Research Article, Arch Med Biotechnol Vol: 1 Issue: 2
Inhibitory Action of Essential Oils on Quorum Sensing Activity of Pseudomonas Aeruginosa by Effecting on Pyocyanin Production
Ali Abdul Hussein S AL Janabi*
Department of Microbiology, College of Medicine, University of Karbala, Karbala, Iraq
*Corresponding Author : Ali Abdul Hussein S AL-Janabi
Department of Microbiology, College of Medicine, University of Karbala, Karbala, Iraq
E-mail: aljanabi_bio@yahoo.com
Received: May 22, 2018 Accepted: June 12, 2018 Published: June 19, 2018
Citation: Janabi AAHSAL (2018) Inhibitory Action of Essential Oils on Quorum Sensing Activity of Pseudomonas aeruginosa by Effecting on Pyocyanin Production. Arch Med Biotechnol 1:2.
Abstract
- Background: Quorum sensing (QS) is considered an important behavior of bacterial cells that communicate with each other. Pyocyanin produced by Pseudomonas aeruginosa is one QS activity. Its synthesis can affect in the presence of essential oils.
- Methods: Four types of essential oils of hermal seeds, black seeds, radish, and linseeds were used against the growth and pyocyanin production of P. aeruginosa.
- Results: All four essential oils showed antibacterial activity against aeruginosa with a variable MIC value. In comparison with hermal oil, pyocyanin production was more inhibited by the other types of essential oils.
- Conclusions: Essential oils of radish and linseeds have more antibacterial action against aeruginosa and its production of pyocyanin. Hermal oil has no effect on the QS pathway as indicated by pyocyanin production with less effect on bacterial growth.
Keywords: Pyocyanin; Radish; Linseeds; Hermal; Black seeds; Qs
Introduction
Pyocyanin is one of extracellular pigments produced mainly by clinical and environmental isolates of P. aeruginosa [1,2]. It plays an important role as a virulence factor during pathogenicity of this bacteria [3] and as an antimicrobial agent against several types of other bacteria and fungi [4,5]. Regulation of pyocyanin production is usually under the control of quorum sensing (QS) activity that has an important role in bacterial communication through several signal molecules [6]. Synthesis of pyocyanin is mainly encoded by phzM and phzS genes of phz operons through regulation of transcriptional activity of MvfR gene [3,7].
Essential oils of different plants are recorded to have an ability to inhibit pyocyanin synthesis through an effect on QS. Ferula oil prevents QS of P. aeruginosa from producing pyocyanin and other products at 25 μg/ml [3,8]. Lower concentration of other oils could also perform such type of inhibition as with cinnamon oil at 0.1-0.2 μl/ml [9]. Meanwhile, reduction of pyocyanin expression needs a higher concentration (15 mg/ml) of Ocimum gratissimum leaf oil [10].
QS activity of P. aeruginosa represented by pyocyanin production was investigated after treatment with different types of essential oils.
Materials and Methods
Bacteria isolate
Pseudomonas aeruginosa was isolated from male (44 years) with urinary tract infection while admitted in Al-Ammam Al-Hussein Medical city of Karbala in January 2017. The isolate was cultured on Mueller Hinton agar (MHA) (Himedia, India) for 24 h at 37°C. Pyocyanin with blue color was clearly observed in bacterial culture media. P. aeruginosa was diagnosed based on biochemical characters of API 20 E (BioMérieux, France).
Essential oils
Essential oils of hermal seeds (Peganum harmala L.), black seeds (Nigella sativa L.), radish (Raphanus sativus L.), and linseeds or flax (Linum usitatissimum L.) were purchased from Hemeni-Karachi, Pakistan.
Antibacterial assay
The isolate was cultured in Luria-Bertani (LB) medium for 24 h at 37°C. Cell density of bacteria was adjusted to 1.5 × 108 cells in each ml of sterilized normal saline after matching with the 0.5 McFarland standard [11]. Different concentrations of essential oils (1, 3, 5, and 10 μl/ml) were prepared by mixing with ethanol. Well-diffusion method was used for antibacterial assay [12]. A 0.1 ml of standard bacteria was spread on MHA plate and 6 mm wells were made by sterile cork borer. Four wells were made in a single inoculated plate. In each well, 100 μl of essential oil concentration was added. Media with either of an essential oil or distilled water were used as a negative control. Gentamicin dissolved in sterile D.W (10 μg/ml) was used as a positive control. Cultures were incubated for 24 h at 37°C and inhibition zones were measured.
Determination of minimum inhibitory concentration (MIC)
Determination of MIC was performed according to NCCLS method, M7-A5 [13]. A 100 μl of standard bacterial suspension was inoculated into each single well of microtiter plate (96 wells). Serial two-fold concentrations of essential oil were prepared and 100 μl of each one was added into inoculated wells. For each test plate, two essential oil-free controls were included, one with medium alone and the other with medium inoculated with bacteria. Plates were incubated at 37°C and read visually after 24 h of incubation.
Pyocyanin determination
A vial with 2 ml of LB medium and 100 μl of essential oil concentration was inoculated with 100 μl of standardized bacteria. Cultures were incubated for 48 h at 37°C. Pyocyanin concentration was measured according to protocol mentioned by Kalia et al. [9]. Briefly, supernatants from P. aeruginosa culture grown in the presence or absence of essential oil were collected. Pyocyanin was extracted using chloroform, followed by 0.2 M HCl. The absorption was measured by a UV-spectrophotometer (ABEL, Japan) at 520 nm.
Statistical analysis
All of the experiments were triplicated. The data were analyzed statistically by analysis of variance (ANOVA) test. The minimum level of (p) value was <0.01 which was considered significant.
Results
The effect of essential oils on the growth and QS activity of P. aeruginosa as indicated by pyocyanin production was investigated. Bacterial growth was inhibited at concentration up to 3 μl/ml of three tested essential oils. Meanwhile, hermal oil showed no activity against P. aeruginosa up to 10 μl/ml (Table 1). The MIC value of essential oils against P. aeruginosa were 7.5, 4.5, 4, and 3.5 μl/ml for hermal, black seeds, radish, and linseed oils, respectively (Table 2).
| Essential oils | Zone of inhibition (mm) | |||
|---|---|---|---|---|
| Concentrations (µl/ml) | ||||
| 1 | 3 | 5 | 10 | |
| Hermal oil | - | - | - | 12 |
| Black seeds oil | 21 | 18 | 12 | 11 |
| Radish oil | 20 | 19 | 12 | 11 |
| Linseed oil | 14 | 12 | 10 | 10 |
| Gentamicin (10 µg/ml) | 19 | |||
Table 1: Zone of inhibition of essential oils on Pseudomonas aeruginosa growth.
| Essential oils | MIC (µl/ml) |
|---|---|
| Hermal oil | 7.5 |
| Black seeds oil | 4.5 |
| Radish oil | 4 |
| Linseed oil | 3.5 |
Table 2: MIC value of essential oils on Pseudomonas aeruginosa growth.
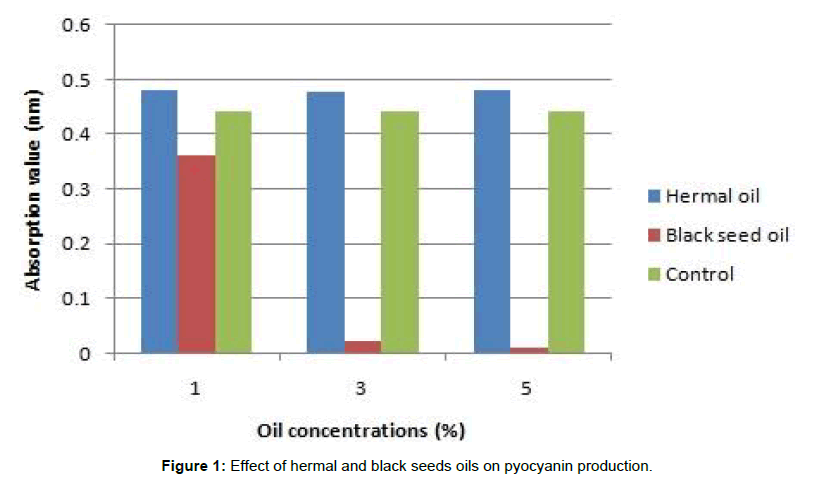
Pyocyanin production was completely inhibited at 1 μl/ml of radish and linseed oils, while hermal oil exhibited no effect on production of pyocyanin at 1 and 3 μl/ml. Black seeds oil showed initial reduction of pyocyanin production at 1 μl/ml until complete inhibition of pigment production at higher concentration (3 μl/ml) (Table 3 and Figure 1).
| Essential oils | Concentrations (µl/ml) | |
|---|---|---|
| 1 | 3 | |
| Hermal oil | +++ | +++ |
| Black seeds oil | + | - |
| Radish oil | - | - |
| Linseed oil | - | - |
Table 3: Production of pyocyanin in media with essential oils.
Discussion
Quorum sensing (QS) is a communication path connecting microbial cells with each other by special signals in order to coordinate motility, virulence factor production, and biofilm formation [6]. In P. aeruginosa, QS stimulates production of several types of pigments as virulence factors such as pyocyanin and pyovardine [8]. Pyocyanin is a blue extracellular-pigment that is synthesised under the control of phz operons and MvfR gene through the synthesis of QS quinolone molecules [3,7]. About 25% of clinical and 38.2% of environmental isolates of P. aeruginosa have the ability to produce pyocyanin, while 3.2% of these are non-pigmented [2]. Antimicrobial activity is the first function of pyocyanin against a wide range of microorganisms giving P. aeruginosa a competitive advantage over other organisms [4,5]. The second function of pyocyanin is represented by its role as a virulence factor through increased intracellular levels of reactive oxygen species (ROS) [14].
QS was found to be affected by many natural essential oils. Pyocyanin production as one of QS activities is also reduced by some of these essential oils such as with the oils of Ferula, Cinnamon, pungent (6-gingerol), Ocimum gratissimum, peppermint and clove [3,8-10,15-17].
In the present study, radish, linseeds, and black seeds oils exhibited an inhibitory action on pyocyanin production. Antibacterial activity of different parts of the radish plant against P. aeruginosa was recorded by several studies [18-20]. This inhibitory activity may relate to the plant contents of alkaloids, flavonoids, glycosides, phenols, tannins, saponin, sterols and proteins [19,20]. Essential oil of radish that blocked QS activity as indicated by inhibition of pyocyanin production is mainly composed of pentyl hexyl, 4-methylpentyl isothiocyanate, dimethyl disulfide, methyl methanethiosulfinate and 1-methylthio-3-pentanone [18].
Linseed oil showed antibacterial effect on P. aeruginosa with prevention of QS activity through inhibition of pyocyanin production. Antibacterial activity of linseed oil recorded by previous studies was variable [21]. Gawad et al. found a negative effect of linseed oil on bacteria of lumen, while other studies found a good activity of linseed oil against some bacteria [22,23]. P. aeruginosa was less affected by linseed oil proteins [24]. However, linseed oil contains several compounds that have a potential activity against microorganisms such as fats, flavonoids, glycosides, phenols and tannins [25]. Semicarbazide fatty acid and phenylpropanoids of linseed oil exhibited an inhibitory effect against many types of bacteria [26,27].
Black seeds oil was found to have antibacterial activity against different types of bacteria and fungi [28-33]. P. aeruginosa and its multi-drug resistant isolates revealed more sensitivity to black seeds oil which has a bacteriostatic action [28,30,31,33-36]. Essential oil of black seeds is composed of large amounts of fixed oil and less of volatile oil [32]. Generally, the main chemical components of black seed oil, which are related to its antibacterial activity are thymoquinone, thymohydroquinone, dithymoquinone, thymol, carvacrol, nigelline-N-oxide, nigellicine, nigellidine and alphahederin, oleic acid, monoterpenes, sesquiterpenes, polyphenols and aromatic aldehydes [30].
Hermal oil revealed no activity against the growth of P. aeruginosa or its production of pyocyanin, even at high concentrations. These results were also proved by another study [37]. Extraction of Peganum harmala seeds exhibited antibacterial effects on various bacteria, including P. aeruginosa [38-40]. The MIC value of seeds extract against P. aeruginosa ranged from a low value (15-50 μg/ml) to a high value (50-400 mg/ml) [38,39,41]. However, hermal oil contains many components with a potential antibacterial activity such as oxygenated monoterpenes and sesquiterpenes [41].
Conclusion
Essential oils of radish and linseeds have antibacterial action against the growth of P. aeruginosa and its production of pyocyanin. Hermal oil has no effect on pyocyanin production with less effect on bacterial growth.
Funding and Conflict of Interesting
There is no conflict of interest and funding for this manuscript.
References
- El-Fouly MZ, Sharaf AM, Shahin AAM, EI-Bialy HA, Omara AMA (2015) Biosynthesis of pyocyanin pigment by Pseudomonas aeruginosa. J Radi Res Applied Sci 8: 36-48.
- Maroui I, Aboulkacem A, Timinouni M, Belhaj A (2016) Virulence profiles of clinical and environmental Pseudomonas aeruginosa isolates from central Morocco. Afri J Microbiol Res 10: 473-480.
- Khambhala P, Verma S, Joshi S, Seshadri S, Kothari V (2016) Inhibition of bacterial quorum-sensing by Ferula asafetida essential oil. Adv Genet Eng 5: 1-2.
- Parvin W, Othman R, Jaafar H, Wong M (2016) Detection of phenazines from UPMP3 strain of Pseudomonas aeruginosa and its antagonistic effects against Ganoderma boninense. Int J Agric Biol 18: 483-487.
- Gharieb MM, Sheekh EIMM, Sabbagh EISM, Hamza WT (2013) Efficacy of pyocyanin produced by Pseudomonas aeruginosa as a topical treatment of infected skin of rabbits. BTAIJ 7: 184-193.
- Rasamiravaka T, Labtani Q, Duez P, EI Jaziri M (2015) The formation of biofilms by Pseudomonas aeruginosa: A review of the natural and synthetic compounds interfering with control mechanisms. BioMed Research International. Hindawi Publishing Corporation 1-17.
- Nowroozi J, Sepahi AA, Rashnonejad A (2012) Pyocyanin biosynthesis genes in clinical and environmental isolates of Pseudomonas aeruginosa and detection of pyocyanin s antimicrobial effects with or without colloidal silver nanoparticles. Cell J 14: 7-18.
- Sepahi E, Tarighi S, Ahmadi FS, Bagheri A (2015) Inhibition of quorum sensing in Pseudomonas aeruginosa by two herbal essential oils from Apiaceae family. J Microbiol 53: 176-180.
- Kalia M, Yadav VK, Singh PK, Sharma D, Pandey H, et al. (2015) Effect of cinnamon oil on quorum sensing-controlled virulence factors and biofilm formation in Pseudomonas aeruginosa. PLOS ONE 1-18.
- Iwalokun BA, Owuh IG, Ronke SA (2004) Effect of essential leaf oil of Ocimum gratissimum on clinical isolates of Pseudomonas aeruginosa. Afri J Clin Experi Microbiol 5: 265-271.
- Brown AE (2012) Bensons microbiological applications: laboratory manual in general microbiology complete version. (12 edtn), McGraw-Hill International edition. USA
- Jahangirian H, Haron J, Ismail MH, Moghaddam RR, Hejri AL, et al. (2013) Well diffusion method for evaluation of antibacterial activity of copper phenyl fatty hydroxamate synthesized from canola and palm kernel oils. Digest J Nanomater Biostruc 8: 1263-1270.
- Clinical and Laboratory Standards Institute (CLSI) (2003) Methods for Dilution Antimicrobial Susceptibility Tests for Bacteria that Grow Aerobically, (6th edtn), National Committee for Clinical Laboratory Standards, USA.
- Hall S, McDermott C, Dukie AS, McFarland AJ, Forbes A, et al. (2016) Cellular effects of pyocyanin, a secreted virulence factor of Pseudomonas aeruginosa. Toxins 8: 1-14.
- Kim H, Lee S, Byun Y, Park H (2015) 6-gingerol reduces Pseudomonas aeruginosa biofilm formation and virulence via quorum sensing inhibition. Sci Rep 5: 1-11.
- Husain FM, Ahmad I, Khan MS, Ahmad E, Tahseen Q, et al. (2015) Sub-MICs of Mentha piperita essential oil and menthol inhibits AHL mediated quorum sensing and biofilm of Gram-negative bacteria. Frontiers Microbiol 6: 1-12.
- Husain FM, Ahmad I, Asif M, Tahseen Q (2013) Influence of clove oil on certain quorum-sensing-regulated functions and biofilm of Pseudomonas aeruginosa and Aeromonas hydrophila. J Biosci 38: 835-844.
- Gutiérrez RM, Perez RL (2004) Raphanus sativus (Radish): Their chemistry and biology. Scientific World J 4: 811-837.
- Ahmad F, Hasan I, Chishi DK, Ahmad H (2012) Antibacterial activity of Raphanus sativus Linn. Seed extract. Global J Medi Res 12: 24-34.
- Janjua S, Shahid M, Abbas F (2013) Phytochemical analysis and in vitro antibacterial activity of root peel extract of Raphanus sativus L. var niger. Advan Medi Plant Res 1: 1-7.
- Mathkhury HJF, Dhamin AS, Taie KL (2016) Antibacterial and antibiofilm activity of Flaxseed oil. Iraqi J Sci 57: 1086-1095.
- Gawad RM, Kattab HM, Strabel M, Nor Abo EISA, Cieślak A, et al. (2015) Effect of different level from Linseed oil and Linseed oil beads on rumen fermentation and microbial parameters using gas production system and rumen simulation technique. Asian J Anim Vet Adv 10: 97-118.
- Khan BA, Warner P, Wang H (2014) Antibacterial properties of hemp and other natural fiber plants: a review. Bio Resources 9: 3642-3659.
- Tehrani MHH, Batal R, Kamalinejad M, Mahbubi A (2014) Extraction and purification of flaxseed proteins and studying their antibacterial activities. J Plant Sci 2: 70-76.
- Joshi Y, Garg R, Juyal D (2014) Evaluation of synergistic antimicrobial activity of gemifloxacin with Linum usitatissimum seed oil. J Phytopharmacology 3: 384-388.
- Borhade S (2014) Synthesis, characterization and antimicrobial activity of essential fatty acid of semicarbazide. Int J Chem Sci App 5: 46-55.
- Czemplik M, Żuk M, Kulma A, Kuc S, Szopa J (2011) GM flax as a source of effective antimicrobial compounds. FORMATEX 1216-1224.
- Ismaeil AS (2011) Effect of Black seed alkaloids against some pathogenic bacteria. Raf J Sci 22: 9-16.
- Adam ME (2013) Antimicrobial activity of bee honey, black cumin oil and green tea against multi-drug resistant pathogenic bacteria. Int J Curr Microbiol Appl Sci 2: 58-63.
- Aljabre SHM, Alakloby OM, Randhawa MA (2015) Dermatological effects of Nigella sativa. J Dermatol Dermatol Surg 19: 92-98.
- Maryam A, Fatimah A, Ebtesam A, Abdulrahman A, Ineta BL (2016) In-vitro studies on the effect of Nigella sativa Linn., seed oil extract on multidrug resistant Gram positive and Gram negative bacteria. J Medicinal Plants Studies 4: 195-199.
- El-Tahir KE, Bakeet DM (2006) The black seed Nigella sativa Linnaeus-A mine for multi cures: a plea for urgent clinical evaluation of its volatile oil. JTU Med Sci 1: 1-19.
- Hasan NA, Nawahwi MZ, Ab Malek H (2013) Antimicrobial activity of Nigella sativa seed extract. Sains Malaysiana 42: 143-147.
- Delaimi AMS (2012) Antimicrobial activity of black seed oil & water extracts on multidrug resistant Pseudomonas aeruginosa. J University of Anbar for Pure Sci 6: 1-8.
- Shouny EWA, Magaam S (2009) Sensitivity of multi-drug resistant Pseudomonas aeruginosa isolated from surgical wound-infections to essential oils and plant extracts. World J Med Sci 4: 104-111.
- Amina B, Rachida A (2013) Molecular composition and antibacterial effect of essential oil of Nigella sativa. African J Biotechnol 12: 3006-3012.
- Kamel GM, Ezz eldeen NA, Mishad EMY, Ezzat RF (2011) Susceptibility pattern of Pseudomonas aeruginosa against antimicrobial agents and some plant extracts with focus on its prevalence in different sources. Global Veterinaria 6: 61-72.
- Jahani S, Saeidi S, Javadian F, Akbarizadeh Z, Sobhanizade A (2016) Investigating the antibacterial effects of plant extracts on Pseudomonas aeruginosa and Escherichia coli. Int J Infect 3: 1-4.
- Darabpour E, Bavi AP, Motamedi H, Nejad SM (2011) Antibacterial activity of different parts of Peganum harmala L. growing in Iran against multi-drug resistant bacteria. EXCLI J 10: 252-263.
- Fatma B, Fatiha M, El attafia B, Noureddine D (2016) Phytochemical and antimicrobial study of the seeds and leaves of Peganum harmala L. against urinary tract infection pathogens. Asian Pacific J Tropical Disease 6: 822-826.
- Apostolico I, Aliberti L, Caputo L, De Feo V, Fratianni F, et al. (2016) Chemical composition, antibacterial and phytotoxic activities of Peganum harmal seed essential oils from five different localities in Northern Africa. Molecules 21: 2-13.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi