Research Article, J Regen Med Vol: 7 Issue: 1
Interferon Regulatory Factor-2 Regulates Hematopoietic Stem Cells
Atsuko Masumi*
Department of Safety Research on Blood and Biological Products, National Institute of Infectious Diseases, Aomori 030-0943, Japan
*Corresponding Author : Atsuko Masumi
Department of Molecular pharmacology, Faculty of Pharmaceutical Sciences, Aormori University, 2-3- 1, Kobata, Aomori-shi, Aomori 030-0943, Japan
Tel: +81 177382001
E-mail: amasumi@aomori-u.ac.jp
Received: September 10, 2018 Accepted: October 12, 2018 Published: October 18, 2018
Citation: Masumi A (2018) Interferon Regulatory Factor-2 Regulates Hematopoietic Stem Cells. J Regen Med 7:1. doi: 10.4172/2325-9620.1000143
Abstract
Objective: Interferon regulatory factor-2 (IRF-2) is highly expressed in hematopoietic stem cells (HSCs) in mouse bone marrow cells. The population of bone marrow Lin-c-Kit+Sca-1+(KSL) cells is increased in IRF-2-/- mice; however, there are less hematopoietic stem cells due to a reduction of the CD150+ population and type I interferon (IFN) signaling. We studied how HSCs lacking IRF-2 differentiate in bone marrow and peripheral blood.
Methods: To study the function of IRF-2 in HSCs, bone marrow cells from IRF-2-/- mice were transplanted into wild-type mice.
Results: Lineage cell population analysis showed that the B220 and CD19 populations from IRF-2-/- mouse bone marrow were enhanced in recipient mice. Conclusion: Bone marrow cells from IRF-2-/- mice can survive by lineage cell re-population. IRF-2 is important for the long-term repopulation of HSCs through not only the type I IFN response but also via the regulation of lineage-specific cell populations.
Keywords: Interferon regulatory factor-2; Hematopoietic stem cells; Bone marrow; Noncompetitive reconstitution; Lineage cells; Transplantation; Chimerism
Abbreviations
HSCs: Hematopoietic Stem Cells; IFN: Interferon; IRF: Interferon Regulatory Factor; KO: Knockout; KSL: Lin- c-Kit+ Sca-1+; SLAM: Signaling Lymphocytic Activation Molecule; WT: Wild Type.
Introduction
Hematopoiesis is regulated by intrinsic gene regulatory networks, ensuring the rapid production of differentiated blood cells for the immediate generation of definitive hematopoietic stem cells (HSCs). HSCs are thought to be quiescent and are referred to as long-term reconstituting HSCs or dormant HSCs [1-4]. Under certain conditions, such as an infection, HSCs differentiate into progenitor cells with a reduced capacity for self-renewal, and they can be stimulated to differentiate into all cell types found in peripheral blood. Under an inflammatory state, such as during a bacterial or viral infection, an apparent expansion of Lin- c-Kit+ Sca-1+ (KSL) cells has been observed, but the HSCs in the increased KSL population have reduced activity [5,6].
Interferon regulatory factor-2 (IRF-2), a negative regulator of the interferon (IFN) response, is highly expressed in mouse HSCs (KSL) [7]. Previously, an enhanced population of Sca-1+ cells and reduced HSC activity in the KSL fraction was detected in bone marrow cells of IRF-2-/- mice [8]. The HSC abnormalities observed in IRF-2-/- mice have been demonstrated to be due to increased type I IFN signaling [9]. In IRF-2-/- mice, the reduced expression of CD150 and enhanced expression of Sca-1 have been detected. In IRF-2-/- IFNAR-/- double knockout (KO) mice, which lack IFN signaling, the expression of CD150 was comparable with that observed in wild-type (WT) mice [8]. However, the activity of HSCs in IRF-2-/- IFN-/- double KO mice was not comparable with that in WT mice, suggesting that IRF-2 partly regulates HSC markers directly [9]. We suggested that HSC-like cells, which may be isolated using cell surface markers distinct from those of KSL cells, are present in the bone marrow cells of IRF-2-/- mice [8].
