Research Article, J Vet Sci Med Diagn Vol: 12 Issue: 2
Genetic and Non-Genetic Factors Affecting Prevalence of Peste Des Petits Ruminants in West African Dwarf Sheep and Goat
Ismaila Muritala1*, Martha N Bemji1, Grace F Farayola1, Mubaraq A Busari1, Basirat O Sodimu1, Eniope B Oluwayinka2, Michael O Ozoje1, Adekayode O Sonibare2, Adedayo O Sosina3, Olusola L Ajayi4, Samuel G Moses1, Damilola J Lawal1, Seun M Kayode1, Oluwatosin A Osifeso1, Oluwaseye E Kayode1 and Eveline A Ibeagha-Awemu2
1Department of Animal Breeding and Genetics, Federal University of Agriculture Abeokuta, Ogun State, Nigeria
2Department of Veterinary Medicine and Surgery, Federal University of Agriculture Abeokuta, Ogun State, Nigeria
3Department of Agricultural Development, Ajayi Crowther University, Oyo State, Nigeria
4Department of Pathology, Federal University of Agriculture Abeokuta, Ogun State, Nigeria
*Corresponding Author: Ismaila Muritala
Department of Animal Breeding and
Genetics, Federal University of Agriculture Abeokuta, Ogun State, Nigeria
E-mail: ismailmurtala4@gmail.com
Received date: 31 October, 2022, Manuscript No. JVSMD-22-78646; Editor assigned date: 02 November, 2022, PreQC No. JVSMD-22-78646 (PQ); Reviewed date: 15 November, 2022, QC No. JVSMD-22-78646; Revised date: 02 February, 2023, Manuscript No. JVSMD-22-78646 (R); Published date: 09 February, 2023, DOI: 10.4172/2325-9590.100040
Citation: Muritala I,Bemji MN, Farayola GF, Busari MA, Sodimu BO, et al. (2023) Genetic and Non-Genetic Factors Affecting Prevalence of Peste Des Petits Ruminants in West African Dwarf Sheep and Goats. J Vet Sci Med Diagn 12:2
Abstract
After the groundbreaking eradication of rinderpest, peste des petits ruminants is currently the target for global eradication by 2030. Understanding the factors affecting its prevalence is imperative for effective, evidence-based planning for the eradication of the disease. Genetic and non-genetic factors affecting PPR prevalence in sheep and goats (403) kept by smallholder farmers were investigated using the positivity of faecal samples to Haemagglutination Assay (HA). Data on positivity to HA, HA titre and rectal temperature of the animals were statistically analyzed. Overall PPR prevalence of 86.0% was recorded for West African Dwarf sheep and goats. Effect of species and coat color were significant on PPR prevalence with an indication that goats had a higher (P<0.05) estimate (84.9%) compared to the PPR prevalence (65.9%) in sheep. Location affected PPR prevalence with highest prevalence (87.6%) recorded in Oyo compared to Ogun (67.9%), while Osun state had 76.2%. The effects of gender and age were not significant (p>0.05). PPR is endemic in three southwestern States of Nigeria and its prevalence in small ruminant may be influenced by coat color, species and location, thus farmer sensitization, advocacy and public enlightenment on the PPR control require urgent attention.
Keywords: PPR disease; Predisposing factors; Haemagglutination assay; Sheep and goats
Introduction
Small ruminant plague, widely recognized as Peste des Petits Ruminants (PPR), an acute and highly contagious viral disease plaguing both domestic and wild small ruminant species. It is characterized by high fever, oculo-nasal discharge, pneumonia, necrotic, erosive and ulcerative stomatitis and inflammation of the gastrointestinal tract leading to severe diarrhea and death [1]. It causes severe economic loss to some developing countries (e.g. Nigeria, Ghana, Cote d’Ivoire, Mali, Mauritania, Liberia, Republic of Benin, Cameroon, Sudan, Saudi Arabia etc.) where the disease is endemic [2,3]. Annual economic loss in Nigeria due to the PPR outbreak in small ruminant was estimated at 1.50 million USD [4]. Generally, the disease is more pathogenic in goats than sheep [5].
Significant differences exist in the epidemiology of PPR in diverse ecological and geographical areas. The arid and semi-arid regions may present low PPR fatality which may exist as a subclinical or mild infection, giving chances to many opportunistic infections as pasteurellosis [6]. Some risk factors have been identified to contribute to the transmission of PPR disease. Among these are the migration and exchange of animals between flocks, gender, age, species, breed and season [7-9]. Idowu and Adelabu reported coat color variations (black, brown, badger face, grey, buckskin, lateral belly, white and black, white and brown, white and black marking, black and white marking, spotted white, spotted black mash, tan, white, bezoar, spotted brown lateral stripes and swiss marking) in WAD goats. However, the effect of such important trait with huge variations, on PPR prevalence has not been reported. Such information could help in the selection for improved resistance to PPR in small ruminants. The enzootic form of PPR in recovered animal may be responsible for how PPR virion is maintained between seasons of varying prevalence. A reason why the introduction of new sheep or goats into healthy flocks often leads to fresh outbreaks of PPR [10].
After the eradication of Rinder Pest (RP), PPR is target for global eradication by 2030 [11]. The eradication program would benefit from understanding and reporting of the factors influencing its prevalence. It could guide in planning the effective control measure, targeting eradication of the disease. In the light of this, the current study aimed at evaluating the genetic factors and non-genetic factors affecting PPR prevalence in small ruminant across different species, coat colors, genders, location and ages in three Southwestern states of Nigeria.
Materials and Methods
Ethics statement
The animal experiment was carried out in accordance with the ethical standards of the Federal university of agriculture Abeokuta, college of animal science and livestock production committee on animal experimentation for the care and handling of experimental animals.
Animals sampled and sampling techniques
A multi-stage sampling procedure was adopted to investigate the effect of genetic and non-genetic factors affecting PPR prevalence in 403 randomly selected West African Dwarf (WAD) breeds of small ruminants consisting of 216 goats and 107 sheep of both genders, 3 different age groups (≤ 1 year, >1-2 years and >2 years) and 7 categories of coat colours reared under different management systems in the 3 southwestern States (Oyo, Osun and Ogun State), Nigeria. Faeces were collected from the animals directly from the rectum, transferred into a plain bottle and kept in a cold box before transporting to the laboratory. A total of 3 ml of Phosphate Buffer Saline (PBS) of pH 6.8 was added to 1 g of faecal sample and incubated for 12 hrs at -20°C. Homogenate was thawed and centrifuged at 2000 xg for 10 min. The supernatant was used as an antigen source. A digital thermometer was disinfected, lubricated and inserted into the rectum of the animal to measure the rectal temperature.
PPR viral antigen detection
As an indicator in Haemagglutination Assay (HA), chicken whole blood was collected in EDTA bottles and washed three times using PBS. A 1% concentration of the washed Chicken Red Blood Cells (CRBCs) was prepared and used for HA [12]. A total of 50 μl of PBS was added to the U-bottom wells, equal volume of antigen source was added to well 1, two-fold serially diluted across the row to well 11, leaving only well 12. Fifty microliter of 1% CRBCs was added to each well and incubated at 4°C for 20 minutes. A well was considered positive when CRBCs were suspended in the reaction, while a well with settled CRBCs was considered negative.
Data collection and analysis
Data on HA titre and rectal temperature were collected from each animal and positivity to HA was used for PPR prevalence determination. Analysis of variance was used to test for significance in the prevalence between genders and species, among age groups, locations and coat colours. Univariate analysis of genetic and nongenetic effects on HA titre and rectal temperatures were determined using factorial design included in SAS version 9.1.3 [13]. Pearson correlation (r) between rectal temperature and HA titre was determined.
Results
Inspection of PPR clinical signs among the sampled sheep and goats in the Southwestern region of Nigeria showed, most domesticated small ruminants which appeared healthy were infected with the virus as revealed by the result of heamagluttination assay and the rectal temperature measured in the sampled animals. Generally, a PPR prevalence of 86% was recorded in WAD sheep and goats in the studied areas, which occurred at varying levels of prevalence among different genetic classes and non-genetic components.
Effects of genetic factors on PPR prevalence
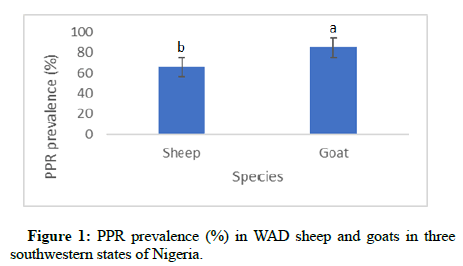
Effect of species on the PPR prevalence in WAD small ruminants was significant for the three southwestern states of Nigeria. The prevalence (84.98%) was higher (p<0.05) in goats compared to the prevalence (65.95%) in sheep (Figure 1).
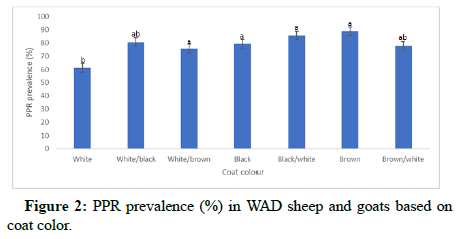
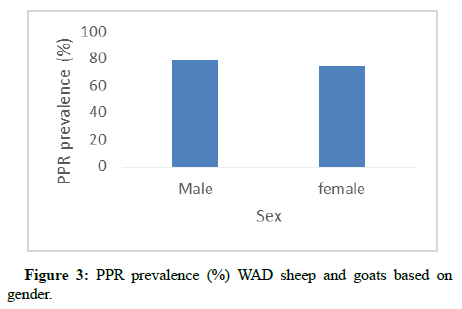
Coat color had a significant effect (p<0.05) on the PPR prevalence among sheep and goats. The prevalence of 88.89% was recorded for brown coat color and 85.71% for black/white coat color which were significantly higher than the prevalence (61.26%) recorded for completely white coat color. However, brown, black/white; white/ black; white/brown; completely black and brown/white were statistically similar (p>0.05) as shown in Figure 2. Also, prevalence in sheep and goats with white coat color was slightly lower but statistically similar (p>0.05) with the prevalence in white/black; white/ brown; black and brown/white. As shown in Figure 3, both males and females showed statistically similar (p>0.05) PPR prevalence (78.57 and 74.82%, respectively).
Effects of non-genetic factors on PPR prevalence
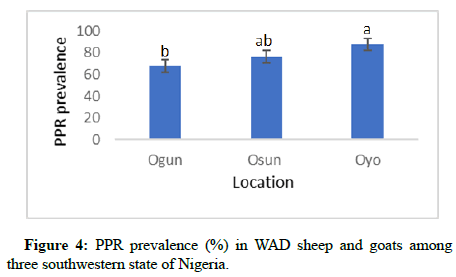
Location had a significant effect (p<0.05) on the PPR prevalence in small ruminants. The highest prevalence (87.63%) was observed in Oyo state, which was significantly higher than the estimate (67.88%) in Ogun state. PPR prevalence (76.22%) in Osun state was statistically similar (p>0.05) with the prevalence in both Oyo and Ogun state as shown in Figure 4. The effect of age was not significant (p>0.05) on the PPR prevalence among sheep and goats in the southwestern states of Nigeria (Figure 5).
Estimates of HA titre and rectal temperature in small ruminants as affected by genetic factors
As shown in Table 1 Least Square Means (LSM) of HA titre in sheep was estimated at 16.03 HAU, statistically similar (p>0.05) to the value (23.78 HAU) observed in goats, thus, effect of species on HA titre in WAD small ruminants was not significant (p>0.05). Brown/white coat color had highest LSM of HA titre (73.78 HAU) significantly (p<0.05) different from HA titre for other coat colors. The effect of coat color on the HA titre of sheep and goats in this study was significant (p<0.05). Males and females had statistically similar (p>0.05) estimates of LSM of HA titre (17.77 HAU and 21.12 HAU, respectively). LSM of rectal temperature in goats (39.62°C) was significantly (p<0.05) different from the value recorded for sheep (39.05°C). Sheep and goats with entirely black and solely brown coat colors had rectal temperature (39.70°C and 39.66°C, respectively) significantly (p<0.05) different from the observed values in sheep and goats with completely white and, white/black coat colors. There was no significant (p>0.05) difference in the LSM of rectal temperature between male (39.30°C) and female (39.38°C) of WAD sheep and goats.
Estimates of HA titre and rectal temperature in small ruminant as affected by non-genetic factors
There was no significant difference (p>0.05) in the LSM of HA titre of PPR infected domesticated small ruminants in terms of age groups as shown in Table 1. Sheep and goats from Osun state had significantly (P<0.05) higher rectal temperature (39.51°C) than estimates (39.26°C) from Ogun State and Oyo State (39.17°C). Animals below one year of age had the highest (P<0.05) rectal temperature (39.66°C) than animals in the older age groups (>1-2 and >2 years).
Pearson Correlation between rectal temperature and HA titre
Pearson correlation estimate (0.11) between HA titre of the faucal samples collected from sheep and goats and the rectal temperature was significant (p<0.05).
| Haemagglutination assay titre (HAU) | Rectal temperature (°C) | ||||
|---|---|---|---|---|---|
| Factor | Sub-classes | N | LSM ± SE | N | LSM ± SE |
| State | Ogun | 137 | 19.61 ± 4.11 | 140 | 39.26 ± 0.05 |
| Osun | 164 | 17. 94 ± 2.52 | 164 | 39.51 ± 0.09 | |
| Oyo | 97 | 24.78 ± 10.70 | 57 | 39.17 ± 0.12 | |
| Species | Sheep | 185 | 16.03 ± 5.80 | 163 | 39.05 ± 0.05 |
| Goat | 213 | 23.78 ± 2.98 | 198 | 39.62 ± 0.07 | |
| Gender | Male | 112 | 17.77 ± 3.26 | 103 | 39.30 ± 0.08 |
| Female | 286 | 21.12 ± 4.18 | 258 | 39.38 ± 0.06 | |
| Age group | ≤ 1 | 117 | 18.31 ± 3.02 | 103 | 39.64 ± 0.07 |
| >1-2 | 142 | 22.36 ± 7.71 | 131 | 39.11 ± 0.10 | |
| >2 | 139 | 19.53 ± 3.52 | 127 | 39.39 ± 0.06 | |
| Coat color | White | 111 | 11.96 ± 3.07 | 97 | 38.99 ± 0.06 |
| White/black | 31 | 7.81 ± 1.82 | 21 | 39.02 ± 0.17 | |
| White/brown | 33 | 12.73 ± 3.21 | 30 | 39.23b ± 0.14 | |
| Black | 107 | 25.21 ± 4.54 | 103 | 39.70 ± 0.07 | |
| Black/white | 35 | 26.11 ± 9.87 | 34 | 39.24 ± 0.13 | |
| Brown | 63 | 17.49 ± 3.28 | 60 | 39.66 ± 0.19 | |
| Brown/white | 18 | 73.78 ± 56.09 | 16 | 39.25 ± 0.16 | |
Table 1: Least Square Means (LSM) of HA titre and rectal temperature by species, gender, age and coat color.
Discussion
An overall PPR prevalence of 86.0% in domesticated small ruminants in the three Southwestern states of Nigeria studied was considerably higher compared to estimates ranging from 16.7% to 76.2% reported in different parts of the country [14-16]. Based on information from extension agents and the farmers in Ogun and Oyo States, there is annual vaccination of small ruminants (with coverage limited to rural settings). On this account, PPR prevalence is expected to decrease with time. Contrary to this, PPR control has not been very effective. This could be a result of uncontrolled movement of animals from one part of the country to the other and inconsistencies in implementation of policy on the control of PPR in most parts of the areas where sheep and goats are reared. Another possibility is the reintroduction of the virus by the wildlife reservoir [17].
PPR prevalence of 84.9% in goats was relatively higher than the prevalence (65.9%) in sheep. This could be an indication of differences in species susceptibility to the PPR virus which may not be unconnected with their differences in genetic make-up. PPR is largely seen as a severer disease in goat than sheep [18]. However, a higher seroprevalence in Sudanese sheep (62.1%) than in goat (53.5%) have been reported [19]. Dissimilarities in the observations by different authors could be as a result of differences in the climates where the studies were carried out. Some climates may favour one species over the other in susceptibility to PPRV infection due to their differences in their physiology and genetic composition.
Effect of coat colour showed that sheep and goats with brown coat colour had highest PPR prevalence (88.9%) than animals with other coat colors, while the least PPR prevalence (61.3%) was recorded for individuals with completely white coat colour. This is an indication that there may be association between coat colour determinant gene and sensitivity to PPR infection which needs further investigation. To the best of our knowledge, no report on the effect of coat colour on sheep and goats’ susceptibility to PPR has been documented. Coat colour in animals does not only play an aesthetic role, but also has substantial impact on many factors, indispensable for survival [20].
PPR prevalence in male and female were equally high in this study (78.6% and 74.8%, respectively). Gender has previously been mentioned as a risk factor for PPR, with females mostly identified to be at higher risk [21]. However, some studies have reported a higher risk for males than females [22,23]. Based on the findings from this study both males and females were equally at risk of PPR infection.
Effect of location on PPR prevalence revealed higher prevalence (87.6%) in Oyo state compared to Osun and Ogun states. This might not be unconnected with the fact that Oyo state serves as a major transit and sales point for sheep and goats transported from the North to the South, from where they are distributed to other states in the Southwest. Thus, high influx of livestock from Northern states could serve as potential carriers of the virus, thereby contributing to the higher PPR prevalence recorded in Oyo state. The observed estimate for Oyo state was also higher than previously reported estimate of 75.0%, which is likely expected over time with high influx of potential carriers of the virus and limited coverage in vaccination concentrating in rural than urban and peri-urban areas. The least PPR prevalence (67.9%) among ruminants in the rural settings was recorded in Ogun state, which was lower than 80.0% earlier reported from animals under semi-intensively and intensively managed systems within the urban and peri-urban settings. Osun had 76.2% PPR prevalence which is considerably higher than 24.5%. This increase in PPR prevalence overtime may be due to factors such as uncontrolled movement of animals from other states and lack of proper awareness for PPR disease control among the farmers in the state.
Generally, the LSM of HA titre was lower in this study (7.81 HAU to 73.8 HAU) compared to the HA titre range of 8.00 HAU to 256 HAU reported. This could be a result of the fact that most animals sampled in this study were healthy but carriers of the virus and thus expected to have low HA titre. Lower LSM HA titre could mean a reduction in the PPR antigen in the faeces during the recovery stage or the animals acquired the virus newly. Noted that there could be a reduction in mean titre of PPR virus haemagglutinins shed in the faeces with time.
LSM of rectal temperature of sampled goats revealed some were pyrexia as they had rectal temperature of greater than 39.0°C, according. Pyrexia is recognized as one of the clinical signs of PPR in infected animal which can be accompanied by depression, anorexia and dryness of muscle. Correlation between the HA titre and rectal temperature showed that increase in HA titre will increase in rectal temperature (pyrexia) and could be a reliable means of suspecting PPR infection in animal. This is in agreement with who classified a temperature range of 39.8°C-41°C (pyrexia) as a sign of PPR infection.
Conclusion
Findings from this study revealed that peste des petits ruminants virus is endemic in the study areas. Species, coat colour and location were observed to be risk factors for the susceptibility of small ruminant to PPR infection. Sensitization of farmers, advocacy, public enlightenment on the PPR disease control measures and mass vaccination require urgent attention.
Acknowledgement
The authors are grateful to the following institutions and individuals for allowing use their facilities and animals for the investigation: Veterinary teaching hospital, directorate of university farms and professor James dairy goat research farm, all of federal university of agriculture abeokuta, Nigeria; national centre for genetics and biotechnology (NACGRAB) and Institute of Agricultural Research and Training, Obafemi Awolowo University (IAR and T), moor plantation, Ibadan, Nigeria; Oyo, Ogun and Osun state Agricultural Development Programmes (ADPs); International Livestock Research Institute (ILRI), Ibadan, Nigeria and all the farmers who participated in the survey.
References
- Parida S, Selvaraj M, Gubbins S, Pope R, Banyard A, et al. (2019) Quantifying levels of Peste des Petits Ruminants (PPR) virus in excretions from experimentally infected goats and its importance for nascent PPR eradication programme. Viruses 11:249.
[Crossref] [Google Scholar] [PubMed]
- Otsyina HR, Arthur CT, Ayim-Akunnor M, Obese FY (2013) Sero-prevalence of Pestes des Petits Ruminants (PPR) in sheep, goats and cattle in Ghana. Bull Anim Health Prod Afr 61:473-479.
- Kaukarbayevich KZ (2019) Epizootological analysis of PPR spread on African continent and in Asian countries. Afr J Trop Agric 7:001-004.
- Banyard AC, Parida S, Batten C, Oura C, Kwiatek O, et al. (2010) Global distribution of peste des petits ruminants virus and prospects for improved diagnosis and control. J Gen Virol 91:2885-2897.
[Crossref] [Google Scholar] [PubMed]
- Lefevre PC, Diallo A (1990) Peste des petits ruminants. Rev Sci Tech 9:951-965.
[Crossref] [Google Scholar] [PubMed]
- Elsawalhy A, Jeffrey C, Mariner J, Dickens C, Henry W, et al. (2010 ) Pan African strategy for the progressive control of PPR. Bull Anim Health Prod Afr 185-193.
- Abubakar M, Hadder AK, Muhammed JA, Manzoor H, Qurban A (2011) Review: Peste des Petits Ruminants (PPR): Disease appraisal with global and Pakistan perspective. Small Ruminants Research 96:1-10.
- Idowu PA, Adelabu OA (2018) An investigation of coat colour distribution of WAD goats. J Agric Sci 10:228.
- Ezeibe MCO, Eze IC, Ngene AA (2008) Persistent detection of pestes de petits ruminants antigen in the faeces of recovered goats. Trop Anim Health Prod 40:517-519.
[Crossref] [Google Scholar] [PubMed]
- Latif A, Akhtar Z, Ullah RW, Zahur AB, Ullah A, et al. (2014) Evaluation of Haemagglutination Assay (HA) for the detection of Peste des Petits Ruminants Virus (PPRV) in faecal samples of recovered goats. Res J Vet Pract 2:11-13.
- Abdullahi US, Shehu LM, Shuaibu MT (1999) The epidemiology of peste des petits ruminants in Bauchi State. Niger Vet Med Assoc 26-29.
- Luther NJ, Umoh JU, Majiyagbe KA, Shamaki D, Nwosu CI, et al. (2005) Studies on the prevalence of antibodies to Peste de Petits Ruminants Virus (PPRV) among Goats in Bauchi State. Niger Vet J 27:17-22.
- Bukar BA, El-Yuguda A, Said L (2020) The first seroprevalence investigation of peste des petits ruminants virus among sahel goat in Yobe State, Nigeria. Asian J Med Health 18:33-38.
- Albina E, Kwiatek O, Minet C, Lancelot R, de Almeida SR, et al. (2013) Peste des petits ruminants, the next eradicated animal disease? Review. Vet Microbiol 165:38-44.
[Crossref] [Google Scholar] [PubMed]
- Quantifying levels of Peste des Petits Ruminants (PPR) virus in excretions from experimentally infected goats and its importance for nascent PPR eradication programme.
- Ishag OM, Intisar KS, Ali YH (2014) Detection of antibodies to peste des petits ruminants virus using passive haemagglutination test and cELISA in the White Nile state-Sudan, comparative study. Afr J Microbiol Res 8:3475-3481.
- Charon KM, Lipka KR (2015) The effect of a coat colour-associated genes polymorphism on animal health: A review. Ann Anim Sci 15:3-17.
- Kivaria FM, Kwiatek O, Kapaga AM, Swai ES, Libeau G, et al. (2013) The incursion, persistence and spread of peste des petits ruminants in Tanzania: Epidemiological patterns and predictions. Onderstepoort J Vet Res 80:593-604.
[Crossref] [Google Scholar] [PubMed]
- Mahajan S, Agrawal R, Kumar M, Mahan A, Pande N (2012) Risk of seroconversion to Peste des Petits Ruminants (PPR) and its association with species, gender, age and migration. Small Rumin Res 104:195-200.
- Mbyuzi AO, Komba EV, Kimera SI, Kambarage DM (2014) Sero-prevalence and associated risk factors of peste des petits ruminants and contagious caprine pleuro-pneumoniain goats and sheep in the Southern zone of Tanzania. Prev Vet Med 116:138-144.
[Crossref] [Google Scholar] [PubMed]
- Lawal A, Lasisi OT, Emikpe BO, Ogundipe GAT (2011) Outbreak of peste des petits ruminants in West African dwarf goats in Eruwa, Southwestern Nigeria. Niger Vet J 32:331-335.
- Okwelum N, Adewumi OO, Akinduti PA, Mshelbwala FM, Williams TJ (2017) Prevalence of suspected peste des petits ruminants infection and complicating bacteria in goats in Abeokuta, Ogun State, Nigeria. Niger J Anim Prod 44:43-48.
- Bamouh Z, Fakri M, Jazouli O, Safini F, Tadlaoui N, et al. (2019) Peste des petits ruminants pathogenesis on experimental infected goats by the Moroccan 2015 isolate. BMC Vet Res 15:452.
[Crossref] [Google Scholar] [PubMed]
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi