Research Article, J Clin Immunol Res Vol: 1 Issue: 1
Interleukin 25 and Systemic Lupus Erythematosus
Dalia A Nigm1* and Marwa A A Galal2
1Department of clinical pathology ,Faculty of medicine, Assiut University, Egypt
2Department of Rheumatology, Rehabilitation and Physical medicine, Faculty of medicine, Assiut University, Egypt
*Corresponding Author : Dalia Ahmad Nigm
Department of Clinical Pathology, Assiut University, Egypt
E-mail: dodonigma@yahoo.com
Received: January 31, 2018 Accepted: March 10, 2018 Published: March 15, 2018
Citation: Nigm DA, Galal MAA (2018) Interleukin 25 and Systemic Lupus Erythematosus. J Clin Immunol Res 1:1.
Abstract
Objectives: We identified that type 2 immunity stimulation has been associated with the pathogenesis of Systemic lupus Erythematous (SLE), so we studied serum level of IL 25 and its relation to SLE activity, clinical and laboratory characteristics. Methods: Serum from 90 patients who met the American College of Rheumatology (ACR) criteria for SLE and 40 healthy controls was tested for IL 25 level by Enzyme Linked Immunosorbent Assay (ELSIA). Clinical and laboratory characteristics and disease activity using SLE Disease Activity Index (SLEDAI) were also assessed. Results: IL 25 serum level was significantly higher among SLE patients than healthy controls. It has statistically significant correlation with disease activity. Conclusion: Our data indicates that IL 25 level is elevated in SLE patients in comparison to healthy controls and is correlated to high disease activity, potentially being a biomarker predictive for disease activity in SLE.
Keywords: Interleukin 25; Systemic lupus erythematous; Lupus nephritis; Systemic lupus erythematous disease activity index.
Introduction
Systemic lupus erythematous (SLE) is a chronic autoimmune disease happen with diverse autoantibodies against self-antigens and affects different organ systems. It has been identified that dysfunctions of the immune system, such as imbalance between Th1/Th2 cytokines is an important factor in SLE pathogenesis [1]. There are contradictory data about predominance of responses by Th1 or Th2, but a rise in Th2 responses is apparent in SLE [2].
Interleukin-17 (IL 17) is a member of a new cytokine family that has recently gained prominence due to its involvement in autoimmune diseases in both human and mouse. There are six members in IL-17 family involving IL 17A, IL 17B, IL 17C, IL 17D, IL 17E (known as IL 25), and IL 17F [3]. While IL 25 has a unique function and structure [4].
Nearly all IL-17 family members have pro-inflammatory role, whereas IL 25 has a different function which stimulate the development of type 2 immune responses (Th2) [5]. IL 25 has receptors (IL-17BR) which are greatly expressed in vivo by stimulation of Th2 cells [6].
Fort et al., [7] was the first one who determines IL 25 by a study performed to analyze the sequence homology in cytokine IL-17 DNA. The sequence of IL 25 is 16-20% consonant to that of IL-17, exposing its particular function. Actually, this cytokine has a stimulatory effect for Th2 cells [8]. IL 25 can also have stimulated the secretion of other cytokines. For example, production of IL 25 in human and mice or injection and induction of IL 25 to animals caused the secretion of high levels of Th2 cytokines, involving IL 13, IL 4, and IL 5 [9]. In addition, by stimulation of transcription factor NF-KB, IL 25 activates the production of IL 8 cytokine in kidney cells. However, the function of this cytokine in renal diseases is still not being interpreted [10]. This cytokine also increases serum secretion of IgA, IgG1 and IgE antibodies, blood eosinophil’s, and eosinophil aggregation in liver [11].
As most autoimmune diseases arise from disruption of balance between the immune cells, in case the autoimmune disease arises from Th1 cells, injection of IL-25 advances differentiation of Th0 to Th2 cells and assembly of their cytokines can be effective in amelioration of the disease [12].
In this study, we investigated the expressions of IL 25 in the serum of SLE patients and evaluated the correlation between serum IL 25 levels and clinical and laboratory values, as well as severity of SLE patients.
Subjects and Methods
Subjects
Ninety SLE cases and 50 healthy controls were included in this work. All cases were admitted to Rheumatology and Rehabilitation Department, Assiut University, Egypt. All subjects met the 1982 and 1997 American College of Rheumatology (ACR) criteria for SLE [13] at the time of recruitment, and were divided into two groups: 40 patients with LN and 50 patients with SLE according to the laboratory data. Controls were sex and age matched, and had no apparent history of SLE, all controls had normal ESR, meaning that they represent a non-inflamed subject. The patients’ written consent was obtained according to the declaration of Helsinki and the study has been approved by local ethics committee prior to their inclusion in the study.
SLE clinical and laboratory characteristics
SLE disease activity was measured by the same physician (SM) in all patients, using the Systemic Lupus Erythematosus Disease Activity Index (SLEDAI) [14]. Active LN was described as active urinary sediment and cellular casts, and/or worsened glomerular filtration rate (GFR) >25 % above baseline/normal range caused by active LN and/or proteinuria ≥ 0.5 g/day, and/or C3 hypocomplementemia. At least two of the previous criteria had to be found and renal biopsy showing lupus nephritis. Sixty five patients (72.2%) had arthritis, 40 (44.4%) patients had a diagnosis of SLErenal disease, 4 (4.4%) patients had Seizures, 5 (5.5%) patients had psychosis 31(34.4%) patients had Malar rash, 16(17.7%) patients had oral ulcers, 9(10%) patients had alopecia (Table 1). Additional measurements included complete blood picture(Beckman Coulter, California, United States), urine analysis (Sysmex UF-1000i™ Automated Urine Particle Analyzer), (urinary 24 hour protein, urea, creatinine, creatinine clearance, glomerular filteration rate, liver function test, lipogram){ using chemiluminescence technology (Dimension ®EXL ™200)}, antinuclear antibodies were quantified using indirect immunofluorescence assay (IFA, ANA-HEp2-IgG, VIRO-IMMUN Labor Diagnostika, GmbH, Oberursel, Germany) and were considered significant when titers ≥ 1:80; anti-dsDNA antibodies were quantified using indirect immunofluorescence assay (NOVA Lite® dsDNA Crithida luciliae, Inova Diagnostics, San Diego, California) and were considered significant when titers ≥ 20 IU/, serum complement 3(C3), complement 4(C4) using nephelometric assay (Behring Nephelometer II, Dade Behring, Marburg, Germany) and high sensitivity C-Reactive protein(hsCRP) using nephelometric assay (Behring Nephelometer II, Dade Behring, Marburg, Germany).
| Characteristics | SLE n=(90 ) |
|---|---|
| Disease duration, years, mean ± SD | 6.13 ± 1.25 |
| SLEDAI categories, n (%) Mild Moderate Severe |
15(16.7%) |
| 34(37.7%) | |
| 41(45.6%) | |
| Anti-dsDNA Ab positive, n (%) | 42(46.6%) |
| C3 consumed, n (%) | 39(43.3%) |
| C4 consumed, n (%) | 36(40%) |
| CRP mean ± SD | 5.3 ± 1.3 |
| Proteinuria ≥ 0.5 mg/24 h, mean ± SD | 205 ± 99 |
| Hematuria, n (%) | 11(12.2%) |
| Urinary casts, n (%) | 18(20%) |
| Arthritis, n (%) | 65(72.2%) |
| Malar Rash, n (%) | 31(34.4%) |
| Oral ulcer, n (%) | 16(17.7%) |
| Seizures, n (%) | 4(4.4%) |
| Psychosis, n (%) | 31(34.4%) |
| Alopecia, n (%) | 9(10%) |
| Leucopenia, n (%) | 8(8.8%) |
| Thrombocytopenia, n (%) | 18(20%) |
| Anemia, n (%) | 42(46.6%) |
| Lupus nephritis, n (%) | 40(44.4%) |
Table 1: Clinical and laboratory characteristics of SLE patients.
Serum cytokine quantification
Serum samples were withdrawn from each patient and control stored at –20ºC until tested. Serum IL-25 levels were analyzed by ELISA kits according to the manufacturer’s protocols (Human IL-25 ELISA reagent kit [Elab Science], Hon- gshan district, wuhai province, china). The results were represented as picograms per milliliter (pg/ mL). All samples were tested in duplicates.
Statistics
The data were tested for normality using the Anderson-Darling test and for homogeneity variances prior to further statistical analysis. Categorical variables were described by number and percent (N, %), where continuous variables described by mean and standard deviation (Mean, SD). Chi-square test and fisher exact test used to compare between categorical variables where compare between continuous variables by t-test and Independent-Samples T test, Mann-whitney U, kruskal wallis test. A two-tailed p < 0.05 was considered statistically significant. All analyses were performed with the IBM SPSS 20.0 software.
Results
The age of 90 subjects with SLE included in the study ranged from 22.5 to 43 years with a median of 28years. All of them are females (100%) for SLE patients. No statistically significant difference was observed between SLE patients and controls concerning age and sex.
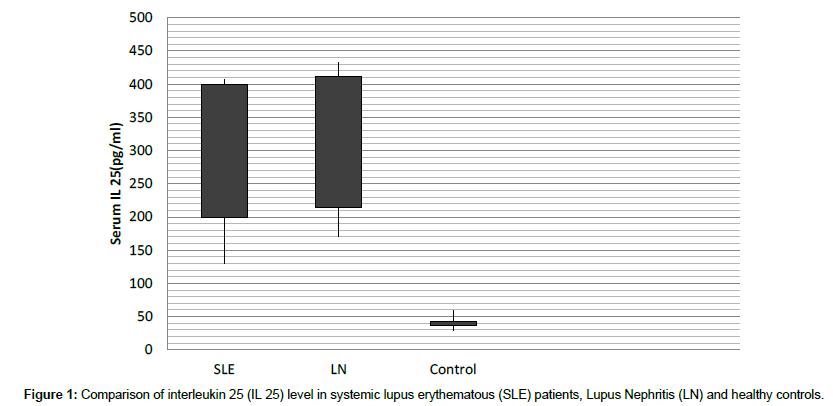
We tested IL 25 in 90 SLE patients and 50 controls. Serum levels of IL 25 were significantly increased in the SLE group (268.24 ± 138.03 pg/mL) compared to the control group (43.83 ± 15.42 pg/mL, P=0.001) (Figure 1).
In our study, we have detected significant positive correlation between high serum IL 25 and Anti ds DNA and C3 (p=0.025, r=0.456) and (p=0.021, r=0.388) respectively, while there is negative correlation between high serum IL 25 and CRP (p=0.027, r=-0.392) (Table 2). Furthermore, linear regression stated that high IL 25 is associated with Anti ds DNA also in an additive model, which remained strongly significant after adjustment for accompanying characteristics, the results given as odds ratio {95% Confidence interval}: 2.9{1.88-5.4} P=0.002 (Tables 3 and 4).
| SLE | LN | Controls | P value | |
|---|---|---|---|---|
| Serum IL25(pg/mL) | 268.24 ± 138.03 | 291.3 ± 120.5 | 43.83 ± 15.42 | 0.001** |
Table 2: Comparison of interleukin 25 (IL 25) level in systemic lupus erythematous (SLE) patients, Lupus Nephritis (LN) and healthy controls.
| Characteristic | High ˃200pg/ml | Low ˂200pg/ml | P.value |
|---|---|---|---|
| Median(Range) | Median(Range) | ||
| Age(years) | 32.5(27.25 - 45) | 34(32 - 42) | 0.257 |
| Duration (years) | 6(4 - 6) | 3.1(1.5 - 11.5) | 0.549 |
| ESR(1sthr)(mm/hr) | 64(21.5 - 90) | 50.5(30.75 - 67.5) | 0.419 |
| ESR (2ndhr)(mm/hr) | 83(42.5 - 115) | 71(49.75 - 98.5) | 0.234 |
| CRP(mg/L) | 2.5(1.55 - 11.35) | 4.5(1.35 - 13.3) | 0.027* |
| ANA(AI) | 34(31.7 5 - 52.45) | 14.4(3.78 - 42.77) | 0.285 |
| Anti-ds DNA(IU/ml) | 123(17.3 - 200) | 63.5(18.9 - 167.5) | 0.025* |
| Hb(g/dL) | 10.5(9.35 - 12.05) | 11.5(8.55 - 12.53) | 0.828 |
| WBCs(103/µl) | 5.78(2.95 - 9.5) | 5.47(5 - 7.16) | 0.878 |
| PLTs (103/µl) | 205.4(166.4 - 259) | 231.5(167.25 - 329) | 0.721 |
| AST(U/L) | 25(19 - 29.5) | 25(14 - 35.5) | 0.852 |
| ALT(U/L) | 18(14 - 29) | 17(7 - 31) | 0.689 |
| S. Cholesterol(mg/dl) | 181.5(122.5 - 197.75) | 202(149.5 - 223) | 0.444 |
| Triglycerides(mg/dl) | 111(63 - 172.75) | 141(124 - 208) | 0.285 |
| HDL(mg/dl) | 57.5(50.5 - 69.75) | 48(37 - 51) | 0.141 |
| LDL(mg/dl) | 121.15(72.75 - 133) | 127(99.3 - 148.65) | 0.543 |
| C3(g/L) | 0.55(0.35 - 1.12) | 0.81(0.82 - 0.97) | 0.021* |
| C4(g/L) | 0.14(0.08 - 0.2) | 0.26(0.26 - 0.7) | 0.052 |
| Urine WBCs(hpf) | 10(0 - 28.75) | 0(0 - 32.5) | 0.633 |
| Urine RBCs(hpf) | 1(0 - 3.75) | 5(0 - 31) | 0.438 |
| Urine 24hr Protein(mg/24hr) | 191.5(28.5 - 730.5) | 200(98.8 - 1655.5) | 0.960 |
| Createnine Clearance(ml/min) | 98.15(67.5 - 129.5) | 98.5(62 - 143.3) | 0.496 |
| S. creatinine(µm/L) | 64.5(33.13 - 88.5) | 58(47.5 - 74.52) | 0.894 |
| eGFR (ml/min/1.73m²) | 99.5(65.25 - 118.25) | 108(81.5 - 141.5) | 0.515 |
Table 3: Laboratory characteristics of SLE patients with regard to the serum IL 25 level.
| Disease activity score | Mean (SD) pg/mL | P-value |
|---|---|---|
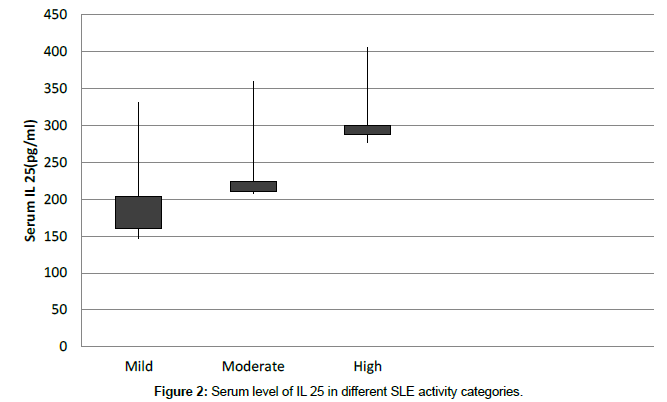
| Mild | 239.12 ± 92.33 | 0.031* |
| Moderate | 283.81 ± 76.34 | |
| High | 341.57 ± 64.63 |
Table 4: Interleukin 25 serum levels in different Systemic Lupus Erythematous Disease Activity Index categories.
However, no significant associations were observed between high serum IL 25 and either the following laboratory and clinical characteristics: ANA, ESR, complete blood picture, Lipogram,C4, creatinine, oral ulcers, arthritis, vasculitis, pericarditis, pleurisy and psychosis (Table 2).
A significant correlation was observed for elevated serum IL 25 with clinical disease activity scores (P = 0.011, r = 0.55) as well as different SLEDAI categories (Table 3 and Figure 2).
Further analysis was made for the data of SLE patients to investigate the association of serum IL 25 with renal involvement. SLE patients were divided into two groups according to the presence of renal involvement, but we did not find a significant correlation between serum IL 25 and lupus nephritis.
Discussion
Currently, many studies reported the role of IL 25 in autoimmune diseases as an inhibitor of both adaptive and innate immunity and an anti-inflammatory cytokine [15]. In organ-specific autoimmunity, the equivalence of cytokines is an essential determinant of susceptibility and resistance. There is an important argument in SLE which is the imbalance between Th1/Th2 cytokines. However, there are conflicting opinions about superiority of responses by Th1 or Th2, a rise in Th2 responses is noticeable in SLE [16]. In SLE, the secretion of prion flammatory cytokines, for instance IFN γ, TNF α, IL 6 and IL 17 is observant to associate with disease susceptibility. Nevertheless, Th2 cytokines, like IL 13, IL 4 and IL 5, have been revealed to take an important role in alleviating and even prohibiting autoimmune diseases [17].
In the present study, all SLE cases are females which can be explained by the increased susceptibility may be due to differences in hormones and sex chromosomes [18]. We studied serum IL 25 level, a Th2 stimulating cytokine in SLE patients and analyzed their correlations with laboratory and clinical characteristics. Our results found significant high level of IL 25 serum in SLE patients when compared to controls (Table 2).
To study the associations between disease biomarkers and serum IL 25 levels, we studied correlations between laboratory markers and IL 25, including anti-dsDNA, ANA, CRP, consumed C3 and C4. We found significant positive correlation between serum IL 25 and ds DNA and consumed C3. While there is significant negative correlations with CRP levels. On the other hand, we did not find significant correlation between serum IL 25 and the other markers (Table 5).
| Serum IL 25 | Crude OR (95% CI) p- value | Adjusted OR (95% CI) p- value |
|---|---|---|
| High serum IL 25 | 2.9(1.88-5.4) 0.012 | 2.1(1.45-4.5) 0.015 |
| Low serum IL 25 | 0.84(0.35-1.9) 0.456 | 0.76(0.25-1.5) 0.332 |
Table 5: The effect of high IL 25 on the risk of SLE analyzed by logistic regression.
Stated that IL 25 level was increased in both of serum and synovial fluid of Rheumatoid Arthritis (RA) patients when compared to osteoarthritis (OA) patients and controls [19]. In this study we obviously found that IL 25 levels were significantly higher in SLE patients with active disease than in patients with inactive disease. Also, revealed that higher serum and synovial fluid levels of IL 25 were found in RA patients with severe and moderate disease activity when compared to those patients with low disease activity and with obvious comparison to patients in remission [20].
Previous study reported that IL 25 has an important role on treatment of autoimmune diseases in kidney patients [21], for example Adriamycin nephropathy (AN) which is a kidney disease characterized by proteinuria. This disease is aggravated by Adriamycin. Qicao et al., 2011 found that treatment of the cases with IL 25, the urinary loss of proteins significantly lower than in the mice without treatment [22].
So, other studies are needed to be done on SLE to further explain the role of IL 25 in the pathogenesis and treatment of these diseases.
References
- Apostolidis SA, Lieberman LA, Kis-Toth K, Crispin JC, Tsokos GC (2011) The dysregulation of cytokine networks in systemic lupus erythematosus. J Interferon Cytokine Res 31: 769-0779.
- Perl A (2010) Systems biology of lupus: mapping the impact of genomic and environmental factors on gene expression signatures, cellular signaling, metabolic pathways, hormonal and cytokine imbalance, and selecting targets for treatment. Autoimmunity 43: 32-47.
- Huang SH, Frydas S, Kempuraj D, Barbacane, RC, Grilli A, et al. (2004) Interleukin-17 and the interleukin-17 family member network. Allergy Asthma Proc 25: 17-21.
- FossiezF, BanchereauJ, Murray R, Van Kooten C, Garrone P, et al. (1998) Interleukin-17. Int Rev Immunol 16: 541-551.
- Iwakura Y, Ishigame H, Saijo S, Nakae S (2011) Functional specialization of interleukin-[1] 17 family members. Immunity 34: 149-162.
- Rouvier E, Luciani MF, Mattei MG, Denizot F, Golstein P (1993) CTLA-8, cloned from an activated T cell, bearing AU-rich messenger RNA instability sequences, and homologous to a herpesvirus saimiri gene. J Immunol 150: 5445-56
- Fort MM, Cheung J, Yen D, Li J, Menon S, et al. (2001) IL-25 induces IL-4, IL-5, and IL-13 and Th2- associated pathologies in vivo. Immunity 15: 985-95.
- Ikeda K, Nakajima H, Suzuki K, Kagami SI, Hirose K, et al. (2003) Mast cells produce interleukin- 25 upon Fc epsilon RI mediated activation. Blood 101: 3594-3596.
- Gossner A, Peers A, Venturina V, Hopkins J (2011) Expressed gene sequences of two variants of sheep interleukin 25 Vet Immunopathlol 139: 319-323.
- Ikeda k, Nakajima H, Suzuki K, Kagami SI, Hirose K, et al. (2003) Most cells produce interlukin 25 upon activation. Blood 101: 4-6.
- Létuvé S, Lajoie-Kadoch S, Audusseau S, Rothenberg ME, Fiset PO, et al. (2006) I-17E upregulates the expression of proinflammatory cytokines in lung fibroblasts. J Allergy clin Immunol 117: 590-596.
- Valizadeh A, Khosravi A, Zadeh LJ, Parizad EG (2015) Role of IL 25 in immunity. Journal of Clinical and Diagnostic Research 9: 1-4.
- Hochberg MC (1997) Updating the american college of rheumatology revised criteria for the classification of systemic lupus erythematosus. Arthritis Rheum 40: 770-771.
- Liang MH, Socher SA, Larson MG, Schur PH (1989) Reliability and validity of six systems for the clinical assessment of disease activity in systemic lupus erythematosus. Arthritis Rheum 32: 1107-1118.
- Murst SD, Muchamuel T, Gorman DM, Gilbert JM, Clifford T, et al. (2002) New IL 17 family member's promote Th1 or Th2 responses in the lung: in vivo function of the novel cytokine IL-25. J Immunol 169: 443-453.
- Namazi S, Ziaee V, Rezaei N (2015) The role of cytokines in systemic lupus erythematosis: review article. Tehran Univ Medical J 73: 397-404.
- Sharkhuu T, Matthaei K I, Forbes E, Mahalingam S, Foster PS, et al. (2006) Mechanism of interleukin-25 (IL-17E)-induced pulmonary inflammation and airways hyper-reactivity. Clin Exp Allergy 36: 1575-1583
- Kasama T, Maeoka A, Oguro N (2016) Clinical features of neuropsychiatric syndromes in systemic lupus erythematosus and other connective tissue diseases. clinical medicine insights. Clin Med Insights Arthritis Musculoskelet Disord 9: 1-8.
- Gumus P, Buduneli E, Buduneli N, Aksu K,Sarac F, et al. (2013) Gingival crevicular fluid, serum levels of receptor activator of nuclear factor-kappaB ligand, osteoprotegerin, and interleukin-17 in patients with rheumatoid arthritis and osteoporosis and with periodontal disease. J Periodontol 84: 1627-1637.
- Kaiwen W, Zhaoliang S,Yinxia Z, Siamak SS, Zhijun J, et al. (2012) Changes and significance of IL-25 in chicken collagen II-induced experimental arthritis (CIA). Rheumatol Int 32: 2331–2338.
- Saenz SA, BC Taylor, D Aritis (2008) Welcome to the neighbodhood epithelial cell- derived cytokines license innate and adaptive immune responses atmucosal sites. Immunol Rev 226: 172-190.
- Angkasekwinai P, H park, YH wang, Wang YH, Chang SH, et al. (2008) Inretleukin 25 promotes the initiation of proallergic type 2 responses. J Exp Med 204: 1509-1517.
 Spanish
Spanish  Chinese
Chinese  Russian
Russian  German
German  French
French  Japanese
Japanese  Portuguese
Portuguese  Hindi
Hindi