In the present study, we investigated how HSCs derived from IRF-2-/- mice differentiate into peripheral blood cells.
Experimental
Mice
IRF-2-/- mice were obtained from Professor T. Mak (Amgen Institute, Toronto, Canada) [10]. All mice were maintained in the animal facility of the National Institute of Infectious Diseases under specific pathogen-free conditions and used at 8-20 weeks of age according to the institutional guidelines for animal experimentation.
Antibodies and cytokines
Antibodies against rat IgG2b anti-mouse lineage markers (FITCCD45.2, APC-CD45.1, APC-c-Kit, FITC-Sca-1, FITC-CD48, APCCD150, and APC780-CD45.2) were purchased from eBioscience (San Diego, CA).
Isolation of lineage cells and HSCs
Mouse HSCs were purified from bone marrow cells of 8–20-weekold C57BL/6 strain mice. In brief, low-density cells were isolated on Histopaque (Sigma, St. Louis, MO). The cells were stained with an antibody cocktail consisting of PE-conjugated anti-Gr1, anti-Mac1, anti-B220, anti-CD4, and anti-CD8 monoclonal antibodies (BD Biosciences, San Jose, CA). Lineage-positive cells were depleted with anti-PE microbeads (Miltenyi Biotec, Bergisch Gladbach, Germany). The remaining lineage-depleted cells were further stained with APC780-CD45.2, FITC-Sca-1, or FITC-CD48 and APC-c-Kit or APC-CD150 antibodies for CD45.2-KSL or CD45.2-signaling lymphocytic activation molecule (SLAM) population analysis. Color analysis and sorting were performed using flow cytometry (JSAN Bay Bioscience, Kobe, Japan).
Noncompetitive reconstitution and chimerism analysis
Noncompetitive bone marrow transplants were performed by the intravenous injection of CD45.2 donor whole bone marrow cells (1.0 × 107, 2.0 × 106, or 1.0 × 105 cells) from either 8–12-week-old IRF-2 KO or WT controls (C57BL/6) into CD45.1 WT C57BL/6 recipients that had been lethally irradiated with a dose of 9.5 Gy. After transplantation, peripheral blood cells or bone marrow cells were isolated and chimerism was analyzed by staining with FITC-CD45.1 and APC-CD45.2 antibodies.
Results
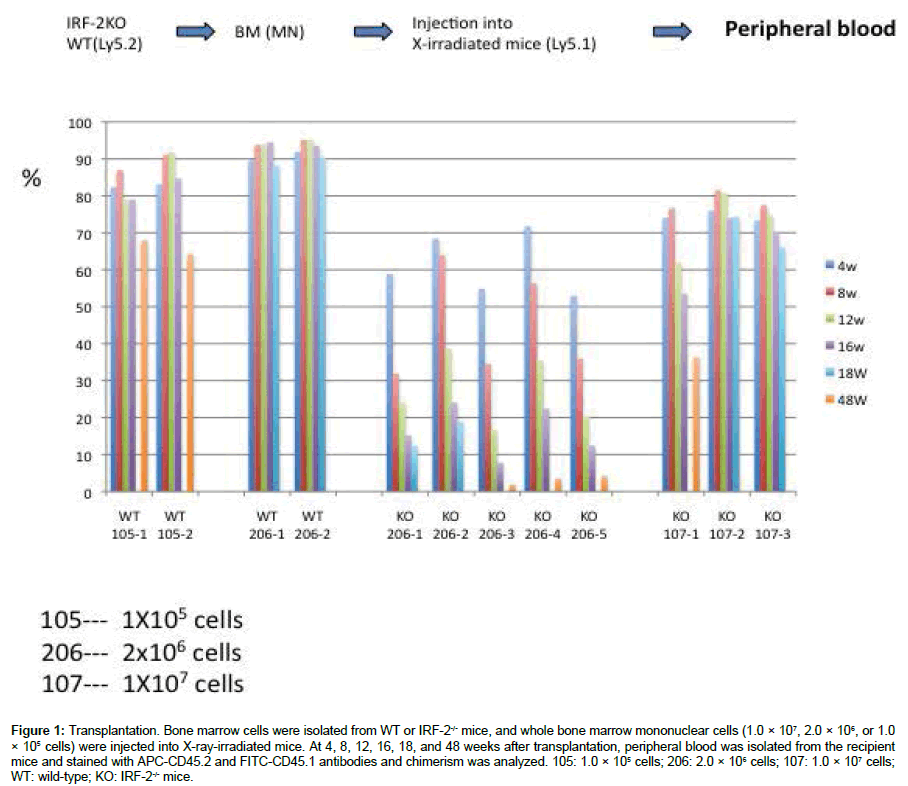
To examine how bone marrow HSCs from IRF-2-/- mice differentiate, whole bone marrow cells were injected into recipient mice and noncompetitive transplant assays were performed. Transplantation of 1.0 × 105 WT bone marrow cells is normally sufficient to rescue and fully repopulate the hematopoietic system of lethally irradiated recipients. However, this dose of cells from IRF-2 KO donors failed to rescue any recipients from lethal irradiation (Figure 1), indicating the impaired self-renewal of HSCs in whole bone marrow cells derived from IRF-2-/- mice. A dose of 2.0 × 106 cells from IRF-2-/- mice rescued the recipients from lethal irradiation, although the engraftment efficacy was poorer than that of WT cells at 1 month after transplantation (Figure 1). A dose of 1.0 × 107 cells is normally sufficient to rescue and fully repopulate the hematopoietic system of some lethally irradiated recipients.
Figure 1: Transplantation. Bone marrow cells were isolated from WT or IRF-2-/- mice, and whole bone marrow mononuclear cells (1.0 × 107, 2.0 × 106, or 1.0 × 105 cells) were injected into X-ray-irradiated mice. At 4, 8, 12, 16, 18, and 48 weeks after transplantation, peripheral blood was isolated from the recipient mice and stained with APC-CD45.2 and FITC-CD45.1 antibodies and chimerism was analyzed. 105: 1.0 × 105 cells; 206: 2.0 × 106 cells; 107: 1.0 × 107 cells; WT: wild-type; KO: IRF-2-/- mice.
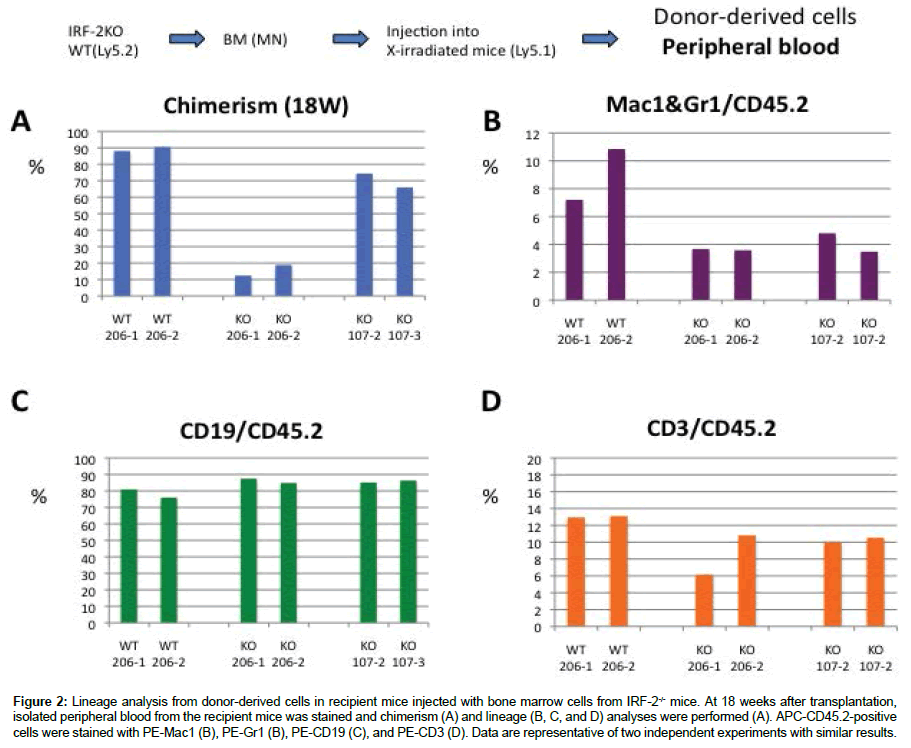
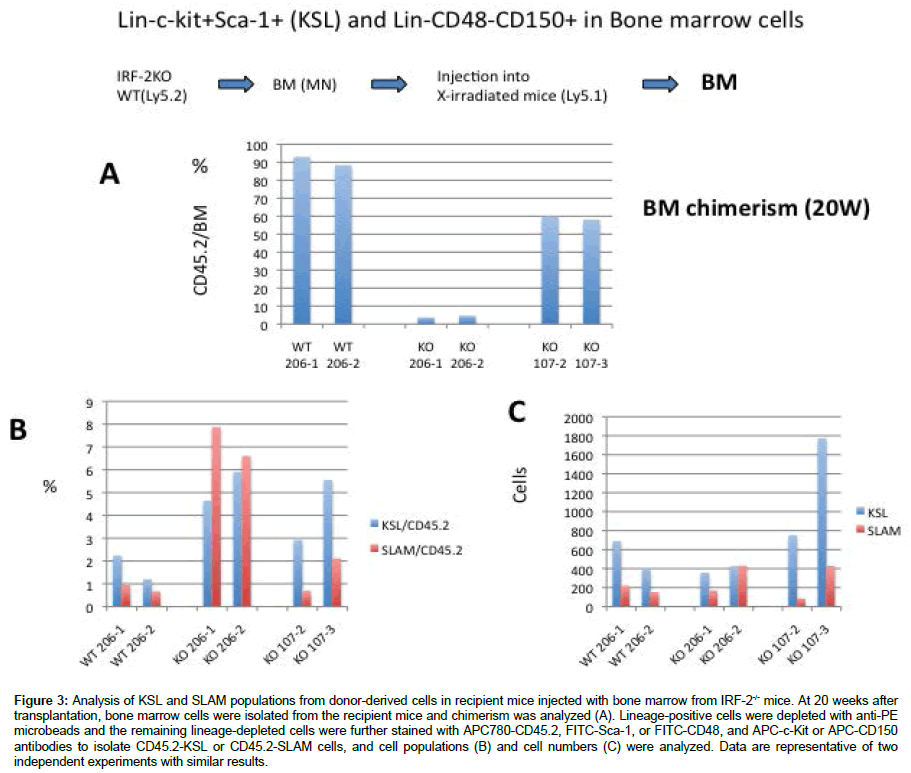
At 18 weeks after transplantation, we analyzed the lineage populations of donor-derived cells from peripheral blood. Donorderived peripheral blood analysis showed a reduction of the Mac1/ Gr1 and CD3 populations, but not the CD19 population (Figure 2). At 20 weeks after transplantation, KSL and Lin- CD48- CD150+ populations from donor bone marrow cells were analyzed in recipient mice injected with 2.0 × 106 or 1.0 × 107 cells from IRF-2-/- mice. The ratio of the populations of these cells was enhanced in recipient mice injected with cells from IRF-2-/- mice compared with recipient mice injected with cells from WT mice (Figure 3).
Figure 2: Lineage analysis from donor-derived cells in recipient mice injected with bone marrow cells from IRF-2-/- mice. At 18 weeks after transplantation, isolated peripheral blood from the recipient mice was stained and chimerism (A) and lineage (B, C, and D) analyses were performed (A). APC-CD45.2-positive cells were stained with PE-Mac1 (B), PE-Gr1 (B), PE-CD19 (C), and PE-CD3 (D). Data are representative of two independent experiments with similar results.
Figure 3: Analysis of KSL and SLAM populations from donor-derived cells in recipient mice injected with bone marrow from IRF-2-/- mice. At 20 weeks after transplantation, bone marrow cells were isolated from the recipient mice and chimerism was analyzed (A). Lineage-positive cells were depleted with anti-PE microbeads and the remaining lineage-depleted cells were further stained with APC780-CD45.2, FITC-Sca-1, or FITC-CD48, and APC-c-Kit or APC-CD150 antibodies to isolate CD45.2-KSL or CD45.2-SLAM cells, and cell populations (B) and cell numbers (C) were analyzed. Data are representative of two independent experiments with similar results.
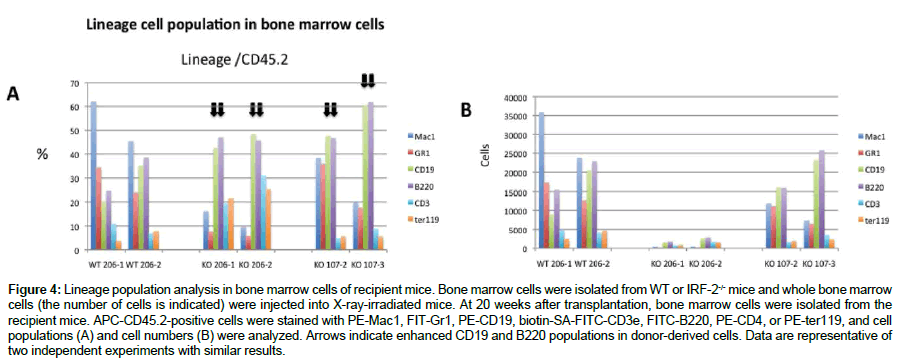
We investigated the lineage populations in bone marrow cells from recipient mice at 20 weeks after injection with bone marrow cells from IRF-2-/- mice. As shown in (Figure 4), the CD19 and B220 cell populations were enriched in mice injected with 2.0 × 106 or 1.0 × 107 bone marrow cells from IRF-2-/- mice, although Mac1+ cells were enriched in recipient mice injected with bone marrow cells from WT mice.
Figure 4: Lineage population analysis in bone marrow cells of recipient mice. Bone marrow cells were isolated from WT or IRF-2-/- mice and whole bone marrow cells (the number of cells is indicated) were injected into X-ray-irradiated mice. At 20 weeks after transplantation, bone marrow cells were isolated from the recipient mice. APC-CD45.2-positive cells were stained with PE-Mac1, FIT-Gr1, PE-CD19, biotin-SA-FITC-CD3e, FITC-B220, PE-CD4, or PE-ter119, and cell populations (A) and cell numbers (B) were analyzed. Arrows indicate enhanced CD19 and B220 populations in donor-derived cells. Data are representative of two independent experiments with similar results.
Discussion
In this study, the function of bone marrow cells from IRF-2-/- mice was investigated. The injection of a large number of bone marrow cells (2.0 × 106 – 1.0 × 107) is, in part, normally sufficient to rescue and fully repopulate the hematopoietic system of some lethally irradiated recipients. The populations of lineage cells from donorderived cells in recipient peripheral blood were different between WT donors and IRF-2-/- donors. A reduction of the Mac1/Gr1 and CD3 populations and slightly higher CD19 population were detected in peripheral blood cells from KO donors. An increase in Mac1+ and Gr1+ cells and a decrease in B220+ cells have been detected in the bone marrow and spleen of IRF-2-/- mice [8]. A reduction of the Mac1/Gr1 population in recipient peripheral blood cells leads to an increase in these populations in bone marrow cells of IRF-2-/- mice. In Table 1, Mac1/Gr1 and CD19 or B220 population in peripheral blood or bone marrow from IRF-2-/- mice or IRF-2-/- donor-derived cells were compared with those of WT mice or WT donor-derived cells. The populations of lineage cells in bone marrow were different between IRF-2-/- mice and recipient mice injected with IRF-2-/- donors. In bone marrow cells of IRF-2-/- mice, the impaired function of HSCs might be maintained by the regulation of lineage cell populations in bone marrow cells.
| Mac1/ Gr1 | CD19 or B220 | |||
|---|---|---|---|---|
| Peripheral Blood | Bone Marrow | Peripheral Blood | Bone Marrow | |
| IRF-2-/- [8] | No Change | Incharge | No Change | Incharge |
| IRF-2-/- Donors | Decrease | Decrease | Slightly Incharge | Incharge |
Table 1: The population in peripheral blood or bone marrow from IRF-2-/- mice and IRF-2-/- donor-derived cells were compared with those of WT mice and WT donorderived cells each other. IRF-2-/-: IRF-2-/- mice, IRF-2-/- donors: recipient mice injected with IRF-2-/- bone marrow cells.
Conclusion
In summary, we found that the blood cell populations of IRF-2-/- mice are rearranged to maintain blood homeostasis. In a clinical field, IRF-2 serves as an essential regulator of HSCs and hematopoiesis including stem cell homeostasis.
Ethics approval and consent of participate
All animal protocols were approved by the Institutional Animal Care and Use Committees of the National Institute of Infectious Diseases.
Acknowledgments
I thank Dr. Kazuya Takizawa for help with carrying out the animal study and FACS analysis, and I also thank Dr. Isao Hamaguchi for his support.
Funding
This work has been done in Department of Safety Research on Blood and Biological Products, National Institute of Infections Diseases, Gakuen, Musashimurayama- Shi, Tokyo 208-0011, Japan. This study was supported in part by a Grant-in-Aid for Scientific Research from the Ministry of Education, Culture, Sports, Science and Technology of Japan.
References
- Papathanasiou P, Attema JL, Karsunky H, Hosen N, Sontani Y, et al. (2009) Self-renewal of the long-term reconstituting subset of hematopoietic stem cells is regulated by Ikaros, Stem Cells 27:3082-3092.
- Gudmundsson KO, Oakley K, Han Y, Du Y (2014) Analyzing gene function in adult long-term hematopoietic stem cells using the interferon inducible Mx1-Cre mouse system, Methods Mol Biol 1194:313-325.
- Wilson A, Laurenti E, Trumpp A (2009) Balancing dormant and self-renewing hematopoietic stem cells, Curr Opin Genet Dev 19:461-468.
- Wilson A, Oser GM, Jaworski M, Blanco-Bose WE, Laurenti E, et al. (2007) Dormant and self-renewing hematopoietic stem cells and their niches, Ann N Y Acad Sci 1106:64-75.
- Scumpia PO, Scumpia KM, Delano MJ, Weinstein JS, Cuenca AG, et al. (2010) Cutting edge: bacterial infection induces hematopoietic stem and progenitor cell expansion in the absence of TLR signaling, J Immunol 184:2247-2251.
- Singh P, Yao Y, Weliver A, Broxmeyer HE, Hong SC, et al. (2008) Vaccinia virus infection modulates the hematopoietic cell compartments in the bone marrow, Stem Cells 26:1009-1016.
- Masumi A, Hamaguchi I, Kuramitsu M, Mizukami T, Takizawa K, et al. (2009) Interferon regulatory factor-2 induces megakaryopoiesis in mouse bone marrow hematopoietic cells, FEBS Lett 583:3493-3500.
- Masumi A, Miyatake S, Kohno T, Matsuyama T (2012) Interferon Regulatory Factor-2 Regulates Hematopoietic Stem Cells in Mouse Bone Marrow, In tech book.
- Sato T, Onai N, Yoshihara H, Arai F, Suda T, et al. (2009) Interferon regulatory factor-2 protects quiescent hematopoietic stem cells from type I interferon-dependent exhaustion, Nat Med 15:696-700.
- Matsuyama T, Kimura T, Kitagawa M, Pfeffer K, Kawakami T, et al (1993) Targeted disruption of IRF-1 or IRF-2 results in abnormal type I IFN gene induction and aberrant lymphocyte development, Cell 75:83-89.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi