Research Article, J Pulm Med Vol: 5 Issue: 2
Influence of Breathing Exercises on the Thoracoabdominal Wall Volume of Healthy Adults
Catherine Corrêa Peruzzolo1, Stefani dos Santos Marcelino1, Maryne Ramos da Silva1, Wellington Pereira dos Santos Yamaguti2, Danielle Soares Rocha Vieira3, Dayane Montemezzo1 and Elaine Paulin1*
1Department of Physiotherapy, Santa Catarina State University, Santa Catarina, Brazil
2Instituto Sírio Libanês de Ensino e Pesquisa, Hospital Sírio Libanês, São Paulo, São Paulo state, Brazil
3Federal University of de Santa Catarina (UFSC), Araranguá, Santa Catarina, Brazil
*Corresponding Author: Elaine Paulin Department of Physiotherapy, Santa Catarina State University, Santa Catarina, Brazil, Tel: (48) 3664-8602; E-mail: elaine.paulin@udesc.br.
Received Date: 29 April, 2021; Accepted Date: 13 May, 2021; Published Date: 20 May, 2021
Citation: Catherine CP, Stefani dos SM, Maryne RS, Wellington Pereira dos SY, Danielle Soares RV, Dayane M et al. (2021) Influence of Breathing Exercises on the Thoracoabdominal Wall Volume of Healthy Adults. J Pulm Med 5:2
Abstract
Objective: There is little scientific evidence on the effects of breathing exercises (BE) on chest wall (CW) movement. The aim of this study was determine and compare the influence of diaphragmatic breathing (DB), fractionated breaths (FB), sniff inspiration (SI) and breathing from functional residual capacity (BFRC) on thoracoabdominal wall volumes in healthy adults in the sitting position and in dorsal decubitus at 30° trunk inclination. Methods: Chest wall volumes (Vcw) were assessing by optoelectronic plethysmography (OEP) were assessed in the sitting position and 30° trunk inclination.
Keywords: Breathing exercises; Chest wall; Rehabilitation; Optoelectronic plethysmography; Breathing pattern; Thoracoabdominal motion; Position
Introduction
Breathing strategies are used by physiotherapists in different healthcare levels to optimize and change respiratory muscle activity, primarily the diaphragm, and influence breathing patterns and thoracoabdominal motion [1,2]. These include breathing exercises, which are widely used in clinical practice, diaphragmatic breathing (DB) being the most widely studied and commonly applied to improve ventilation in the lower chest region. The aim is to increase diaphragmatic activity, promote greater abdominal motion and ventilation in the lung bases, increase tidal volume, decrease respiratory rate and dyspnea, and enhance functional capacity. Sniff inspiration (SI) exercises, fractionated breaths (FB) and breathing from functional residual capacity (BFRC) aim at stimulating the action of the diaphragm and improving the expansion of lung bases and abdominal motion. However, few studies have shown the effects of these exercises on pulmonary ventilation and thoracoabdominal motion [1-11].
Breathing exercises influence respiratory kinematics, but the position in which they are performed may change thoracoabdominal wall kinematics during exercise, since gravity has a direct effect on ventilatory muscle functionality [9].
Studies that assessed thoracoabdominal mobility using three dimensional analysis of movement (Vicon MX) and optoelectronic plethysmography (OEP) showed changes in thoracoabdominal motion during slow deep breathing, when the subject is in the sitting or orthostatic position. Rib cage displacement was greater in the sitting or orthostatic position and abdominal motion was higher in dorsal decubitus (DD) or at 30° trunk inclination [12-14].
However, despite the fact that DB, FB, SI and BFRC are relatively well known and widely used exercises in clinical practice, few studies have investigated their effect on thoracoabdominal motion during these interventions. There are also few studies that determined the influence of position used during these exercises. As such, the aim of this study was to determine whether exercise and position (sitting or in dorsal decubitus with 30° trunk inclination) influences chest wall movement and volume in healthy adults.
Methods
Subjects
Non probability sampling included healthy individuals of both sexes, aged between 18 and 65 years, with body mass index (BMI) of 18.5 ≤ BMI normal ≥ 29.9 kg/m2, non-smokers, with no ventilatory disturbances of any type as determined by the lung function test, no selfreported neuromuscular or respiratory diseases, recent surgery, history of cardiovascular disease and not pregnant [15,16].
Excluded were uncooperative subjects, those with difficulty executing the breathing exercises and individuals with heart rate (HR) more than 85% above resting HR, less than 90% of peripheral oxygen saturation (SpO2) and resting blood pressure (BP) above 180/110 mmHg [16].
Study design and ethical aspects This is an observational study with a quantitative approach, approved by the institutional Human Research Ethics Committee with protocol number 69929517.7.0000.0118.
Collection procedures
After providing written informed consent, the participants were asked about their health history and age, measured for height, weight, and body mass index (BMI) and rated for perceived exertion using the Borg scale. The following cardiorespiratory parameters were also measured: BP, HR, respiratory rate (f), and SpO2, and the lung function test were applied according to the methods and criteria recommended by the American Thoracic Society (ATS) and European Respiratory Society (ERS) [17]. The variables were reported in absolute values and in percentage of predicted values and the function test were considered normal when forced vital capacity (FVC) and forced expiratory volume in 1 second (FEV1) parameters were greater than or equal to 80% of the predicted value and the FEV1/FVC ratio was higher than 0.7.16 next, the subjects were familiarized with the breathing exercises, using a set of ten repetitions of each exercise, according to the examiner’s instructions.
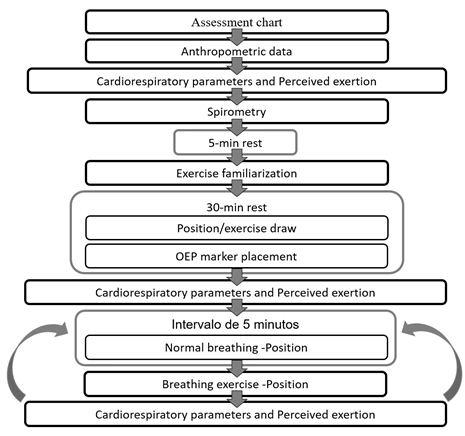
After familiarization, a 30 minute rest period allowed cardiorespiratory parameters to return to basal levels. During this time a draw was made to determine the initial position (sitting or at 30° trunk inclination), and the exercise sequence (DB, FB, SI or BFRC) in addition to attaching the reflexive markers to the subject’s chest using OEP. The OEP assessment protocol followed the recommendations of Aliverti et al. for the use of 89 markers, attached to the skin in the sitting position, and of Aliverti et al. and Romei et al. for 52 markers in dorsal decubitus (DD) at 30° trunk inclination. The markers were arranged in horizontal and vertical lines, according to the following anatomical reference points: collarbones, angle of Louis, nipples, xyphoid process, inferior coastal margin, supra umbilical line and anterior superior iliac spine [12,18- 20]. The cameras were positioned to capture the entire chest, and focus, zoom and image clarity were adjusted.
During slow breathing, respiratory cycles (DB, SI, FB and BFRC) were recorded in the sitting position and in DD at 30° trunk inclination. Participants breathed normally for three minutes, followed by a set of 12 repetitions for each exercise. They rested for 5 minutes between exercises and HR and SpO2, as well as the feeling of tiredness and dyspnea, were recorded. Once the cardiorespiratory parameters returned to basal values, they performed the next exercise drawn, and so on until executing all four exercises in both positions (Figure 1). The verbal command was standardized for each exercise. All the procedures were conducted on a single day by the same researcher.
DB was carried out via deep nasal inspiration, prioritizing anterior displacement of the abdominal region, avoiding displacing the rib cage (RC) and basal oral expiration [4]. For SI, short, successive slow nasal inspirations were performed, the last orally, until reaching near total lung capacity [5,8]. FB exercise consisted of 4 smooth nasal inspirations followed by post inspiratory pauses of approximately 2 to 3 seconds, near total lung capacity, concluding with oral expiration [9,20]. BFRC involved the subject’s performing slow oral expiration, near the resting expiratory level, followed by an inspiration until reaching approximate inspiratory reserve volume (IRV). [8,9].
Variables analyzed
The following variables were considered in each breathing exercise: chest wall volume (Vcw), respiratory rate (f), minute ventilation (VE), pulmonary rib cage volume (Vprc), abdominal rib cage volume (Varc), abdominal volume (Vab), percentage contribution of the pulmonary rib cage (%Vprc), percentage contribution of the abdominal rib cage (%Varc) and percentage contribution of the abdomen (%Vab).
Data reduction
The tidal volume variables of all the compartments were determined after 180 seconds during normal breathing, with the first and last 30 seconds of each recording excluded. Twelve repetitions were performed, 10 of which were used for analysis, with the first and last repetition of each exercise excluded.
Statistical analysis
The data were presented as measures of central tendency and dispersion. The variables age, sex, BMI and spirometric parameters were used to characterize the sample. The Shapiro Wilk test was used to analyze normal data distribution. For normally distributed data, repeated measures two ways ANOVA with Bonferroni’s post-hoc were applied to determine the effects of the exercises, positions and exercise position interaction. The sample was calculated based on a pilot study with the first 10 subjects. The following OEP related variables were considered: Vcw, f, VE, Vprc, Varc and Vab. The significance level was set at 5%, with a statistical power of 0.80, resulting in 33 subjects [5,21].
The data were processed in the SPSS (Statistical Package for Social Sciences) program, version 20.0.
Results
A total of 52 subjects were recruited, 33 of whom participated in the study, 7 refused to take part, 2 did not appear on the assessment day, and 2 were excluded for not meeting inclusion criteria on the lung function test and 8 for technical problems with OEP (complications during data collection, system malfunctioning and inconsistent data at processing).
Table 1 shows participant characterization, according to demographic, anthropometric and spirometric data. The sample consisted of a similar number of men and women, age ranged from 22 to 59 years, and BMI and spirometric data remained in the normal range.
| Variables | Mean ± SD |
|---|---|
| Sex | 15M/18W |
| Age (years) | 31.1 ± 11.05 |
| BMI (Kg/m²) | 23.6 ± 3.1 |
| FEV1 (L) | 3.6 ± 0.76 |
| FEV1 (% predicted) | 100.0 ± 11.41 |
| FVC (L) | 4.29 ± 0.9 |
| FVC (% predicted) | 98.25 ± 9.89 |
| FEV1/FVC | 0.85 ± 0.05 |
| Data presented as mean and standard deviation, except for sex. H: Men. W: Women. BMI: Body mass index. FEV1: forced expiratory volume in 1 second. FVC: forced vital capacity. FEV1/FVC: ratio between forced expiratory volume and forced vital capacity | |
Table 1: Subject characterization in terms of demographic, anthropometric and lung function data.
Breathing pattern variables (Vcw, f and VE) at rest and during breathing exercises, in the sitting position and DD at 30° trunk inclination
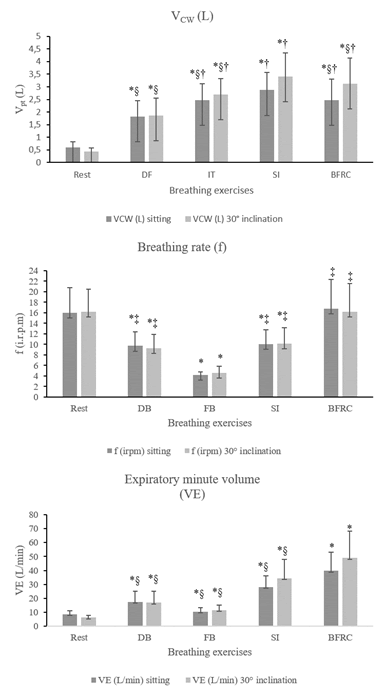
Vcw exhibited a significant increase during all the exercises in relation to at rest for the sitting position and DD at 30° trunk inclination. FB, SI and BFRC displayed a higher Vcw when compared to DB in the two positions (P<0.001). Moreover, in the SI exercise, Vcw was higher than that of the other exercises in both positions (P<0.001).
During the exercises, DB, FB and SI showed a reduction in f in relation to at rest in both positions (P>0.001); only BFRC exhibited no difference (P=1), and the highest f (P<0.001), with the FB exercise obtaining the lowest f (P<0.001).
VE displayed a significant increase in all the exercises when compared to at rest in both positions. It was higher in BFRC than in the other exercises in both positions (P<0.001). The FB exercise showed a lower VE compared to the others in the two positions studied (P<0.001) (Figure 2).
Figure 2: Breathing pattern at rest and during the four breathing exercises. Data presented as mean (X) and standard deviation. Vcw: chest wall volume; f: respiratory rate and VE: minute ventilation. *p<0.005 for rest x breathing exercises; † p<0.005 for diaphragmatic exercise × fractionated breaths, sniff inspirations and BFRC; ‡ p<0.005 for fractionated breaths × diaphragmatic exercise, sniff inspirations and BFRC; § p<0.005 for sniff inspirations × fractionated breaths and BFRC.
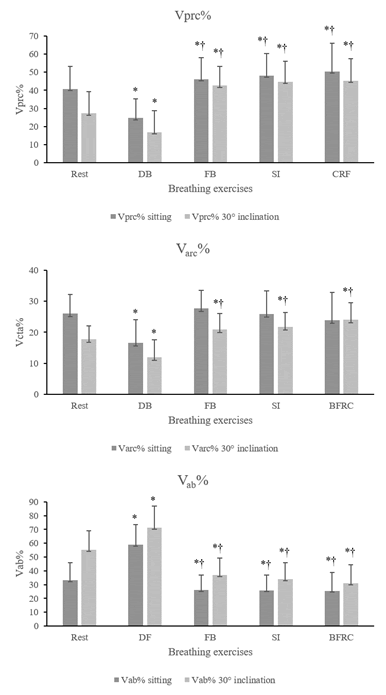
Percentage contribution of each compartment, at rest and during breathing exercises, in the sitting position and DD at 30° trunk inclination %Vprc was significantly lower in DB and higher than the other exercises in relation to at rest, in the sitting position and DD with 30° trunk inclination. DB showed a significantly lower %Vprc than that of the other exercises in the two positions studied (P<0.001).
With respect to %Varc, only DB showed a lower contribution from this compartment in relation to at rest, in the sitting position (P<0.001). For trunk inclination at 30°, DB exhibited a lower contribution (P<0.001) and the other exercises greater contribution from this compartment in relation to at rest (FB: P=0.011; SI: P=0.001; FRC: P<0.001).
In the sitting position, %Vab was higher in DB (P<0.001) and lower in the other exercises in relation to at rest, with P=0.004 in the FB exercise, P=0.002 in SI and P=0.012 in BFRC. The same occurred in the inclined position, highest in DE, with P<0.001 and lower in the others, with P<0.001. In the sitting position %Vab was significantly higher in DB when compared to the other exercises in both positions (P<0.001) (Figure 3).
Figure 3: Percentage contribution of each chest wall compartment. Data presented as mean (X) and standard deviation. %Vprc: percentage contribution of the pulmonary rib cage to tidal volume; %Varc: percentage contribution of the abdominal rib cage to tidal volume and %Vab: percentage contribution of the abdomen to tidal volume. *P<0.005 for at rest x × breathing exercises; † P<0.005 for diaphragmatic exercise × fractionated breaths, sniff inspirations and BFRC.
Comparison of volumes and the relative contribution between the sitting position and DD at 30° trunk inclination (Δ variation) in breathing exercises
For inclination at 30°, Vcw increased in most of the exercises, with P=0.004 in the FB exercise, >0.001 in SI and FRC, and P=0.418 for DB. Additionally, Vprc was higher for the BFRC and SI exercises, with P=0.001 and P=0.004, respectively, and lower in DB (P=0.001), with no difference for FB (P=0.696). In the same position, Varc decreased in DB (P=0.005) and FB (P<0.001) and increased in BFRC (P=0.001). However, there was no increase in Varc in SI (P=0.766) between the positions. Finally, Vab increased in the inclined position compared to its sitting counterpart in all the exercises (p<0.001), demonstrating a significant rise in the volume of this compartment at 30° trunk inclination, irrespective of the exercise performed. In relation to volume contribution between the positions, %Vprc showed no difference only in SI (P=0.053), while in the others, there was an increased contribution in DD at 30° trunk inclination, where P<0.001 in DB, 0.007 in FB and 0.013 in BFRC. The %Varc showed no difference only in BFRC (P=0.911), and declined in the other exercises, with P<0.001. Finally, %Vab rose in all the exercises, where DB, FB and SI obtained P<0.001 and FRC, P=0.002. DB obtained the highest increase in abdominal contribution in the sitting position (Table 2).
| Sitting | 30° inclination | |||||||
| Volume (L) | DB | FB | SI | BFRC | DB | FB | SI | BFRC |
| TD | 1.83 (1.02-2.11) | 2.48 (0.95-2.69) | 2.87 (1.09-3.06) | 2.49 (1.33-3.16) | 1.87 (0.88-2.73) | 2.7 (1.26-2.02)* | 3.41 (1.75-3.25)* | 3.12 (1.46-4.47)* |
| Vprc | 0.44 (0.23-0.7) | 1.13 (0.44-1.35) | 1.35 (0.52-1.62) | 1.21 (0.68-1.86) | 0.31 (0.31-0.87)* | 1.14 (0.46-1.53) | 1.52 (0.71-2.25)* | 1.39 (0.69-1.99)* |
| Varc | 0.31 (0.29-0.69) | 0.69 (0.43-0.9) | 0.77 (0.47-1.5) | 0.62 (0.46-1.45) | 0.22 (0.15-0.5)* | 0.57 (0.35-0.92)* | 0.76 (0.43-1.34) | 0.77 (0.41-1.69)* |
| Vab | 1.08 (0.71-1.8) | 0.66 (0.49-1.15) | 0.75 (0.56-1.43) | 0.65 (0.55-1.54) | 1.34 (0.83-2.58)* | 0.99 (0.54-1.34)* | 1.14 (0.73-1.77)* | 0.98 (0.91-1.9)* |
| Volume (%) | ||||||||
| Vprc | 24.6 (12.2-40.9) | 46.2 (19.9-43.9) | 48.2 (18.5-49.9) | 50.5 (17.7-62.9) | 16.9 (12.9-45)* | 43.6 (12.2-47.1)* | 44.8 (16.9-43.6) | 45.3 (14.7-52)* |
| Varc | 16.5 (9.8-32) | 27.6 (7.4-24.7) | 25.9 (8.8-36.4) | 23.9 (10.2-35.4) | 11.8 (8.3-21.8)* | 20.9 (6.3-22.7)* | 21.7 (5.9-22.7)* | 24.1 (5.5-29.5) |
| Vab | 58.9 (22.6-59.3) | 26.1 (12.7-43.5) | 25.8 (12.8-43.9) | 25.5 (14.9-59.5) | 71.3 (22.2 – 62.2)* | 36.8 (17.5-60.3)* | 33.9 (18.9-55.1)* | 30.9 (18.3-64.2)* |
| Data presented as mean (interquartile range). *<0.05: Significant difference between the positions adopted. | ||||||||
Table 2: Effects of position on the volumes of each thoracoabdominal cavity compartment.
Discussion
The following were the main findings of this study
(1) All the exercises caused an increase in chest wall volume and VE in both positions when compared to at rest, with DB exhibiting the smallest increase; DB, FB, and SI exercises showed a decline in f when compared to at rest in both positions
(2) DB was the main exercise to mobilize the abdominal region, represented by Vab in both positions
(3) FB, SI and BFRC exercises demonstrated greater mobility in the upper chest wall regions, represented by Vprc
(4) SI obtained the highest increase in Vcw in both positions
(5) All the exercises displayed an increased contribution of %Vab for trunk inclination at 30° and a decline in the contribution of %Vprc.
Breathing exercises are used to positively influence respiratory patterns, causing a rise in TV, and decrease in f, among others. As expected, all the exercises improved this pattern, except BFRC, which showed no decline in f, possibly due to the pattern of deep rapid inspirations during exercise [1,2,5,6]. Although most of the exercises exhibited a decline in f, VE rose in all the exercises, which could be justified by the significant increase in TV, compensating the decrease in f. Slow deep breathing improves the ventilation/perfusion ratio and increases trans pulmonary pressure, which contributes to recruiting possibly collapsed alveoli and raising overall alveolar recruitment [1,5]. DB obtained the highest increase in TV, and the predominant abdominal motion pattern imposed by this exercise limits complete expansion of the chest wall, unlike the other exercises, where there is no movement pattern, thereby approaching total lung capacity [8,9,20].
DB aims to improve ventilation in the lung bases, promoting greater diaphragmatic motion, ventilation in the abdominal region and predominance of abdominal motion. [3-5,7] In the present study, DB predominantly mobilized Vab in both positions studied, with 57.7% and 70.3% contribution of Vcw in the sitting and inclined positions, respectively. Thus, DB caused the distribution of air to be predominantly in the abdominal region, improving pulmonary ventilation in the bases, irrespective of the position used [5,22,23].
Other studies investigated the effect of DB on thoracoabdominal mobility, [3,5,7] and found that DB mobilizes the abdominal region more, but only one of these used analysis of three compartments, only possible with OEP [5]. Vieira et al. assessed thoracoabdominal mobility with OEP during DB, SI, maximal sustained and intercostal inspiration in healthy subjects, and found that %Vprc exhibited the lowest contribution compared to the other exercises studied and that %Vab was significantly higher in DB. Yamaguti et al. used respiratory inductance plethysmography, demonstrating an increase in abdominal and diaphragmatic mobility after DB in patients with CPOD. Rodrigues et al. [7] used thoracic cirtometry to analyze thoracoabdominal motion after a DB protocol performed in various positions, showing that the lower thoracic and abdominal regions increased during exercise and after the protocol. The results of the aforementioned articles corroborate the findings of the present study.
Among the methods used in the studies, thoracic cirtometry is a simple technique performed with a tape measure that quantifies thoracoabdominal mobility at three circumference levels (axillary, xyphoid and abdomen), but is not accurate in measuring lung volume [24,25]. Respiratory inductance plethysmography makes it possible to measure lung volume and quantify thoracoabdominal motion, but displacement is only measured in two thoracic regions (axillary and umbilical level) and volumetric variation is not assessed [26]. OEP evaluates thoracoabdominal motion and volume in three compartments, Vprc, Varc and Vab, as well as Vcw, providing greater details of thoracoabdominal mechanics compared to cirtometry and respiratory inductance plethysmography, in addition to being a more accurate instrument that also measures volumetric variation in different situations, [20,26]. This is why OEP was selected to assess participants in the present study.
The other exercises (FB, SI and BFRC) showed a predominance of Vprc, characterizing movement in the upper chest wall regions, unlike DB, with SI reaching the highest TV. This pattern may occur because during these exercises the subject is not asked to predominantly perform a movement in any chest wall region, in contrast to DB, where mobilization of the abdomen is prioritized. In addition, these exercises reach levels approaching total lung capacity, promoting a greater increase in TV when compared to DB. Thus, the movement pattern is distinct between them. [8,9] Vieira et al. also compared SI and other exercises with DB, obtaining a predominance of Vprc and a larger increase in Vcw in these exercises, when compared to DB. Trevisan, Soares and Rondinel [10] used respiratory inductance plethysmography and studied the effect of FB exercises after abdominal surgery, and also observed an improvement in thoracoabdominal mobility, increase in Vcw and greater mobility in the upper CW region.
FB, SI and BFRC achieved volumes near total lung capacity and are described in the literature as exercises that increase the expansion of lung bases [8,9]. In the present study, TD increased, not only in the basal regions. FB and BFRC exercises behave similarly in relation to compartment and CW volumes, primarily in the sitting position, since SI differs from the others, causing a greater increase in Vcw. As such, although the study was conducted in healthy subjects, not patients, we believe that these exercises should be applied in situations where a physiotherapist needs an overall increase in lung ventilation, with SI the most indicated to raise the TV of the chest wall.
In relation to the difference in volume and contribution of the volume of exercises when the position is changed from sitting to DD at 30° trunk inclination, all the exercises increased %Vab in the inclined compared to the sitting position and decreased %Vprc in the inclined position, with only BFRC not reducing %Varc.
Some studies that investigated the effect of changing position on thoracoabdominal mobility found a predominance of movement in the abdominal region in DD and upper thoracic movement when sitting or in the orthostatic position [1,27]. Romei et al. investigated thoracoabdominal mobility at 5 different trunk inclinations, from the sitting to the dorsal decubitus position. They corroborate the findings of the present study, where Vprc predominated in the sitting position, and Vab predominated as trunk inclination increased towards dorsal decubitus. Although this study investigated mobility during normal breathing, we demonstrated that this pattern of different distributions also occurs during breathing exercises [12].
Another study with healthy individuals during slow deep breathing also found that the rib cage moves more in the sitting position and the abdomen predominates in dorsal decubitus [13]. Priori et al. and Cavalcanti et al. studied changes in position, but in subjects with CPOD, and also found that the PRC predominates in the sitting position and the AB in the dorsal decubitus [14,28].
Irrespective of the intervention or population, position influences air distribution in the CW, and some studies report that these changes are due to the action of gravity, which provides greater displacement of the diaphragm muscles in the craniocaudal direction. The weight of viscera in this position displaces the diaphragm in the cephalic direction, decreasing the curvature radius of this muscle and providing a more favorable length tension ratio, improving mobility and causing greater abdominal region displacement. In the sitting position with no support, abdominal muscles need to contract, in order to maintain the position, and prevent the abdomen from moving as much as the rib cage [12,13,29].
Study limitations included operational difficulties, OEP errors during the recording of exercises and processing errors, resulting in some participants being excluded. Another limitation was not performing exercises in the dorsal decubitus position.
Conclusion
The results of the present study reveal the specific characteristics of each exercise and the important effects of position on chest wall movement, thereby contributing to better application of these exercises and position in physiotherapy practice. We investigated breathing exercises in healthy subjects. Nevertheless, the effects observed may be similar when implemented in subjects with respiratory disorders, but more research with these individuals is needed to confirm this hypothesis.
The results of this study demonstrate the effects of diaphragmatic and fractionated breathing, sniff inspirations and breathing from functional residual capacity on chest wall movement and volume, and may contribute to their proper use in clinical practice, alone or associated with the sitting position and trunk inclination at 30°. The aforementioned exercises promoted an increase in tidal volume and minute volume, and only the BFRC exercise did not cause a decline in respiratory rate. Position influenced volumes and the contribution of CW compartment volume in diaphragmatic and fractionated breathing, sniff inspirations and BFRC.
References
- Feltrim MIZ, Jardim JRB (2004) Thoracoabdominal movement and breathing exercises: literature review. Rev Fisioter Univ São Paulo 11(2):105-13.
- Holland AE, Hill CJ, Jones AY, McDonald CF (2012) Breathing exercises for chronic obstructive pulmonary disease. Cochrane Database of Systematic Reviews Issue 10. Art. No.: CD008250.
- Tomich GM, França DC, Diniz MTC, Britto RR, Sampaio RF, et al (2010) Effects of breathing exercises on breathing pattern and thoracoabdominal motion after gastroplasty. J Bras Pneumol 36(2):197-204.
- Yamaguti WP, Claudino RC, Neto AP, Chammas MC, Gomes AC, et al. (2012) Diaphragmatic breathing training program improves abdominal motion during natural breathing in patients with chronic obstructive pulmonary disease: A randomized controlled trial. Archives of Physical Medicine and Rehabilitation 93(4):571-577.
- Vieira DSR, Mendes LPS, Elmiro NS, Velloso M, Britto RR, et al. (2014) Breathing exercises: influence on breathing pattern and thoracoabdominal motion in healthy subjects. Braz J Phys Ther 18(6):544-552.
- Borge CR, Hagen KB, Mengshoel AM, Omenaas E, Moum T, et al (2014) Effects of controlled breathing exercises and respiratory muscle training in people with chronic obstructive pulmonary disease: Results from evaluating the quality of evidence in systematic reviews. BMC Pulm Med 14(1):184.
- Rodrigues CP, Alves LA, Matsuo T, Gonçalves CG, Hayashi D (2012) The effect of an exercise program directed to the thoracic mobility in COPD. Fisioter Mov 25(2):343-349.
- Cuello GA, Masciantonio L, Cuello AF (1982) Patrones respiratorios en distintas afecciones. Corde 3:48-60.
- Azeredo (2002) Fisioterapia respiratória moderna, 4th edition. São Paulo: Manole 361-367.
- Trevisan ME, Soares JC, Rondinel TZ (2010) Efeitos de duas técnicas de incentivo respiratório na mobilidade toracoabdominal após cirurgia abdominal alta. Fisioterapia e Pesquisa 17(4)322-326.
- Gastaldi AC, Magalhães CMB, Baraúna MA, Silva EMC, Souza HCD (2008) Benefits of postoperative respiratory kinesiotherapy following laparoscopic cholecystectomy. Rev Bras Fisioter 12(2):100-106.
- Romei M, Mauro AL, D’angelo MG, Turconi AC, Bresolin N, et al (2010) Effects of gender and posture on thoraco-abdominal kinematics during quiet breathing in healthy adults. Respiratory Physiology and Neurobiology 172(3):184-191.
- Kaneko H, Horie J (2012) Breathing movements of the chest and abdominal wall in healthy subjects. Respiratory Care 57(9):1442-1451.
- Cavalcanti AGL, Rattes lima CSF, Barros SR, Reinaux CMA, Braz JDS, et al. (2014) Influence of posture on the ventilatory pattern and the thoraco-abdominal kinematics of patients with chronic obstructive pulmonary disease (COPD). Physiotherapy Theory and Practice 30(7):490-494.
- WHO-World Health Organization. Global database on body mass index: an interactive surveillance tool for monitoring nutrition transition. Geneva. Geneva: WHO 2004. American Thoracic Society/European Respiratory Society, 2002.
- Miller MR, Hankinson J, Brusasco V (2005) Standardisation of spirometry. Eur Respir J 26(2):319-338.
- Aliverti A, Rodger K, Dellacà RL, Stevenson N, Lo Mauro A, et al (2005) Effect of salbutamol on lung function and chest wall volumes at rest and during exercise in COPD. Thorax 60(11):916-924.
- Aliverti A, Dellacà R, Pelosi P, Chiumello D, Gattinoni L, et al (2001). Compartmental analysis of breathing in the supine and prone positions by optoelectronic plethysmography. Annals of Biomedical Engineering 29(1):60-70.
- Cala SJ, Kenyon CM, Ferrigno G, Carnevali P, Aliverti A, et al. (1996) Chest wall and lung volume estimation by optical reflectance motion analysis. J app physio 81(6):2680-2689.
- Chinali C, Busatto HG, Mortari DM, Rockenbach CWF, Leguisamo CP (2009) Inspirometria de incentivo orientada a fluxo e padrões ventilatórios em pacientes submetidos a cirurgia abdominal alta. ConScientiae Saúde 8(2):203-210.
- Portney LG, Watkins MP. (2008) Foundations of clinical research: Applications to practice. 3rd ed. New Jersey: Pearson Prentice Hall.
- Cahalin LP, Braga M, Matsuo Y, Hernandez ED (2002) Efficacy of diaphragmatic breathing in persons with chronic obstructive pulmonary disease: A review of the literature. J Cardiopulm Rehab 22(1):7-21.
- Caldeira VS, Staling CCD, Britto RR, Martins JÁ, Sampaio RF, et al (2007). Reliability and accuracy of cirtometry in healthy adults. J Bras Pneumol 33(5):519-526.
- Pedrini A, Gonçalves MA, Leal BE, Yamaguti WPS, Paulin E (2013) Comparação entre as medidas de cirtometria tóraco-abdominal realizadas em decúbito dorsal e em ortostatismo. Fisioter Pesq 20(4):373-378.
- Pereira MC, Porras DC, Lunardi AC, Silva CCBM, Barbosa RCC, et al. (2017) Thoracoabdominal asynchrony: Two methods in healthy, COPD, and interstitial lung disease patients. PLoS One 12(8):1-15.
- Konno K, Mead J (1967) Measurement of the separate volume changes of rib cage and abdomen during breathing. J Appl Physiol 22(3):407-422.
- Priori R, Aliverti A, Albuquerque AL, Quaranta M, Albert P, et al (2013). The effect of posture on asynchronous chest wall movement in COPD. J Appl Physiol 114(8):1066-1075.
- Yamaguti WP, Paulin E, Shibao S, Kodaira S, Chammas MC, et al (2007) Avaliação ultra-sonográfica da mobilidade do diafragma em diferentes posturas em sujeitos saudáveis. J Bras Pneumol 33(4):407-413.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi