardiographic findings of these 6 sloths and those of 7 young Bradypus variegatus kept in outdoor enclosures at the Kids Saving the Rainforest rescue center were reported.
Sedation protocol
Sedation was performed only in adults using a combination of IM alfaxalone (Alfaxan®, Jurox, Crawley West Sussex RH10 1DD, United Kingdom; 2-4 mg/kg) and IM midazolam (Hypnovel®, Roche, 30, Cours De L’ile Seguin. 92100 Boulogne Billancourt. France; 0.5 mg/ kg). Heart rate, respiratory rate, and body temperature in the ear were recorded every 5 min throughout sedation.
All animals underwent a complete physical examination under general anesthesia. Cardiac auscultation was performed on animals in both right and left lateral recumbency.
Echocardiographic examinations
Transthoracic echocardiograms (TTE) were performed by the same trained observer (VC) using a portable cardiovascular ultrasound system (Vivid I ultrasound unit, General Electric Medical System, Waukesha, Wisconsin, USA) equipped with a 7S (3.5-8.0 MHz) phased-array transducer, in accordance with previously published international recommendations [12].
For the assessment of TTE feasibility, different positions of the sloths (left and right lateral decubitus, ventral and dorsal recumbency, upright standing position) were investigated in a pilot study in an attempt to optimize image quality. After 4 sessions of trials, the most suitable animal position for obtaining correct to good quality 2D images was judged to be a lying position (Figure 1) on left or right lateral recumbency, using two boxes. The animal was gently restrained by two operators, with its head lying on one box, its hindquarters on the other, and its thorax pending between the two boxes.
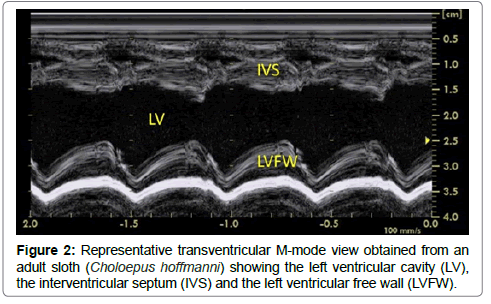
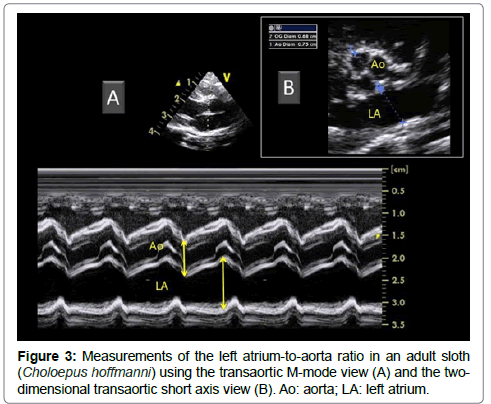
Once the appropriate position was found, the variability study was undertaken. A mean of 3 measurements was obtained for each M-mode ventricular parameter on 3 consecutive cardiac cycles on the same frame. Ventricular measurements (i.e., left ventricular (LV) end-diastolic and end-systolic diameters, LV free wall and interventricular septal thicknesses at end-diastole and end-systole) were taken from the right parasternal location using the 2D-guided M-mode (Figure 2), and the LV shortening fraction was then calculated [13-19]. Heart rate was also calculated from this M-mode view. The E point-to-septal separation was also measured using the 2D guided M-mode at the level of the mitral valve. Measurements of the aorta (Ao) and left atrium (LA) were made by two different methods (Figure 3), and the corresponding LA:Ao ratios were then calculated. One method consisted in measuring LA and Ao diameters at end-diastole, using the 2D short-axis right-sided parasternal view obtained at the level of the aortic valve where the commissures of the cusps could be visualized. The internal short-axis Ao diameter was measured along the commissure between the non-coronary and left coronary aortic valve cusps. The LA diameter was measured using the same frame, in a line extending from and parallel to the commissure between the non-coronary and left coronary aortic valve cusps. The second method consisted in using the 2D-guided M-mode from the 5-chamber long-axis view from the right parasternal location. The internal Ao diameter was measured from the corresponding M-mode view at end-diastole, and the internal LA diameter was measured at end-systole.
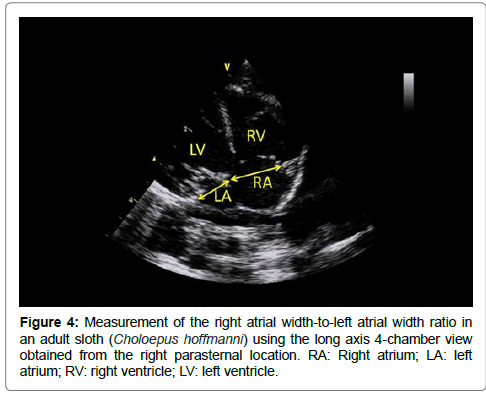
The long axis 4-chamber view obtained from the right parasternal location was used to measure the end-diastolic right atrial width-toleft atrial width ratio at the level of the tricuspid annulus and mitral annulus, respectively (Figure 4). Finally, the maximal systolic aortic and pulmonary velocities as well as the maximal early and late diastolic mitral velocities as well as E deceleration time were determined by pulsed-wave Doppler mode. The mitral E:A ratio was then calculated. A total of 18 M-mode, 2D, and Doppler variables were thus measured at each examination, i.e., 8 M-mode variables (including 6 ventricular variables measured 3 times and 2 others M-mode measured once), 5 2D and 5 Doppler variables, thus corresponding to 30 different measurements associated with the subsequent calculation of 4 ratios (LV shortening fraction, 2 LA:Ao and 1 mitral E:A ratio).
Within-day and between-day variability protocol
For the variability protocol, only the 6 healthy adult Choloepus hoffmanni were used. All examinations were randomized (i.e., for the order of examinations, animals were randomly drawn), and the same animal could not be used for 2 consecutive examinations. The study was performed on 4 different days over a one-week period. Each day, one observer did 3 echocardiographic examinations on 3 animals. Therefore, 9 echocardiographic examinations/day were scheduled, representing a total of 36 examinations with 1080 measurements and calculation of 144 ratios for the whole study period. All measurements were performed by the same observer (VC).
Statistical analysis
Data are expressed as mean ± SD and minimum-maximum values. A software program (Systat, version 10.0, SPSS Inc, Chicago, Ill) was used to perform the statistical analysis as previously described [13].
To determine the within-day and between-day variability, the following general linear model was used for each echo-Doppler variable:
Yijk = μ + dayi + slothj + (day X sloth)ij + εijk
where Yijk is the kth value measured for sloth j on day i, μ is the mean of the observed values, dayi is the differential effect of day i, slothj is the differential effect of slothj, (day X sloth)ij is the interaction term between day and sloth, and εijk is the model error. The SD of repeatability was estimated as the residual SD of the model, and the SD of reproducibility was estimated as the SD of the differential effect of day. Values of p<0.05 were considered significant.
Results
Study population characteristics
The study population was composed of 13 sloths, i.e., 6 adult female Hoffmann’s two-toed sloths (Choloepus hoffmanni) with a mean age of 5.1 ± 1.3 years [4.0-7.5 and a mean body weight of 4.6 ± 0.7 kg [3.5-5.5 and 7 young brown-throated sloths (Bradypus variegatus), 4 sexually intact males and 3 sexually intact females, with a mean age of 4.7 ± 0.5 months [4.0-5.0] and a mean body weight of 0.8 ± 0.2 kg [0.6-0.9.
Sedation inductions were rapid and uneventful, and allowed adequate handling. All animals but one were healthy and in excellent body condition, based on physical examination. Cardiac auscultation of one sloth (a 5 month-old male Bradypus variegatus) revealed a right systolic grade IV/VI heart murmur without any other abnormalities on physical examination.
Within-day and between-day variability of TTE variables
The 36 echocardiographic examinations could be well performed on the 6 healthy adult Choloepus hoffmanni for the variability protocol, including 30 different M-mode, 2D and Doppler measurements each time, representing therefore a total of 1080 interpretable measurements with deductive calculation of 144 ratios. The withinand between-day coefficients of variation (CV) and the corresponding SD for all measurements are presented in Table 1. Most within- and between-day CV values (27/30, 90%) were <15%, the lowest being observed for the right atrial width-to-left atrial width ratio (2.2%) and the highest for the E-point-to-septal separation (26.8%). For the later variable, a significant interaction between animal and day (p = 0.023) was also noted.
| Variable | Within-day | Between-day | ||
|---|---|---|---|---|
| SD | CV (%) | SD | CV (%) | |
| Left atrium/aorta-2D | 0.1 | 11.1 | 0.1 | 8.4 |
| Left atrium/aorta -M-mode | 0.1 | 7.8 | 0.1 | 12.2 |
| Left ventricular diastolic diameter (mm) | 0.8 | 4.5 | 1.4 | 7.3 |
| Left ventricular systolic diameter (mm) | 0.6 | 5.1 | 1.2 | 10.0 |
| Interventricular septum in diastole (mm) | 0.1 | 4.8 | 0.2 | 7.8 |
| Interventricular septum in systole (mm) | 0.2 | 4.7 | 0.5 | 10.6 |
| Left ventricular free wall in diastole (mm) | 0.1 | 3.9 | 0.1 | 2.3 |
| Left ventricular free wall in systole (mm) | 0.2 | 4.3 | 0.8 | 15.7 |
| Fractional shortening (%) | 1.9 | 5.5 | 1.5 | 4.2 |
| E point to septal separation (mm) | 0.2 | 19.8 | 0.2 | 26.8 |
| Right atrial/Left atrial height | 0.2 | 12.2 | 0.03 | 2.2 |
| Maximal ejection velocity during systole (m/s) | ||||
| Aorta | 0.04 | 5.7 | 0.1 | 9.8 |
| Pulmonary artery | 0.04 | 6.2 | 0.03 | 4.4 |
| Doppler variables during diastole | ||||
| Mitral E/A ratio assessed by pulsed wave Doppler | 0.1 | 6.8 | 0.1 | 8.6 |
| Deceleration time (ms) | 7.5 | 9.2 | 7.5 | 9.3 |
Table 1: Within-day and between-day standard deviations (SD) and coefficients of variation (CV) for echo-Doppler variables performed in 6 healthy adult sloths (Choloepus hoffmanni).
Echocardiographic findings in the whole study population
Regarding the TTE findings, no 2D, M-mode and Doppler abnormality was detected in the clinically healthy adult population used for the variability study (n=6 Choloepus hoffmanni). Concerning the young sloth population from the other rescue center (n=7 Bradypus variegatus), no abnormality was also observed, except for the 5 month-old male Bradypus variegatus with the right systolic grade IV/VI heart murmur. A small perimembranous ventricular septal defect (2.8 mm) with a left-to-right shunt was detected using 2D and color-flow Doppler mode. Echocardiographic measurements from this animal were excluded from mean ± SD calculations presented in Table 2. As compared with the other young sloths of similar age (Table 2), this animal showed a mildly dilated left ventricle, with a LV diameter in diastole and systole of 13.1 and 7.8 mm, respectively and LA dilation, characterized by a 2D LA-to-Ao ratio of 1.2.
| Choloepus hoffmanni (n = 6) | Bradypus variegatus (n = 6) | |||
|---|---|---|---|---|
| Variable | Mean ± SD | Minimum - Maximum | Mean ± SD | Minimum-Maximum |
| Heart rate (beats per minute) | 102 ± 16 | 135 – 75 | 126 ± 33 | 81 – 159 |
| Left atrial diameter -2D (mm) | 6.1 ± 0.6 | 4.8 – 7.4 | 3.5 ± 0.7 | 2.6 – 4.3 |
| Aortic diameter-2D (mm) | 8.1 ± 0.4 | 7.2 – 8.9 | 5.1 ± 0.4 | 4.7 – 5.7 |
| Left atrium/aorta-2D | 0.76 ± 0.08 | 0.59 – 0.91 | 0.69 ± 0.16 | 0.46 – 0.85 |
| Left atrial diameter -M-mode (mm) | 8.0 ± 0.5 | 6.9 – 9.3 | 4.3 ± 1.6 | 2.6 – 6.7 |
| Aortic diameter-M-mode (mm) | 7.5 ± 0.5 | 6.6 – 8.8 | 4.8 ± 0.8 | 3.4 – 5.5 |
| Left atrium/aorta -M-mode | 1.07 ± 0.11 | 1.31 – 0.89 | 0.89 ± 0.23 | 0.70 – 1.22 |
| Left ventricular diastolic diameter (mm) | 18.6 ± 0.9 | 17.1 – 20.7 | 9.2 ± 1.0 | 7.6 – 10.5 |
| Left ventricular systolic diameter (mm) | 12.1 ± 0.6 | 11.1 – 13.6 | 4.9 ± 0.9 | 3.2 – 5.6 |
| Interventricular septum in diastole (mm) | 3.0 ± 0.1 | 2.8 – 3.3 | 2.5 ± 0.5 | 1.9 – 3.1 |
| Interventricular septum in systole (mm) | 5.0 ± 0.3 | 4.2 – 5.3 | 3.9 ± 0.9 | 2.8 – 5.1 |
| Left ventricular free wall in diastole (mm) | 3.1 ± 0.1 | 2.9 – 3.3 | 2.6 ± 0.4 | 2.2 – 3.1 |
| Left ventricular free wall in systole (mm) | 5.1 ± 0.3 | 4.4 – 5.5 | 4.1 ± 0.6 | 3.5 – 5.3 |
| Fractional shortening (%) | 35.0 ± 2.3 | 30.3 – 40.3 | 46.8 ± 7.4 | 35.4 – 58 |
| E point to septal separation (mm) | 0.9 ± 0.2 | 0.3 – 1.1 | 0.6 ± 0.2 | 0.4 – 0.7 |
| Right atrial height (mm) | 10.8 ± 0.9 | 9.0 – 13.2 | ||
| Left atrial height (mm) | 7.5 ± 1.1 | 5.0 – 9.8 | ||
| Right atrial/Left atrial height | 1.46 ± 0.17 | 1.20 – 1.81 | ||
| Maximal ejection velocity during systole (m/s) | ||||
| Aorta | 0.79 ± 0.04 | 0.68 – 0.84 | 0.80 ± 0.20 | 0.51 – 1.15 |
| Pulmonary artery | 0.73 ± 0.06 | 0.62 – 0.88 | 0.90 ± 0.30 | 0.55 – 1.50 |
| Doppler variables during diastole | ||||
| Mitral E wave (m/s) | 0.81 ± 0.06 | 0.71 – 0.90 | 0.90 ± 0.30 | 0.55 – 1.30 |
| Mitral A wave (m/s) | 0.56 ± 0.06 | 0.44 – 0.68 | 0.50 ± 0.20 | 0.33 – 0.66 |
| Mitral E/A ratio assessed by pulsed wave Doppler | 1.46 ± 0.11 | 1.24 – 1.73 | 1.40 ± 0.20 | 1.26 – 1.67 |
| Deceleration time (ms) | 81 ± 7 | 70 – 96 | 85 ± 40 | 49 – 137 |
Table 2: Mean, standard deviation (SD), and minimum-maximum ranges of echocardiographic measurements obtained in 6 healthy adult Choloepus hoffmanni (5.1 ± 1.3 years, [4.0-7.5]) and 6 healthy young Bradypus variegatus (4.7 ± 0.5 months [4.0-5.0]). 2D: bi-dimensional echocardiography.
Discussion
To the best of the authors’ knowledge, this is the first study investigating feasibility and variability of echocardiographic exploration in sloths. The present data suggest that TTE is feasible and reliable in these species when performed by an experienced cardiologist (Dipl. ECVIM-CA Cardiology). Moreover, some normal values of echocardiographic measurements on healthy sedated individuals were obtained. Although these values cannot be used as reference intervals due to the low number of included animals, they may be of help in the future when evaluating those species for heart diseases.
Heart diseases in sloths have never been described. In the Pilosa order including sloths and anteaters, one case of dilated cardiomyopathy and amebic gastritis was diagnosed by echocardiography in a giant anteater (Myrmecophaga tridactyla) [14]. Additionally, Trypanosoma cruzi, responsible for Chagas disease in humans with potential associated myocarditis, has been isolated in blood from different species belonging to the superorder Xenarthra [15,16], including one sloth, although no case of myocarditis has been described to date in those species. In fact, in one study on myocardial lesions of Trypanosoma cruzi in opossums and armadillos from southern Louisiana [17], characteristic myocarditis lesions were only isolated in opossums, but similar studies from other endemic areas including sloths are missing. Therefore, normal echocardiographic values for sloths may be of help if those animals are evaluated for the presence of myocarditis lesions in the future.
Interestingly, a ventricular septal defect was fortuitously diagnosed in a baby sloth during our study. This case illustrates that congenital heart diseases may be present in sloths, as is the case for other mammalians, and that more routine use of TTE would be optimal in these species. Unfortunately this animal died some months after diagnosis probably due to his heart defect as a left heart dilatation was already present at diagnosis (unfortunately no necropsy was undergone). A recent study performed by our group in dogs and cats suggests that most isolated ventricular septal defects are associated with a good long-term prognosis and do not impact either the quality or duration of life, except for some reported exceptions with cardiac death related to left-sided congestive heart failure [18]. However no data are currently available on such congenital heart defect in sloths.
Echocardiography is a major diagnostic tool in human and veterinary cardiology developed within the last 35 years, allowing non-invasive morphological and functional exploration of the heart. Nevertheless, a limitation factor regarding the technique remains its accuracy and reliability, that is to say, if the method is repeatable and reproducible, and able to be used in repeated manner in animals. This was precisely the purpose of the proposed study. Accurate echocardiographic measurements are essential to the diagnosis of different cardiovascular diseases and also for the follow-up.
Our group has conducted several studies assessing repeatability and reproducibility of conventional echocardiography [18] and other ultrasound imaging techniques such as tissue Doppler imaging [13,19] in dogs and cats, using the similar variability protocols as the one used in this study with CVs close to the ones reported here. Unlike in the later reports dedicated to conventional echocardiography [18], sedation was used in the present study, but the protocol chosen for sedation is reported to have minimal cardiovascular effect in dogs with a wide margin of safety [20].
Although circadian rhythm may influence heart rate and blood pressure in those species [21], the fact that echocardiographic examinations were performed in different moments of the day did not influence variability in a negative way as the great majority of 2D, M-mode and Doppler measurements were considered as repeatable and reproducible. Indeed 90% of all variables were <15%, which represents the usual cut-off value for an acceptable inter- or intraday variability regarding quantitative imaging variables [13,18,19]. Interestingly, the right atrial width-to-left atrial width ratio was >1, which is different from what is usually observed in companion animals.
The present study has several limitations. First of all, reference intervals could not be established because of the low number of animals included. Moreover, the echocardiographic values provided in this study can only be applied to animals sedated using the same protocol and restrained in the same position. Sedation may play a role in TTE measurements in all species, and it is well known that the position of sloths has a role in blood pressure [6,7].
In conclusion, TTE may be performed with good repeatability and reproducibility by trained observers in anesthetized sloths. Further studies are needed in large sloth populations to both establish reference intervals and assess the prevalence of cardiovascular diseases.\
Acknowledgments
This study was supported by the Fondation Un Coeur (Foundation under the aegis of the Fondation de France) and by the Yaboumba Association. The authors would also like to thank the Costa Rica Animal Rescue Centre and the Kids Saving the Rainforest rescue center for their welcome and for providing the animals. A special and friendly thanks to the members of the vet team, Dylan Duby, Vincent Segalini, Leonor Camacho Sillero, Julie Malheu-Bravard, Laurie Virolle and Ernesto Rojas.
References
- Wetzel R (1985) The identification and distribution of recent Xenarthra (= Edentata). In: Montgomery GG. The Evolution and Ecology of Armadillos, Sloths and Vermilinguas. Smithsonian Institution Press, Washington.
- Superina M, Plese T, Moraes Barros N, Abba AM (2010) The 2010 Anteater List Assessment. Edentata 11: 96-114.
- http://www.scielo.br/scielo.php?script=sci_arttext&pid=S0100-879X2000000200001
- Cingolani HE, Moita MM, da Costa CE (1979) Mechanical characteristics of the myocardium of the sloth (Bradypus tridactylus). Acta Physiol Lat Am 29: 271-274.
- Duarte DP, da Costa CP, Cabral AM, Silva EM, Gilmore DP (2007) Blood pressure regulation in the three-toed sloth, Bradypus variegatus. Comp Biochem Physiol A Mol Integr Physiol 147:697-702.
- Duarte DP, Da Costa CP, Huggins SE (1982) The effects of posture on blood pressure and heart rate in the three-toed sloth, Bradypus tridactylus. Comp Biochem Physiol A Comp Physiol 73: 697-702.
- Duarte DP, Huggins SE, Da Costa CP, Leal AM (1989) The effects of posture changes on blood pressure and heart rate of anesthetized and reserpinized sloths. Comp Biochem Physiol A Comp Physiol 94: 159-165.
- Duarte DP, Jaguaribe AM, Pedrosa MA, Clementino AC, Barbosa AA, et al. (2004) Cardiovascular responses to locomotor activity and feeding in unrestrained three-toed sloths, Bradypus variegatus. Braz J Med Biol Res 37: 1557-1561.
- Meinertz T (1975) Further studies on the sinus coronarius cordis, vena cordis media and arcus aortae as well as on the ductus (lig.) Botalli in various mammalian hearts. Gegenbaurs Morphol Jahrb 121:139-154.
- Silva EM, Duarte DP, da Costa CP (2005) Electrocardiographic studies of the three-toed sloth, Bradypus variegatus. Braz J Med Biol Res 38: 1885-1888.
- Valentinuzzi ME, del Valle Ruiz E, da Costa CP (1984) Ventricular fibrillation threshold in the three-toed sloth (Bradypus tridactylus). Acta Physiol Pharmacol Latinoam 34: 313-322.
- Thomas WP, Gaber CE, Jacobs GJ, Kaplan PM, Lombard CW, et al. (1993) Recommendations for standards in transthoracic two-dimensional echocardiography in the dog and cat. Echocardiography Committee of the Specialty of Cardiology, American College of Veterinary Internal Medicine. J Vet Intern Med 7: 247-252.
- Chetboul V, Athanassiadis N, Carlos C, Nicolle AP, Tissier R, et al. (2004) Quantification, repeatability, and reproducibility of feline radial and longitudinal left ventricular velocities by tissue Doppler imaging. Am J Vet Res 65: 566-572.
- Coke RL, Carpenter JW, Aboellail T, Armbrust L, Isaza R (2002) Dilated cardiomyopathy and amebic gastritis in a giant anteater (Myrmecophaga tridactyla). J Zoo Wildl Med 33: 272-279.
- de Thoisy B, Michel JC, Vogel I, Vié JC (2000) A survey of hemoparasite infections in free-ranging mammals and reptiles in French Guiana. J Parasitol 86:1035-1340.
- Fernandes O, Mangia RH, Lisboa CV, Pinho AP, Morel CM, et al. (1999) The complexity of the sylvatic cycle of Trypanosoma cruzi in Rio de Janeiro state (Brazil) revealed by the non-transcribed spacer of the mini-exon gene. Parasitology 118: 161-166.
- Barr SC, Brown CC, Dennis VA, Klei TR (1991) The lesions and prevalence of Trypanosoma cruzi in opossums and armadillos from southern Louisiana. J Parasitol 77: 624-627.
- Bomassi E, Misbach C, Tissier R, Gouni V, Trehiou-Sechi E, et al. (2015) Signalment, clinical features, echocardiographic findings, and outcome of dogs and cats with ventricular septal defects: 109 cases (1992-2013). J Am Vet Med Assoc 247: 166-175.
- Chetboul V, Athanassiadis N, Carlos Sampedrano C, Nicolle A, Zilberstein L, et al. (2004) Assessment of repeatability, reproducibility, and effect of anesthesia on determination of radial and longitudinal left ventricular velocities via tissue Doppler imaging in dogs. Am J Vet Res 65: 909-915.
- Seo JI, Han SH, Choi R, Han J, Lee L, et al. (2015) Cardiopulmonary and anesthetic effects of the combination of butorphanol, midazolam and alfaxalone in Beagle dogs. Vet Anaesth Analg 42: 304-308.
- Duarte DP, Silva VL, Jaguaribe AM, Gilmore DP, Da Costa CP (2003) Circadian rhythms in blood pressure in free-ranging three-toed sloths (Bradypus variegatus). Braz J Med Biol Res 36: 273-278.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi